Abstract
Significant interobserver variability in the diagnostic interpretation of endoscopic gastrointestinal (GI) specimens exists even with the use of World Small Animal Veterinary Association (WSAVA) standardization criteria. Chi-square analyses compared the extent of pathologists’ agreement for microarchitectural features of inflammation in endoscopic specimens obtained from 253 animals of the original WSAVA study. Patterns of agreement between pathologists were classified as broad (3/4 pathologists agreed), dichotomous (2/4 pathologists agreed), or divergent (no agreement between pathologists). The simplified model for GI inflammation was based on those parameters for which the pathologists had either broad or minimally divergent opinions of histopathologic significance. In this model, the parameters chosen were as follows: gastric parameters (intraepithelial lymphocytes [IELs], lamina propria [LP] infiltrates, and mucosal fibrosis), duodenal parameters (villus atrophy, epithelial injury, IELs, crypt changes, and LP infiltrates), and colonic parameters (epithelial injury, crypt dilation, fibrosis, LP infiltrates, and goblet cell depletion). Preliminary data using this simplified model showed excellent correlation between pathologists in defining the presence and extent of GI inflammation in dogs.
Keywords
The diagnosis and treatment of chronic enteropathy in dogs and cats often requires histopathologic assessment of mucosal biopsy specimens collected from the gastrointestinal (GI) tract. While endoscopic procedures are commonly performed, endoscopic biopsy interpretation is notoriously subjective and suffers from the technical constraints of small specimen size and procurement/processing artifacts that confound accurate diagnostic evaluation. 1,6 Complicating these potential limitations is the fact that histopathologic criteria for the diagnosis of GI tract inflammation in dogs and cats are inconsistent. The World Small Animal Veterinary Association (WSAVA) GI Standardization Group recently developed a standardized histology monograph that pictorially and textually defined inflammatory and morphologic features in endoscopic biopsy specimens obtained from the stomach, duodenum, and colon. 3 However, succinct well-defined parameters for defining GI inflammation based on the extent of interpathologist agreement (when using this template) were not defined. The aim of the present study was to design a simplified histopathologic model that might improve the consistency of interpreting GI inflammation between pathologists.
Materials and Methods
Retrospective Analysis of the Original WSAVA Histopathology Data Set
Comprehensive statistical analysis of the histopathologic findings (n = 253 animals that were a mixture of dogs and cats) from the original WSAVA data set was performed. In brief, 1 member of the group (A.E.J.) reviewed and collated the summary histopathologic findings rendered by the 4 individual pathologists who participated in that study (M.J.D., J.M., B.W., and T.B.). Chi-square analyses were performed and compared the extent of agreement between the 4 study pathologists for each inflammatory (eg, intraepithelial lymphocytes [IELs], lamina propria [LP] infiltrate, etc) or morphologic (eg, villous atrophy, duodenal crypt changes, etc) feature. The resultant P values were then used to impart relative significance as to the extent of interobserver agreement between the 4 pathologists for a single inflammatory or morphologic abnormality. The 5 histopathologic features that showed the least interobserver variability (based on chi-square analyses) in defining GI inflammation involving the stomach, small intestine, and colon comprised the simplified model.
Prospective Trial Using the Simplified Histopathologic Model for GI Inflammation
The simplified histopathologic model for GI inflammation was next tested in a prospective trial involving diagnostic evaluation of duodenal biopsy specimens obtained endoscopically from 20 dogs (15 dogs diagnosed with idiopathic inflammatory bowel disease [IBD] and 5 healthy dogs free of GI signs) over a 3-month period. The diagnosis of IBD was based on established clinical criteria and included confirmation of histopathologic inflammation involving the intestines. 1,6 Two experienced, board-certified veterinary pathologists (M.A. and J.H.) independently reviewed and scored hematoxylin and eosin (HE)–stained endoscopic small intestinal biopsy specimens from all dogs using the simplified histopathologic criteria and in accordance with the WSAVA guidelines. The presence and severity of each histopathologic change was numerically scored 0 to 3, where 0 = absent, 1 = mild, 2 = moderate, and 3 = marked. Only diagnostically “adequate” tissue specimens were included in this study. Microscopic scores obtained by each pathologist were statistically analyzed for their extent of interobserver agreement for all 5 small intestinal parameters.
