Abstract
Stringently controlled in vitro experiments are a necessary part of translational research. Cell lines are useful for exploring the underlying biology of cancer. Very few canine soft tissue sarcoma cell lines exist. This report describes the establishment of a new canine soft tissue sarcoma cell line (MBSa1) derived from a high-grade, metastatic neurofibrosarcoma. The primary tumor tissue was obtained from a 12-year-old neutered male German Shepherd Dog and was maintained in tissue culture for a minimum of 20 passages over 7 months. MBSa1 was injected into athymic mice to determine tumorigenicity. Five million cells were injected into the subcutis of the right flank of athymic nude mice. Nine of the 10 mice grew tumors 1 cm or larger within 8 weeks of cell injection. The large number of in vitro passages coupled with solid tumor formation in athymic nude mice demonstrates that MBSa1 has been immortalized and is tumorigenic.
Cancer is typically found to be either the most common or second most common cause of death in dogs and is a significant concern of pet owners. Comparative species research is important given that cancer affects humans and dogs at equally high rates. The similar biologic behavior, phenotypic characteristics, and clinical course of canine and human cancer underscore the importance of comparative species research. Although the incidence of soft tissue sarcomas is greater in the dog population than in the human population, approximately 15% and less than 1%, respectively, the behavior, treatment and outcomes are very similar. 1,12 –14
Stringently controlled in vitro experiments are a necessary part of translational research. Cell lines are useful for exploring the underlying biology of cancer. A fair number of canine mammary tumor and osteosarcoma cell lines are available. 5 –8 However, very few canine soft tissue sarcoma cell lines exist. Here we describe the establishment and basic characteristics of a canine soft tissue sarcoma cell line derived from a patient with a high-grade neurofibrosarcoma at presentation. The patient had metastatic disease at the time of presentation, but metastatic potential of the derived cell line was not assessed.
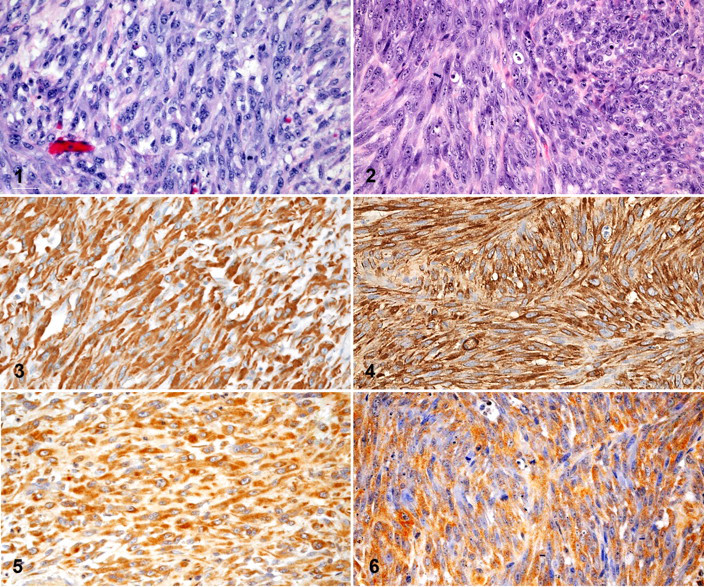
A punch biopsy of the primary tumor, located on the right mandible, was obtained from a 12-year-old neutered male German Shepherd Dog seen by the Oncology Service at North Carolina State University. A portion of the primary tumor was submitted for histologic diagnosis and an additional portion was saved for cell line establishment. Histology and immunohistochemical characterization of the primary tumor were consistent with a soft tissue sarcoma. The nonencapsulated, invasive mass was composed of sheets of intersecting fascicles of large, pleomorphic spindle cells that were supported by small amounts of fibrovascular stroma (Fig. 1 ). Mitoses were moderately common, ranging between 0 and 6 per 1 high-power (400×) microscopic field. Diffuse strong vimentin and moderate neuron specific enolase (NSE) immunohistochemical immunoreactivity was detected (Figs. 3 and 5 , respectively). Immunohistochemical reactivity for S100, Melan-A, smooth muscle actin, muscle specific actin, or desmin was not detected (images not shown). The histopathologic appearance of the tumor combined with the lack of reactivity for S100, Melan-A, smooth muscle actin, muscle specific actin, and desmin excluded leiomyosarcoma, rhabdomyosarcoma, and melanoma from the diagnosis.
