Abstract
In dogs, primary bone tumors can be difficult to distinguish with histopathology. Of those tumors, osteosarcoma (OSA) is the most common and aggressive. In this study, 4 immunohistochemistry markers—alkaline phosphatase (ALP), osteonectin (ON), osteopontin (OP), and runx2—were evaluated for their ability to distinguish OSA from other primary bone tumors. The 42 formalin-fixed, paraffin-embedded, primary canine bone tumors included 15 OSAs, 8 chondrosarcomas, 11 fibrosarcomas, and 8 histiocytic sarcomas. All 4 antibodies were highly sensitive for detection of osteosarcoma. ALP was the most sensitive at 100% and runx2 the most specific at 78%. Running ALP and runx2 in series resulted in a sensitivity of 87% and a specificity of 85%. This combination of immunomarkers resulted in a diagnostic panel for distinguishing osteosarcoma from other primary bone tumors.
Primary bone tumors in dogs often have similar clinical and radiographic appearances. Osteosarcoma (OSA), chondrosarcoma (CSA), fibrosarcoma (FSA), and histiocytic sarcoma (HS) represent 90% of all primary bone tumors in dogs, with OSA being the most common.11,17 Definitive diagnosis of a bone tumor requires invasive techniques, including fine needle aspiration or biopsy, to rule out non-neoplastic lesions, such as osteomyelitis, trauma, and dysplastic lesions. 13
Differentiation of OSA from other primary bone tumors is clinically important due to its high risk of metastasis and aggressive biologic behavior. Cytology is a practical and relatively non-invasive diagnostic technique; however, cytologic features of primary bone tumors can overlap, which hinders definitive diagnosis. The addition of staining for alkaline phosphatase (ALP) activity can increase the sensitivity of diagnosis of OSA to 100% and specificity to 87%. 5 Complementing routine cytology, histopathologic diagnosis can be made without additional methods if there is visible production of osteoid by the neoplastic cells. Unfortunately, sections of tissue submitted for biopsy are often small, and osteoid may be minimal and difficult to discern from collagenous fibrous connective tissue or fibrin. 17 Further complicating diagnosis, OSA can exhibit histologic diversity, producing fibrous connective tissue and cartilaginous matrix in addition to osteoid. Difficulty in distinguishing primary bone tumors is underscored by 1 study where histopathology was found to be 72% accurate with 13 of 18 malignant tumors correctly diagnosed. Of the 5 tumors originally misdiagnosed, 3 tumors were initially misdiagnosed as less aggressive tumors, although they were ultimately diagnosed as OSA. 13
Different markers have been evaluated via immunohistochemistry to assist in the diagnosis of OSA with samples derived from small biopsies. Osteonectin (ON) and osteopontin (OP) are extracellular matrix proteins produced by well-differentiated osteoblasts, 17 and both have been shown to be highly sensitive markers for diagnosis of OSA. 17 Additionally, ALP activity has been identified as a differentiating marker for OSA 5 and in humans, ALP is considered an osteoblast-specific marker. 3 Recent studies have shown expression of ALP in OSA cell lines 18 but no studies evaluating ALP expression in other primary bone tumors have been evaluated.
Runx2 is a member of the runx family of transcription factors, which are responsible for many cellular processes including cell proliferation, differentiation, and cell lineage specification. 9 There are 3 members of this family which include runx1, runx2, and runx3. Runx1 and runx3 are critical for hematopoiesis. Runx2 is important in skeletal development, is required for osteogenesis, and functions to activate genes associated with osteoblast differentiation.9,16 Abnormal expression of all 3 runx genes have been associated with cancer development, including proliferation and epithelial-to-mesenchymal transition. 9 Expression of runx2 appears increased in most human OSA cell lines,2,3 and comparatively, runx2 and ALP have also been found to be consistently expressed in canine OSA. 3
Because of the overlap in the morphologic appearance of primary bone tumors, there is justification to evaluate the differential expression of these osteoblast-associated markers by primary bone tumors, as well as determine the marker sensitivity and the specificity. The aim of this study was to characterize the expression patterns of 4 proteins (ALP, runx2, ON, and OP) through immunohistochemistry and determine if the expression patterns will assist the pathologist in differentiating osteosarcoma from other bone tumors.
