Abstract
Amyloid-producing odontogenic tumors (APOTs) of the facial skin were diagnosed in 3 domestic cats. The neoplasms had the histopathological characteristics of the odontogenic tumor. The neoplastic cells were present in irregular islands, strands, and sheets. The peripheral neoplastic cells of the islands and strands were arranged in a palisading fashion, while the central cells were polyhedral to stellate and randomly arranged. Multiple spherules of homogeneous eosinophilic material were closely apposed to the neoplastic epithelial cells. The spherules stained with Congo red and produced an apple green birefringence under polarization microscopy, indicative of amyloid. Immunohistochemically, amyloid materials of the neoplasms reacted with polyclonal antibodies for ameloblastin, amelogenin, and sheathlin antibodies. Neoplastic epithelial cells also reacted with antiameloblastin, amelogenin, and sheathlin antibodies, with varied intensity. The histopathological and immunohistochemical characteristics of dermal neoplasms of the 3 cats were analogous to those of APOTs reported in the dog and the cat.
Keywords
Amyloid-producing odontogenic tumor (APOT) is characterized by the proliferation of odontogenic epithelium, accompanied by intercellular deposition of amyloid materials. 4 APOTs have been reported in a few cases of dogs, 5,9 cats, 1,2,9 a goat, 7 and a tiger. 6 APOTs usually show expansile growth, form solid or cystic masses, and typically have benign behavior, but can invade locally, incorporate teeth, and may gradually destroy the jaw and facial bones in the long term. 4 Tumors occasionally recur following excision. 4 APOTs in the dog and the cat are believed to be neoplasms, and reported cases have been restricted to the gingiva of the mandible and maxilla. 4
Biochemical and immunohistochemical examination of canine APOT revealed that the amyloid proteins in the neoplasm are composed of enamel protein lineage (such as ameloblastin, amelogenin and sheathlin) secreted by neoplastic ameloblasts. 5 In addition, the neoplastic epithelial cells express enamel proteins. 5 The neoplastic epithelial cells and intratumoral amyloid are considered to be of ameloblastic lineage in canine and feline APOTs. 2,5
To the best of our knowledge, there have been no reports of APOTs occurring on the facial skin of any animals. This report describes the histologic and immunohistochemical features of APOT occurring in the supraorbital and labial dermis of 3 cats.
Case 1, a 5-year-old domestic male cat, had a small solid mass (0.8 cm diameter) on the left supraorbital skin (Fig. 1). Case 2, a 10-year-old domestic female cat, had a small solid mass (1.2 cm diameter) on the left labial skin. Case 3, a 4-year-old domestic castrated cat had a small mass (2 cm diameter) on the left labial skin. The solid dermal masses were clearly demarcated from the surrounding tissue and were easily excised. The neoplasms have not recurred after their removal. There were no tumors in the oral cavity of these 3 cats.