Statistical Analysis of Prospective Trial
Weighted κ was calculated to evaluate agreement between the 2 evaluators for each duodenal parameter. Test of symmetry was performed to test for significant difference between the scores of the 2 evaluators. Interpretation of κ values was as follows: values <0.2 indicated slight agreement, values between 0.2 and 0.4 indicated fair agreement, values between 0.4 and 0.6 indicated moderate agreement, values between 0.6 and 0.8 indicated substantial agreement, and values >0.8 indicated almost perfect agreement. 2
Results
Both the number of interpretable tissues per slide (mean, 9.9; range, 9–11) and specimen quality (86% of total tissues ranked as “adequate”) of WSAVA samples were suitable for diagnostic evaluation. Chi-square analyses indicated distinct patterns of agreement between the 4 WSAVA pathologists that were classified as broad (3/4 pathologists agreed on the histopathologic abnormality), dichotomous (2/4 pathologists agreed on the histopathologic abnormality), or divergent (the agreement for the presence of a histopathologic abnormality varied widely between all 4 pathologists; Table 1). The simplified model for defining GI inflammation was based on using only those parameters in which WSAVA pathologists had broad agreement for or minimally divergent opinions of histopathologic significance. Those WSAVA parameters for which only a single study pathologist interpreted the tissues as abnormal were excluded from this new model. The resultant simplified model comprises 5 easily assessed inflammatory and morphologic changes for use in defining inflammation involving the gastric, small intestinal, and colonic mucosa in dogs and cats (Figs. 1–5). These criteria included the following: gastric parameters (IELs, LP infiltrates, and mucosal fibrosis, which included atrophy and glandular “nesting”), duodenal parameters (villus atrophy, epithelial injury, IELs, crypt changes, and LP infiltrates), and colonic parameters (epithelial injury, crypt dilation, fibrosis, LP infiltrates, and goblet cell depletion) (Table 2).