Materials and Methods
Selection of Cases
The University of Illinois Diagnostic Laboratory database was searched from 2006 to 2017 for cases of primary bone tumors with a histologic diagnosis of OSA, HS, FSA, and CSA. A criterion for case selection for OSA included a sufficient sample size that allowed for unequivocal identification of a malignant spindle cell population (determined by presence of nuclear atypia and mitoses) producing osteoid. 15 Sections with minimal to no necrosis were selected for immunohistochemistry. OSA cases selected may have had other matrix present in the tissue section, but osteoid was the predominant matrix and the osteosarcomas were characterized as osteoblastic. Cases of HS, FSA, and CSA were of a sample size that allowed for as confident determination as possible in this retrospective study, that there was no evidence of osteoid production within the tumor. All efforts were made to limit cases to amputations/excisional samples, but this was not always possible to confirm given the retrospective nature of the study and exact tumor dimensions for each case were not available. Therefore, a minimum of 4 sections (approximately 2 cm x 2 cm each) from the tumor were required for case inclusion to minimize the possibility that osteoid was present. Histologic criteria for diagnosis of HS, FSA, and CSA were as previously described. 15 Briefly, HSs consisted of a malignant spindle to round cell population and exhibited CD18 immunoreactivity. FSAs were well differentiated or moderately differentiated, with presence of malignant spindle cells and collagen production. CSAs were composed of malignant spindle cells with production of chondroid matrix and no osteoid production by neoplastic cells.
Histopathology
All tissues were decalcified before processing for histopathology. Bone was placed into a formic acid-based decalcification solution (DeltaForm) and then tested daily to determine sufficient demineralization per the manufacturer’s recommendation. Briefly, 5 ml of the decalcifying reagent was removed from the bottom of the specimen container and added to a small beaker. To that, 5 ml of 5% ammonium oxylate and 5 ml of 5% ammonium hydroxide were added. The solution was mixed. If a precipitate formed, the sample was not completely decalcified and would remain in the decalcifying solution. Samples remained in decalcification solution a variable amount of time; however, no sample required decalcification for more than 8 days. Once the sample was decalcified, samples were processed overnight, embedded in paraffin, sectioned at 5 microns, and stained with hematoxylin and eosin. One pathologist (E.D.) confirmed the original diagnosis.
Western blot analysis
Osteosarcoma cell lines from human OSA (LM7 and SaOS2) and canine OSA (HMPOS and KOO3) were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin and were cultivated at 37°C with 5% CO2 humidified air. Cell pellets were lysed on ice in Mammalian Protein Extraction Reagent (MPER) (Thermofisher) containing Halt Protease Inhibitor Cocktail (Thermofisher). Samples were also briefly sonicated on ice for 30 seconds before protein of the resultant lysates were quantified using the Pierce BCA Protein Assay kit (Thermofisher). In all, 30 μg samples were then denatured (95°C for 5 minutes), separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) (4%–20%), and transferred to a polyvinylidene difluoride (PVDF) membrane. Membranes were blocked in 5% non-fat milk and dissolved in tris-buffered saline-tween 20 (TBST) for 1 hour at room temperature prior to primary antibody incubation. ALP (Abcam) (1:500) and RUNX2 (Santa Cruz) (1:500) antibodies were incubated overnight at 4°C, followed by secondary labeling using species specific HRP-linked antibodies (Cell Signaling) for 1 hour at room temperature. Membranes were then developed using SuperSignal West Femto Maximum Sensitivity Substrate (Thermofisher) and ChemiDoc XRS+ molecular imager system. Membranes were re-probed for β-actin (Abcam) (1:10,000) for use as loading control.
