Abstract
Lymph node (LN) metastasis in canine mast cell tumor (MCT) can affect prognosis and postsurgical treatment recommendations; however, routine histological single-section examination may underestimate the incidence of metastases. This prospective study aimed at determining whether longitudinal step-sectioning of the entire LN allows for a more reliable detection of metastases. Dogs with MCT undergoing resection of the primary tumor and regional lymphadenectomy were enrolled. Formalin-fixed LNs were bisected longitudinally, both halves were embedded in paraffin and histological sections prepared at 200 μm steps. The nodal mast cells were classified according to the Weishaar classification. First-section evaluation (FSE; ie, examination of the first section obtained from the blocks) and whole LN step-section evaluation (SSE) were compared. Fifty-eight LNs were included. The median number of sections per LN was 6 (range, 3–28). FSE with toluidine blue (TB) revealed 27 (47%) nonmetastatic (HN0), 14 (24%) premetastatic (HN1), 9 (15%) early metastatic (HN2), and 8 (14%) overtly metastatic (HN3) LNs. SSE with TB resulted in upgrading the LN status in 2 cases (HN2 to HN3; HN0 to HN1). Evaluation of the first section plus an additional step-section resulted in 100% accuracy. Compared with SSE with TB, the accuracy of FSE with HE was 98% for HN3 LNs and 74% for HN2 LNs. FSE appears to reliably allow for the detection of LN metastasis in MCT, although examination of a further parallel section at a 200 μm step may increase the accuracy. A metachromatic stain is recommended for the identification of early metastases.
Keywords
The status of regional lymph nodes (LN) in dogs with mast cell tumors (MCT) can affect prognosis and postsurgical treatment recommendations.1,15,17,19 The cytologic assessment of LN metastases remains very subjective and, although a more standardized system has been proposed, it is of little help in the decision-making process, mainly due to the nebulous categories of possible metastasis and probable metastasis. 13
Histologic evaluation of surgically removed LNs allows for a better characterization of mast cell arrangement and is therefore generally considered more accurate. 14 To standardize the assessment of nodal metastatic disease in canine MCT, a 4-tiered histologic classification system has been proposed, and it has been demonstrated to correlate with the clinical outcome. 27 Nevertheless, the number of evaluated histologic sections represents a further element of variability and may affect diagnostic accuracy. Although there is some variability in the methods of LN evaluation among veterinary pathology laboratories, one of the most common techniques involves examining a single section, obtained by LN bisection through the longest axis, but this may underestimate the true incidence of metastases. 24
In human oncology, several studies have proposed different techniques for histological LN processing. However, it is generally accepted that a proper evaluation requires histologic and immunohistochemical analysis of multiple microtome sections performed at regular steps, no wider than 2 mm, to increase the likelihood of detecting neoplastic cells.4,5,6,18,25 This procedure has also been recommended for the staging of small animal tumors, including MCT.3,10,11
The purpose of this prospective study was to determine whether step-sectioning of the whole LN would result in a higher probability of detecting MCT metastases when compared with a more traditional technique of single sectioning via LN bisection through its longest axis.
Materials and Methods
All LNs of dogs with histologically confirmed cutaneous or subcutaneous MCT were prospectively and consecutively included over a 1-year period (March 2020–March 2021). Patients had been referred to the Oncology Unit at the Department of Veterinary Medical Sciences (University of Bologna, Italy) and had undergone surgical resection of the primary tumor with concurrent or subsequent regional lymphadenectomy. Recorded data included demographic and clinical information (breed, sex, age, weight, tumor site, removed LNs, LN size on physical/ultrasound examination [ie, normal or enlarged]) and pathological information (cutaneous/subcutaneous location of MCT, Patnaik et al 22 grade, Kiupel et al 12 grade).
Formalin-fixed LNs were bisected along their longest axis through the hilum, and both halves were included in paraffin (bivalving protocol).4,18 Smaller LNs were included in a single block, whereas larger nodes required one half or one quarter of the LN per block.
The first 2 consecutive sections (4 µm) of the paraffin blocks, corresponding to the macroscopic LN cut surface, were stained with hematoxylin and eosin (HE) and toluidine blue (TB; ready-to-use staining solution; Bio-Optica, Milan, Italy), respectively. Multiple 4-µm parallel sections were then cut at 200 µm steps until exhaustion of the paraffin blocks and stained with TB.
