Abstract
Mammary neoplasms are the most common neoplasm in female dogs. This article describes the embryologic development, normal anatomy, and histology of the canine mammary gland from the onset of first estrous and the changes that occur in the mammary gland during the estrus cycle. The clinical features of canine mammary gland tumors and their relation to prognosis are discussed, including age, hormones, breed, diet, and obesity. Additional clinical prognostic factors including clinical presentation, tumor size, and lymph node status at the time of presentation are discussed in relation to diagnosis and tumor staging. Immunohistochemical evaluation of the cell differentiation markers of the normal and neoplastic canine mammary gland is described and compared with similar studies in humans; the ways these markers may be used to assist with the prognosis of canine mammary neoplasms are discussed.
The mammary gland is a modified apocrine sweat gland found only in mammals. It consists of a network of ducts surrounded by a fibrovascular and adipocyte-rich stroma. The development of this gland is unique, as the last stages of development occur in the adult female only during pregnancy. With each pregnancy there is proliferation of the ductal tissue, differentiation to milk-producing acini, secretion of milk by the acinar cells, and, at the end of lactation, involution of the secretory component of the gland with preservation of the ductal structures.
Development of the Mammary Gland
Mammary development can first be recognized during embryologic development by the appearance of 2 ventral linear thickenings (ridges) of ectoderm, below which are specialized regions of mesoderm. The ridges, also referred to as milk lines, extend from the axillary to the inguinal region. The ectodermal cells migrate along each milk line and coalesce to form a placode, which eventually becomes individual mammary glands. The formation of the placode is a complex interaction, involving several signal pathways between the epithelial cells of the ectoderm and mesenchymal cells of the mesoderm.
The epithelial cells of the placode form a solid cord of cells that grow into the underlying mesenchyme to form the mammary buds, which subsequently branch to form a mammary sprout. Within each sprout a lumen forms via a process of cavitation, which communicates externally via a region of specialized epithelium called the nipple sheath and which becomes the raised teat in the adult dog. Each mammary sprout will eventually form the papillary duct of the adult mammary gland.
Most dogs develop 5 pairs of mammary glands, although 4 or 6 pairs have been found in a few animals. There are 2 thoracic (M1 and M2), 2 abdominal (M3 and M4), and 1 inguinal (M5) pair of mammary glands. 86 Each teat has between 7 and 16 duct openings, and each of these ducts will eventually form a lobe of the adult gland and act as an independent functional unit within the gland. The mammary glands continue to grow in proportion to the rest of the body, and until the time of puberty the ducts extend only a short distance from the teat.
At puberty, mammary development is initiated by the release of estrogens from the ovary. At the terminal ends of the ducts, cell proliferation occurs with the formation of terminal end buds. With the onset of pregnancy and a rise in progesterone levels, growth occurs by lengthening and tertiary branching of the ductal system of the lobes. The ducts give rise to multiple lobules from which will develop the alveoli, the secretory units of the mammary gland (lobuloalveolar unit). Under the influence of prolactin, the presecretory alveolar cell changes to a secretory alveolar cell. Thus, at parturition, the mammary gland consists of a ductular–lobular–alveolar structure, whereby the products of the tubuloalveolar unit are conveyed via ductules to ducts to the teat sinuses for removal by the neonate.
Ten days post partum there is the onset of alveolar regression, which is completed by 40 days post partum, and at this stage only the ducts of the preexisting complex tubuloalveolar unit can be found. With pseudopregnancy the same changes occur within the mammary gland except there is less alveolar secretory development. 86
Microscopic Anatomy of the Mammary Gland
The epidermis covers the teat but there is a lack of most of the adnexal structure within the dermis. The epidermis can have modest rete ridge formation. Melanocytes are present between the basal epidermal cells and often have a vacuolated cytoplasm. Opening onto the teat surface are the teat ducts that are lined by a stratified squamous epithelium and often have a plug of keratin within their lumina. Surrounding the opening of the teat duct is a smooth muscle sphincter. The teat duct opens into the teat sinus that is lined by a bilayered columnar epithelium, external to which are fusiform myoepithelial cells. Emptying into the teat sinus are the ducts; the larger ducts are lined by a bilayered cuboidal epithelium and surrounded by fusiform myoepithelial cells, whereas the smaller ducts have a single layer of cuboidal epithelium and fusiform myoepithelial cells. The luminal epithelial cells and basal/myoepithelial cells are in a constant state of flux with new ductal growth (elongation and branching) emanating from medially located epithelial cells that penetrate through gaps between the myoepithelial cells.
The nonsecretory alveoli are similar to the small ducts. The secretory alveolar cells vary from cuboidal to columnar and have variable numbers of intracellular fat droplets that accumulate in the alveolar lumina. Surrounding the alveolus in a basket-like fashion are star-shaped myoepithelial cells.
The mammary gland epithelium is surrounded by a continuous basement membrane, principally composed of type IV collagen, laminin, nidogen, and heparin sulfate proteoglycan. Normal luminal epithelial and myoepithelial cells deposit the basement membrane, particularly the α3 and α5 chains of laminin 1, but only myoepithelial cells deposit α1 laminin chains.
The epithelial component of the mammary gland is supported by mesenchymal tissue that has developed from the embryonic mesoderm. This includes fibrous connective tissue, adipose tissue, blood vessels, nerves, and lymphatics. Occasional histiocytes, mast cells, and lymphoid cells also occur within the stromal tissues. The fibrous connective tissue may be subdivided into 2 components: the intralobular component that surrounds the intralobular ducts consists of finer collagen fibers surrounded by a more extensive extracellular matrix, whereas the interlobular component that separates the lobules has larger collagen fibers with less extracellular matrix. The amount of mammary and adipose tissue present is very variable and is more abundant in the abdominal and inguinal glands than in the thoracic glands.