Immunohistochemistry (IHC)
Immunolabeling was performed on each sample using 4 antibodies, mouse monoclonal anti-alkaline phosphatase (Abcam), rabbit polyclonal anti-osteonectin (Abcam), rabbit polyclonal anti-osteopontin (Abcam), and mouse monoclonal anti-runx2 (Santa Cruz). Anti-runx2 was prepared as previously described in the dog. 8 Sections (4-μm) of formalin-fixed, paraffin-embedded tissues were deparaffinized and rehydrated through graded alcohol baths, endogenous peroxidase was quenched with 3% hydrogen peroxide in methanol at room temperature. Target antigens were retrieved by immersion of the tissue in a citrate buffer (pH 6.0) and microwaving. Antibodies were optimized and dilutions for anti-ALP, anti-osteonectin, anti-osteopontin and anti-runx2 were 1:500, 1:500, 1:300, and 1:100, respectively. The primary antibodies were incubated with the tissue slides for 30 minutes, at room temperature and immunoreactivity detected with a non-avidin biotin immunoperoxidase diaminobenzidine detection method. Canine renal tubular epithelium was used as a positive control for anti-ALP and anti-osteopontin, canine cartilage for anti-osteonectin, and paraffin embedded HMPOS cells (a canine osteosarcoma cell line) for anti-runx2.9,18 Negative control tissues for ALP and runx2 were stromal cells from the canine ovary 1 and canine kidney, respectively. An additional negative control included the primary antibody replaced by an isotype-matched non-relevant antibody.
IHC Scoring
Immunoreactivity of cytoplasmic labeling was determined on all slides, using a semiquantitative scale evaluating the labeling intensity and percent positive cells, by 1 pathologist (AB) without knowledge of the previous diagnosis. 12 Briefly, a score of 0 indicated no labeling in any cells, 1 indicated weak labeling in <25% of cells, 2 indicated strong labeling in <25% of the cells or weak labeling in >25 of cells, and 3 indicated strong labeling in >25% of cells; if membranous labeling was noted, the score was increased by 1 giving a possible score of 0 to 4. A score of 0 or 1 was considered negative; a score of ≥2was considered positive. For runx2, nuclear labeling was scored on a 0 to 4 scale evaluating the percentage of positive nuclei. 12 Briefly, a score of 0 indicated no nuclear labeling, 1 indicated minimal labeling 1-15% of nuclei, 2 indicated 16% to 40% positive nuclei, 3 indicated 41-75% nuclei, and 4 indicated a strong positive with positive labeling of 76% to 100% of nuclei. A score of 0 or 1 was considered negative; a score of ≥2 was considered positive.
Evaluation of Effects of Decalcification
An additional 4 cases of OSA that had a soft tissue component to the tumor were evaluated with and without decalcification and were labeled for ALP and runx2. These tumors were scored as described above.
Statistical Analysis
For statistical comparisons, all tumor samples were dichotomously categorized as OSA and non-OSA. Within these 2 categories, contingency tables were created with each immunolabeling category to evaluate the likelihood of the label correctly identifying the OSA samples. Samples were considered positive if scored 2 or greater on a 0 to 4 scale. Within each of the 4 categories of immunolabeling, sensitivity, and specificity were determined. Specificity was defined as the proportion of histologically diagnosed OSAs with positive immunolabeling; specificity was defined as the proportion of histologically diagnosed non-OSA tumors with negative immunolabeling. Tests of significance with these contingency tables used either Chi square or Fisher exact tests depending on sample number. Additionally, sensitivity and specificity for running these tests in parallel or in series were performed. For measures of significance, alpha was set at 0.05.
Results
Western blot analysis for ALP and runx2 revealed bands at the expected molecular weight (70 and 55 kDa) and were comparable between human and canine cell lines (Supplemental Fig. S1).
Seventy-two hematoxylin and eosin-stained histological samples obtained from necropsy or surgical biopsy were reviewed. Thirty samples were omitted due to lack of sufficient tissue for confident definitive diagnosis or presence of abundant necrosis that would hinder ability to accurately score immunoreactivity. Forty-two formalin-fixed, paraffin embedded, primary canine bone tumors were selected and included 15 OSAs, 8 CSAs, 11 FSAs, and 8 HSs.
All tumor samples were evaluated using IHC for ALP, runx2, osteonectin, and osteopontin (Supplemental Figs. S6–S15, Supplemental Table 1). Positive and negative control tissues labeled as expected (Supplemental Figs. S2–S5). Representative labeling of tumors is shown in Figs. 1 –4. For OSA, all 15 had positive labeling for ALP and 13/15 had strong membranous labeling (Fig. 1). For runx2, 13/15 exhibited prominent nuclear labeling and 2 were negative. There was positive labeling for OP and ON in 12/15 cases with no evidence of membranous localization.