Different Patterns of Interobserver Agreement Between World Small Animal Veterinary Association Study Pathologists.
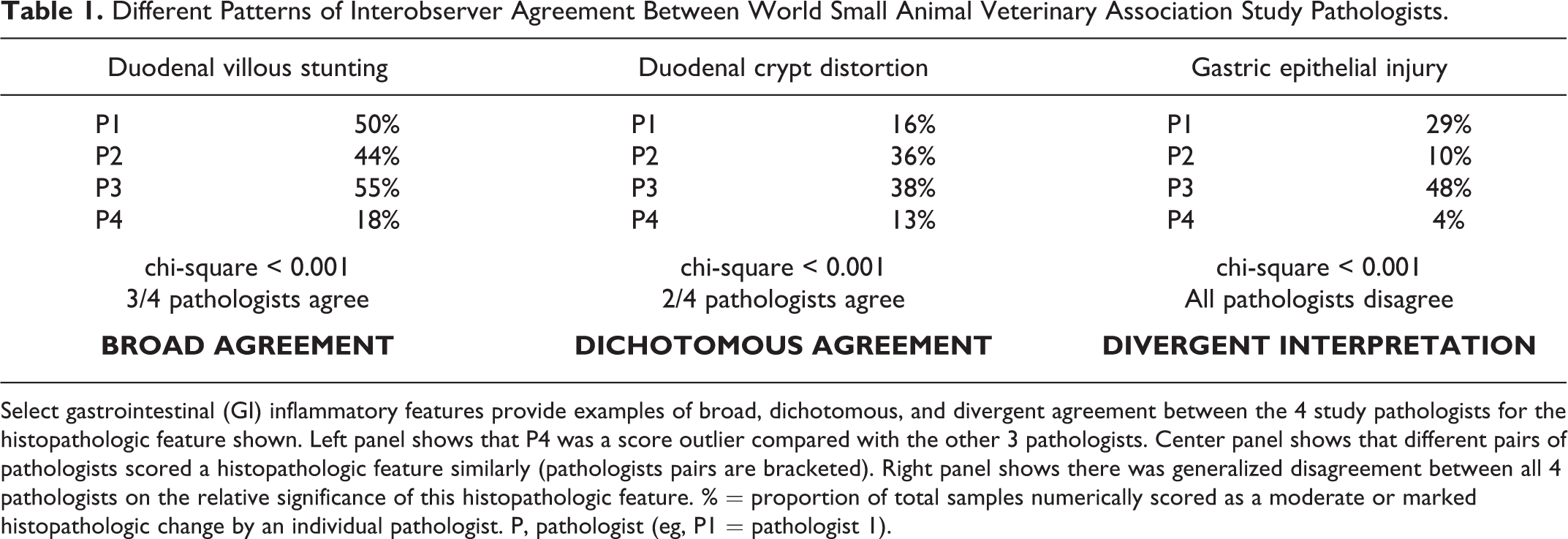
Select gastrointestinal (GI) inflammatory features provide examples of broad, dichotomous, and divergent agreement between the 4 study pathologists for the histopathologic feature shown. Left panel shows that P4 was a score outlier compared with the other 3 pathologists. Center panel shows that different pairs of pathologists scored a histopathologic feature similarly (pathologists pairs are bracketed). Right panel shows there was generalized disagreement between all 4 pathologists on the relative significance of this histopathologic feature. % = proportion of total samples numerically scored as a moderate or marked histopathologic change by an individual pathologist. P, pathologist (eg, P1 = pathologist 1).
Inflammatory and Morphologic Features of the Simplified Gastrointestinal Model Used in Defining Inflammation Involving the Gastric, Small Intestinal, and Colonic Mucosa.
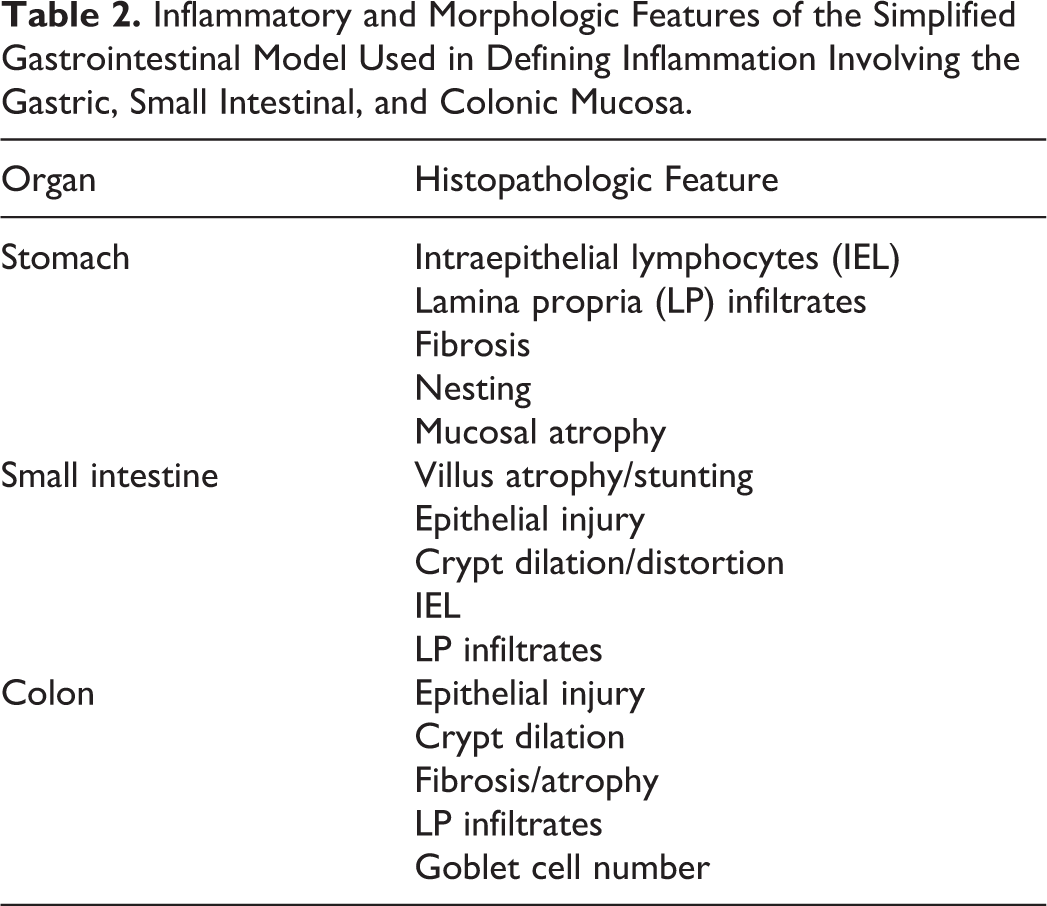
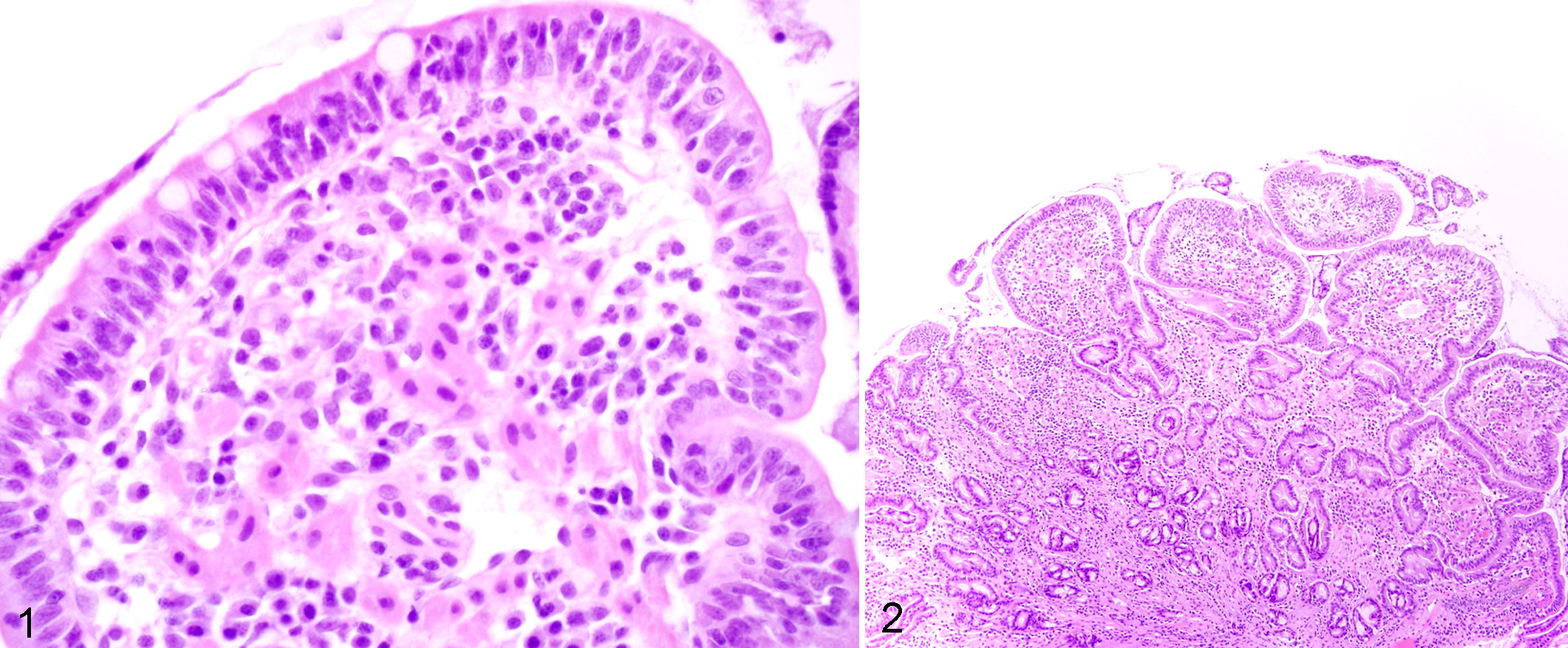