Rehm et al 73 and Santos et al 78 describe the histology of the canine mammary gland during the estrous cycle, which is helpful when evaluating pathologic changes that occur in these glands. In prepubertal dogs in the proestrus phase of their ovarian cycle, only large ducts (interlobular ducts) lined by a double layer of epithelial cells and a single layer of myoepithelial cells are found within a dense interlobular connective tissue stroma. Projecting laterally from the wall of these ducts are bulbous epithelial structures (end buds), consisting of tightly packed cells with large euchromatic nuclei, 1–2 prominent nucleoli, and indistinct cell borders.
In adult dogs in the proestrous stage, the gland is inactive and consists primarily of interlobular ducts and occasional small lobules surrounded by extensive amounts of interlobular and intralobular connective tissue. Occasional alveolar structures from the previous estrus cycle, lined by cells with flattened or cuboidal vacuolated epithelial cells, may still be present in the small lobules. Macrophages that contain lipofuscin are found within the lumina of these alveoli and also within the loose intralobular connective tissue.
The onset of estrus results in the proliferation of the intralobular ductal epithelium with the formation of multiple small ductules lined by a multilayered epithelium within the loose connective tissue of the lobules. The cells of the multilayered epithelium are similar to the cells of the end buds (see above).
In early diestrus, when progesterone levels are high, there is greater development of the ducts with the formation of lobules. The epithelium lining these ducts is multilayered, and the cells are loosely packed with round to elongated euchromatic nuclei, 1–2 prominent nucleoli, and numerous mitotic figures. The fibroblasts within the interlobular stroma have more prominent nuclei and may show increased mitotic activity. They are present within a mucinous matrix that contains numerous small congested blood vessels.
In late diestrus, the development of the canine mammary gland has reached its peak with the formation of secretory alveoli at the terminal end of the intralobular ducts. The alveoli are filled with a brightly eosinophilic proteinaceous secretion and lined by cells that vary from cuboidal to a more attenuated epithelium. Surrounding the alveolar epithelium, the myoepithelial cells are elongated and form a continuous layer around the less developed alveoli and a discontinuous layer around the fully developed and distended alveoli. The latter finding may be attributable to the stellate morphology of these alveolar myoepithelial cells. The intralobular ducts also have a brightly eosinophilic secretion within their lumina and are lined by a single layer of low cuboidal to flattened epithelium and a continuous layer of fusiform myoepithelium. There is a minimal amount of intralobular stroma around the alveoli, but bands of collagen and reticulin fibers form the more extensive interlobular stroma that surrounds the secretion-filled interlobular ducts.
Early anestrous is characterized by alveoli that contain less secretion. The alveoli are lined by vacuolated epithelial cells supported by a more prominent basement membrane. Some lobules show changes associated with regression of the mammary gland, with increased amounts of intralobular connective tissue and an infiltrate by lymphocytes and plasma cells.
In late anestrus, the changes associated with regression of the mammary gland become more prominent. Duct lumina are decreased in diameter and lobules decrease in size. Cells with pyknotic nuclei often line ducts/alveoli. Eosinophilic concretions (corpora amylacea) are seen within ductal lumina. The interstitium is more abundant, the collagen fibers appear more compact, and there may be an infiltrate by lymphocytes, plasma cells, and lipofuscin-containing macrophages. The interlobular stroma appears more abundant and compact.
The neural innervation of the canine mammary gland is mainly associated with the vasculature and consists of peptidergic nerves, which may be involved in the regulation of local blood flow. The presence of sensory neuropeptides in nerves supplying the mammary nipple suggests that these peptides may also play a role in the afferent pathway of the milk ejection reflex. 67
Lymph Drainage of the Mammary Gland
Patsikas et al 59 –61 investigated the lymph drainage of the normal and neoplastic mammary glands in the bitch; these results are presented in Table 1 . In normal healthy dogs, lymphatics drain into the ipsilateral lymph nodes; there is no drainage to the contralateral gland or lymph node, but drainage may be altered in cases of mammary neoplasia. 62,63
Lymphatic Drainage of the Normal and Neoplastic Mammary Gland in the Bitch

LN, lymph node.
By convention most malignant epithelial neoplasms (carcinomas) metastasize via lymphatic whereas malignant mesenchymal neoplasms (sarcomas) metastasize via capillaries and veins. The findings of Patsikas 61 are helpful in determining which nodes should be evaluated for neoplastic spread when a mammary carcinoma is suspected clinically. Those investigators' findings also reinforce the impression of clinicians that mammary carcinomas that occur in the inguinal (M5) gland may show retrograde metastasis via the lymphatic plexus in the subcutis of the inner thigh and to the popliteal lymph nodes. Occasional cases of retrograde metastatic spread by mammary carcinomas to the vagina have also been reported in bitches. 76
Clinical Features of Canine Mammary Gland Tumors and Prognosis
The classical presentation of a dog with mammary tumors is an older female dog of a wide range of breeds that is sexually intact or spayed later in life with 1 or more palpable tumors in the mammary chain(s). In addition to describing a typical patient with mammary tumors, the preceding sentence captures the 2 most important known risk factors for mammary tumor development in dogs: age and hormonal exposure. Several additional factors, including breed, diet, and obesity, have been associated with an increased risk for mammary tumor development.