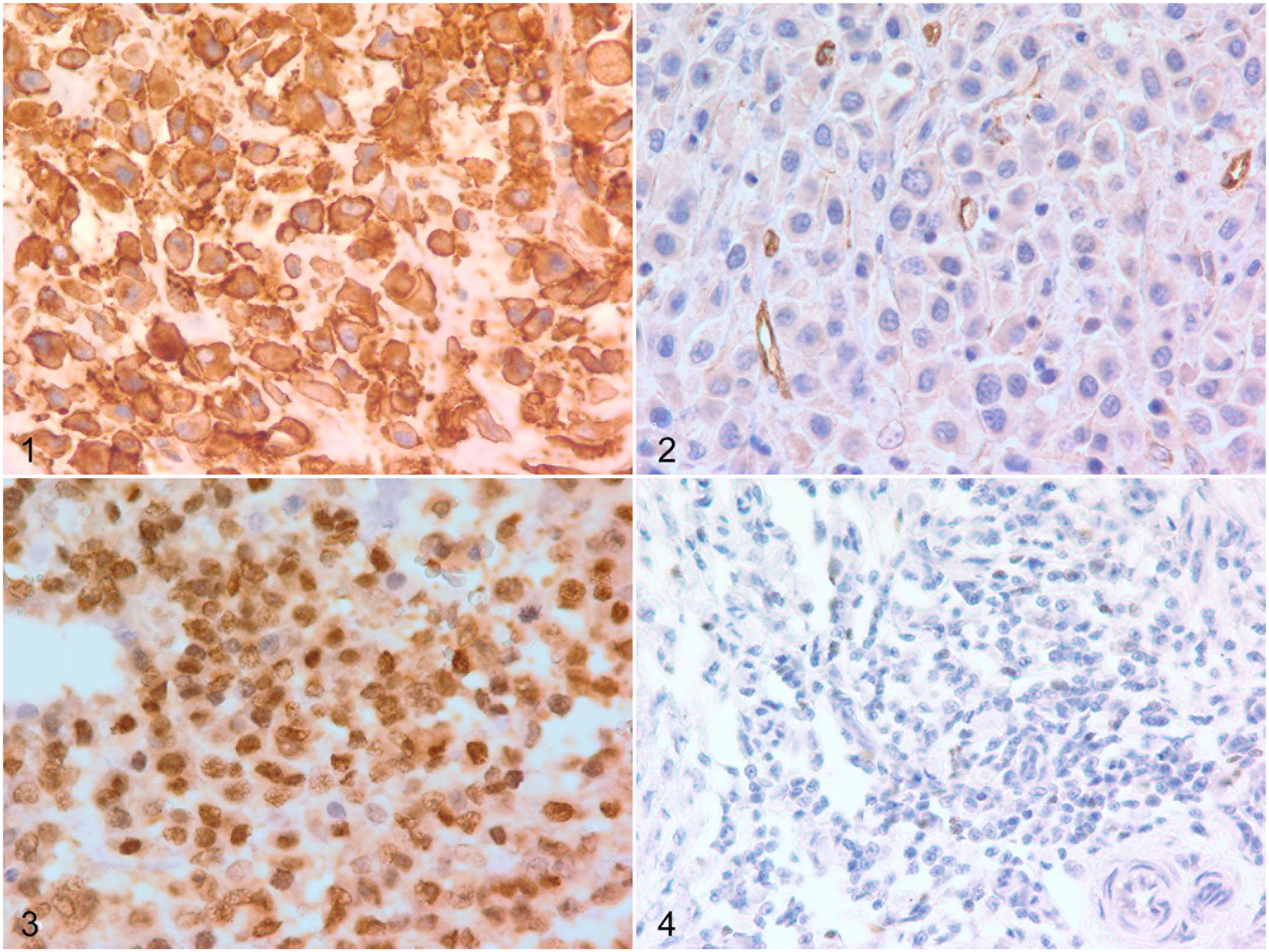
Osteosarcoma, humerus, dog. There is membranous immunolabeling for alkaline phosphatase. Figure 2. Histiocytic sarcoma, humerus, dog. There is a lack of cytoplasmic immunolabeling for alkaline phosphatase. Benign endothelial cells in vessels are strongly labeled. Figure 3. Osteosarcoma, femur, dog. There is strong nuclear immunolabeling for runx2 (IHC score of 4). Figure 4. Histiocytic sarcoma, humerus, dog. There is minimal nuclear immunolabeling for runx2 (IHC score of 1). Histiocytic sarcoma, humerus, dog. There is a lack of cytoplasmic immunolabeling for alkaline phosphatase. Benign endothelial cells in this image are staining strongly. Osteosarcoma, femur, dog. There is strong nuclear immunolabeling for runx2 (IHC score of 4). Histiocytic sarcoma, humerus, dog. There is minimal nuclear immunolabeling for runx2 (IHC score of 1).