The nodal mast cells were blindly evaluated according to Weishaar et al’s 27 classification. Results obtained from first-section evaluation (FSE) on HE-stained slides and those obtained from FSE on TB-stained slides were each compared with step-section evaluation (SSE) of the entire LN (including the first section) on TB-stained slides. Accuracies, defined as the percentages of correctly classified cases, were calculated considering SSE with TB as the gold standard. Sensitivity (ie, the likelihood of detecting a positive LN) and negative predictive value (ie, the likelihood that a LN with a negative result is truly unaffected) in evaluating the absence (HN0/HN1) or presence (HN2/HN3) of metastatic disease were also assessed on TB- and HE-stained slides.
Histologic evaluations were performed by a board-certified veterinary pathologist (SS) for TB-stained slides and by a board-certified veterinary pathologist and a resident in veterinary pathology (AR2, AR1) for HE-stained slides. All pathologists were blinded to the results of the primary tumor evaluation, the depth of the section, and the results of the differently stained section. For HE-stained slides, the 2 operators were required to reach a consensus for the final diagnosis. All evaluations were performed with light microscope at a maximum magnification of 400x.
For descriptive statistics, numerical variables were expressed as median and range; categorical variables were summarized as frequency (percentage).
Results
Demographic and Clinicopathologic Information
Fifty-eight LNs from 18 MCT-bearing dogs were prospectively included in the study. There were 3 mixed breed dogs and 15 purebred dogs. Most represented breeds included American Staffordshire terrier (n = 3) and Boxer (n = 3). There were 10 (56%) spayed females, and 8 (44%) males, of which 2 neutered. Median age was 8 years (range, 4–12 years), and median weight was 28 kg (range, 5–41 kg).