Age
This is one of the most important risk factors for developing a mammary tumors in dogs; a comprehensive longitudinal study of a large Beagle colony with naturally occurring mammary tumors found that the risk starts to become significant around age 8 and increases linearly with age thereafter. 92 Similarly, a progressive increase in mammary tumor incidence with advancing age was documented in the Alameda-Contra Costa Counties Animal Neoplasm Registry, which included privately owned dogs of various breeds, with a peak incidence of mammary tumors between 9 and 11 years in this population. 22 The age of onset, however, may vary depending on the natural life span of the breed of dog, and most authors report an average age range of 8–11 years, indicating that this is a malignancy of the middle-aged or older dog. 10,38,41,69,83,85 Younger dogs are more likely to have benign tumors than older dogs, 35,51,54,92 and a recent study reported a significant difference in age: a mean age of 8.5 years in dogs with benign tumors versus 9.5 years in dogs with malignant tumors. 88 Mammary tumors are rare in dogs under 5 years of age, unless the dogs have been treated with exogenous hormones (progestins). 83,92
Hormones
Exposure to endogenous ovarian hormones early in life is a more important source and cause of mammary tumor development in dogs. The landmark publication by Schneider et al 82 in 1969 reported that a dog’s risk for developing mammary tumor increases dramatically during the first few estrus cycles, as illustrated by the fact that a dog that is ovariohysterectomized prior to the first estrus has only a 0.5% risk of developing mammary tumors, compared with 8% and 26% in dogs ovariohysterectomized between the first and the second estrus or after the second estrus, respectively. 82 This study did not find any significant protective effects from performing ovariohysterectomy (OHE) after the second estrus. However, subsequent studies have shown that later OHE may provide some protection. The results from the large Beagle colony study discussed above concluded that performing OHE after the age of 4 did not confer any protection against mammary tumors. 92 This is similar to the finding by Sonnenschein et al 87 that documented significant protection associated with OHE prior to the fourth estrus cycle and showed that OHE between the 4th and 6th estrus cycle was protective but not statistically significant. A study from the Netherlands provided evidence that there might be some protection associated with even later OHE. 51 Thus, the protective benefit from ovarian hormonal ablation appears to be highly dose dependent; the effect is clearly strongest in patients with no to minimal exposure and diminishes relatively quickly after the first few years, suggesting that the most crucial events that predispose the mammary glands to tumor development occur early in a dog’s life. Other reproductive variables such as estrus irregularity, pseudo-pregnancy, and pregnancy that may result in physiological changes in endogenous hormone levels have not been found to significantly influence mammary tumor risk in dogs. 9,82,92 Thus, it is exposure to ovarian hormones during the first 2 estrus cycles that represents the greatest risk; additional exposure, or lack thereof, later in life appears to be significantly less important. Studies that involve a large cohort of dogs will be required to detect such additional effects. Exogenous exposure to pharmacological doses of hormones, both progestins and estrogens, has, however, been associated with an increased risk for mammary tumors. These findings are based on a significant body of evidence from research in colony dogs treated long term with various doses and combinations of synthetic progestins and estrogens and monitored for tumor development. 17,32,34,43,84 Most of these studies found earlier and more frequent tumor development and significantly more benign tumors than malignant tumors in progestin-treated dogs, with an increased risk of malignancies in dogs treated with a combination of progestins and estrogens. 17,32,34,43,84 Administration of progestins in privately owned animals in order to delay or prevent estrus also increases the risk of tumor development. A Dutch study on privately owned dogs found that dogs with mammary tumors were more likely to have received progestins than age-matched controls, 51 and data from the Norwegian canine cancer tumor registry reported that progestin-treated dogs had a significantly increased risk, with an odds ratio 2.3 when compared with nonexposed dogs. 90
Breeds
Mammary tumors can occur in any female dog of any breed, but certain types of dogs and breeds, such as miniature and toy breeds, are consistently overrepresented in epidemiological studies. Some of the larger purebred dogs are also at increased risk for mammary tumors; these reports vary somewhat depending on where these studies originate, but the major breeds most consistently listed include English Springer Spaniel, Brittany, Cocker Spaniel, English Setter, Pointer, Afghan Hound, and German Shepherd Dog, in addition to the typical smaller breeds such as Miniature and Toy Poodle, Maltese, Chihuahua, Beagle, Dachshund, West Highland White Terrier, Yorkshire Terrier, and Bichon Frise. 11,16,23,35,52,53,69,75,96 These studies show that the risk for mammary tumors has some breed association and, therefore, likely a genetic component. The genetic component is further underscored by the fact that some studies have shown that certain lines or families within specific breeds are more at risk than others. For example, Boxers are noted to be underrepresented in the reports from the United States, 10,35,69 whereas they are noted to be at increased risk according to many of the reports from several European countries. 11,53,75 A Beagle colony study also documented a familial variation within the same breed; one line had an increased risk whereas the other had a decreased risk. 81
Diet and Obesity
A large case-control study investigating the association between diet, body conformation, and the risk for developing mammary tumors found a significantly decreased risk of developing mammary tumors in dogs that were thin at 9–12 months of age compared with the control dogs. 87 However, dogs in this study that were obese 1 year prior to mammary tumor diagnosis or were fed a high-fat diet were not found to have an increased risk for developing mammary tumors. A recent case-control study found an association between diet and mammary cancer risk: dogs fed a diet high in red meat and dogs that were obese at 1 year of age were significantly more likely to develop mammary tumors and dysplasias when multivariate analysis was used. 64 Both of these studies show that the effect of obesity on mammary tumor risk is strongest early in a dog’s life; increased body weight closer to the time of tumor diagnosis seemed not to be significant in both these studies. These findings are consistent with the findings that it is the exposure to ovarian hormones early in a dog’s life that has the greatest effect on tumor risk: obesity is thought to mediate breast cancer risk in postmenopausal women by increasing circulating free estrogen levels as well as through increased local estrogen production by aromatases. 14,37,93,95 It is possible that obesity increases the dogs' risk for mammary tumors though similar mechanisms; therefore, the effect is strongest early in life during the time when the hormonal effects on mammary tissue are most damaging.