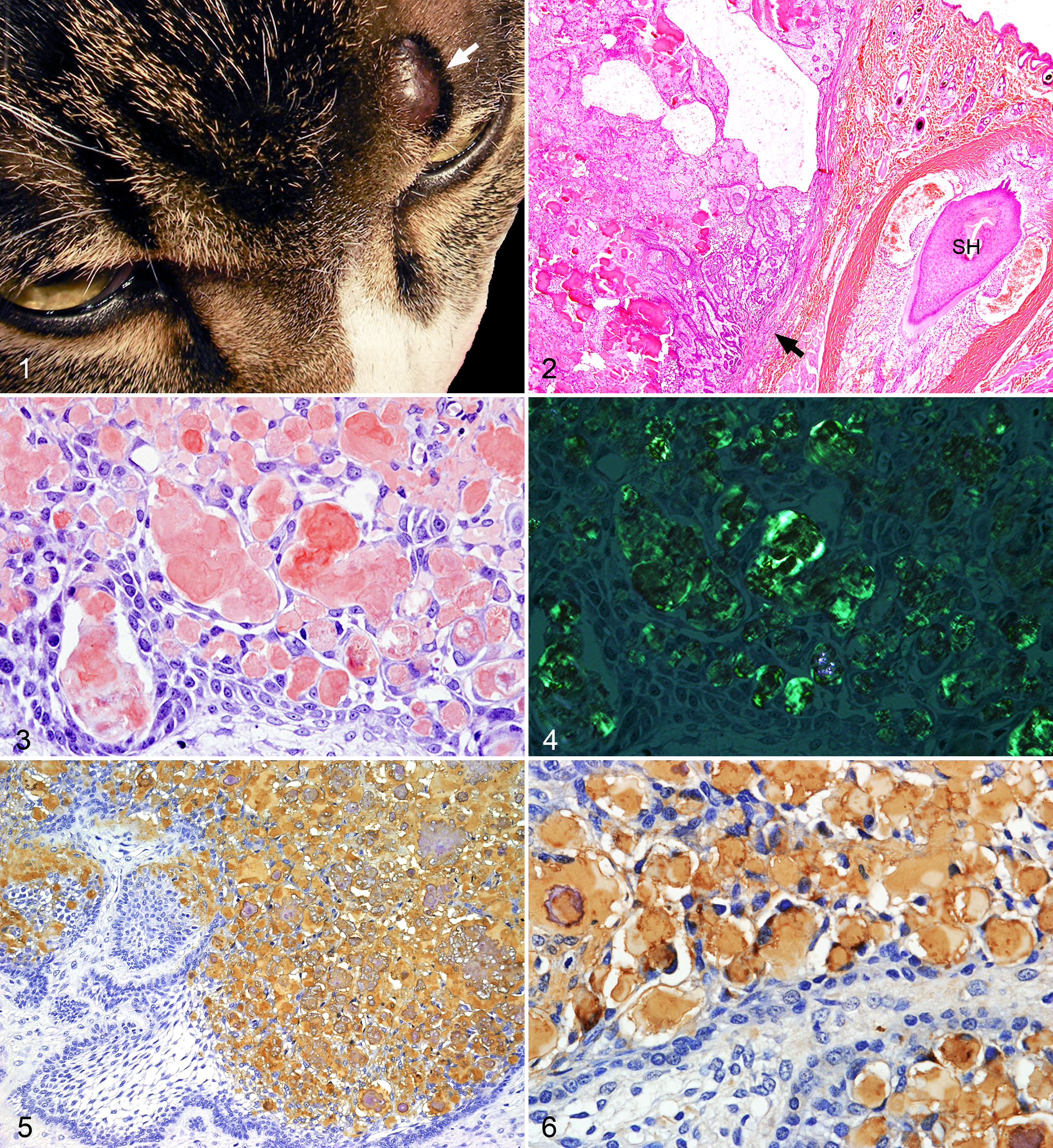
The neoplastic tissues removed surgically were fixed in 10% formalin solution, decalcified as needed, processed routinely, and stained with hematoxylin and eosin (HE). Additional sections were stained with Congo red with and without prior 5% potassium permanganate treatment. Congo red–stained sections were microscopically examined under polarized light.
Histological sections of the neoplasms from 3 patient cats were examined immunohistochemically using the avidin-biotin-peroxidase complex procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA, USA). Endogenous peroxidase was blocked in deparaffinized sections by incubation in 3% H2O2 for 10 minutes. All sections were incubated with primary antibody (described below) at 4°C for 16 hours, with biotinylated secondary antibody for 30 minutes at room temperature, and with ABC for an additional 30 minutes. The immunologic reaction was detected with 0.05% 3,30-diaminobenzidine as chromogen. For positive controls, normal tooth germ tissues from 3 canine and 2 feline fetuses were used. Negative control procedures included the use of normal mouse and rabbit serum instead of the primary antibody.
Antirat ameloblastin (residues 98–107), antiporcine amelogenin (25 kDa), and antiporcine sheathlin (N-terminal) polyclonal antibodies were donated. Commercial antibodies against human ameloblastin and human amelogenin proteins were purchased (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Polyclonal antiporcine vimentin, antihuman desmin, and antihuman smooth muscle actin (SMA) antibodies, and monoclonal antihuman cytokeratin (CK) AE1/AE3, CK10, CK13, CK14, and CK19 antibodies were also used for immunohistochemical evaluations of in situ amyloid protein, neoplastic epithelial cells of the neoplasms, and the normal tooth germ tissues. Details of the antibodies used are summarized in Supplemental Table 1.
APOTs of the facial skin in the 3 cats examined were clearly demarcated from surrounding dermal tissues by fibro-vascular connective tissue. Infiltration of the neoplasm into the surrounding soft tissue was not observed (Fig. 2). The neoplasm was composed of neoplastic epithelial cells arranged in islands, strands and sheets. The peripheral epithelial cells were arranged in a palisading fashion. The central cells of some islands consisted of polyhedral stellate reticulum cells and were randomly arranged. Intercellular eosinophilic materials were congophilic with and without 5% potassium permanganate pretreatment (Fig. 3), and had apple-green birefringence under polarized light microscopy (Fig. 4). Mineralized amyloid materials and small bone tissue were frequently observed. Small cysts lined by the cuboidal or flat neoplastic epithelial cells were observed in 3 cats.