Tumor sites included trunk (n = 6; 33%), head and neck (n = 4; 22%), limbs (n = 4; 22%), inguinal area (n = 3; 17%), and mammary region (n = 1; 6%).
Twelve of the 18 (67%) MCTs were cutaneous, whereas 6 (33%) were subcutaneous. Among cutaneous MCTs, there were 10 (83%) Patnaik grade II tumors, and 2 (17%) Patnaik grade III tumors. According to Kiupel’s classification, there were 7 (58%) low-grade tumors, and 5 (42%) high-grade tumors. Removed LNs included 20 (34%) inguinal, 11 (19%) prescapular, 10 (17%) axillary, 5 (9%) medial iliac, 5 (9%) submandibular, 4 (7%) popliteal, and 3 (5%) retropharyngeal. Forty-three (74%) LNs were normal sized and 15 (26%) were clinically enlarged.
LN Evaluation
The complete list of removed LNs and final Weishaar’s score (based on SSE of TB-stained slides) is provided in Supplemental Table S1.
The median number of sections per LN was 6 (range, 3–28).
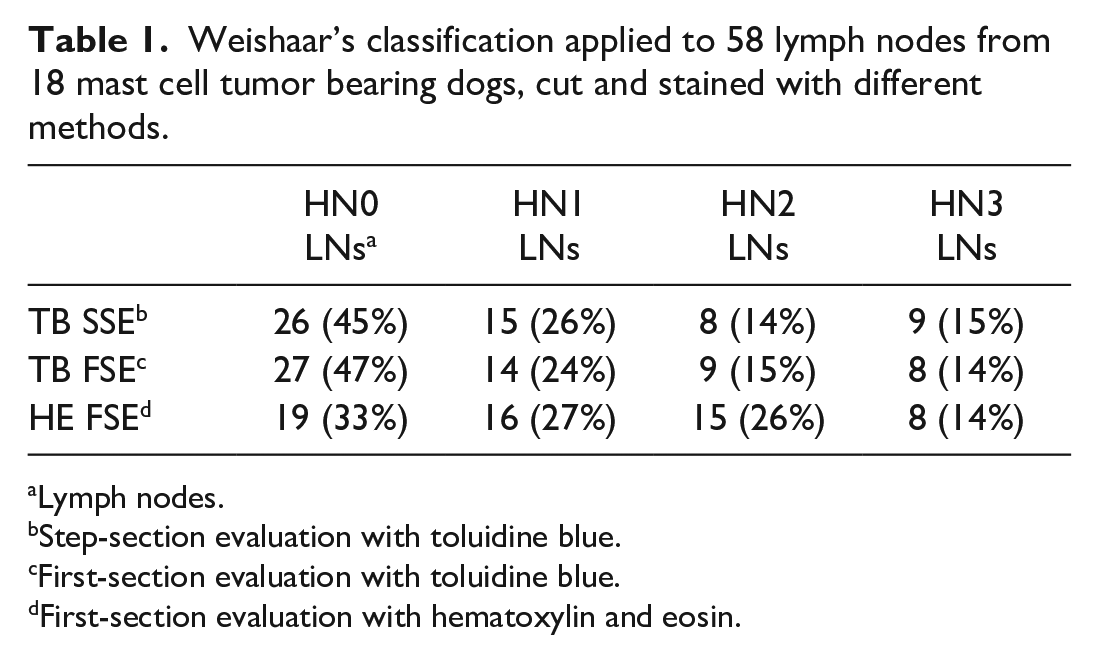
According to FSE on TB-stained slides, there were 27 (47%) HN0, 14 (24%) HN1, 9 (15%) HN2, and 8 (14%) HN3 LNs. SSE resulted in upgrading of Weishaar’s classification in 2 cases (HN2 to HN3 and HN0 to HN1, respectively). Upgrading occurred at the first step section for both LNs (Fig. 1; Table 1). Considering SSE on TB-stained slides as the gold standard, FSE on HE-stained slides allowed identification of 8/9 (89%) HN3 LNs, 4/8 (50%) HN2 LNs, 6/15 (40%) HN1 LNs, and 15/26 (58%) HN0 LNs. No HN0, HN1, or HN2 LN were misdiagnosed as HN3, whereas 10/41 (24%) nonmetastatic (HN0/HN1) LNs were misdiagnosed as early metastatic (HN2; Fig. 2; Table 1).
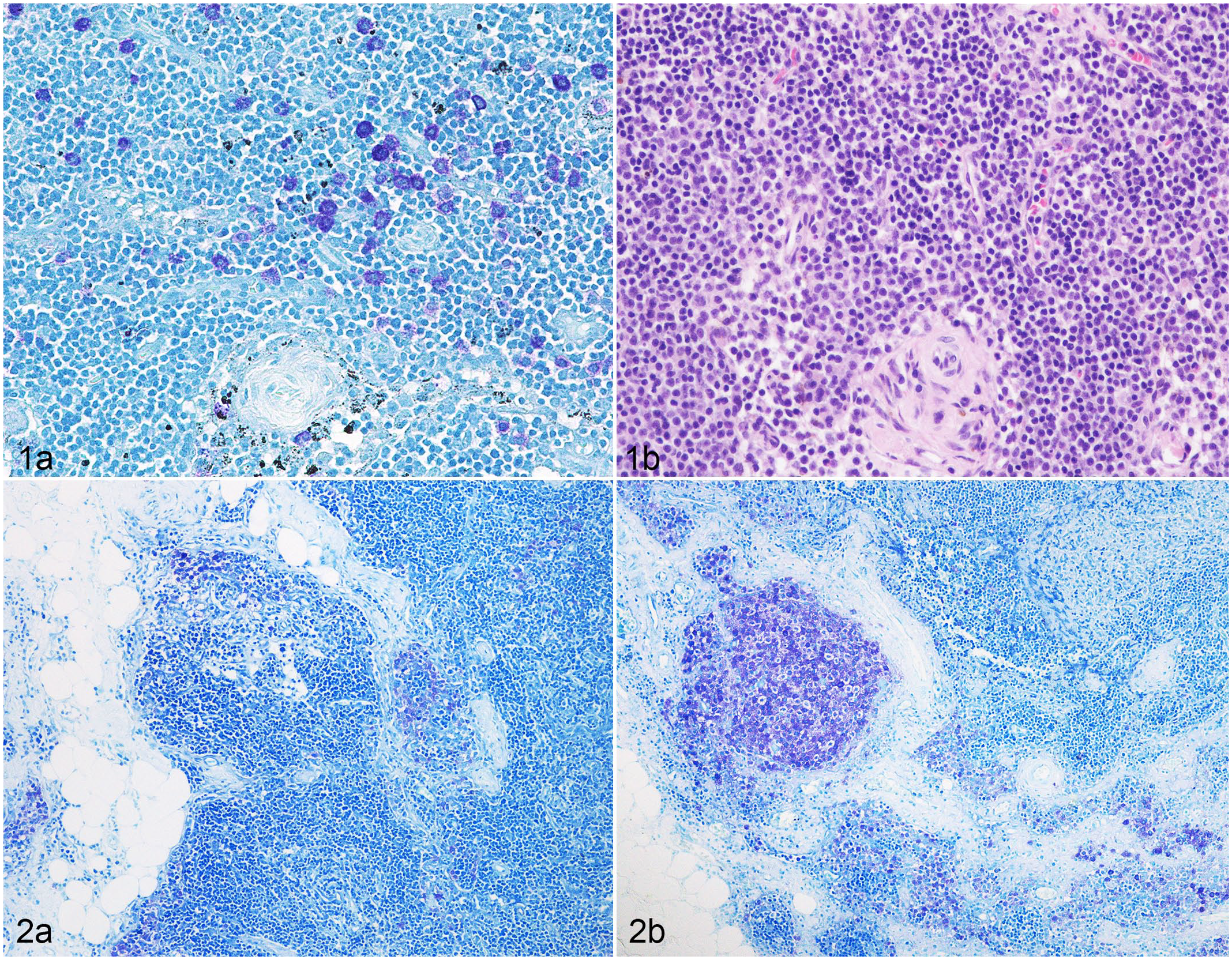
Weishaar’s classification applied to 58 lymph nodes from 18 mast cell tumor bearing dogs, cut and stained with different methods.