Clinical Presentation
Dogs with mammary tumors commonly present with more than 1 tumor. A recent prospective study on canine mammary tumors in privately owned dogs reported that close to 70% of the dogs had more than 1 tumor. 88 Multiple synchronous tumors are also commonly reported by many other authors, including several of the large observational studies conducted in Beagle colonies. 5,6,54,92 The 2 caudal pairs of glands are most commonly affected, and the axillary mammary glands are often not involved as a first occurrence. 5,6,35,54,92 Mammary tumors are relatively easy to detect on clinical exam and typically appear as 1 or more discrete masses in the glands. The specific clinical findings depend on the biological behavior of the tumor and at what stage of disease they are brought in for veterinary care: Tumors can be small or large, fixed or freely movable, ulcerated, and single or multiple. Lymph nodes may or may not be clinically enlarged, but signs of system illness due to distant metastasis are rare, especially in privately owned dogs for which veterinary care is readily available and used. One exception to this is the inflammatory carcinoma, a relatively uncommon, extremely aggressive type of mammary carcinoma. Histologically, this carcinoma is characterized by extensive invasion of the dermal lymphatics with a strong inflammatory response; clinically it may be difficult to distinguish from severe acute mastitis. 45,65 These dogs have extensive involvement of their mammary glands, edema, swelling, and pain and show systemic signs of illness, including anorexia, weight loss, and weakness. Most of these dogs are poor surgical candidates because of extensive tumor involvement and a high incidence of distant metastasis. Survival is poor despite treatment. 45,65
As noted above, the majority of dogs will have more than 1 tumor when presenting for evaluation, and additional smaller tumors may be detected when the mammary chains are palpated more carefully as part of the initial workup. These synchronous tumors are often of various sizes and of various histopathologies. Smaller tumors are significantly more likely to be benign than larger tumors, and the risk of malignancy increases with increasing tumor size. 88 A histological progression toward malignancy with increasing tumor size has been noted in many dogs with multiple synchronous tumors. 88 These findings reinforce the theory that canine mammary tumors transform over a protracted period of time from benign to malignant as a histological continuum where carcinomas are the end stage of the process. Previous researchers have also suggested such a transformation and noted that there might be an association between tumors of benign and malignant histology: Moulton et al 54 noted early on that dogs with carcinomas also often had concurrent benign tumors of the same histological type, Bender et al 5 reported that dogs with a diagnosis of a benign mammary tumors had a significant increased risk of developing malignant tumors later, Sorenmo et al 88 noted that dogs with carcinoma in situ and malignant tumors were more likely to develop new tumors in other mammary glands than dogs with benign tumors, and Gilbertson et al 33 found that dogs with malignant tumors were significantly more likely to later develop de novo carcinomas in other glands. This latter study also noted a high incidence of carcinoma in situ adjacent to invasive carcinomas. The association between intraepithelial lesions with atypia and carcinoma in situ and invasive carcinoma was reported and described in more detail in a more recent study by Antufermo et al. 2 Last, Benjamin et al 6 noted a histological progression from less aggressive carcinomas to anaplastic carcinomas in a large lifetime morbidity Beagle study. All these findings fit well into and support the proposed theory that canine mammary tumors develop as a dynamic histological continuum where malignant tumors develop from areas within benign tumors. Clinically they appear to be separate entities: they present as discrete tumors of various sizes and histologies but etiologically they are related and associated. Therefore, much can be learned about tumorigenesis by studying dogs with naturally occurring mammary tumors.
Diagnosis and Staging
A thorough history and a complete physical exam including careful palpation of the mammary glands should be performed in all dogs with mammary gland tumors. These dogs are often middle-aged to older dogs and may have concurrent medical problems that need treatment. In general, however, most dogs with mammary gland tumors are systemically healthy except for dogs with advanced-stage disease and dogs with inflammatory mammary carcinomas. These dogs are typically systemically ill and may have changes in their blood work results, including coagulopathies, consistent with systemic and metastatic disease. 33,45,89 In dogs with multiple synchronous tumors, all tumors should be excised, their location (left or right, glands 1–5) and size should be noted, and the tumors should be submitted for histopathological examination, which remains the gold standard for diagnosing and classifying mammary gland tumors.
Mammary tumors are staged according to the TNM system: tumor, lymph node, metastasis. Therefore, to stage a dog with mammary tumors, information regarding tumor size, lymph node status, and presence of metastasis needs to be collected and recorded. The greatest diameter of the tumor is used to classify the tumor according to the staging system. Stage assignment should be performed according to the largest malignant tumor in dogs with more than 1 tumor. The draining lymph nodes (see section on lymph node drainage, Table 1) should be identified, and if they are palpable and/or clinically enlarged, fine-needle aspirates for cytological evaluation should be performed. Cytological evaluation of the draining lymph nodes has been found to be highly sensitive for detecting metastatic disease in patients with solid tumors. 44 If the cytology is negative for metastatic disease, further lymph node assessment such as surgical biopsies may not be necessary for additional staging unless the node is removed as part of the tumor resection. This is often the case if the tumor involves the caudal mammary gland and the inguinal lymph nodes are included as part of the mastectomy and therefore available for histopathological evaluation. Dogs with malignant mammary tumors should also be evaluated for metastatic disease. Three-way thoracic radiographs remain the mainstay for staging the thorax/lungs for metastatic disease in veterinary medicine. The lungs are the most common site for distant metastasis in dogs with mammary tumors, 25,26 but additional staging tests, including abdominal ultrasound, skeletal radiographs, or other imaging modalities, may be indicated if clinical examination identifies other sites suspicious for metastasis.