The tumor fragment identified for cell line establishment was placed into 0.05% trypsin/EDTA (Mediatech, Inc., Manassas, VA) for 10 minutes to allow the cells to disaggregate. The tumor tissue was then cut into 1-mm 3 fragments. The fragments were placed into a 100-mm sterile tissue culture dish containing 5 ml of Dulbecco’s modified Eagle medium (DMEM) (Mediatech) supplemented with 20% fetal bovine serum (FBS) (Mediatech) and 50 μg/μl Primocin (Invivogen, San Diego, CA) and placed in a 37°C humidified 95% air/5% CO2 cell culture incubator. Cells consistent with fibroblastic morphology emerged from the tumor pieces within 2 weeks. The cells were allowed to grow to 90% confluence in the initial 100-mm dish. The cells were passaged into T-75 tissue culture flasks and allowed to grow to 80% confluence for subsequent passages. The cell line, now defined as MBSa1, was maintained in tissue culture for a minimum of 20 passages over 7 months. Once passage 20 had been reached, the concentration of FBS in the media was reduced to 10%. The cells continued to grow at the same rate as with the higher concentration of FBS. The doubling time of the cells was calculated to be approximately 26 hours. One million cells were seeded, and in 72 hours the cell population had expanded to 12 million cells. We have been able to maintain growth of MBSa1 for more than 40 passages.
Tumorigenicity was assessed by solid tumor formation in athymic nude mice. MBSa1 was grown in vitro to passage 39 for injection into athymic nude mice. The cells were collected, and total number and viability were determined with a Cellometer (Nexcelom Bioscience, Lawrence, MA). The cells were found to be 92% viable. The cells were resuspended in equal parts serum-free DMEM and matrigel basement membrane matrix (BD Biosciences, San Jose, CA). Each athymic nude mouse was injected with 5 million cells in a volume of 200 μl into the subcutis of the right flank according to an approved Institutional Animal Care and Use protocol. Tumors were harvested from the mice when they reached 2.5 cm in diameter or 8 weeks had elapsed, whichever came first. Seven of the 10 athymic nude mice had tumors reaching 2.5 cm within 6 weeks following tumor cell injection. Nine of the 10 mice grew at least 1-cm tumors within 8 weeks of the initial injection. The remaining mouse had no visible growth of tumor and was euthanatized. Tumors were harvested and fixed in 10% normal buffered formalin for subsequent analysis.
Five of the 9 xenographs examined shared a similar histological morphology. Upon initial histological review, the remaining 4 tumors had varying degrees of necrosis and were determined to contain too little viable tissue for an accurate morphological diagnosis. Histological morphology of the xenografts is consistent with the neurofibrosarcoma identified in the original surgical biopsy from the canine patient (Fig. 2 ). The 5 assessed tumors were nonencapsulated and invasive masses. They measured 1.0–2.0 cm in diameter and were composed of solid sheets of large neoplastic spindle cells, which were arranged in intersecting fascicles and were supported by small to moderate amounts of collagenous stroma containing few small caliber blood vessels. The moderately pleomorphic cells had moderately abundant amphophilic cytoplasm with indistinct cell borders and oval to spindle nuclei with 1–3 prominent nucleoli. There was twofold to threefold anisokaryosis. Mitotic figures were common and ranged between 0 and 12 per high-power field (HPF) (400×). Multifocal areas of lytic necrosis were common and involved 5–30% of the large tissue sections. Similarly, strong diffuse vimentin and moderate patchy NSE immunohistochemical immunoreactivity was detected in xenografts (Figs. 4 and 6 , respectively).
