Abstract

Keywords
Clinical History, Laboratory Results, and Gross Findings
A 5-year-old spayed female Portuguese water dog was presented to a veterinary teaching hospital on referral for marked liver enzyme elevation (aspartate transaminase [AST] 1473 IU/L [ref: 15–66 IU/L], alanine transaminase [ALT] 1152 IU/L [ref: 12–118 IU/L], alkaline phosphatase [ALP] 1907 IU/L [ref: 5–131 IU/L]), hyperbilirubinemia (total bilirubin 35.9 µmol/L [ref: 1.71–5.13 µmol/L]), a leukocytosis characterized by a mature neutrophilia of 15.7 x 109 cells/L (ref: 2.1–10.6 x 109 cells/L), coughing and gagging. Hematocrit was 0.53 L/L (ref: 0.36–0.60 L/L) and total protein was 64 g/L (ref: 50–74 g/L). Four days prior to presentation, the dog began vomiting and became lethargic and hyporexic with the development of a hacking cough. Physical examination showed bilaterally enlarged mandibular lymph nodes and salivary glands, pain on opening of the mouth, reduced right cranial bronchovesicular sound on auscultation, and a cough on tracheal palpation. Cytology of the salivary glands and lymph nodes showed a neutrophilic to lymphohistiocytic sialadenitis and reactive lymphoid hyperplasia with neutrophilic infiltration. Thoracic radiographs revealed a diffuse bronchointerstitial pattern, and the cough, gag, and auscultatory findings were attributed to presumed aspiration pneumonia. Pertinent history included being fed a raw diet and living with four other dogs in a rural/agricultural setting.
An initial abdominal ultrasound was consistent with pancreatitis and possible hepatitis (patchy hyperechoic pancreatic regions and peripancreatic adipose tissue, and mild hypoechoic liver) with a normal gallbladder appearance. The dog was treated with ampicillin, enrofloxacin, methadone, metronidazole, N-acetylcysteine, and gastrointestinal supportive medications. The liver enzymology and hyperbilirubinemia worsened, so serial ultrasounds were performed which revealed progressive gallbladder wall thickening with no evidence of bile duct obstruction, and resolution of pancreatic changes. Antibiotics were switched to clindamycin and ceftazidime, and ursodiol, vitamin E, and an anti-inflammatory dose of dexamethasone SP were added to the treatments. Four wedge laparoscopic liver biopsy samples were obtained and revealed moderate to marked midzonal to centrilobular hepatocellular swelling and clearing (consistent with a steroid hepatopathy), canalicular bile stasis, and neutrophilic and lymphoplasmacytic portal infiltrates (Fig. 1). Copper quantification was normal (125 ppm), and additional diagnostics for infectious agents—including a liver culture, fecal float and sediment examination, Toxoplasma titers, and vector-borne disease panel (Anaplasma, Babesia, Bartonella, Ehrlichia, hemotrophic Mycoplasma, Rickettsia, Borrelia burgdorferi, Dirofilaria immitus)—were negative. At the time of presentation, the patient was negative for IgM antibodies to Leptospira canicola, L. grippotyphosa, L. icterohaemorrhagiae, and L. pomona (WITNESS canine leptospira antibody test). Six days following presentation, Leptospira polymerase chain reaction and titers (L. pomona, L. icterohaemorrhagiae, L. canicola, L. grippotyphosa, L. hardjo, L. autumnalis, and L. bratislava) were negative, and convalescent titers 7 days later were 1:50 for L. icterohaemorrhagiae.
