Abstract
Mast cell tumor (MCT) is a common canine cutaneous neoplasm with variable biological behavior. A 2-tier histologic grading system was recently proposed by Kiupel et al to reduce interobserver variation and eliminate prognostic uncertainty of the Patnaik system. This study compared the ability of these 2 grading systems to predict survival in a cohort of dogs with MCTs. However, surgical margins were unknown, and the risk of developing new/metastatic MCTs was not studied. Histologic grade was assessed according to both systems for 137 surgically resected cutaneous MCTs. The relationship between grade and survival was evaluated. According to the Patnaik system, 18 MCTs (13.1%) were classified as grade I, 83 (60.6%) as grade II, and 36 (26.3%) as grade III. Grade III was associated with a poorer prognosis (P < .001), but no significant difference between grades I and II was detected. Grading according to the Patnaik system was based on consensus grading among 3 pathologists, and interobserver variability was not considered. All grade I MCTs were low grade in the Kiupel system, and all grade III were high grade. Among grade II, 71 (85.6%) were low grade, and 12 (14.4%) were high grade, with a 1-year survival probability of 94% and 46%, respectively (P < .001). The 2-tier system had a high prognostic value and was able to correctly predict the negative outcomes of some grade II MCTs. Data also confirm that histologic grading cannot predict biological behavior of each MCT and should be supplemented with molecular methods for more accurate prognostication.
Mast cell tumors (MCTs) are the most common cutaneous tumors in dogs, accounting for 16% to 21% of all skin neoplasms. Canine MCTs vary widely in their biological behavior, ranging from nearly benign to highly invasive and metastatic. 1,11
It is acknowledged that histologic grading is a prognostic tool for canine cutaneous MCTs. 5,9,10 Various grading systems have been proposed, 2,8 and among them, the classification by Patnaik et al is the most widely used. 1,8 The Patnaik system (PS) classifies MCTs into 3 grades based on histologic characteristics that include cellularity, cellular morphology, invasiveness, mitotic activity, and stromal reaction. 8 According to this classification, well-differentiated MCTs (grade I) carry an excellent long-term prognosis and are usually cured by surgical excision alone, while poorly differentiated (grade III) MCTs are locally invasive and more likely to metastasize, so chemotherapy is generally recommended in addition to surgery. 1 Conversely, the behavior of intermediate (grade II) MCTs is more difficult to predict. The majority of grade II MCTs are cured with wide surgical resection, but between 5% and 22% metastasize. 1 Additionally, the PS is influenced by subjective interobserver variations. 4,6,7
To improve concordance among pathologists and reduce the prognostic uncertainty of the intermediate grade in the PS, a 2-tier histologic grading system was proposed in 2011 by Kiupel et al. 4 According to the Kiupel system (KS), the diagnosis of a high-grade (HG) MCT is characterized by any of the following criteria: at least 7 mitotic figures in 10 high-power fields (HPFs), at least 3 multinucleated cells in 10 HPFs, at least 3 bizarre nuclei in 10 HPFs, and karyomegaly. 4 All other tumors are considered low grade (LG). In the original study, this novel grading system demonstrated a 96.8% consistency among pathologists and a high prognostic power. 4
Many MCTs originate in the dermis and extend into the subcutis, but there is a subset that is restricted to the subcutaneous fat. The work by Patnaik et al did not consider primarily subcutaneous MCTs; nevertheless, many pathologists include them with cutaneous MCTs and ascribe them as being grade II because of their location. 12 It was recently demonstrated that the majority of subcutaneous MCTs have a favorable prognosis compared to most dermal tumors, with extended survival times and low rates of recurrence and metastasis. 12
In the present study, the Patnaik and Kiupel grading systems were comparatively evaluated in the same cohort of dogs with cutaneous MCTs to determine which system is the best predictor with regard to tumor mortality. Additionally, the behavior of subcutaneous MCTs with respect to dermal MCTs was investigated.