Based on results from the above assessment, a dog will be assigned a clinical stage. Two staging systems have been proposed and published for canine mammary tumors: the original World Health Organization (WHO) staging system 57 and the modified WHO staging system. 77 Table 2 depicts both staging systems with a direct comparison of the TNM criteria involved in stage assignments. As Table 2 illustrates, there are important differences between the 2 systems: according to the modified system, stages I–III classify dogs according to increasing tumor sizes, stage IV represents dogs with lymph node metastasis, and stage V represents distant metastasis. In contrast, the original staging system assigned the same stage of disease, specifically stage II, to a dog with a small tumor and positive regional lymph node (N1+) as was assigned a dog with a larger tumor (T2: 3–5 cm) regardless of lymph node status (N0+ or N1+), suggesting that a larger tumor size has equal prognostic significance to that of a positive lymph node. Furthermore, according to the original WHO staging system, a dog with a large tumor (T3) would be assigned a stage III status, regardless of lymph node status (any N), again suggesting that tumor size is more important than lymph node status (Table 2).
Staging Systems for Canine Mammary Tumors
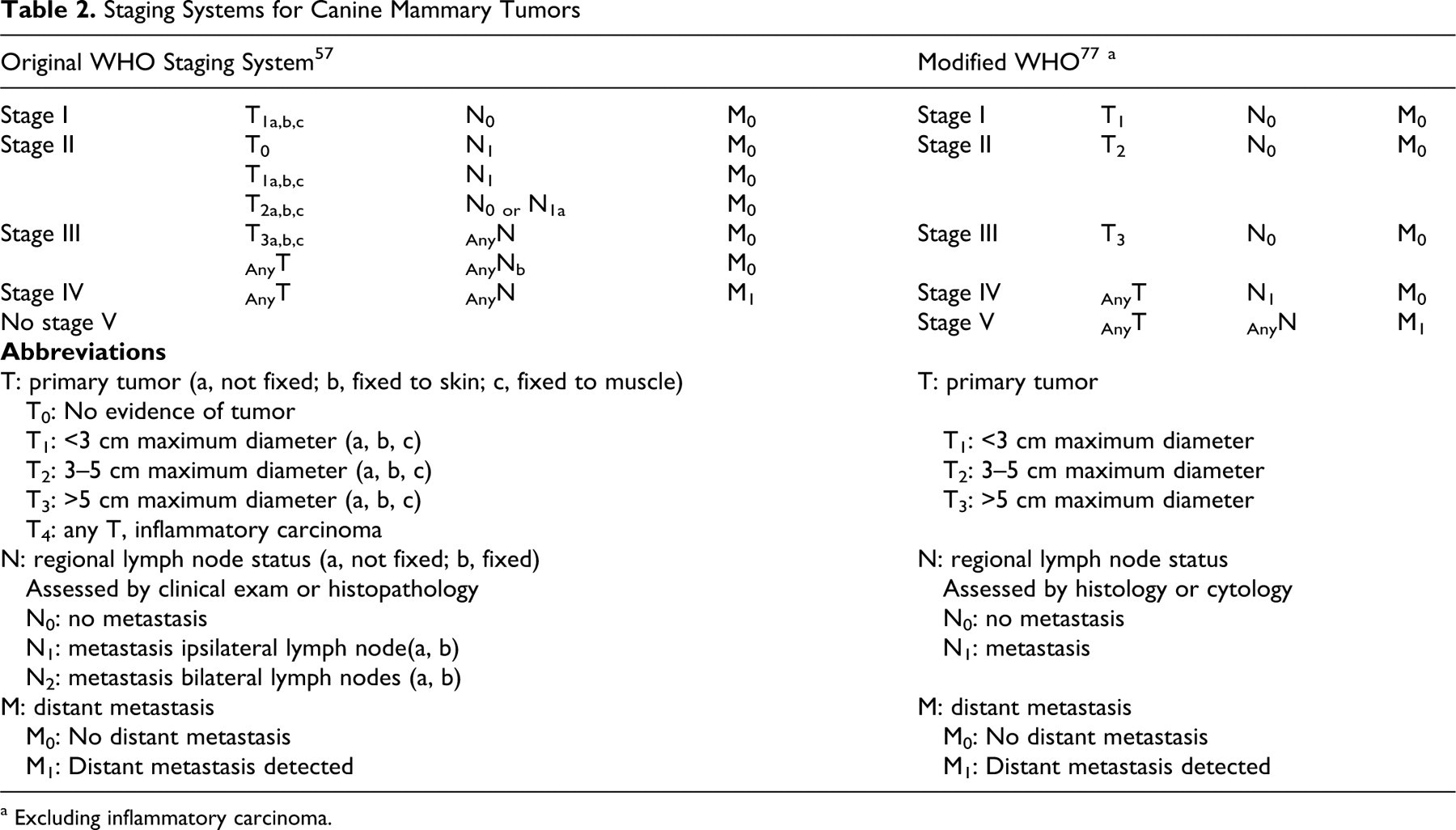
a Excluding inflammatory carcinoma.