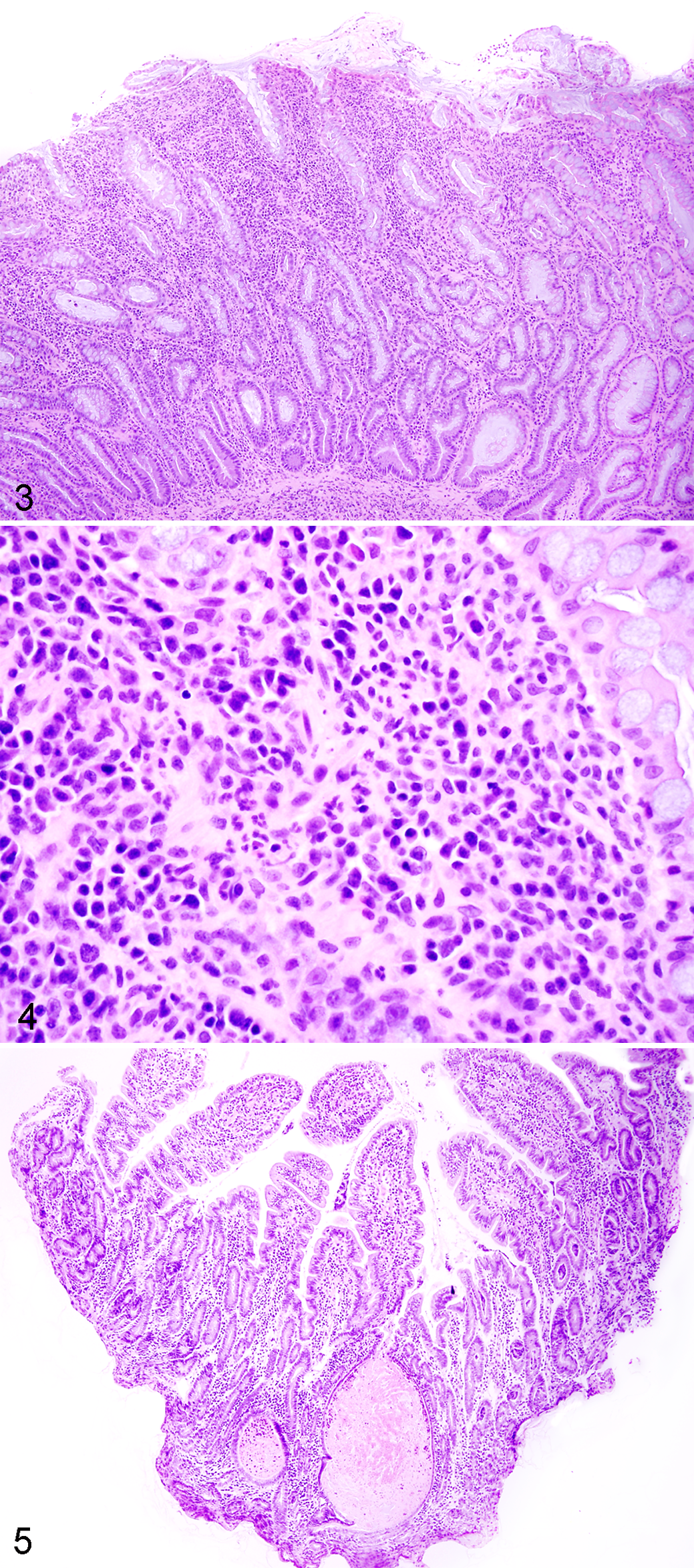
The IBD dogs evaluated in the prospective trial were predominantly middle-aged, exhibited chronic GI signs, and had moderate to severe disease activity (median canine IBD activity index [CIBDAI] 5 score = 7.8) and histopathologic evidence of intestinal inflammation (Table 3). All duodenal tissues evaluated included at least 3 contiguous villi and contained subvillus lamina propria that extended down to the mucosa-muscularis mucosa border. Preliminary data using the simplified model showed excellent agreement between pathologists in defining the presence and extent of duodenal inflammation (Table 4). Using these refined criteria, the pathologists agreed on all 5 histopathologic features of small intestinal inflammation in dogs with IBD (Fig. 1). The presence of crypt changes showed the most interobserver variability but still indicated substantial agreement (κ = 0.78) between the 2 trial pathologists.
Baseline Patient Characteristics in Dogs With Inflammatory Bowel Disease of the Pilot Study.