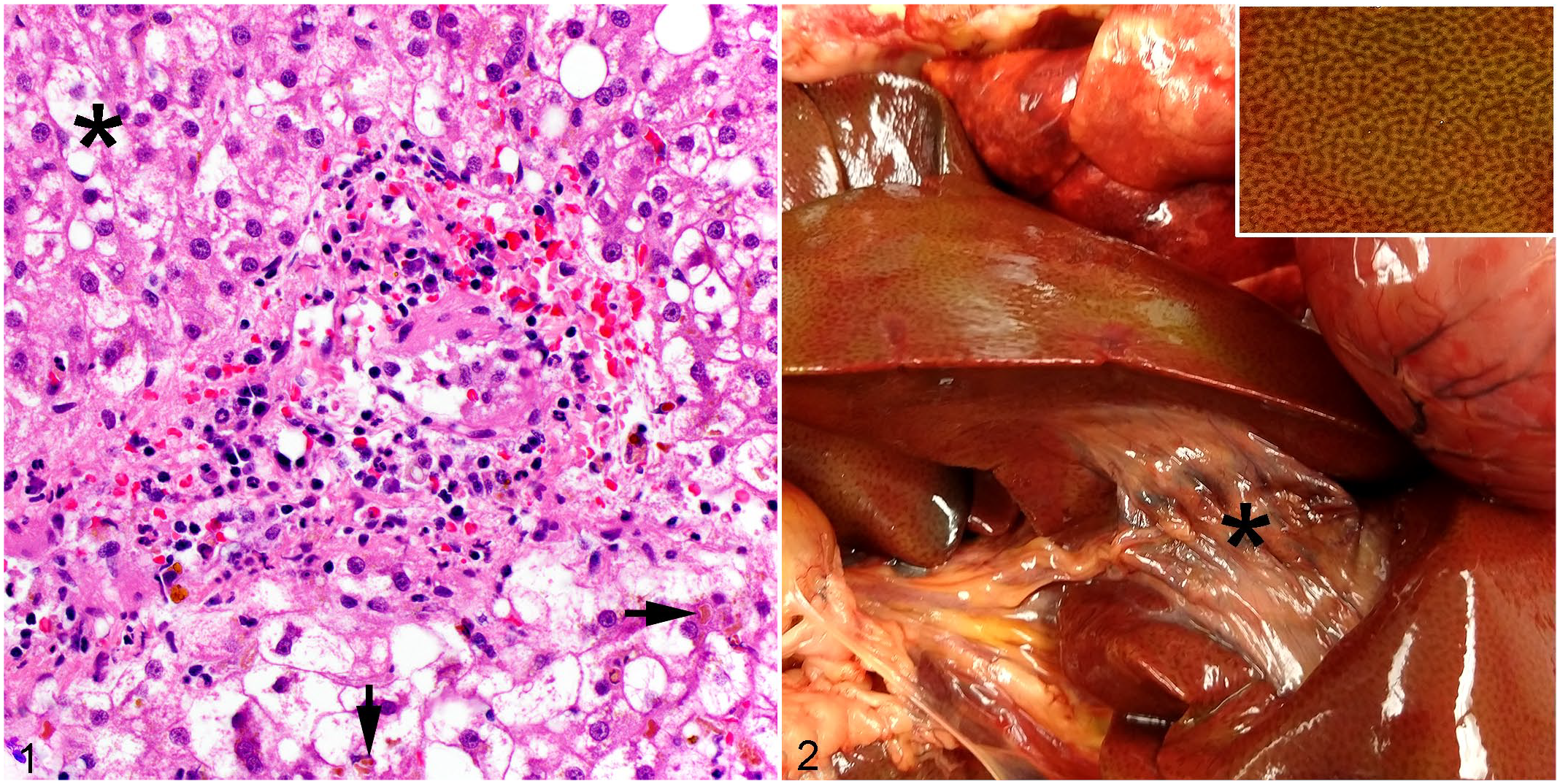
Liver, dog. There is moderate to marked midzonal to centrilobular hepatocellular swelling and clearing (asterisk), with canalicular bile stasis (arrows), and neutrophilic and lymphoplasmacytic portal infiltrates. Hematoxylin and eosin.
