Abstract
Lacrimal gland tumors (LGTs) in dogs and cats are rare neoplasms that can affect either the nictitans (NLG) or the main lacrimal gland (MLG). A consistent classification scheme for canine and feline LGTs is lacking; however, the importance of a classification scheme for LGTs has been emphasized in the human literature, and an update to the World Health Organization (WHO) classification has recently been published. The aim of this study was to investigate the occurrence of different subtypes of canine and feline LGTs in accordance with the human WHO classification system. Epithelial LGTs (n = 46 tumors; 38 dogs, 8 cats) were reviewed and immunophenotyping for p63, CK14, SMA, calponin, CKAE1/AE3, and CK19 was performed. Consistent with previous literature reports, lacrimal carcinomas outnumbered adenomas in dogs and cats. Based on the WHO classification of human LGTs, the most common subtypes identified in dogs were pleomorphic, ductal, adenoid cystic, and epithelial-myoepithelial carcinoma. In cats, a lower number of subtypes was observed, and adenocarcinoma “not otherwise specified” (NOS) was the most frequent diagnosis. An uncommon case of feline epithelial-myoepithelial carcinoma was also observed. The application of the human WHO-LGT classification scheme to canine and feline tumors increased the diversity of diagnoses and allowed for the identification of numerous subtypes. Further studies to identify possible correlations between pathological subtypes and prognosis are warranted.
Lacrimal gland tumors (LGTs) in dogs and cats are rare but well-known tumors that can affect either the nictitans (NLG) or main lacrimal gland (MLG). The MLG is a diamond-shaped structure in the periorbital tissues at the dorsolateral aspect of the globe. The MLG provides most of the tear secretion in both humans and carnivores. 30,32 The NLG is an ovoidal gland (about 10 mm) surrounding the ventral portion of the third eyelid cartilage. 26,29 No NLG or third eyelid are present in humans, in whom minor/accessory lacrimal glands (Wolfring and Krause glands) located near the superior conjunctival fornix contribute to tear secretion. 32 Accessory lacrimal glands are probably also present in dogs and cats, although specific anatomical studies are lacking. 21,30
The MLG and NLG have similar histological features and are affected by the same type of tumors. They are exocrine glands, organized in lobules separated by loose connective tissue that contains interlobular ducts, vessels, and nerve fibers. Each lobule is composed of acini and intralobular ducts lined by polygonal epithelial cells. Myoepithelial cells are distributed around the acini and intercalated ducts. MLG and NLG produce seromucous secretion in dogs and predominantly serous secretion in cats and humans. 15,28,30
Recently, a new classification has been published for human LGTs, 12 and the human literature emphasizes the importance of a detailed classification that distinguishes subtypes of LGTs for both study and prognostic purposes. 1,2,4,35 Benign tumors constitute about half of human epithelial LGT tumors and malignant epithelial tumors comprise the other half. Human LGTs are currently classified based mainly on neoplastic cell morphology, reflecting their differentiation to acinar, ductal, or myo-epithelium. Pleomorphic adenoma is the most common LGT in humans, representing 20% of all LGTs. It is composed of both epithelial and myoepithelial cells, along with areas of mesenchymal tissue (fibrous, bone, or cartilage). Pleomorphic adenoma is benign but can recur if incompletely excised, and a small number of cases give rise to carcinoma (named carcinoma ex-pleomorphic adenoma). The most common malignant epithelial LGT in humans is adenoid cystic carcinoma, comprising 20% to 30% of all malignant LGTs. It is composed of both epithelial and myoepithelial cells organized in various patterns of growth (cribriform, tubular, or solid). The histopathological features of adenoid cystic carcinoma have prognostic implications: the solid pattern is associated with a worse prognosis, and perineural invasion is associated with local recurrence, while bone invasion is associated with a fatal outcome. Additional rarer malignant epithelial LGTs include epithelial-myoepithelial and ductal carcinoma. 1,2,4,35 Epithelial-myoepithelial carcinoma is described as a low-grade malignant tumor histopathologically characterized by a “biphasic arrangement of inner luminal cuboidal cells and outer myoepithelial cells.” 12 High-grade transformation of epithelial-myoepithelial carcinoma with anaplasia, increased mitotic rate, and loss of distinctive biphasic pattern has been described. Ductal carcinoma of lacrimal glands, similarly to ductal carcinoma of salivary gland, is described in humans as an aggressive epithelial tumor characterized by comedonecrosis and a cribriform growth pattern. 22,24 Other rare LGTs are listed in Table 1.
World Health Organization classification of human lacrimal gland epithelial tumors. 12

A consistent morphological classification of LGTs in dogs and cats is lacking. A few reports have been published describing what have been called “atypical epithelial tumors” or “pleomorphic tumors” of the canine lacrimal glands. 5,16,23,34 These tumors were essentially characterized by a myoepithelial or basal phenotype. In the single extensive descriptive study, 6 which reviewed 145 canine and feline LGTs, no information was provided concerning specific histopathological features and/or immunophenotype.
The aim of the present study was therefore to describe the morphological features of canine and feline LGTs, applying the new human World Health Organization (WHO) classification system in order to identify a common and consistent nomenclature for different neoplastic subtypes. For the purposes of this study, the histopathological diagnosis was supported by an immunohistochemical investigation mainly directed toward the identification of myoepithelial cells.