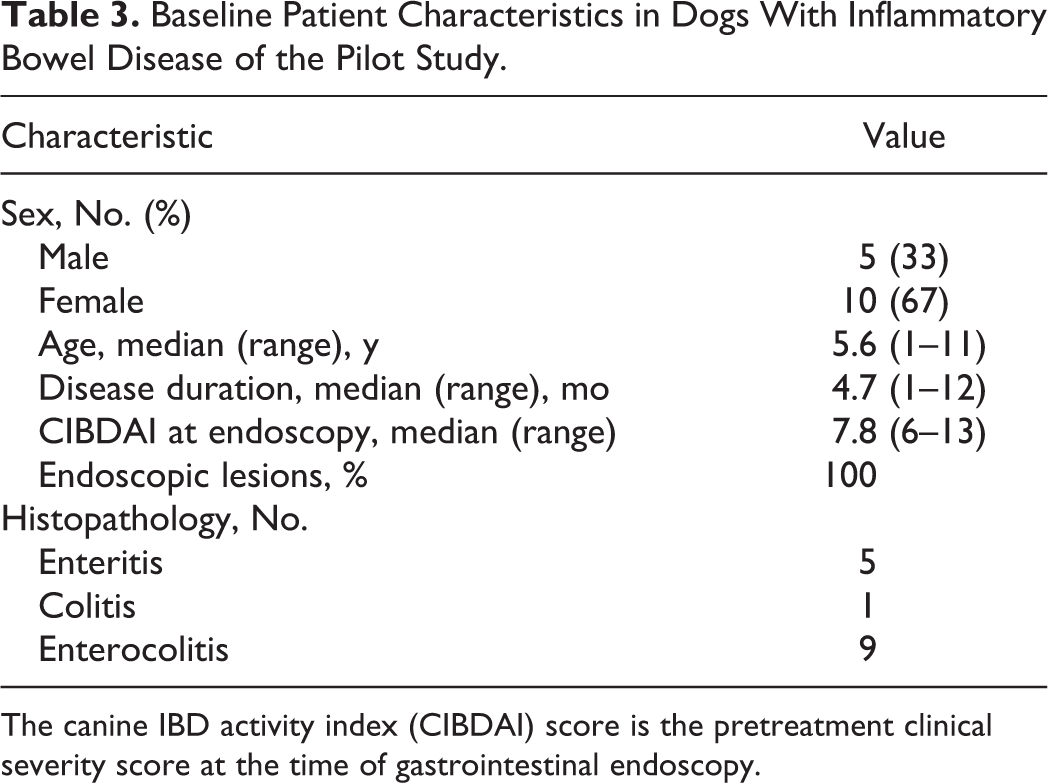
The canine IBD activity index (CIBDAI) score is the pretreatment clinical severity score at the time of gastrointestinal endoscopy.
Preliminary Data Showing Interpathologist Agreement for Diagnosis of Duodenal Inflammation in Dogs Using the Simplified Gastrointestinal Model.
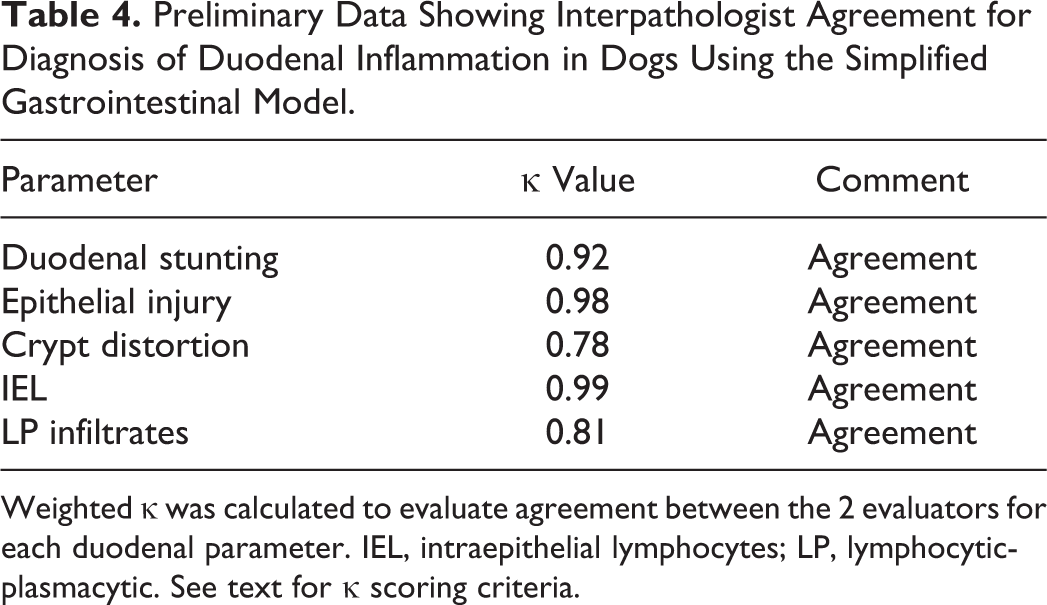
Weighted κ was calculated to evaluate agreement between the 2 evaluators for each duodenal parameter. IEL, intraepithelial lymphocytes; LP, lymphocytic-plasmacytic. See text for κ scoring criteria.