Materials and Methods
Study Overview and Case Inclusion Criteria
A retrospective study was performed on tissue samples submitted to the diagnostic laboratories of the Animal Health Trust (Newmarket, UK) and the Department of Veterinary Medical Sciences (University of Bologna, Italy) in the period between 2004 and 2011 with a diagnosis of canine primary cutaneous MCT.
Cases were ultimately included if (1) surgical excision had been the only treatment modality, (2) the surgical sample was submitted for histological examination, and (3) follow-up information (overall survival and cause of death) was available for a minimum period of 12 months after surgery.
Patients receiving chemotherapy or radiotherapy were excluded to better evaluate the ability of grading in predicting the biological behavior of MCTs without the interference of treatment. Information about tumor stage and histologic margin status was not available for all cases, so these parameters were not considered in the statistical analysis.
All tumor samples were fixed in 10% neutral buffered formalin and paraffin embedded at the time of submission. Five-micrometer sections were cut and routinely stained with hematoxylin and eosin for histologic evaluation. For each sample, the primary tumor location (dermal or subcutaneous) was assessed. MCTs were defined as subcutaneous if no invasion of the dermis was observed. All dermal tumors were graded following PS and KS, by consensus of 3 pathologists (F.S., S.S., G.B.).
Statistical Analysis
Data were analyzed by use of commercial software programs (SPSS Statistics v. 19, IBM, Somers, NY; Prism v. 5.0, GraphPad, San Diego, CA); P values ≤ .05 were considered significant.
The prognostic significance of the 2 grading systems was investigated by means of Kaplan-Meier survival curves; the log-rank test was used to compare the survival among groups. The date of surgery, as the day of submission of the sample to the laboratory, was used as the entry point. The last reported date that the patient was seen alive by the referring practice was used as the date of censor. Dogs that were reported dead because of MCT were recorded as events.
Both grading systems were included in a multivariable Cox proportional hazards model. For each variable, the risk (hazard ratio) of not surviving during the follow-up period was estimated with corresponding 95% confidence intervals (CIs) and P values.
Results
A total of 162 dogs met the inclusion criteria. Sixty-nine (42.6%) patients were still alive at the end of the study, and 51 (31.5%) died for causes not correlated with MCT. The median follow-up time for this group of dogs was 1183 days (range, 365–2756). Forty-two (25.9%) dogs died or were euthanized for progressive MCT disease; the mean survival time in those cases was 153 days (range, 2–698 days).
In sum, 137 (84.6%) tumors were primarily dermic. According to PS, 18 (13.1%) were classified as grade I, 83 (60.6%) as grade II, and 36 (26.3%) as grade III. The mortality rate due to MCT-associated disease was 0% in grade I MCTs, 12% (10 dogs) in grade II MCTs, and 83.3% (30 dogs) in grade III MCTs. The survival probability at 12 months was 100% for grade I MCTs, 87% for grade II MCTs, and 16% for grade III MCTs. The median survival time was not reached for grade I and II MCTs, while it was 108 days (95% CI, 34–182) for grade III MCTs. The PS resulted prognostically significant (log-rank test; P < .001), although the survival curves of grade I and grade II MCTs were not significantly different (Fig. 1).