Staging systems generally facilitate accurate and consistent medical record keeping and as such help clinicians communicate efficiently with each other regarding a patient’s stage of disease. These systems allow for comparison of patients with similar tumor burden, which may be important when evaluating the effectiveness of new treatments. Last, stage may have prognostic significance. In the most clinically useful staging systems, stage correlates directly with outcome and therefore has implications for treatment: an advanced stage confers a worse prognosis and a poor prognosis typically requires an escalation in therapy. Therefore, complete staging provides crucial prognostic information, which subsequently is implemented in the patient’s treatment plan. Neither of the above staging systems, however, has been validated for prognostic significance in controlled studies. This would require prospective studies in which all the dogs receive the same treatment and then are followed at regular intervals to determine outcome. Such studies would allow for a comparison of the prognostic significance of advancing stage according to the 2 systems and determination of the prognostic significance of tumor size versus positive lymph node status. Even though most studies on prognostic factors in canine mammary tumors do not comply with the standards recently set forth by the American College of Veterinary Pathologists Oncology Subcommittee Recommended Best Practices on the Conduction and Evaluation of Prognostic Studies in Veterinary Oncology, the results from several of the larger retrospective studies that identify tumor size, lymph node status, and WHO stage as prognostic factors are relatively consistent and biologically plausible: more aggressive tumors grow faster, may therefore be larger, and are more likely to harbor metastatic subclones, and tumors that have already metastasized to the regional lymph nodes have already proven their metastatic ability. Therefore, staging systems based on tumor size and lymph node status are likely to confer some prognostic significance and provide guidance to clinicians when making treatment decisions.
Clinical Prognostic Factors
Numerous retrospective studies have been published on prognostic factors in mammary tumors in dogs. The factors found to be most consistently associated with outcome according to the largest, best-controlled studies include tumor size, lymph node status, and clinical stage. Only these 3 factors will be addressed here.
Tumor size
Three different size categories are described in the staging systems: T1: <3 cm, T2: 3–5 cm, and T3: >5 cm. 57,77 Most of the more recent studies use these size categories when evaluating size as a prognostic factor. Even though there seems to be a general agreement that size confers prognostic significance, there is conflicting evidence at which size category the prognosis changes significantly for the worse; is it when the tumor reaches 3 cm or 5 cm in diameter? Kurtzman and Gilbertson 42 found a significant change in the risk of recurrence in dogs with mammary tumors larger than 3 cm. According to their study, dogs with T1 tumors had a better outcome than dogs with T2 and T3 tumors, and there were no significant difference between dogs with T2 and T3 tumors. 42 Philibert et al 66 also found a significant difference in survival in dogs with tumors smaller than 3 cm (T1) and dogs with tumors larger than 3 cm (T2 and T3). Yamagami et al, 96 however, found similar outcome in dogs with T1 and T2 tumors, but the prognosis was significantly worse in dogs with T3 tumors. Chang et al 12 also found that dogs with tumors larger than 5 cm had a significantly worse survival than dogs with tumors smaller than 5 cm. Some of the other larger studies on prognostic factors in canine mammary tumors did not classify tumor size according to the same size categories described above. Despite using very different size categories (<5, 5–10, 11–15, >15), Misdorp and Hart 50 reported that stage based on size alone was prognostic but only in dogs with complex carcinomas when corrected for other variables. Hellmen et al 38 and Shofer et al, 85 did not find that tumor size was prognostic but did not classify the tumor according to the size categories (I–III) in their studies. Even though there are some discrepancies regarding the importance of size as a prognostic factor and at which T-stage the prognosis changes, most studies support the usefulness of a classification system in which the stage is classified according to tumor size, given that tumor size likely has prognostic significance. It is likely that the prognosis changes gradually as the tumor size increases, but the modified system seems to capture crucial changes in prognosis based on tumor size alone. Many of the above studies, however, have been univariate analyses, which do not take into account how tumor size and lymph node status might be associated. Conducting a multivariate analysis, Chang et al 12 found that both tumor size and stage remained prognostic even though dogs with tumors larger than 5 cm were significantly more likely to have lymph node metastasis.
Lymph node status
According to the modified staging system, lymph node status is classified as N0 (no metastasis) or N1 (metastasis) based on cytology or histopathology. 77 A positive lymph node (N1) is consistent with stage IV disease in the modified system. In the original WHO system the status of the lymph node is determined based on clinical exam or histopathology, and it also specifies whether the metastatic lymph node is ipsilateral/bilateral and fixed/not fixed; this additional information influences whether the patient is assigned a stage II or III disease. 57 Lymph node status has been reported as a prognostic factor in canine mammary tumors in several publications; 49 however, the results from other studies have found that lymph node status is not prognostic. 50 This may be attributable in part to the fact that these studies were retrospective in nature: the staging and the surgical procedures were often not standardized and might have been incomplete in terms of histological lymph node evaluation, resulting in failure to detect metastasis; thus underestimating the stage. Additionally, most of these studies were not stratified for tumor type; primary mammary gland sarcomas are high-grade tumors generally associated with a poor prognosis, but as sarcomas they typically metastasize via the hematogenous route and bypass the lymph node. These cases have a poor outcome despite a negative regional lymph node. 38 The choice of end points also varies; ideally the end points used to determine the importance of a prognostic variable should be tumor specific, such as remission duration, time to progression, or tumor-specific survival. Many dogs with mammary tumors are old and die from other causes; not censoring these cases may result in failure to identify true prognostic factors, that is, factors associated with aggressive biological behavior. In an analysis conducted as part of a large retrospective study using tumor recurrence as the end point and including only dogs with carcinomas whose lymph node was excised as part of the original surgery, lymph node metastasis was found to be a significant prognostic factor. 42 Lymph node status has also been found to be associated with prognosis in several other larger studies, some of which have included multivariate analysis. 12,38,56,96
Tumor stage
Information regarding tumor size and lymph node status is included in both of the staging systems and provides the information necessary for assigning tumor stage. Tumor stage, according to both the original WHO staging system and the modified system, has been evaluated for prognostic significance. Yamagami et al 96 used the original staging system and found a significant difference between stages 1, 3, and 4. There were insufficient numbers of dogs with stage 2 disease in this particular study. The modified staging system was used in 2 more recent studies; Chang et al 12 found a significant difference between dogs with stage 1, 2, or 3 compared with dogs with stage 4 and 5; however, detailed comparisons between the various stages were not provided. Philibert et al 66 also found stage to be prognostic for survival, with dogs with stage 1 disease live significantly longer than dogs with more advanced disease. Studies on stage and prognosis often suffer from the same limitations as discussed in the section on lymph node status and stage, including lack of standardization of staging procedures, surgical approach, and follow-up monitoring. These limitations in data collection and variations in patient care may result in stage being underestimated or overestimated and therefore result in flawed analyses.