Immunoreactivity of sarcomas other than OSA had variable results. Of the CSAs, 4/8 were positive for ALP (only one exhibited membranous labeling), 1/8 was positive for runx2, and 5/8 were positive for both ON and OP. For the FSAs, 8/11 expressed ALP (only 1/11 exhibited membranous labeling), 1/11 was positive for runx2, 9/11 FSAs were positive for ON, and 5/11 were positive for OP. For the HSs, 5/8 were positive for ALP (2 exhibited membranous labeling), 4/8 were positive for runx2, and 6/8 were positive for both ON and OP.
All 4 antibodies were highly sensitive for detection of OSA, with ALP being the most sensitive at 100% followed by runx2 at 87%, OP at 87%, and ON at 80%. Samples diagnosed as OSA were significantly more likely to have positive labeling for ALP and runx2. None of the antibodies had a high specificity; however, runx2 was the most useful at distinguishing OSA from other bone tumors with a specificity of 78%, followed by ON at 37%, ALP at 30%, and OP at 26%. If ALP and runx2 were run in series (i.e. ALP run first, and with a positive result, runx2 was evaluated), this resulted in a sensitivity of 87% and specificity of 85%. This was a better overall result than running these 2 tests in parallel (sensitivity 100% and specificity 23%). Both tests met standards for independent tests based on kappa statistic.
Four additional osteoblastic OSAs that had a soft tissue component were collected to evaluate the effects of the decalcification process on ALP and runx2 labeling. All 4 cases were scored as 4 for ALP in the decalcified and nondecalcified tissues. Similarly, for runx2, all 4 cases were scored as 3 for both the decalcified and nondecalcified tissues. The decalcification process did not produce any appreciable effects on the intensity of the immunohistochemical labeling (Supplemental Figs. S16–S19).
Discussion
In the absence of osteoid, OSA is difficult to distinguish histopathologically from other common primary bone tumors. This scenario may occur due to small sample size, which is a common problem encountered with bone biopsies in veterinary medicine. 13 Cytologic diagnosis of OSA has been shown to be more useful with the addition of staining for alkaline phosphatase activity; however, due to the epimembranous location of the ALP enzyme, formalin-fixed tissues are not useful for detection of ALP activity. 5 ALP staining in cytology is a cytochemical stain, detecting the enzyme activity rather than the enzyme itself. IHC for ALP labels the actual protein rather than the active enzyme. A single, reliable, sensitive, and specific marker has yet to be identified to differentiate osteoblastic cells in formalin-fixed tissues. Given the utility of ALP staining in cytologic applications, the authors proposed that ALP IHC may prove similarly useful in the histopathologic differentiation of OSA from other primary bone tumors. As shown in this study, the specificity of ALP immunoreactivity is limited, thus necessitating the use of a panel of markers for diagnosis. The epimembranous location of the ALP enzyme in cells may explain the prominent membranous labeling observed in the current investigation. OSA had more prominent membranous labeling than the other tumor types, and this identified subcellular localization for ALP might assist in confirming the diagnosis.