Lymph nodes.
Step-section evaluation with toluidine blue.
First-section evaluation with toluidine blue.
First-section evaluation with hematoxylin and eosin.
Overall, the accuracy of FSE on TB-stained slides compared with SSE on TB-stained slides was 97%. The accuracy of FSE on HE-stained slides compared with SSE on TB-stained slides was 98% for the detection of HN3 LNs and 74% for the detection of HN2 LNs. When generally considering the absence (HN0/HN1) or presence (HN2/HN3) of metastatic disease, sensitivity and negative predictive value of FSE were 100% on TB-stained slides, and 76% and 91% on HE-stained slides, respectively.
Five out of 8 (62%) early metastatic and 6/9 (67%) overtly metastatic LNs were of normal size.
Discussion
The microscopic evaluation of surgically removed LNs of MCT-bearing dogs is a crucial step for clinicians, who must decide whether to declare the surgery as curative or not.1,9,15,17,19 Nevertheless, the optimal processing method for the histologic examination of regional LNs is yet to be established in veterinary oncology.
In the present study, we compared 2 protocols of LN processing, with the aim to determine whether whole LN longitudinal step-sectioning can increase the probability of detecting MCT metastasis when compared with the sole examination of the cut surface after bisection. The latter method resulted in a very high accuracy of 97%. This is not surprising, as cutting an LN longitudinally across the hilum exposes the largest possible tissue surface and allows for a through simultaneous examination of all LN structures (cortex, medulla, sinuses). Neoplastic cells reach the LN from the subcapsular sinuses through afferent capsular lymph vessels; later, the tumor spread continues from the cortical parenchyma to deeper zones of the LN, frequently following the medullary sinuses.4,21 Thus, the majority of metastases are located in a central plane through the hilum.4,18 Nevertheless, one could argue that evaluating more sections per LN could improve the accuracy of microscopic assessment, since single mast cells or small foci of mast cells could hide in peripheral zones of the organ. In human cancer patients, the optimal processing for the pathology of sentinel lymph is still a matter of debate. It is generally accepted that cutting LNs into multiple 2.0-mm slices should allow for the detection of all macrometastases (defined as neoplastic cells foci >2.0 mm), which are known to relevantly affect prognosis. 16 However, closer steps might be required to identify micrometastases (>0.2–2.0 mm) and isolated tumor cells (≤0.2 mm or 200 cells). 26 Although an increasing attention is being given to the detection of these low-volume metastases, the number of sections required and the interval between them has not been standardized. In addition, their clinical significance is still unclear. While there is some evidence that they may be associated with worse long-term prognosis, overall survival data with 10 or more years of follow-up are limited and there are still many reservations regarding the need for adjuvant chemotherapy in these patients.2,5,7,8,18,23
In canine MCT, the HN3 nodal status is comparable with the human macrometastatic category, but a comparison between HN2 and micrometastases or between HN1 and isolated tumor cells may be less accurate both in terms of histologic features, biologic behavior, and clinical outcome. Despite the uncertainties regarding their true clinical significance, nodal aggregates of 3 or more mast cells are presently recognized in dogs as early metastasis (HN2) and should be reported. 27 To increase the likelihood of identifying such small aggregates, a step-sectioning at 0.2-mm intervals (corresponding to the minimum size of micrometastases according to human pathology) has been applied in the present study. A shorter interval has been judged too expensive and hardly applicable in routine veterinary diagnostics. With the proposed method, the histological node status was increased in only 2 of 18 cases. While this upgrading would not have influenced postsurgery recommendations in one case (HN0–HN1), it might have led to undertreatment in the other one (HN2–HN3). Indeed, according to a recent study, surgery can be considered curative for low-grade MCTs with a surgically removed regional HN2 LN, whereas adjuvant medical therapy is recommended for HN3 LNs. 17 The upgrading was identified in the first step section in both cases. As reported before, the median longitudinal section allows for the best evaluation of the LN, but tissue-processing artifacts, such as tearing, partial detachments, or fragmentation, may sometimes alter the quality of this section. Furthermore, bisecting an LN exactly through its hilum is an ideal, which is hardly ever achieved in practice. The first step section has similar features to the middle section (ie, the section corresponding to the bisected LN cut surface) and may help minimize the impact of technical and processing errors. Based on our results, the extent of LN dissection may be safely limited to 2 sections at a 200-µm step, since evaluation of the middle section plus an additional step-section reached an accuracy of 100% in this study.
