Abstract
Aspiration of lytic bone lesions is an excellent diagnostic test in the initial evaluation of primary bone neoplasia. However, cytologically, it can be difficult to differentiate osteosarcoma (OSA) from other bone neoplasms, including fibrosarcoma, chondrosarcoma, synovial cell sarcoma, and plasma cell myeloma. The purpose of this study is to determine the sensitivity and specificity of alkaline phosphatase (ALP) staining to differentiate OSA from other tumors that express vimentin by immunocytochemistry or immunohistochemistry. ALP is a hydrolytic enzyme present in multiple tissues including liver, kidney, intestine, placenta, and bone. Hypothetically, neoplasms actively producing bone should be specifically positive for ALP staining. Unstained, cytologic specimens were incubated for 8-10 minutes with nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate toluidine salt-phosphatase substrate. A positive reaction stains the membrane of the cells gray to black. Samples were counterstained with a Romanowsky's stain to determine whether the sample was of representative cellularity. A total of 61 vimentin-positive neoplasms have been evaluated and confirmed histopathologically. Tumors that expressed vimentin and were positive for ALP included 33 OSAs, one multilobular tumor of bone, one amelanotic melanoma, and one chondrosarcoma. Tumors that expressed vimentin and were negative for ALP included chondrosarcomas (three of four), multiple fibrosarcomas, and multiple synovial cell sarcomas. The sensitivity is 100%, and the specificity is 89%. In conclusion, ALP appears to be a highly sensitive and fairly specific marker in the diagnosis of OSA.
Osteosarcoma (OSA) is the most common primary bone tumor of dogs, accounting for 85% of reported skeletal malignancies. 4, 14 Biologically, the tumor is locally aggressive, with a high metastatic rate. 16 The majority of OSAs are appendicular, 75% of which originate from the distal radius or proximal humerus. 7 Histopathologically, OSA is described as a malignant spindle cell tumor characterized by the production of an osteoid matrix by tumor cells. 16 The gold standard for diagnosis is considered biopsy with histopathologic evaluation. 7 Bone biopsy is an invasive procedure, often with a delayed diagnosis because of the decalcification process. In addition, bone biopsies can have complications, such as an increased risk of pathologic fracture at the biopsy site. Fine needle aspiration of lytic bone lesions is becoming more common in human medicine. 1, 9, 18 In one study, fine needle aspiration and cytology of bone tumors revealed a sensitivity of 86% and a specificity of 94.7%, with histopathology as the gold standard. 1 In veterinary medicine, aspiration of lytic lesions is gradually being used more frequently. In a recent study by Cohen et al., the overall sensitivity and specificity of cytology was evaluated. Although this study was not limited to bone, nine bone aspirations were evaluated, with partial agreement with histopathology in all nine cases. 5 Cytology offers the advantage of rapid turnaround time for diagnosis and limited complications because of the minimally invasive procedure required to obtain a sample. Limitations of cytology may include low cellularity because of the poorly exfoliative nature of the tissue, difficulty in differentiating reactive bone from neoplastic bone and, finally, difficulty in distinguishing the different tumors of bone. 8 Cytologically, OSA can appear similar to chondrosarcoma, synovial cell sarcoma, and fibrosarcoma. 11 Each of these tumors can have comparable cytologic features including eccentrically placed nuclei and varying amounts of an extracellular eosinophilic matrix. The diagnostic power of cytology would likely be improved with cytologic stains specific for particular cell types. Primary bone sarcomas comprise a heterogeneous group of cancers with differing biologic behavior. OSA is the most common, malignant primary bone tumor and possessing a high-micrometastatic rate (≈ 90%) at diagnosis. In general, other primary bone sarcomas such as fibrosarcoma and chondrosarcoma are less metastatic. Because OSA has a high metastatic rate, conventional treatment options include the combination of surgical resection and adjuvant chemotherapy. For less metastatic bone sarcomas, the requirement for systemic chemotherapy may not be necessary. The histologic discrimination among primary bone sarcomas allows for the most rational implementation of local and systemic therapeutic modalities. Therefore, a diagnosis of sarcoma is not enough, and the ability to differentiate OSA is important.