Materials and Methods
A retrospective search (2005–2020) through the archive of the Department of Veterinary Medicine (DIMEVET) of the Università degli Studi di Milano was performed. The search criteria included primary epithelial neoplasms arising from the glandular tissue of the NLG or the MLG in dogs and cats, as diagnosed from formalin-fixed, hematoxylin and eosin (HE)-stained 5-µm paraffin sections of submitted tissues. Surface-neoplasms affecting the NLG (eg, conjunctival squamous cell carcinoma) were excluded as they were not considered primary glandular neoplasms. Moreover, cases that lacked available material for histopathological review (HE-stained slides or paraffin blocks) were excluded from this study.
The collected patient data included species, age, sex, breed, recurrence (confirmed or suspected), and metastatic spread (histologically confirmed or diagnosed with imaging techniques). Veterinary ophthalmologists were contacted via email for patient follow-up information. Recurrence was confirmed when neoplastic tissue was noted clinically at the surgical site and diagnosed via cytology or histology.
The tumor size was measured on the HE-stained sections as the 2 greatest tumor dimensions. Complete longitudinal sections of the specimens were available for all cases.
All HE-stained sections of the tumors were reviewed (CG, LN). Surgical margins were defined as complete (tissue margins free of neoplastic cells for a distance of greater than 1 mm), or incomplete (neoplastic cells extended to the cut tissue margin). When the tissue margins free of neoplastic cells were <1 mm, surgical margins were indicated as “close.” Tumors were classified based on the most recent WHO classification of human tumors of the lacrimal gland (Table 1). The main criterion for the definition of malignancy was the presence of an infiltrative (overt or micro-infiltrative) growth pattern. According to the current literature, 6,9,11,12 micro-infiltrative growth was recorded when a tumor was surrounded by an incomplete thin connective tissue capsule, multifocally infiltrated by neoplastic cells.
Mitotic counts were determined in all cases evaluating 10 contiguous high-power field (HPF; 40× objective, 10× ocular FN 22 mm). Counts were determined starting in the most proliferative area and avoiding areas with necrosis or severe inflammation.
Serial 4-μm-thick sections were obtained from paraffin blocks and stained with Alcian blue pH 2.5 (Diapath Staining Kit) to highlight the presence of mucus-secreting cells and ground substance.
For immunohistochemistry, serial 4-µm sections were mounted on positively charged glass slides (Superfrost Plus; Thermo Fisher Scientific), and dried at 60 °C for 20 minutes. The following antigens were analyzed: pan-cytokeratins (CKAE1/3), cytokeratin 19 (CK19), cytokeratin 14 (CK14), calponin, α-smooth muscle actin (SMA), and p63 (Table 2). Immunohistochemistry was manually performed applying the standard ABC method. Briefly, the sections were dewaxed in xylene and rehydrated through graded alcohols; the endogenous peroxidase activity was blocked with 0.3% H2O2 in methanol for 30 minutes. Blocking was obtained with a 1-hour incubation at RT using normal horse serum. Different antigen retrieval methods were applied (Table 2). The primary antibodies were incubated overnight at 4 °C. The immunohistochemical reaction was revealed through an avidin-biotin complex method using biotinylated horse anti-mouse antibody (Vector Laboratories) and an AEC (3-amino-9-ethylcarbazole) substrate-chromogen kit (Vector Laboratories). The sections were counterstained with Mayer’s hematoxylin. A portion of the normal lacrimal gland included in the specimens served as the positive internal control for all antibodies, and the negative control consisted of a serial section of each specimen, where primary antibodies were replaced with normal mouse serum (non-immune serum, Dako).
Methods used for immunohistochemistry.
Abbreviations: MEC, myoepithelial cells; EC, epithelial cells.
a HIER: heat-induced epitope retrieval; microwave oven for 10 minutes at 567 W in citrate buffer, pH 6.0.
b Also reported in a subpopulation of acinar cells. 20
c PED: pepsin enzymatic digestion (Digest-All Invitrogen; Thermo Fisher Scientific), 15 minutes at 37 °C.
d CK14 also stains basal layer of stratifying squamous and non-squamous epithelia.
e SMA also stains myofibroblasts and smooth muscle cells.
Interpretation of immunohistochemical results was performed by 2 board-certified veterinary pathologists (CG, LN). For each tumor, the entire section was evaluated and immunolabeling of cells was reported as positive or negative, independent of signal intensity.
Results
Canine LGTs
Thirty-eight LGTs from 38 dogs were retrieved. Case 31 was a local recurrence of case 30, and case 10 was the complete excision of a previously biopsied mass (cases 9; Suppl. Table 1).
Most tumors arose within the NLG (34/38, 90%) and only 2/38 (5%) affected the MLG. Two cases were diagnosed as lobular orbital adenomas and the precise gland of origin was not determined.
Males (19/38, 50%; 4 neutered) and females (17/38, 45%; 8 neutered) were almost equally represented, and in 2 cases the sex was not reported. The age of dogs affected by LGTs varied from 4 to 16 years (mean 10.7; median 7 years). Different pure breeds were usually represented by a single individual (Suppl. Table S1).
Malignant LGTs were diagnosed in 34/38 (90%) cases, and benign LGTs were diagnosed in 4/38 (10%) cases. Details are provided in Supplemental Table S1.
Of the 34 malignant LGTs, the NLG was affected in 32 cases (94%) and the MLG in 2 cases. Males and females were almost equally represented (14 females; 18 males; and in 2 cases the sex was not reported). Several breeds were represented and the dogs’ age varied from 4 to 16 years (mean 11, median 10.5; Suppl. Table S1). The surgical margins were considered complete in 20/34 (60%) cases, close in 4 cases (11%), and incomplete in 10 cases (29%). Follow-up data were available in only 15 cases (cases 2, 11, 12, 13, 17, 18, 19, 21, 23, 24, 25, 27, 29, 30/31, and 34). Follow-up period varied from 3 to 24 months (mean 10, median 12). Among cases for which follow-up was available, one had close margins (case 21) and 4 had incomplete margins (cases 17, 24, 30/31); however, a local recurrence was reported only in cases 30/31 and metastatic spread was not reported.