The patient continued to decline with progression of aspiration pneumonia, progressive acute kidney injury, worsening icterus, and an esophagostomy tube site infection with multiple drug-resistant Enterobacter cloacae complex and methicillin-resistant Staphylococcus pseudintermedius. Over the course of treatment, the sialadenitis resolved, but liver enzymes (ALT 344 IU/L [ref: 16–91 IU/L], AST 94 IU/L [ref: 23–65 IU/L], ALP 1693 IU/L [ref: 20–155 IU/L], γ-glutamyl transferase [GGT] 152 IU/L [ref: 7–24 IU/L]) remained elevated with ALP and GGT greater than 4× the upper reference interval at the time of discharge, 19 days following presentation. Hyperbilirubinemia continued to increase, reaching 513 µmol/L (ref: 1.71–5.13 µmol/L) at the time of euthanasia, 29 days following presentation. An autopsy was performed 2 days later (31 days following presentation), and significant macroscopic changes included marked generalized icterus, mild hepatomegaly with an enhanced reticular pattern and yellow-green discoloration, and a shrunken gallbladder surrounded by increased fibrous connective tissue and devoid of bile (Fig. 2).
Microscopic Findings
Microscopic examination of postmortem liver samples revealed numerous centrilobular to midzonal canalicular bile plugs and severe midzonal to centrilobular hepatocellular swelling and clearing (Fig. 3). Portal regions were infiltrated by low numbers of neutrophils, lymphocytes, and plasma cells with indistinct bile ducts. The gallbladder and common bile duct epithelium had moderate multifocal to coalescing lymphoplasmacytic and neutrophilic infiltrates surrounded by mildly increased fibrous connective tissue. Immunohistochemistry for cytokeratin 7 (CK7), an intermediate filament found in biliary epithelial cells, revealed the loss of bile ducts from nearly all small portal regions, with 2 per 23 portal regions examined having residual ducts demonstrating strong CK7 cytoplasmic labeling (Figs. 4, 5).
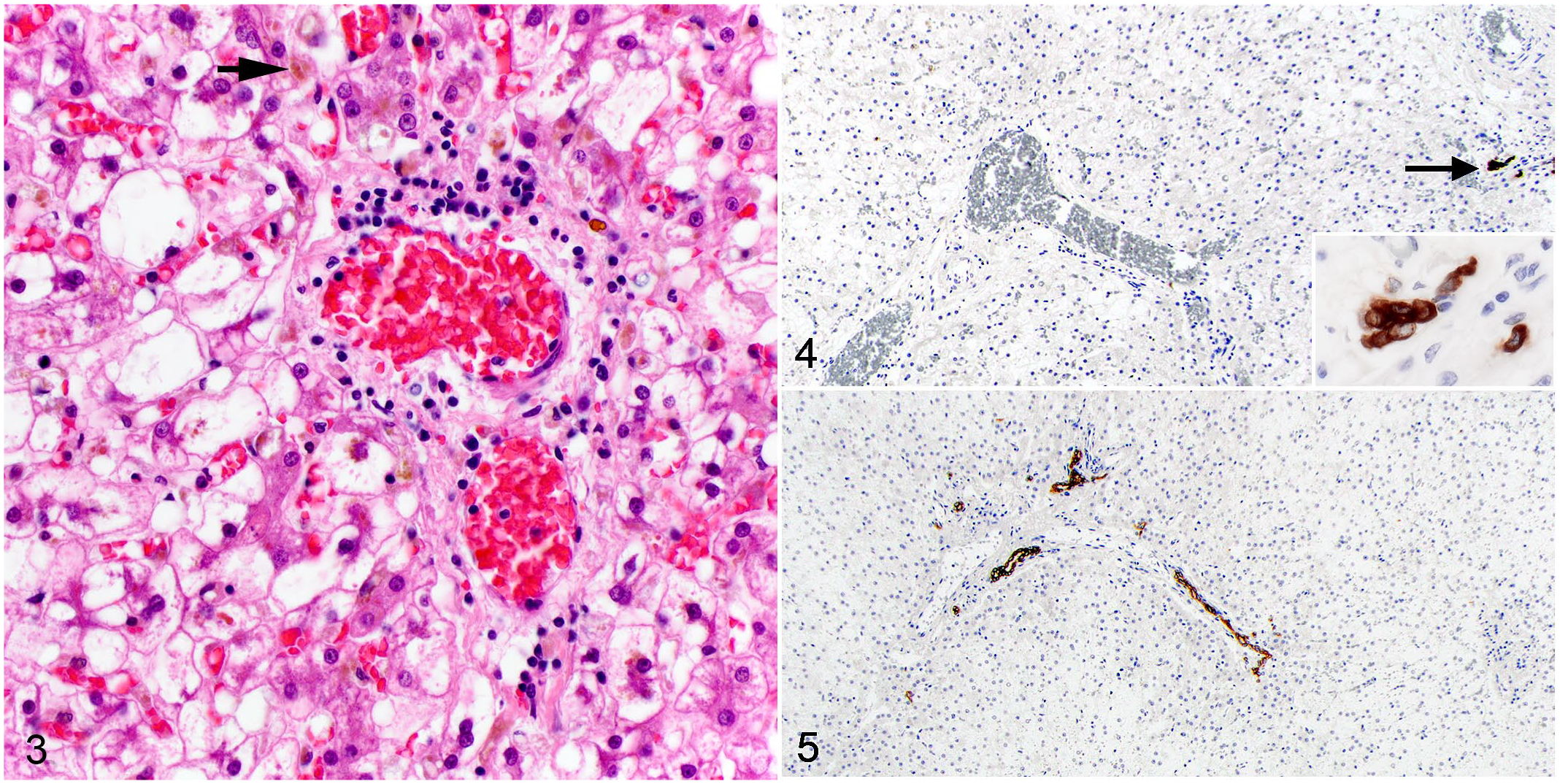