The number of chromosomes contained in MBSa1 cells was determined by karyotype. Briefly, cells were treated for 4 hours with 10 μg/ml colcemid (Karyomax, Gibco, Inc.) followed by fixation with a methanol/acetic acid solution (3 parts methanol to 1 part acetic acid). Fixed cells were applied to glass slides and stained with 4′,6-diamidino-2-phenylindole (DAPI) for visualization of the nuclei. Cells were then examined with a 40× oil immersion objective using an Axioplan 2 imagining microscope (Zeiss, Inc., Maple Grove, MN). Normal canine cells contain 78 chromosomes. Two independent observers assessed chromosome spreads from 14 separate MBSa1 cells (Fig. 7 ). The number of chromosomes per cell averaged 77 ± 0.27 (mean ± SEM).

Fluorescence microscopy, 100×, DAPI labeling of chromosomes from an MBSa1 cell. For assessment purposes the red dot indicated each chromosome to be counted. Fourteen individual MBSa1 cells were assessed for chromosome number. The number of chromosomes per cell averaged 77 ± 0.27 (mean ± SEM).
Discussion
The expanding field of veterinary oncology coupled with the expanding interest in using spontaneously occurring canine tumors as translational models of disease necessitates corresponding tumor cell lines. The establishment of MBSa1 as a canine neurofibrosarcoma cell line is one tool for the study of soft tissue sarcomas in canine patients and for both human and canine translational research.
A number of investigators have found a strong similarity between canine and human cancers of the same histological type, both genetically and in presentation. 11 Additionally, soft tissue sarcomas can be difficult to diagnose because they often are painless and occur in somatic areas of the body. 4 The tumors may be allowed to grow to a larger size before detection compared with their carcinomatous counterparts.
Although soft tissue sarcomas are much less common in the human population, both human and canine tumors are most often treated with a combination of surgery and radiation therapy. For tumors designated as high grade, adjuvant chemotherapy is often recommended. 3 In canines, soft tissue sarcomas are slow growing, are locally invasive, and have a high rate of recurrence. 3 Surgical margins free of tumor cells are prognostic for local recurrence, and the mitotic rate (≥9 mitoses per 10 HPFs) of soft tissue sarcoma is prognostic for development of metastatic disease. 2,9
Behavior of canine cancer cells is similar to that of human cancer cells. Often, malignant cells have abnormal chromosome numbers or the chromosomes may be morphologically irregular. We did karyotype analysis of our cells to corroborate that our cells were canine in origin. Identification and differentiation of the smaller canine chromosomes are often difficult in the karyotype analysis of malignant cells. Cancer cells, and MBSa1 in particular, have a high mitotic rate, making identification and differentiation of the smaller canine chromosomes difficult during the karyotyping process. Canine chromosomes are acrocentric, and malignant cells condense dramatically with Karyomax treatment. Additionally, canine chromosomes are very small in size compared with their human counterparts. Identification and banding of canine chromosomes by DAPI are difficult at best. Newer techniques such as chromosome paints and fluorescent in situ hybridization have improved chromosome identification, but these techniques are technically challenging even for experienced canine cytogeneticists. The fact that these cells are malignant in origin and the difficulty of distinguishing between the smaller chromosomes may account for the abnormal chromosomal number. Human, mouse, and rat cells contain 46, 40, and 40 chromosomes, respectively. The only 2 species containing chromosome numbers near 78 are the pigeon and chicken, with 80 and 78 chromosomes, respectively. 10 Therefore, it is unlikely that this tumor is of any other origin than canine.
The established neurofibrosarcoma cell line, MBSa1, grows well in tissue culture as evidenced by more than 40 serial passages, has been demonstrated to be tumorigenic by xenograft growth in athymic nude mice, and has a chromosomal number consistent with cells of canine origin.
Footnotes
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