The most frequent diagnoses were pleomorphic carcinoma and ductal carcinoma (7 cases each), followed by epithelial-myoepithelial carcinoma (6 cases) and adenoid cystic carcinoma (5 cases).
Pleomorphic Carcinoma
Pleomorphic carcinoma is composed of epithelial (malignant) and myoepithelial (benign) neoplastic cells and areas of well-differentiated chondroid, myxoid, fibrous, or bone tissue. There is generally a considerable variation in the proportions of these components.
In the present series, 7 of 34 cases (20%) were diagnosed as pleomorphic carcinoma (cases 1–7; Suppl. Table S1). Tumors were composed of both epithelial/myoepithelial and mesenchymal-like tissues. Epithelial cells were variably arranged in tubules or in solid cords and nests with surrounding myoepithelial cells that trailed out gradually into myxomatous mesenchyme. Mesenchymal tissue was variably represented by areas of hyaline stroma, or islands of bone or chondroid tissue (Fig. 1). Foci and areas of necrosis were present in all cases. Mitoses were observed in both epithelial and myoepithelial components. The mitotic count ranged from 3 to 20 mitoses/10 HPF (mean 10). Of the 7 cases, 5 had complete margins, and in 2 cases margins were considered close. In all cases, Alcian blue intensely stained the tumor stroma.
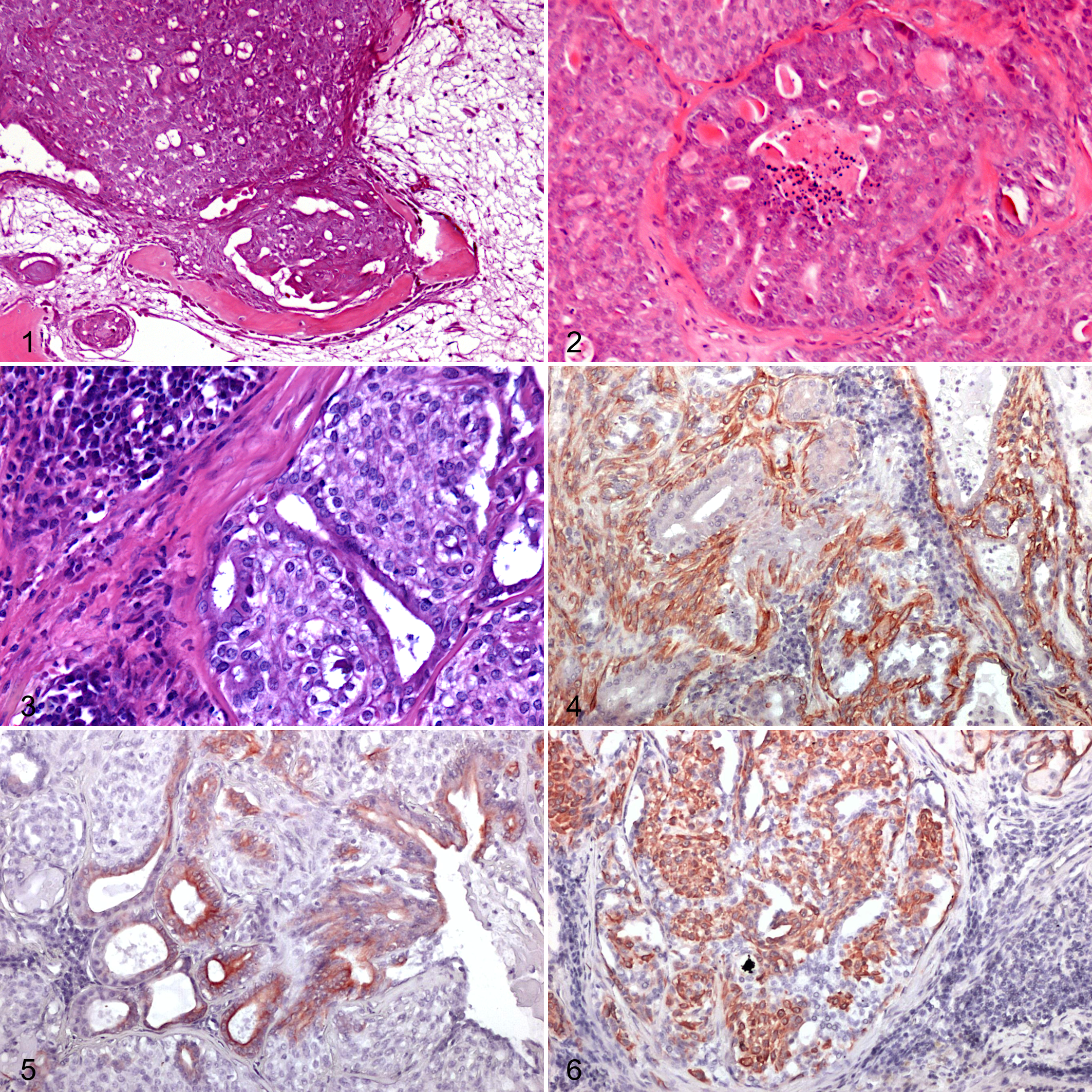
Immunohistochemically, the myoepithelial component stained positively with CK14, CALP, and SMA, except for one case (case 5) where CALP was negative. P63 was positive in 4 cases and negative in 3/36. Tubular structures were highlighted by positive staining for CKAE1/3 and CK19.