By immunohistochemistry, intratumoral amyloid reacted with antibodies against ameloblastin (Fig. 5), amelogenin (Fig. 6), and sheathlin. The cytoplasm of neoplastic epithelial cells were also variably positive with the above antibodies (Figs. 5 and 6). The intratumoral amyloid did not react with antibodies against CKs, vimentin, desmin, and SMA. The neoplastic epithelial cells reacted with antibodies against CK AE1/AE3, CK13 and CK14, but not with those against CK10. The central stellate cells of some islands reacted with anti-CK19 antibody. The neoplastic cells did not express vimentin, desmin, SMA, or GFAP. As controls, the enamel matrix of normal tooth germ from 3 canine and 2 feline fetuses reacted for ameloblastin, amelogenin and sheathlin. Ameloblasts reacted for ameloblastin, amelogenin, sheathlin, AE1/AE3, CK13, CK14, and CK19, but not for CK10, vimentin, desmin, and SMA. The results of immunohistochemical examination were summarized in Table 1.
Immunohistochemical Findings in 3 Feline Amyloid-Producing Odontogenic Tumors of Facial Skin and in Normal Tooth Germ.
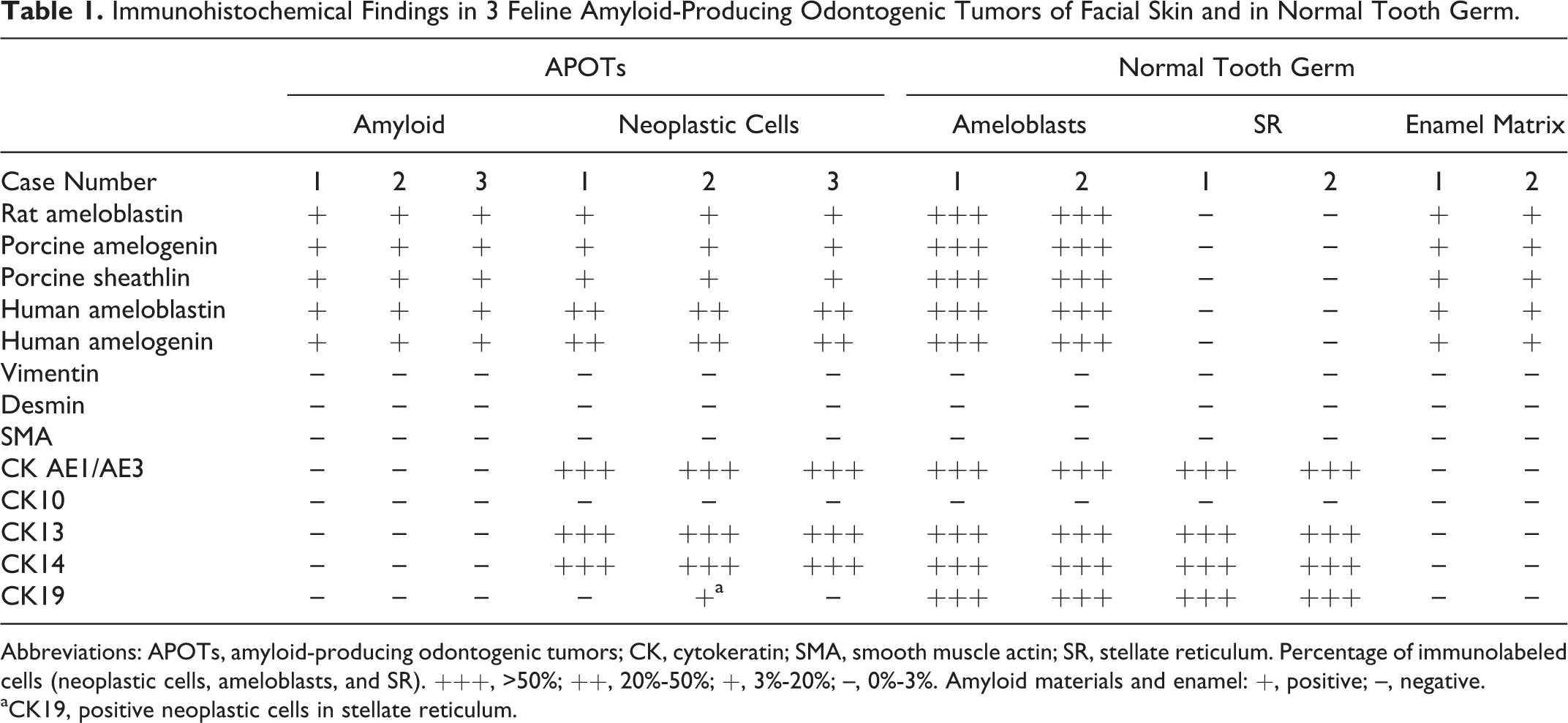
Abbreviations: APOTs, amyloid-producing odontogenic tumors; CK, cytokeratin; SMA, smooth muscle actin; SR, stellate reticulum. Percentage of immunolabeled cells (neoplastic cells, ameloblasts, and SR). +++, >50%; ++, 20%-50%; +, 3%-20%; –, 0%-3%. Amyloid materials and enamel: +, positive; –, negative.