Liver (postmortem sample), dog. There is progressive, severe centrilobular and midzonal hepatocellular swelling with abundant canalicular bile plugs (arrow). Portal regions are infiltrated by fewer neutrophils, lymphocytes, and plasma cells relative to the biopsy, and pigment-laden stellate macrophages (Kupffer cells) are scattered throughout the parenchyma. Bile ducts are inapparent. Hematoxylin and eosin.
Differential Diagnoses
Hyperbilirubinemia can be due to pre-hepatic, hepatic, and post-hepatic causes. Pre-hepatic causes result from increased bilirubin production, typically due to hemolysis or severe internal hemorrhage. These causes were ruled out by lack of significant anemia or internal hemorrhage, and the hepatic enzymology changes. Hepatic causes are due to decreased hepatic uptake or reduced conjugation or secretion of bile, typically in the context of loss of hepatic failure or sepsis. Given the severity of the hyperbilirubinemia, a hepatic cause would require widespread, severe disease such as a diffuse toxic or inflammatory insult or cirrhosis. Steroid hepatopathy can result in minimal to mild cholestasis if the swelling of the hepatocytes is severe and compresses canalicular bile ducts, but would not account for the severity of cholestasis and hyperbilirubinemia in this case. Given the lack of other changes to the hepatocytes, a hepatic cause of hyperbilirubinemia was considered less likely. Post-hepatic causes are due to impaired outflow of bile from the liver, or cholestasis. Cholestasis as the primary cause of hyperbilirubinemia in this case was supported by the severe increases in ALP and GGT, and the histopathologic findings, namely the canalicular bile plugs. The combination of inflammation centered on hepatic portal regions, the gallbladder and common bile duct, was consistent with cholecystitis and cholangitis. While the steroid hepatopathy may have played a contributory role, the severity of the hyperbilirubinemia was supportive of the cholangitis and cholecystitis as the primary cause of cholestasis in this case.