Cell Differentiation Markers and Prognosis of Canine Mammary Tumors
In the normal canine and human mammary glands, ducts and lobules are lined by 2 cell layers, an inner/luminal population of secretory cells and a distinct outer cell layer, juxtaposed to the basement membrane, termed the basal/myoepithelial layer. In both species, the luminal epithelial cells are characterized by the expression of low molecular weight luminal cytokeratins (CKs), including CK8, CK18, CK19, and CK7. 31,36,68,74,94 The outer cell layer is formed by cells that variably express high molecular weight basal CKs such as CK5, CK6, CK14, and CK17 in addition to other markers such as smooth muscle actin (SMA), calponin, vimentin, and p63 (Figs 1-8). 1,3,8,20,28,39,68 Since these basal cells exhibit an immunophenotype of both epithelial and smooth muscle cells, they are therefore referred to as basal/myoepithelial cells. Some authors hypothesize that there is a close relationship between the basal/myoepithelial compartment and the stem/progenitor cells of the mammary gland. 21,68
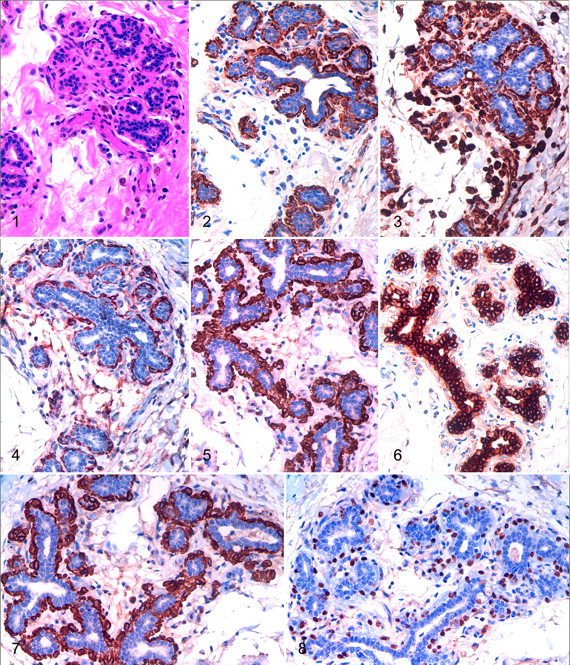
During cell transformation and tumor development, most of the specific cell differentiation markers seem to be maintained. 4,13 This property has enabled these proteins to be used as tumor markers for canine and human mammary tumors.
In the last 20 years the investigation of cell differentiation markers has been used in veterinary medicine primarily to improve our knowledge of the histogenesis of canine mammary tumors. There are conflicting hypotheses on the role of the myoepithelium in the genesis of complex and mixed tumors, the most common mammary neoplasm in dogs. Historically, there were 3 hypotheses on the origin of the mesenchymal elements, such as bone or cartilage that are found in mixed tumors: metaplasia from epithelial cells, metaplasia from stromal connective tissue, and metaplasia from basal/myoepithelial cells. 49 Even if the specific mechanism is still not completely understood, several studies performed with different panels of basal markers reinforced the putative role of the myoepithelium in the metaplastic change to mesenchymal tissue. In both complex and mixed tumors, those myoepithelial cells that were still present within the basement membrane maintained their normal immunohistochemical characteristics, whereas in those portions of the tumor where the myoepithelial cells were proliferating in the interstitum they tended to show decreased expression of CK14, CK5, SMA, calponin, and p63 and enhanced expression of vimentin. Proliferating interstitial myoepithelial cells may eventually become fibroblast-like cells, showing only vimentin immunoreactivity. However, few of these cells and rare chondrocytes in mixed tumors retained expression of basal CKs, SMA, calponin, and p63, supporting the hypothesis that there was a metaplastic mesenchymal change of myoepithelial cells. 7,20,24,28,31,36,72,91 This shift in the myoepithelial immunoprofile was associated with an increased expression of bone morphogenetic protein-6 and chondromodulin-I, proteins that may be involved in ectopic cartilage and bone formation. 40,91 However, we do not know the role of the stromal connective tissue, such as interstitial myofibroblasts or fibroblasts in mesenchymal tissue formation, and how these cells might interact with the myoepithelial cells to produce the connective tissue. 91
In humans, where the majority of breast malignancies are composed of a single population of epithelial cells (corresponding to simple tumors in dogs), most of the antibodies against basal cell markers are also able to identify the preexisting myoepithelial cell layer. In neoplastic lesions, this feature is considered useful to differentiate in situ from invasive carcinomas. 21,97 Similarly, in dogs, the expression of calponin, SMA, and p63 was evaluated and p63 was found to be more specific for myoepithelial cells than the contractile proteins calponin and SMA given the absence of any cross-reaction with stromal myofibroblasts. 7,24,46 In dogs with complex tumors, in areas where there is proliferation of myoepithelial cells that disrupt the basement membrane and infiltrate into the interstitium, immunohistochemistry for these myoepithelial markers is not helpful in differentiating in situ from invasive carcinomas.