Alkaline phosphatase (ALP) is a membrane-bound enzyme that is synthesized by cells in a multitude of tissues. 2, 3 It is found in most organs of all mammalian species. Two genes code for canine ALP; the intestinal gene and the tissue nonspecific gene. 13 The intestinal gene produces the intestinal and corticosteroid ALP, whereas the tissue nonspecific gene produces the liver and bone isoforms. 3, 13 ALP is primarily found in epithelial tissue, liver, kidney, and placenta. Bone is the only connective tissue shown to produce ALP in dogs. 3, 13
Nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate toluidine salt (NBT/BCIP) is an ALP substrate, which undergoes an oxidation/reduction reaction after dephosphorylation by the enzyme, resulting in a color change of the insoluble product at the site of the reaction. This substrate can be used to detect ALP activity. The purpose of this study is to determine whether the NBT/BCIP substrate can differentiate OSA from other mesenchymal neoplasms both in bone and in soft tissue. We hypothesized that OSA would be the only vimentin-positive mesenchymal neoplasm positive for ALP activity.
Materials and Methods
This was a prospective study, with sample accrual from November 2002 to February 2004. Cytologic specimens from dogs with a diagnosis of mesenchymal neoplasia either in bone or soft tissue, which had at least three unstained slides, were included in the study group; a total of 72 cytologies, with a diagnosis of mesenchymal neoplasia, were evaluated. Subsequent histopathologic confirmation of neoplasia was required for inclusion in the study.
Cytologic specimens evaluated included roll preparations from biopsies as well as fine needle aspirations. At least two slides of each sample were stained with Wright's Giemsa for the initial cytologic diagnosis. Unstained slides were stained with vimentin (Dako Corporation, Carpenteria, CA) or NBT/BCIP (Kirkegard and Perry Laboratories Inc, Gaithersburg, MD). Monoclonal mouse anti-human vimentin was used with a Dako automated stainer to confirm the mesenchymal origin of the cells. For the ALP staining, slides were incubated with 300–400 μl (enough to coat the slide) NBT/BCIP solution for 8–10 minutes at room temperature. Slides were rinsed with water, blotted dry, and examined microscopically. Positive staining was indicated by distinct grayish black to brown staining of the cell surface. All samples were counterstained with Diff-Quik (Dade Behring Inc, Newark, DE). Only one dip in each solution was performed for the NBT/BCIP staining to still remain visible. Negative samples were counterstained to evaluate the cellularity and adequacy of the sample. Positive slides were counterstained to confirm that it was the neoplastic cells reacting with the ALP stain. Imprints of canine kidney were used as a positive control. Background red and white blood cells, within the sample itself, were used as an internal negative control. Normal canine neutrophils have been shown to be negative for ALP. 12 Samples were considered positive if > 75% of the neoplastic cells had the typical color change in the cytoplasm. Samples were considered negative, if none of the neoplastic cells reacted with the NBT/BCIP. All cytologies were reviewed by one clinical pathologist (AB), and all tissue biopsies were reviewed by one anatomic pathologist (KB).
Sensitivity and specificity were calculated using histopathologic diagnosis as the gold standard, to evaluate the diagnostic use of this test. All OSAs which were ALP positive were considered true positives, whereas other ALP-positive tumors were considered false positives. Non-OSA mesenchymal tumors staining negative for ALP were considered true negatives, whereas OSAs staining negative for ALP were considered false negatives.
Results
A total of 72 cytologic preparations were evaluated. Eleven cytologies were excluded. Nine cytologies were excluded because of lack of tissue biopsy and two cytologies because only reactive bone and no neoplastic cells were observed on histopathology. In addition, for the latter two specimens, the cytologic diagnosis was not definitive because of low cellularity. A total of 61 vimentin-positive neoplasms with histopathologic diagnoses were included and consisted of 33 OSAs, 4 chondrosarcomas, 4 synovial cell sarcomas, 2 primary bone fibrosarcomas, 9 soft tissue sarcomas, (including hemangiopericytoma, fibrosarcoma, histiocytic sarcoma, and hemangiosarcoma), 2 plasma cell tumors, (1 from bone), 2 lymphomas, 2 amelanotic melanomas, 2 mast cell tumors, and 1 multilobular tumor (MLT) of bone. The OSAs included two extraskeletal OSAs, one chondroblastic OSA, and one fibroblastic OSA. The remaining OSAs were of the osteoblastic type.
ALP staining was performed on all 61 of these neoplasms. Of those, all 33 OSAs including the 2 extraskeletal OSAs were positive for ALP activity (Fig. 1a–c). The MLT of bone, one amelanotic melanoma, and one chondrosarcoma also stained positive for ALP. All other tumors, including two primary bone fibrosarcomas, four synovial cell tumors, and three chondrosarcomas were ALP negative (Fig. 2a–c). These results are summarized in Table 1.