Ductal Carcinoma
Ductal carcinoma is an aggressive tumor with differentiation to the interlobular ducts, typically composed of irregularly shaped cystic nodules with central comedonecrosis, lined with carcinoma cells arranged in a cribriform pattern.
In the present series, 6/34 tumors (18%) from 6 dogs were diagnosed as ductal carcinomas (cases 8–14). Cases 9 and 10 represented an incisional biopsy and subsequent radical surgery of the same tumor. Ductal carcinomas were composed of epithelial cells predominantly arranged in lobules with prominent central necrosis resembling comedonecrosis, and lined by epithelial cells organized either in solid nests or in a cribriform architecture with “Roman bridge” formation (Fig. 2). Neoplastic cells were characterized by high nuclear/cytoplasmic ratio, moderate amount of dark eosinophilic cytoplasm, and oval nuclei with finely stippled chromatin. The mitotic count ranged from 1 to 39 per 10 HPF (mean 21.7). The margins were complete in 4 cases, close in 1 case, and incomplete in 2 cases. No Alcian positive stroma or secretion was present.
In one case (case 9) paraffin-embedded material was insufficient for immunohistochemistry. In all remaining tumors (6/7), epithelial neoplastic cells were consistently positive for CKs. p63 was positive in 4/6 cases, and SMA and CALP were positive in 2/6 cases, where they stained a thin, incomplete rim of myoepithelial cells delineated scattered solid lobules.
Epithelial-Myoepithelial Carcinoma
Epithelial-myoepithelial carcinoma is composed of 2 distinct cell populations, that is, epithelial and myoepithelial. In the present series, 6/34 (17%) cases were classified as epithelial-myoepithelial carcinoma (cases 15–20). One case (case 18) was the recurrence of a MLG tumor excised 2 months before with a marginal excision and originally diagnosed as myoepithelioma. The primary tumor was not included in this series due to lack of available material for the review of the case.
Epithelial-myoepithelial carcinomas were composed of both epithelial and myoepithelial cells organized either in a distinctive pattern of tubules with inner epithelial and outer myoepithelial cells, or in a more solid pattern with myoepithelial overgrowth (Fig. 3). In all cases, both epithelial and myoepithelial cells were considered malignant. Mesenchymal tissue (bone, cartilage, or hyaline stroma) suggestive of pleomorphic carcinoma was never present. The mitotic activity ranged from 0 to 12 mitoses/10 HPF (mean 4.5). Mitoses were observed in both the epithelial and myoepithelial cells. Three epithelial-myoepithelial carcinomas had complete margins and 3/6 incomplete margins.
Alcian blue diffusely stained the neoplastic stroma. Immunohistochemistry was performed in 5/6 tumors (in case 20, paraffin-embedded material was insufficient). The epithelial cells stained intensely CK AE1/3 and CK19 positive while the myoepithelial component was intensely and diffusely positive for CK14, p63, CALP, and SMA (Figs. 4–6).
Adenoid Cystic Carcinoma
Adenoid cystic carcinomas are tumors composed of small, uniform cords, and/or solid nests, frequently surrounded by basophilic mucoid or hyaline material. The same material fills small cystic spaces within cellular aggregates. Neoplastic cells have hyperchromatic nuclei and a small amount of indistinct basophilic cytoplasm. Myoepithelial-like cells are also present in stromal regions.
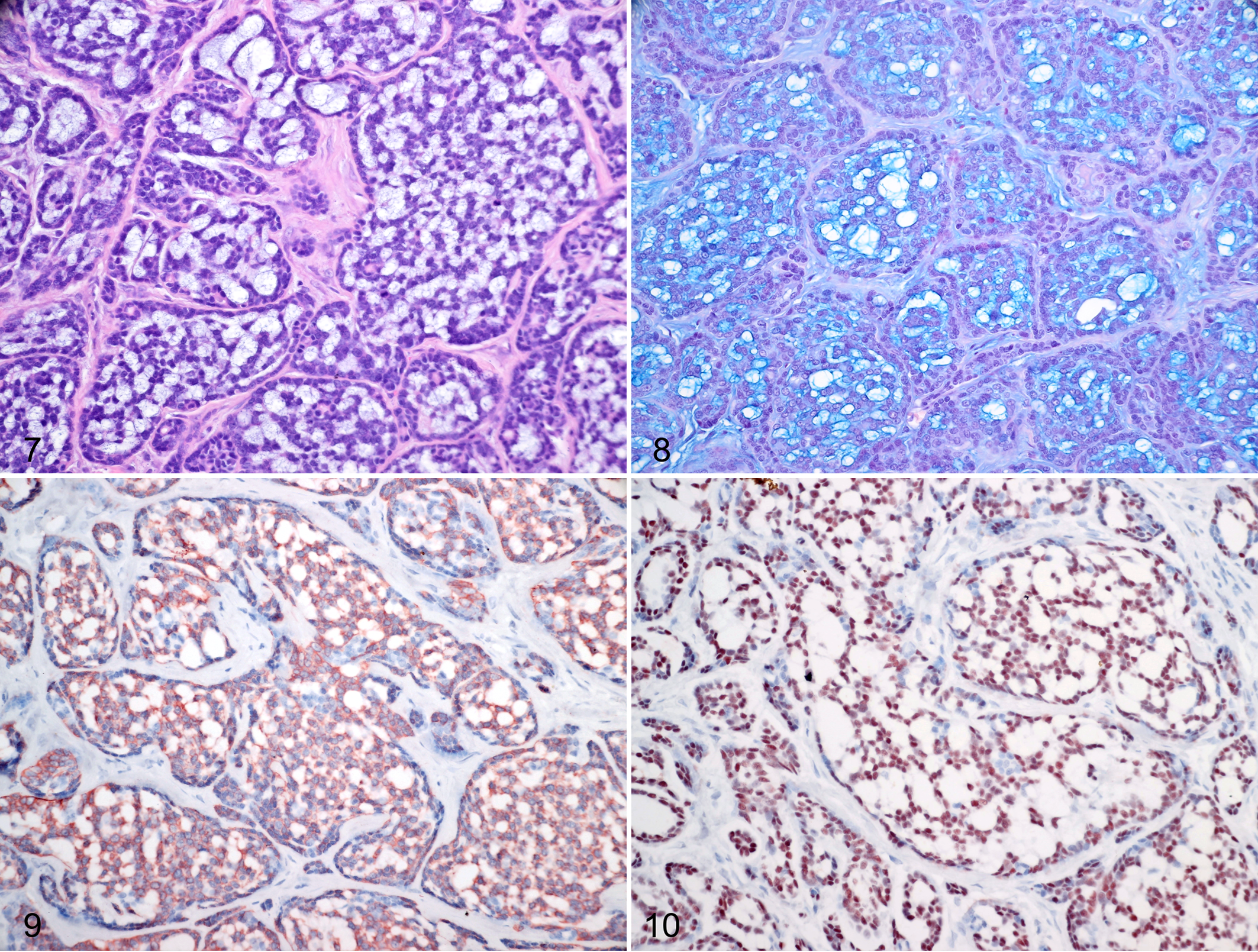
In the present series, 5/34 (15%) cases were classified as adenoid cystic carcinoma (cases 21–25). The tumors were characterized by predominant tubular or cribriform growth pattern, with smaller areas of solid growth pattern (Fig. 7). Necrosis and perineural growth were not prominent features. The mitotic activity ranged from 3 to 19 mitoses/10 HPF (mean 10). Four cases had complete margins and one case had incomplete margins (case 24, Suppl. Table 1).