In the study by Weishaar and colleagues, 27 metachromatic stains were not available for all LNs, thereby limiting the evaluation to HE-stained sections in more than half of cases. In our caseload the evaluation of HE-stained sections was not reliable in identification of early metastasis and led either to underestimating or overestimating the LN status in a significant proportion of cases, with a HN2 detection rate as low as 50%. In HE-stained slides, mast cell granules are not as evident as in TB-stained slides, and the identification and quantification of small numbers of mast cells interspersed among lymphocytes can be time-consuming and more challenging for less-experienced pathologists. Poor mast cell differentiation, excessively thick sections, and variations in the affinity of stains may further adversely affect this assessment. The limit between HN1 and HN2, which relies on the presence of an aggregate of at least 3 mast cells, is very narrow and can easily justify cases of misdiagnosis. Conversely, the presence of discrete foci of mast cells disrupting LN parenchyma (HN3) can be more immediately identified even without the help of metachromatic stains. Noteworthy, in this study, the only HN3 LN misdiagnosed as HN2 on HE-stained slides was the one for which the first step section was needed to upgrade the nodal status from HN2 to HN3. Based on the overall low accuracy rate of HE, the use of TB-stained sections to define the nodal status is strongly encouraged in nonovertly metastatic LNs.
Beyond the standardization of processing methods, the assessment of nodal metastases of canine MCT will remain a challenge, due to the inherent characteristics of the neoplasm. LNs normally contain a low number of mast cells and it is virtually impossible to distinguish neoplastic from non-neoplastic ones, with the exception of few cases where neoplastic mast cells show marked features of cellular/nuclear atypia. Weishaar’s classification is certainly an important step toward the standardization of nodal assessment in MCT, but further studies are needed to better clarify the proposed criteria and fully validate their prognostic relevance. The classification relies more on the arrangement and distribution of the infiltrate, rather than considering the overall number of mast cells, and there is great variability within both HN2 and HN3 categories. The former includes cases with a single cluster of 3 mast cells and others with a diffuse infiltration of numerous mast cells not causing disruption or effacement, whereas the HN3 category includes cases with a single neoplastic focus as well as cases in which the LN is completely replaced by mast cells. Due to this wide range of possibilities, some HN3 LNs may contain a lower overall number of mast cells compared with some HN2 cases. Whether prognosis is ultimately affected by this huge intra-class heterogeneity will have to be the subject of further investigations.
Finally, it should be noted that nearly two thirds of metastatic LNs in this study were not clinically enlarged, as previously reported.9,20 This confirms the lack of utility of physical examination in the assessment of metastatic disease and further underlines the importance of a proper histologic examination.
In conclusion, the evaluation of the cut surfaces obtained from longitudinally bisected LNs allows for high accuracy; nevertheless, an additional parallel section at a 200-μm step can increase the likelihood of identifying MCT metastases with a limited cost increment and a minimum impact on laboratory workload. Furthermore, since HE is not reliable for the detection of early metastases, the use of a metachromatic stain such as TB is encouraged for LN assessment.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221088780 – Supplemental material for Longitudinal lymph node step-sectioning for the identification of metastatic disease in canine mast cell tumor
Supplemental material, sj-pdf-1-vet-10.1177_03009858221088780 for Longitudinal lymph node step-sectioning for the identification of metastatic disease in canine mast cell tumor by Silvia Sabattini, Eugenio Faroni, Andrea Renzi, Giulia Ghisoni, Antonella Rigillo, Giuliano Bettini, Arianna Pasquini, Stefano Zanardi, Dina Guerra and Laura Marconato in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