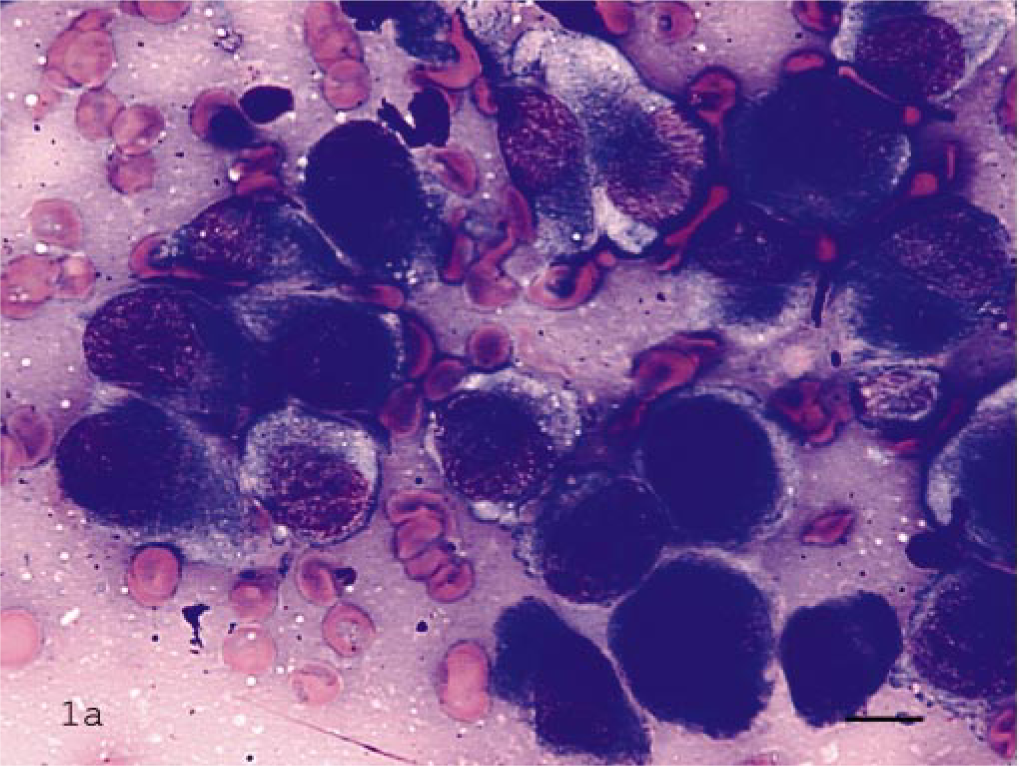
Bone aspirate of osteosarcoma; dog.
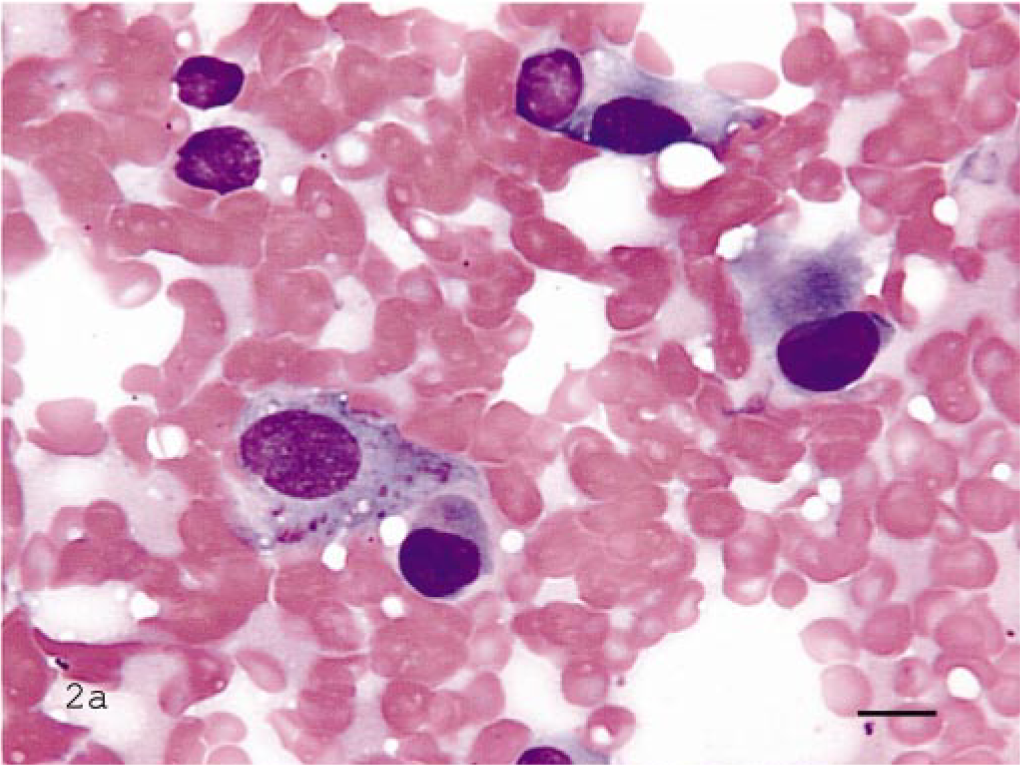
Bone aspirate of synovial cell sarcoma; dog.