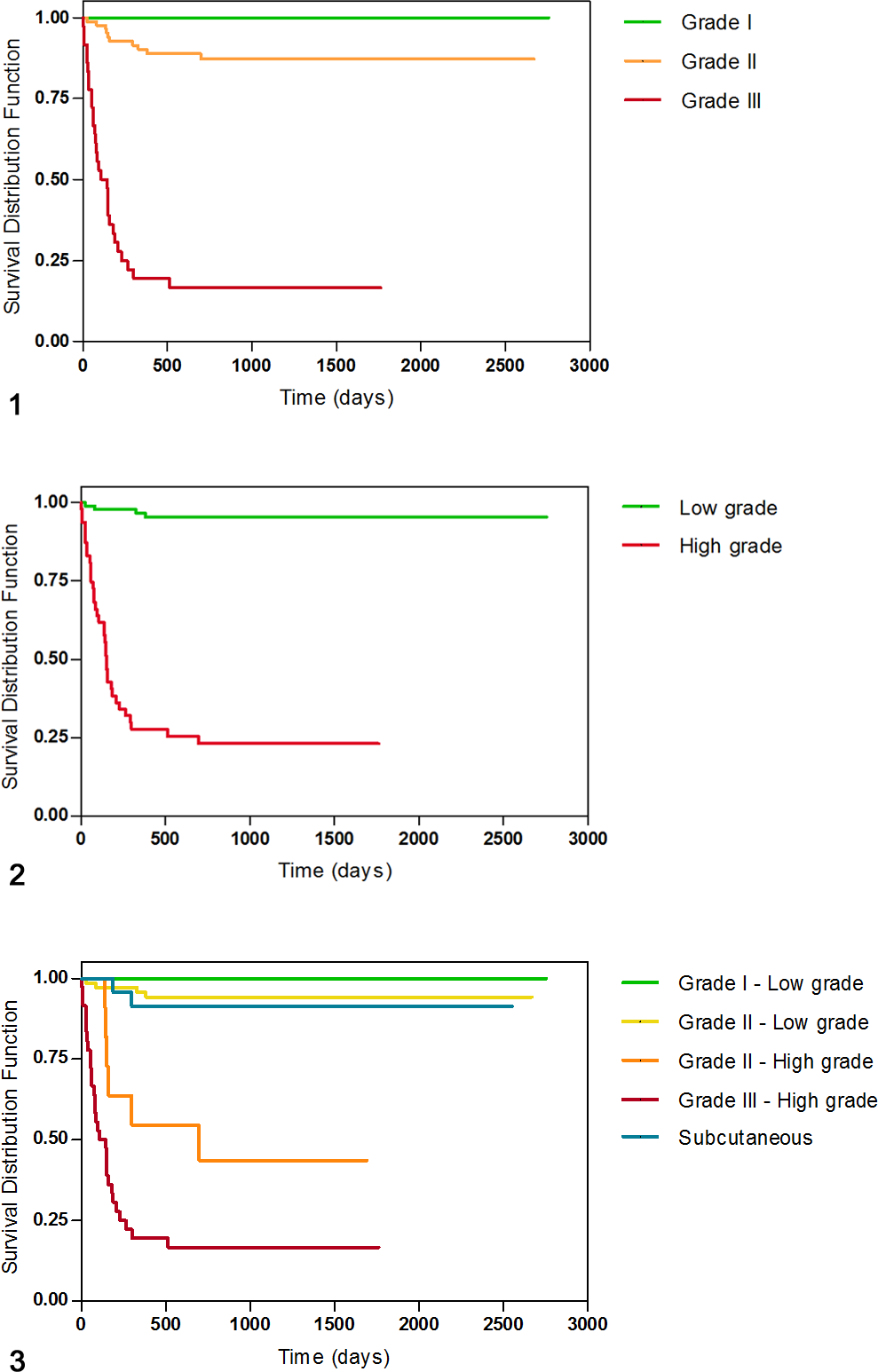
By KS, 89 (65%) MCTs were identified as LG and 48 (35%) as HG. The mortality rate was 4.5% (4 dogs) in LG MCTs and 75% (36 dogs) in HG MCTs. The survival probability at 12 months was 95% for LG MCTs and 24% for HG MCTs. The median survival time was not reached for LG MCTs, while it was 150 days (95% CI, 134–166 days) for HG MCTs. The survival curves of LG and HG MCTs were significantly different (log-rank test, P < .001) (Fig. 2).
On multivariable survival analysis, the Kiupel grading systems and the Patnaik grade 3 versus grade 2 and 1 grading were significantly associated with outcome. The hazard of death for HG tumors was 12.2 times that of LG tumors (95% CI, 3.3–45.5; P < .001). However, PS was still statistically significant in the model (hazard ratio, 2.8; 95% CI, 1.2–6.7; P = .021), showing that it contained additional information about survival. This is in contrast to previously published data. 4
When the 2 grading systems are compared, all grade I MCTs were classified as LG, and all grade III MCTs were classified as HG. Among grade II tumors, 71 (85.6%) were classified as LG and 12 (14.4%) as HG.