aCK19, positive neoplastic cells in stellate reticulum.
APOTs of dogs and cats has been described as multilocular and composed of odontogenic epithelial cells arranged in islands, strands and sheets, separated by variably thickened fibrous connective tissue. In islands and strands of epithelial cells, the peripheral cells are arranged in a palisading fashion, while the central cells are polyhedral with a stellate, sheet-like or rarely random arrangement. Eosinophilic spherules surrounded by neoplastic cells are congophilic both with and without 5% potassium permanganate pretreatment, and have apple-green birefringence under polarized light microscopy. 1,2,5,9 The dermal neoplasms in the current cats had similar histopathological findings to these previously described dog and cat APOTs. The neoplasms were clearly demarcated from the dermis by thick fibrous-vascular connective tissue, and did not invade the peripheral soft tissue. The current neoplasms were diagnosed as dermal APOTs of the facial skin by histopathological examination.
We and others have previously demonstrated, by biochemical and immunohistochemical examinations, that the amyloid deposits in canine APOT are derived from the enamel proteins secreted by ameloblastic epithelium. 5 In the present study, ameloblastin, amelogenin and sheathlin were detected by immunohistochemistry in the amyloid materials of 3 dermal APOTs. These enamel proteins are produced by ameloblasts at different stages of development and are considered to play various roles in the modulation of mineral deposition and crystal growth during tooth morphogenesis. 10 Thus, amyloid materials in the dermal APOTs are derived from proteins with similar amino acid sequence as ameloblastin, sheathlin and amelogenin, like in the gingival APOTs of dog and cat. On the other hand, laminin, vimentin, and odontogenic ameloblast-associated protein (ODAM) have been examined using antibodies specific to these proteins, but positive immunostaining was restricted to a few cats. 1,2,9 Positive labeling of CKs, nerve growth factor receptor, and collagen type I, III and IV have not been detected in the amyloid deposits. 2,9 Therefore, these proteins are not considered to be precursors of amyloid materials, at least in feline APOTs. 1,2,9
Immunohistochemical characterization of the neoplastic epithelial cells of feline APOTs has been performed in a few studies. 1,2,9 Positive signals of pan-CK, AE1/AE3, CK14, and CK19 were always detected in the cytoplasm with variable intensity. 1,2,9 However, vimentin, laminin, GFAP, S100, calretinin, and ODAM were not detected in all feline APOTs. 1,2,9 Also, positive signals of nerve growth factor receptor were found in the neoplastic cells of 2 out of 3 cats. 2 CKs, vimentin, SMA, desmin, and GFAP are cytoplasmic cytoskeletal proteins of intermediate filaments. Neoplastic cells of the dermal APOTs reacted with anti-CK13, CK14 and CK19 (localized in stellate reticulum cells), and did not react with anti-CK10 antibody. By comparison, ameloblasts of normal tooth germ strongly reacted with anti-CK13, CK14 and CK19 antibodies, but did not react with anti-CK10 antibody. 3,8 These results suggest that neoplastic cells of APOTs have some characteristics of ameloblastic epithelial cells of normal tooth germ, but that these cells may have the ability to differentiate into various epithelial cells involved in the development of teeth. 3,8
APOTs are considered to be neoplasms derived from residual odontogenic epithelial cells found near the tooth (central) or in the gingival epithelium (peripheral). 4 APOTs reported in cats and dogs has been restricted to the oral cavity and, to the best of our knowledge, there have been no reports of APOTs in the facial skin of any animals. APOTs of the supraorbital skin and labial skin in the present report had histopathological and immunohistochemical characteristics similar to those in the gingiva of dogs and cats. These results suggest that the residual odontogenic epithelial cells exist in the facial skin outside of the oral cavity.
Footnotes
Acknowledgements
We thank Dr Takashi Uchida of the Department of Oral Anatomy, Hiroshima University, for providing the ameloblastin, sheathlin, and amelogenin antibodies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