Adenoid cystic carcinoma, nictitans lacrimal gland, dog, case 6.
Alcian blue positive stroma was present in 3/5 cases. The neoplastic cells were diffusely positive for all immunohistochemical markers tested (Figs. 8–10), with the exception of case 23 that was CALP-negative.
Myoepithelial Carcinoma (MYO)
Myoepithelial carcinomas are tumors composed entirely of myoepithelial cells, proliferating in trabeculae or sheets. Sparse residual non-neoplastic ductular structures can occasionally be present.
In the present caseload 2 tumors (cases 26 and 27) were almost completely composed of myoepithelial cells and were diagnosed as myoepithelial carcinomas. Histologically, the tumors were densely cellular, composed of spindle, polygonal, or clear cells organized in trabeculae or sheets with a delicate fibrous stroma. Occasionally, small ducts (interpreted as non-neoplastic residual structures) were encountered. Cells had a moderate amount of faintly eosinophilic cytoplasm and normochromatic, round to oval, smoothly bordered nuclei. The mitotic count was 2 and 3 mitoses/10 HPF, respectively. Both cases had complete margins.
Alcian blue diffusely stained the neoplastic stroma in both cases. In both cases neoplastic cells were diffusely positive for p63, CALP, and SMA, and negative for CK14, CK19, and CKAE1/3 (with the exception of occasional scattered ductular structures).
In one case (case 28), an adenocarcinoma with sebaceous differentiation was diagnosed. The tumor was mainly composed of cuboidal epithelial cells arranged in tubules or solid sheets. In addition, there were numerous cells morphologically reminiscent of mature sebocytes scattered throughout the mass or organized in small groups. These cells had abundant, finely vacuolized cytoplasm and small central to paracentral, darkly stained nucleus occasionally scalloped by the intracytoplasmic vacuoles. The surgical margins were complete. The Alcian blue stain was negative. The neoplastic cells were diffusely positive for CKAE1/3 and CK19, while CK14 intensely labeled sebocyte-like cells. P63, CALP, and SMA were diffusely negative.
In one case (case 29), an adenocarcinoma with oncocytic features was diagnosed. The tumor was composed of variably sized tubules lined by 2 distinct cell populations, one composed of cuboidal to columnar luminal cells, that merged in a second population composed of polygonal cells with poorly defined cell borders, a low N/C ratio, bland nuclei, and abundant intensely eosinophilic granular cytoplasm. This second population was prominent enough to warrant the diagnosis of oncocytic differentiation. The surgical margins were complete. Alcian blue stained negative. Neoplastic cells were positive for p63, CKAE1/3, CK14, and CK19, and negative for CALP and SMA.
In one dog, a squamous cell carcinoma of the NLG was diagnosed (cases 30, 31; Suppl. Fig. S2). Histologically, the tumor was composed of distorted cords and solid nests of epithelial cells with prominent squamous differentiation that diffusely effaced the NLG architecture. Occasional keratin pearl formation and extensive areas of necrosis were present. The mitotic count was 12 mitoses in 10 HPF. The tumor extended to the surgical margins (incomplete margins). The tumor recurred 3 months later (case 31), as multiple, large (2 × 1 cm) masses infiltrating and distorting the conjunctival substantia propria of the eyelid. Conjunctival surface epithelium and eyelid epidermis were unaffected. Histologically, tumor recurrence was consistent with squamous cell carcinoma with intense fibroplasia. Mitoses were 41/10 HPF. Neoplastic cells extended to the surgical margins (incomplete margins). Alcian blue stained negative. The neoplastic cells were p63, CKAE1/3, and CK14 positive. CK19 stained scattered groups of cells in the first specimen and was mostly negative in the recurrence. CALP and SMA were consistently negative.