Differential diagnoses for cholangitis in dogs and cats fall into 4 categories, according to the World Small Animal Veterinary Association (WSAVA) guidelines: (1) neutrophilic cholangitis, (2) lymphocytic cholangitis, (3) chronic cholangitis associated with liver fluke infestation, and (4) destructive cholangitis. 5 Chronic cholangitis associated with liver fluke infestation is ruled out by the absence of liver flukes, eggs, and associated bile duct ectasia, bile duct epithelial proliferation, and periductal fibrosis. 5 Lymphocytic cholangitis, which is more common in cats, is ruled out by the lack of abundant small lymphocytes within portal regions centered on or within the biliary epithelium. 5 Neutrophilic cholangitis is the most common type encountered in dogs and is attributed to ascending bacterial infection from the intestine. 2 Histologically, neutrophilic cholangitis is characterized by neutrophilic infiltrates within the biliary epithelium and/or lumen with extension into portal regions. 5 Chronic neutrophilic cholangitis frequently has mixed lymphoplasmacytic and neutrophilic inflammation with bile duct proliferation, and concurrent biliary obstruction is common resulting in bile duct ectasia and periductular fibrosis. 2 Given the relatively mild inflammation, particularly at the time of autopsy, and lack of bile duct ectasia or periductal fibrosis, neutrophilic cholangitis is considered less likely in this case. Destructive cholangitis is characterized by loss of interlobular bile ducts in 50% or more of small portal regions, and infiltrates composed of pigment-laden macrophages and neutrophils.1,5 Chronic lesions may develop portal fibrosis and are often associated with severe cholestasis and icterus. 5 Cholecystitis may be associated with ascending bacterial infection, parasitism, or metabolic and idiopathic processes (e.g., cholelithiasis and mucocele formation).
Further Investigations and Diagnosis
The marked cholestasis and ductopenia—with 2 residual bile ducts per 23 portal regions examined—confirm the diagnosis of destructive cholangitis in this case. Escherichia coli, Streptococcus agalactiae, and Enterococcus faecalis were isolated with aerobic culture of the gallbladder at the time of autopsy. Two of these isolates, E. coli and E. faecalis, are the most common isolates reported in cholangitis and cholangiohepatitis in dogs (though they may also be encountered as postmortem contaminants). 2 Concurrent chronic neutrophilic cholecystitis was also diagnosed. These findings, in addition to significant initial increases in AST and ALT which improved following antimicrobial therapy, and the moderate to marked neutrophilic and lymphoplasmacytic portal infiltrates at the time of biopsy, support a concurrent neutrophilic cholangitis and secondary hepatocellular damage early in the course of disease. The lack of secondary portal lesions attributed to chronic cholecystitis, including bile duct proliferation, ectasia, and periductal fibrosis, is likely due to the destruction of the bile duct epithelium as the destructive cholangitis progressed. The mild hepatomegaly and enhanced reticular pattern noted grossly is attributable to the steroid hepatopathy, likely secondary to a combination of endogenous and exogenous steroids.
Discussion
Destructive cholangitis is a rare form of cholangitis in dogs with only 10 cases reported in the literature.1,3,4,6 The underlying pathogenesis is not fully elucidated, but destructive cholangitis has been associated with possible idiosyncratic drug reactions (potentiated sulfonamides, amoxicillin-clavulanate, amitraz, and milbemycin oxime), toxic insults (paraquat, and 4,4’ diaminodiphenylmethane), and canine distemper virus infection.3,5,6 As this case presented with elevated liver enzyme concentrations and hyperbilirubinemia prior to medication administration, the underlying cause of destructive cholangitis in this case was undetermined. Destructive cholangitis carries a poor prognosis: of the 10 reported cases, 7 survived 7 weeks or less, 2 survived 1 year and 6 months (at the time of publication), and 1 had an unknown survival time.1,3,4,6 Interestingly, in the current case, the biliary epithelium of the gallbladder and major ducts was intact and histologically unremarkable.
Destructive cholangitis is defined by the loss of interlobar bile ducts in 50% or more of small portal tracts. 1 Therefore, histologic examination early in the disease course, prior to complete loss of bile ducts, may preclude a definitive diagnosis. In addition, there is histologic overlap between destructive cholangitis and acute neutrophilic cholangitis, prior to the development of bile duct proliferation and periductal fibrosis, as both are characterized by neutrophilic biliary infiltrates. In this case, the distinction was further complicated by superimposed chronic neutrophilic cholecystitis and aerobic culture results. The discrepancy in aerobic bacterial cultures may be due to the concurrent antimicrobial therapy or the higher sensitivity of biliary culture versus hepatic culture in detecting bactibilia. 7 This case demonstrates the challenges of concurrent disease processes, the benefit of serial histopathologic examination, and the importance for the pathologist to not only describe what is present on a slide, but also hunt for what may be missing.
Footnotes
Acknowledgements
We thank Brona Ranieri for performing the immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