The expression of SMA, calponin, p63, and vimentin has been found in some simple canine mammary tumors. In these cases the immunoreactivity was not confined to the preexisting myoepithelium but also involved the neoplastic epithelial cell population; the basal markers were clearly expressed by a proportion of the neoplastic epithelial cells, an indication of a basal/myoepithelial component that is morphologically indistinguishable from the luminal epithelial component, which stained CK8 and CK18 positive. 20,28,46 Identifying those mammary tumors with and without myoepithelial cell differentiation may be important because the participation of myoepithelial cells in malignant tumors of the mammary gland is considered a favorable prognostic indicator in dogs and women. 27,49 There is evidence to suggest that differentiated myoepithelial cells are natural tumor suppressors because of their inhibitory effect on neoplastic cells, including tumor cell growth, invasion, and angiogenesis. 21,58 Considering these findings, some authors have suggested that basal markers be used as part of the routine diagnosis of canine mammary tumors to identify those cases that have a myoepithelial component that cannot be readily identified on hematoxylin and eosin (HE) evaluation; however, further studies are needed to clarify the prognostic significance of this finding. 20,46
Reports in the veterinary literature describe cases in which there is variable vimentin labeling of the luminal epithelium in canine mammary carcinomas. 15,30,70,94 This phenomenon, associated with a poor prognosis and chemoresistance, has been more widely investigated in humans than in dogs. Some authors interpret this phenomenon as a sign of epithelial–mesenchymal transition (EMT), reflecting the final step of tumor dedifferentiation with loss of epithelial characteristics and polarity, and acquisition of a mesenchymal phenotype with increased migratory behavior and metastatic capability. 41,79 There is no information on the clinical outcome of dogs with vimentin expressing mammary carcinomas. This finding has recently been described in association with the generation of microvascular channels by malignant tumor cells (vasculogenic mimicry). This feature was noted with highly aggressive tumors that showed invasion of dermal lymphatic vessels and had distant metastases (canine inflammatory mammary cancers). 15
In women, the evaluation of cell differentiation proteins in cases of breast cancer is frequently performed in association with other markers such as ER, PR, and HER-2. These immunohistochemical results form the basis for a diagnostic algorithm of tumor subtypes that were identified using gene expression profile studies. These immunohistochemical panels allow breast cancer cases to be subdivided into: (1) luminal tumors, expressing ER and/or PR receptors as well as luminal cytokeratins (CK7, CK8, CK18, CK19), (2) basal-like tumors, negative for hormonal receptors and expressing basal markers (CK5, CK6, CK14, CK17, SMA, calponin, vimentin, and p63), and (3) HER-2–positive tumors, overexpressing the HER-2 receptor. 55,71,74 These breast cancer subtypes are associated with markedly different clinical outcomes, ranging from the best prognosis for the luminal group with a well-differentiated glandular immunoprofile to the worst prognosis for the basal-like phenotype, possibly reflecting a stem/progenitor cell origin. 48,74 Three recent investigations tried to apply similar immunohistochemical panels to canine mammary carcinomas, but the existence of all the immunohistochemical subtypes found in human breast cancer (see above) and the diversity in their biological behavior has been not proven to date. 29,30,80 Dogs with luminal tumors (ER and/or CK19 positive), which usually were low-grade carcinomas with a low proliferation index, had a good prognosis with a long disease-free interval and survival. When this immuhistochemical profile was lost, the tumors demonstrated aggressive histological characteristics and a shorter disease-free interval and survival. 30 There are, however, contradictory data on HER-2–positive tumors and on the definition and biological behavior of basal-like carcinomas in dogs. 29,80 When evaluating these basal-like carcinomas in the dog, the investigators used a different panel of basal markers and established different criteria to define what constituted positivity, which may explain the discrepancies in the results. Furthermore, this immunohistochemical approach, originally created to investigate human breast neoplasias, which rarely show complex and/or mixed patterns, does not distinguish between the expression of the epithelial and the myoepithelial components. One cannot rule out that in dogs, in which myoepithelial proliferation is so frequently a part of the neoplastic process, this may influence the total amount of basal marker expression by the tumor, thereby preventing the identification of the corresponding canine counterpart of human basal-like breast carcinomas.
Canine malignant mammary tumors are a heterogeneous group of neoplasms, which would benefit greatly from a classification system that addresses molecular differences, similar to that described for breast cancer in women. However, caution should be used when applying the human classification system to the dog because further studies are needed to find the appropriate molecular-based taxonomy in dogs and a reliable immunohistochemical surrogate to identify it. 29,80
Finally, in human pathology it is common to use cytokeratin stains to detect occult micrometastases in regional lymph nodes considered unaffected or doubtful when evaluated on routine HE-stained sections. By using these cytokeratin stains investigators have been able to identify patients with a decreased disease-free interval and overall survival. This is particularly important for cases of lobular breast carcinoma composed of noncohesive cells of a similar size to lymphocytes. 18,19 When a cocktail of different cytokeratin clones (AE1, AE3) was applied to 119 regional lymph nodes from dogs with malignant mammary tumors, occult micrometastatic lesions were found in 9.2% of the cases; however, the clinical relevance of this finding in the dog needs to be further investigated. 47
Footnotes
Acknowledgement
This study represents an initiative of the American College of Veterinary Pathologists' Oncology Committee. We thank the ACVP for its support and guidance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