Unclassified Carcinomas
There were 5 cases of carcinoma that did not clearly match the features of any entity in the recent human WHO lacrimal gland tumor classification scheme (cases 32–36).
One case was classified as lacrimal adenocarcinoma NOS (not otherwise specified; case 32). The tumor was an infiltrative neoplasm composed of epithelial cuboidal cells arranged in tubules with a moderate amount of fibrovascular stroma. Neoplastic cells were characterized by relatively distinct cell margins and a moderate amount of pale eosinophilic cytoplasm. Nuclei were oval and normochromic with a single small nucleolus. Anisokaryosis and anisocytosis were moderate, and the mitotic count was 0/10 HPF. The tumor extended to the surgical margins (incomplete margins). The Alcian blue staining was negative. The neoplastic cells were CK AE1/3, CK19, CK14, and p63 positive and calponin and SMA negative.
Three tumors were histologically characterized by a pattern of growth consistent with solid carcinoma (cases 33–35). Histologically, the tumors were composed of epithelial cells arranged in dense solid sheets or cords supported by a moderate amount of fibrovascular stroma. Neoplastic cells were polygonal, with poorly demarcated cell margins and scant, lightly eosinophilic to basophilic cytoplasm. Nuclei were oval and hyperchromatic with coarsely stippled chromatin and a single central prominent nucleolus. Anisokaryosis and anisocytosis were moderate to severe, and mitoses ranged from 10 to 48/10 HPF (mean 34, median 45). Two cases had complete margins, in 1/3 cases the margins were close. Two (2/3) solid carcinomas were investigated immunohistochemically. Neoplastic cells were positive for all markers tested, and Alcian blue stained the neoplastic stroma positive. In case 34, the paraffin-embedded material was insufficient to perform immunohistochemistry.
The last case (case 36) was an extremely pleomorphic NLG tumor, with features of myoepithelial carcinoma, areas of squamous metaplasia and tubulopapillary growth. The surgical margins were incomplete. Neoplastic cells were variably positive for all markers tested: myoepithelial cells were positive for p63, CK14, SMA, and CALP, tubular cells were CKAE1/3 and CK19 positive, and squamous cells were CKAE1/3 and CK14 positive. Alcian blue stained the tumor stroma. The tumor did not clearly match the features of any entity in the lacrimal tumor classification and was defined as an unclassified carcinoma.
Canine Benign LGTs
Four of the 38 canine neoplasms (10%) were benign tumors (cases 37–40).
Two tumors (cases 37 and 38) were clinically and macroscopically similar: they clinically presented as space occupying orbital masses with severe conjunctival swelling, and had a typical botryoid appearance, composed of multiple translucent lobules. Histologically, the tumors were composed of fully differentiated glandular secretory cells with abundant, granular pale eosinophilic cytoplasm. No differentiated ducts were seen. Both cases had incomplete margins. According to the current literature these tumors were classified as canine orbital lobular adenomas. 14 These tumors were included in the cohort of LGTs, because in both cases no clinical or diagnostic imaging features suggestive of a zygomatic gland origin (eg, a dorso-medial displacement of the eye globe or orbital floor discontinuity) were present. Neoplastic cells of lobular adenomas were immunolabeled only with CK19 and CKAE1/AE3, while SMA, CK14, CALP, and p63 were negative.
One tumor (case 39) was composed of epithelial cells arranged in tubules that occasionally contained an amorphous eosinophilic secretion. The tubules were lined by a single layer of columnar cells with a moderate amount of faintly eosinophilic cytoplasm, and round to oval nuclei with finely stippled chromatin. Anisokaryosis and anisocytosis were minimal and mitotic figures were undetected (simple adenoma). Paraffin-embedded tissue was insufficient to test the immunohistochemical features of this case.
The other tumor (case 40) consisted of 2 distinct cell populations. The first population shared the same features described for the previous case, and the second population was composed of spindle-shaped myoepithelial cells arranged in loose bundles separating tubular structure. Myoepithelial cells had poorly demarcated borders, a moderate amount of eosinophilic cytoplasm and round to fusiform nuclei with finely stippled chromatin. Anisokaryosis and anisocytosis were minimal and mitotic figures were undetected. The tumor was classified as adenoma—complex type and had complete margins. The complex adenoma had strong cytoplasmic labeling of lobular/acinar cells with CK19 and CKAE1/3, while the myoepithelial cells were intensely and diffusely positive for CK14, CALP, and SMA and negative for p63, CK19, and CKAE1/3. In all adenoma cases tested, Alcian blue stained the secretory content within tubular lumina. Follow-up data were available for 1 benign tumor (case 40) in which no recurrence was reported at 12 months after surgery.
Feline Lacrimal Gland Tumors
Eight epithelial lacrimal gland tumors were retrieved. Details are provided as Supplemental Table S2. Females were overrepresented (6 females, 2 spayed; 2 males, 1 neutered). Mean age of cats with NLG/MLG tumors was 12 years (median 15, range 6–16 years). Most (7/8) were domestic shorthaired cats and one was Siamese.
Five tumors affected the nictitans gland and 3 affected the main lacrimal gland. All tumors were malignant and had infiltrative growth. Follow-up data were retrieved in only 2 cases where no recurrence was recorded (cases 1 and 8; Suppl. Table S2) at 6 months after surgery.