A variety of IHC markers have previously been evaluated to help differentiate OSA from other primary bone neoplasms, including OP, ON, osteocalcin, and runx2, among others.2,4,14,17 Many of these proteins are synthesized by multiple cell types including osteoblasts, and are important components of bone matrix.2,17 In a study of bone tumors in humans, the sensitivity of ON in the diagnosis of OSA was high (90%), but the specificity was only moderate (54%). 7 In previous studies in dogs, the sensitivity of ON was also very high at 100%, but the specificity was quite low, 17 similar to the current study. Sensitivity and specificity data for OP is lacking; however, claims of its limited value have been published.6,7 The current study evaluated ON and OP IHC in all tumor samples for comparison with the results of the novel application of ALP and runx2 IHC in the diagnosis of OSA. Both ON and OP were highly sensitive (>80%) but poorly specific at 37% and 26% respectively, limiting their singular value in the diagnosis of OSA or their addition in a panel of markers.
ALP was the most sensitive marker at 100% and runx2 had the highest specificity at 78%. Though previous studies evaluating ALP and runx2 in the recognition of osteoblasts have been published, 18 this study is the first to evaluate the specificity of these 2 markers to distinguish OSA from other primary bone tumors. Due to the insufficient specificity of ALP, it is of limited use as a stand-alone diagnostic tool to diagnose OSA, though combination of ALP with another marker may improve the diagnostic utility. In this study, the greatest benefit was found by running ALP and runx2 in series. Recommended use of these 2 antibodies would be as follows. If a sample is negative for ALP expression, the tumor is likely not an OSA, and the addition of runx2 would not be necessary; however, if the tumor expressed ALP, it would be recommended to then perform runx2 labeling for additional characterization. By using these 2 antibodies in series, specificity increased significantly. When the samples were run in parallel, the result was a sensitivity of 100% and a specificity of 23%, negating the improvement of combination.
In the current study, the small sample size for each tumor type was a limitation. OSA is the most common primary bone tumor in dogs, and it was difficult to identify clinical examples of the other primary bone tumors. Care was taken to only include samples that had a clear diagnosis. These factors limited the sample size. Additionally, these samples were large enough to make a diagnosis without IHC. It is important to follow this study with samples of small biopsy specimens to further test the diagnostic utility of these antibodies.
Another limitation to the study is performing IHC on decalcified tissues. Decalcification of tissue specimens is a necessary step in the histopathologic evaluation of bone neoplasia. However, the chemicals used in the decalcification process may irreversibly alter tissue proteins and thereby impair the detection of specific tumor cell antigens. Currently, there is no standardized process for tissue decalcification in veterinary diagnostic medicine. Hydrochloric acid decalcification is often preferred by pathologists since it reduces the processing time and allows for more rapid diagnosis. However, decalcification methods using hydrochloric acid and long-term formic acid solutions can induce false-negative IHC results, particularly for antibodies that target nuclear antigens. 10 In the current study, the tissue specimens were subjected to formic acid decalcification. The duration of tissue exposure to formic acid was variable and depended on the density of the bone in each specimen. In a previous study, mean IHC scores of tissues decalcified by formic acid for less than 8 days were similar to non-decalcified tissues. 10 In the current study, the samples were not exposed to long term (>8 days) decalcification and therefore should not have had major effects on the labeling process. The 4 OSA samples that were evaluated with and without decalcification had comparable results suggesting that <8 days of formic acid decalcification does not seem to negatively impact labeling for ALP and runx2.
The purpose of this study was to examine the utility of ALP and runx2 to distinguish OSA from other bone tumors where the diagnosis was clear. Future studies, using small tissue samples, will help to determine if use of these antibodies can be beneficial when the diagnosis of OSA is in question.
In summary, this study aimed to determine whether ALP and runx2 immunohistochemical detection would be useful in the diagnosis of OSA with IHC. While ALP had excellent sensitivity, its poor specificity limited its utility as a stand-alone diagnostic marker. The addition of runx2, used in series, increased the specificity to 85%. A combination of these 2 antibodies may be a useful tool for diagnosis of OSA.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221083035 – Supplemental material for The use of alkaline phosphatase and runx2 to distinguish osteosarcoma from other common malignant primary bone tumors in dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858221083035 for The use of alkaline phosphatase and runx2 to distinguish osteosarcoma from other common malignant primary bone tumors in dogs by Anne Barger, Kate Baker, Elizabeth Driskell, Will Sander, Patrick Roady, Matthew Berry, Amy Schnelle and Timothy M. Fan in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Rottweiler Health Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