The survival probability at 12 months was 94% for grade II LG MCTs and 46% for grade II HG MCTs. The estimated median survival time for grade II HG MCTs was 698 days (95% CI, 33–1515). The survival curves were significantly different between grade II LG and grade II HG tumors (log-rank test, P < .001) and between grade II HG and grade III HG tumors (log-rank test, P = .027) (Fig. 3).
Twenty-five (15.4%) MCTs were classified as subcutaneous. The mortality rate in those cases was 8% (2 dogs). The survival probability at 12 months was 91%. The survival curves were significantly different between subcutaneous MCTs and grade II HG MCTs (log-rank test, P = .001) but not between subcutaneous MCTs and grade II LG MCTs or grade I LG MCTs (Fig. 3).
Discussion
Despite widespread use, the Patnaik grading system is subject to debate. Assignment of histologic grade in canine MCTs varies greatly among pathologists, and these dissimilarities mostly depend on the subjective parameters of PS, such as invasiveness, cellularity, and cellular morphology. Thus, new proposals for more accurate prediction of the biological behavior of canine cutaneous and subcutaneous MCTs have been formulated. 4,12 Since the proposal of the 2-tier grading system, several studies have confirmed its prognostic relevance and its better interobserver concordance. 3,10,13 KS also has the advantage to eliminate the intermediate grade of PS, which is associated with a fairly unacceptable prognostic uncertainty. For these reasons, many histopathology laboratories have started to routinely apply both systems, waiting for further validation studies to ultimately allow the choice of one of them.
To our knowledge, this is the largest retrospective study comparatively evaluating the 3- and 2-tier grading systems. Both systems proved to be of prognostic value to predict tumor mortality, although our data may be influenced by the low reproducibility of the PS. Furthermore, we studied only survival and did not evaluate the risk for MCT metastasis. Regarding the PS, dogs with grade III MCTs had significantly shortened survival than did those with either grade I or II tumors. However, there were no statistic differences between grade I and II MCTs. Several authors report similar results, 5,10 while other surveys indicate significant differences among the survival curves of each Patnaik grade. 8,13 In the present study, more than 60% of cases fell under the intermediate grade, which is comparable with previous studies. Additionally, the proportion of dogs with grade II MCTs that died of disease (12%) was considerably lower than that reported in the original work by Patnaik et al (56%) and closer to those reported by Kiupel et al (27%) and Vascellari et al (23%). These differences suggest a tendency to overuse the midrange category, even if they could be affected by variations in the case population. 4,8,13 KS had high prognostic value in predicting tumor mortality, which is consistent with other studies, 10,13 and the results of Cox regression analysis demonstrated that it was an independent prognostic factor. By the combined use of both grading systems, KS allowed to properly differentiate those grade II MCTs with good long-term prognosis from the small subset (14%) of tumors behaving more aggressively. The biologic behavior of LG MCTs was almost invariably benign, regardless they were classified as grade I or II in PS. However, among HG MCTs a distinction emerged between grade II and grade III, with significantly longer survival times in grade II. Interestingly, the Kaplan-Meier survival curve of grade II HG MCTs was similar to the curve of grade II MCTs from Patnaik’s original study. This may reflect again the tendency to overrate grade I MCTs in this and other studies.
In conclusion, our results indicated a superior prognostic value of the 2-tier histologic grading for canine cutaneous MCTs, and this system seems able to provide oncologists with the most important information—namely, which dogs require additional therapy to address systemic disease. While dividing HG MCTs according to the PS provided some additional information about survival, the practical use of such an approach to interobserver variation seems rather questionable. Our data also confirm that histologic grading alone is unable to predict the biological behavior of each MCT case and should be supplemented with molecular methods, such as assessment of the proliferation activity or the c-Kit mutation status, for more accurate prognostication. 13 –15
Finally, we confirm the generally indolent clinical course of subcutaneous MCTs, although there is a subset of them behaving more aggressively, so the evaluation of specific histologic criteria 12 and/or molecular markers is also indicated.
Footnotes
This study was presented at the annual congress of the European Society of Veterinary Pathology, September 2013, London, UK.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