The majority of the feline tumors (5/8, cases 1–5; Suppl. Table 2) were classified as adenocarcinoma NOS, simple, tubular (cases 1–3) or tubulopapillary (cases 4 and 5). The tumors were poorly demarcated, infiltrative masses, composed of neoplastic cells predominantly arranged in tubules with a moderate amount of fibrovascular stroma. The tubules were lined by 1 to 2 layers of cells that had variable morphology, from cuboidal to cylindrical, with relatively distinct cell margins and moderate amount of pale eosinophilic cytoplasm. Nuclei were mainly oval, normochromic with a single small nucleolus. In addition to these features, the tubulopapillary carcinomas had neoplastic cells variably arranged in tubules or papillae extending into the tubular lumina and supported by a fine fibrovascular connective tissue. Anisokaryosis and anisocytosis were generally moderate, and mitoses ranged from 1 to 21/10 HPF (mean 7.6; median 3). Alcian blue sparsely stained the luminal content. The neoplastic cells were CKAE1/3 and CK19 positive, with scattered CK14 positive cells. P63, CALP, and SMA were always negative.
Two feline LGTs (cases 6 and 7) were diagnosed as ductal carcinoma. The neoplastic cells were organized in large solid lobules, composed of a thick rim of epithelial cells disposed in a sieve-like pattern and surrounding a large central area of coagulative necrosis (comedonecrosis). The neoplastic cells were polygonal, with a moderate amount of homogeneous eosinophilic cytoplasm and oval nuclei with a single small nucleolus. Anisokaryosis and anisocytosis were moderate, and mitoses were 3 and 22/10 HPF. Neoplastic cells were intensely CKAE1/3 and CK19 positive, while scattered, mainly basally located cells were CK14 and p63 positive. CALP and SMA were negative. No Alcian blue staining was present.
One case (case 8) was diagnosed as epithelial-myoepithelial carcinoma. The tumor was composed of both epithelial and myoepithelial cells and retained the typical pattern of a gland with a distinctive outer myoepithelial and an inner epithelial layer. The epithelial cells were intensely CK AE1/3 and CK19 positive while the myoepithelial component was intensely and diffusely p63, CK14, calponin, and SMA positive. Alcian blue sparsely stained the luminal content.
Normal Lacrimal Gland
Portions of the nonneoplastic lacrimal gland were frequently included in the NLG, and more rarely in MLG tumor specimens. Immunohistochemically, acinar and ductal cells were consistently CKAE1/AE3 and CK19 positive while CK14, SMA, and calponin consistently and intensely stained myoepithelial cells surrounding the acini and ducts. p63 stained scattered nuclei at the base of the acini and ducts (myoepithelial nuclei; Suppl. Figs. S3–S6).
Discussion
In humans, primary adenocarcinomas of the lacrimal gland have only recently been subclassified, allowing for important advancements in the management of tumors. 2,33,34
In the present case series, the WHO classification of human LGTs 12 was applied to canine and feline LGTs and 4 main histopathological subtypes of LGTs were described, namely, pleomorphic, ductal, adenoid cystic, and epithelial-myoepithelial carcinomas. Additionally, rarer tumors that are also reported in humans were described, including myoepithelial carcinoma, squamous cell carcinoma, sebaceous carcinoma, and oncocytoid carcinoma.
In humans, pleomorphic carcinoma is considered to arise as a malignant transformation of the more common pleomorphic adenoma, as evidenced by the presence of residual tumor areas with benign features, and therefore frequently indicated as pleomorphic carcinoma-ex pleomorphic adenoma. 2,12,33,35 No similar areas with benign features were clearly identified in our cases, and we therefore preferred the term of pleomorphic carcinoma. Pleormophic adenoma has been previously diagnosed in dogs, 16 but was not present in this series.
Adenoid cystic carcinoma of the human lacrimal gland is described as a malignant neoplasm composed of both epithelial and myoepithelial cells organized in various patterns, including cribriform, tubular, and solid. 2,12,33,35 The tumor has a high rate of recurrence and can give rise to widespread metastasis. Important histopathological features associated with a worse prognosis include solid pattern of growth and perineural invasion. 12,35 Tumors in dogs of the present series were mostly characterized by a tubular or cribriform growth pattern, and perineural invasion was never observed.
Epithelial-myoepithelial carcinomas and ductal carcinomas are considered rare in humans, 2,12,35 while in the present series, they outnumbered the adenoid cystic carcinoma cases. The epithelial-myoepithelial carcinomas in this caseload were in many cases consistent with those that are considered high-grade epithelial-myoepithelial carcinomas in humans, that is, tumors that retain a clear distinction between the myoepithelial and epithelial components but had increased mitotic rate and loss of the typical pattern of growth (ie, absence of tubules with inner epithelial and outer myoepithelial growth). An epithelial-myoepithelial carcinoma was diagnosed in a cat in this series, to the best of authors’ knowledge for the first time in this species.
Ductal carcinoma is currently classified under adenocarcinomas of the lacrimal gland in the recent WHO classification of LGTs, 12,22,24,31 and it is characterized by prominent features of comedonecrosis and sieve-like pattern of intraductal growth. The last feature was particularly prominent in feline ductal carcinomas in the present series.
In the present caseload, 3 cases of uncommon tumors were also described, that is, squamous cell carcinoma, carcinoma with oncocytic features, and carcinoma with sebaceous differentiation. While not included in the WHO list of LGTs, primary squamous cell carcinoma of the lacrimal gland has been described in humans, 10,17 and a single case has also been previously reported in a dog. 6
Oncocytoma and oncocytic carcinoma are included in the current WHO classification of human LGTs, 12 and to the best of the authors’ knowledge, they have never been described before in dogs. In the present case (case 29), the tumor was not entirely composed of cells with oncocytic features so a diagnosis of carcinoma with oncocytoid features was preferred.