Tumor type, quantity, and number positive for ALP staining.

On the basis of the criteria described in the Materials and Methods, the sensitivity and specificity were calculated with a sensitivity of 100% and a specificity of 89% (Table 2).
ALP staining of tumors characterized as OSA and non-OSA.

Discussion
The novel use of phosphatase substrate to differentiate OSA from other mesenchymal tumors is a useful tool. With a sensitivity of 100%, this is an excellent screening test. If the tumor is ALP negative, it is not an OSA. One of the limiting factors of the ALP substrate is its lack of specificity for neoplastic osteoblasts. The ALP enzyme is located on the membrane of all osteoblasts. 6, 17 Therefore, reactive bone will also be positive. The two instances of reactive bone eliminated from this study were positive for ALP. Therefore, close cytologic scrutinization of the sample for clear evidence of criteria of malignancy and lack of inflammatory cells is essential for appropriate interpretation of the ALP stain.
A few tumors other than OSA stained positive for ALP activity. These included one amelanotic melanoma, one MLT of bone, and one of four chondrosarcomas. The amelanotic melanoma was in the hard palate, resulting in significant bone proliferation and lysis. It is possible that there was reactive bone associated with this tumor. However, osseus metaplasia has been reported in melanoma. 10 It is possible that this tumor was producing bone. One other amelanotic melanoma, also associated with bone, stained negative for ALP.
The MLT of bone (also known as chondroma rodens, multilobular chondroma, and multilobular osteochondroma) also stained positively for ALP. These tumors are considered to be primary tumors of bone and can produce bone or cartilage. 16 Therefore, it is not surprising that this tumor would stain positive for ALP activity. Fortunately, these tumors have fairly distinct radiographic and gross appearance, often arising from the skull. 7 For this reason, clinically, these tumors are rarely confused with OSA.
One of four chondrosarcomas in this study stained positive for ALP activity. Chondrocytes are used as positive controls for detection of ALP activity in flow cytometric assays, so it is not unexpected that chondrosarcomas would be positive for ALP activity. 15 What is unusual is that the majority of chondrosarcomas evaluated in this study (75%) were in fact negative for ALP activity. This may indicate some dedifferentiation of the tumor cells themselves to a more immature phenotype, or that the excessive production of matrix by the tumor cells may interfere with the interaction of the substrate and the enzyme. Chick chondrocytes, used as controls for flow cytometry, are cultured and washed to eliminate the presence of the chondroid matrix. 15 It would be interesting to compare chondrosarcomas and chondromas of dogs with normal canine cartilage for the presence of the ALP gene and enzyme activity.
Staining for ALP activity is a rapid and simple test, which does not require any special equipment. When used carefully and in conjunction with additional testing such as cytology, detection of ALP activity appears to be a useful tool in the diagnosis of OSA. In the absence of cytologic criteria for malignancy, other differentials for positive staining should be considered, especially reactive bone. Only if the cytologic specimen is clearly a sarcoma (e.g., criteria for malignancy present in the absence of inflammation) can the ALP stain by appropriately interpreted because reactive bone will also be ALP positive. ALP staining should only be used as an auxiliary test. Generally, the cytologic and clinical features of the tumors can assist in diagnosis. Often, chondrosarcomas have excessive eosinophilic matrix, melanomas often will have pigment, (even small amounts in amelanotic melanoma), and MLTs of bone have a distinct clinical appearance. The combination of cytology, clinical appearance, location of the tumor, and radiographic changes are all important in the diagnosis of OSA. In addition, detection of ALP activity only improves the ability of the clinician and pathologist to appropriately diagnose primary tumors of bone.
Footnotes
Acknowledgements
We thank the personnel of the Cancer Care Clinic of the University of Illinois Veterinary Teaching Hospital for sample collection.