Discussion
Defining the presence, distribution, and severity of GI disease in endoscopic biopsy specimens from dogs and cats is difficult. Prior studies have failed to detect a convincing association of mucosal histopathology with clinical signs, biomarkers of inflammation, or response to therapy and outcome in dogs with IBD. 10 One study in cats showed that duodenal inflammation was linked to microbial imbalances and clinical disease activity in cats with IBD. 4 Even with standardized grading schemes, the interpretation of GI histopathologic findings may vary widely between pathologists. 9
The present report represents an extension of the original WSAVA template whereby a refined and reductionist (simplified) model is designed for histopathologic evaluation of GI tissues. We used statistical analysis with inferences to identify those WSAVA criteria that showed the least interpretive variation between pathologists in the original report. These excluded parameters included the following: stomach (epithelial injury, epithelial hyperplasia, lymphofollicular hyperplasia), duodenum (lacteal dilatation, mucosal fibrosis), and colon (crypt hyperplasia). The resultant simplified GI model is based predominantly on mucosal architectural and epithelial changes that define mucosal inflammation in the stomach, duodenum, and colon. Changes in goblet cells are included as a new colonic parameter since goblet cell depletion is seen with different causes of colitis, and goblet cell numbers may increase with successful treatment. 11,12 Importantly, our prospective trial demonstrated excellent agreement between the 2 trial pathologists for all 5 features of duodenal inflammation in IBD dogs using this GI model. Even the severity of lamina propria cellularity showed substantial agreement (κ = 0.81) between observers, whereas other reports using different grading schemes have questioned the clinical relevance of this inflammatory feature in canine and feline enteropathy. 7,8
One potential limitation of the present study is the failure to include histopathologic evaluation of ileal biopsy specimens. Until recently, the clinical utility of ileal biopsy has been questioned since ileoscopy is arguably a more technically demanding and time-consuming endoscopic procedure for many clinicians. However, new studies now confirm the need to collect both duodenal and ileal biopsy specimens in dogs and cats having either small or large intestinal diarrhea caused by intestinal inflammation (eg, IBD) or cancer (eg, lymphosarcoma). 2,13 –15 The performance of the original WSAVA standardization trial preceded these new endoscopic recommendations and did not include histopathologic review of ileal mucosal specimens. Our current recommendation is to perform ileoscopy, when possible, and to use the simplified model criteria of duodenal inflammation for microscopic evaluation of ileal biopsy specimens.
Last, the present report evaluated only the diagnostic utility of the simplified model in histopathologic interpretation of duodenal inflammation in dogs. While application of these grading criteria to feline endoscopic specimens needs to be performed, pilot data from our laboratory suggest that this system will be of value in the diagnosis of GI inflammation in this species as well.
Conclusion
In summary, a simplified histopathologic model for defining GI inflammation in dogs and cats has been designed from additional analysis of the original WSAVA GI histopathologic templates. Preliminary data indicate that the simplified model reduced interobserver variability in the diagnostic interpretation of GI inflammation between pathologists. Further testing and validation of this improved model system in a large, multicenter clinical trial using a new team of pathologists is presently under way.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