Sebaceous carcinoma is a rare variant of LGT in humans. It is hypothesized that it derives from the sebaceous metaplasia of ductal epithelium or from sebaceous differentiation within a preexisting tumor. 13,18 Sebaceous LGT has never been described in dogs, while sebaceous mammary carcinomas are known. It is the authors’ opinion that the term “sebaceous” in the present case should be considered to be more descriptive than indicative of a differentiation of neoplastic cells toward sebocytes. In fact, lipid-secreting acini similar to those composing the Harder’s gland have been identified in the canine lacrimal gland, 15 and in the authors’ experience, they can actually be sporadically seen in sections of the normal nictitans gland. The epithelial cells with finely vacuolized cytoplasm observed in the present case could therefore derive from these lipid-secreting acini which are typical of canine NLG, rather than from the metaplasia of ductal epithelium.
A few tumor cases in dogs and numerous tumors in cats remained unclassified. Adenocarcinoma NOS was the most frequent diagnosis in the feline cases (5/8). Unclassifiable cases have also been reported in humans, 27,35 which is most likely due to the extreme pleomorphism of LGTs. However, it is also possible that the human classification of LGT imperfectly fits canine and feline LGTs, and that a more species-specific classification has to be created.
The LGTs were also characterized by immunohistochemistry. In human medicine it is considered important to identify myoepithelial cells in the study of LGTs. 7,20,25 However, it has recently been shown that a subset of cells within the basal layer of lacrimal gland excretory interlobular ducts can also express SMA and it has been hypothesized that some cell lineages retain plasticity after maturation and can trans-differentiate into other cell types upon injury. 3 In the present study, calponin and SMA gave the most consistent and overlapping results. CK14 and p63 labeled myoepithelial cells but also ductal carcinoma cells. Moreover, p63 was unexpectedly negative in benign canine lesions, while it was expressed in myoepithelial cells of normal LGs. A loss of p63 expression is usually associated with tumor progression, and the only other study 23 currently investigating p63 in canine LGTs did not include any benign tumors. Therefore, it cannot be determined to date whether our results represent a lack of reactivity toward a specific p63 isoform, or more simply a problem with fixation.
In general, immunohistochemistry was useful for highlighting different cell components of the tumors, and it supported the histomorphological subtyping of tumors but was not strictly necessary for identification and diagnosis of tumor subtypes. Similarly, Alcian blue staining highlighted the neoplastic stroma mainly in areas of myoepithelial proliferation.
In the current cases, with the exception of squamous cell carcinoma, no canine tumors metastasized or recurred, regardless of their margin status. In the veterinary literature, canine LGTs are generally considered to be at risk of local recurrence but at a low risk of metastasis, especially if radical surgery with removal of the nictitating membrane is performed. 9,11,36 Feline LGTs are generally considered to be more aggressive 6,8 and metastasis was reported. 19 In our series, follow-up data were retrieved for only 2 feline cases, with no recurrence or metastasis observed.
When comparing the biological behavior of LGTs in humans, dogs, and cats, one main anatomical difference should be considered: LGTs generally affect the MLG in humans, but more frequently affect the NLG in animals (the NLG is absent in humans). While the MLG is located in a fossa behind the superotemporal orbital rim, close to orbital bones, and innervated by a main branch of the trigeminal nerve, the NLG has no strict connection with the surrounding orbital tissue apart from the nictitans membrane itself, and its innervation is poorly studied but not provided by major nerve branches. The main consequences of this anatomical difference is that NLG tumors are more easily and completely excised than MLG tumors, and perineural growth and bone invasion—both indicators of recurrence and a worse prognosis in the human literature—are rarely observed in canine and feline LGTs. However, the survival data in dogs and cats of this study are limited by the low number of cases with follow-up data, the relatively short period of follow-up, and the lack of a complete clinical staging for the identification of metastatic disease or cause of death. Thus, the behavior of different LGT histopathological subtypes in domestic species remains unknown.
In conclusion, the present study applied the human WHO classification of LGTs to canine and feline lacrimal tumors to identify different histopathological subtypes. This represents a first attempt to use a consistent and reproducible nomenclature for feline and canine LGTs. Immunohistochemistry has been proven useful for studying the cell components of LGTs but currently it is not considered mandatory for making the diagnosis of LGTs according to the WHO human classification.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211025791 - Epithelial lacrimal gland tumors in dogs and cats: Is the human WHO classification appropriate for animals?
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211025791 for Epithelial lacrimal gland tumors in dogs and cats: Is the human WHO classification appropriate for animals? by Chiara Giudice, Laura Nordio, Micol Cadonici, Maria Novella Perelli and Mario Caniatti in Veterinary Pathology
Footnotes
Acknowledgements
The authors want to thank all those people (students, colleagues, PhD students) that in this difficult period of pandemia, with laboratories sometimes inaccessible, delayed product delivery and no technical support for instrumentation did their very best to help, collecting information, taking pictures, providing documents and sometimes even babysitting. A special thanks to Dr. Marco Rondena, Dr. Matteo Gambini, Dr. Valentina Mezzadri, Dr. Laura Barachetti, Dr. Chiara Paratici, Dr. Anna Bonfanti, Dr. Cinzia Frizzo, Dr. Robert Corvi and Dr. Domenico Multari.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
