Abstract
Talaromyces spp. are soil-dwelling fungi sporadically reported to cause disease in humans and dogs. This study summarized the clinical presentations, histologic findings, and Talaromyces sp. involved in 5 dogs diagnosed through the panfungal polymerase chain reaction service (PCR) at Texas A&M University, with a review of previously reported cases. Of the 5 cases, 3 were Labrador Retrievers, 2 were male, and 3 were female. Three of 5 involved the musculoskeletal or lymphatic systems, and 2 of 5 dogs presented with meningoencephalitis. Talaromyces helicus, Talaromyces aurantiacus, and Talaromyces boninensis were identified based on panfungal PCR, showing 99% to 100% sequence matches in combination with morphologic features. Three of 5 dogs had static disease at the time of publication, 1 was euthanized, and 1 was lost to follow-up. This study describes Talaromyces spp. as a cause of meningoencephalitis in dogs, identifies 2 novel Talaromyces spp. involved in infections, and adds to the existing knowledge of clinical presentations and outcomes.
Talaromyces spp. are soil-dwelling fungi characterized by the production of sac-like reproductive structures containing ascospores. 17 They were first described in 1955 as a sexual state of Penicillium. However, more recent phylogenetic information based on morphology and polymerase chain reaction (PCR) data demonstrates Talaromyces spp. as a distinct clade from Penicillium, resulting in recent reclassification of many clinically relevant species. 19 The genus contains an important human pathogen, Talaromyces marneffei, known to cause fatal systemic mycosis in HIV/AIDS patients in Southeast Asia, with occasional reports in other regions.3,5,9,10,12,13 Talaromyces spp. infections have been sporadically reported in dogs since 1990. Affected dogs present most commonly with lymphadenomegaly and lameness, with only 1 previous case resulting in pneumonia. A summary of the 8 previously reported cases in dogs is provided in Supplemental Table S1. All cases occurred in young to middle-aged adult dogs, 5 of 8 were female, 3 of 8 were male, 2 of 8 were Labrador Retrievers, 2 of 8 were German Shepherd Dogs, and the remaining 4 were a mix of various breeds. A single case survived to the time of reporting, and all other cases died or were euthanized due to progression of disease.4,5,8,11,16,17,18,20 With the exception of 1 case with concurrent canine distemper virus infection and 1 case with a history of treatment with immunosuppressive drugs for atopic dermatitis, the majority of previously reported cases did not have a history of immunosuppression, indicating Talaromyces spp. are capable of causing disease in immunocompetent dogs.5,17 The route of entry for all previously reported cases was unknown; although given that the majority of affected dogs do not present with respiratory signs, direct inoculation with systemic dissemination is considered likely.
Five cases of Talaromyces spp. infections were identified at the Texas A&M University (TAMU) Veterinary Medical Diagnostic Laboratory via panfungal PCR and sequencing through the Panfungal PCR Service. Two cases were from eastern Texas and treated through the TAMU Neurology Service. Three cases were from referral clinics in Florida, North Carolina, and Arkansas. All cases were identified in 2019 and 2020, occurred in dogs, and affected a range of tissues. Formalin-fixed tissues were received in paraffin-embedded blocks and processed routinely for histologic examination and characterization with hematoxylin and eosin (HE) stain and Grocott’s methenamine silver (GMS) stain. Samples were analyzed by PCR and sequence analysis of the internal transcribed spacer 2 (ITS-2) region and ribosomal RNA large subunit (LSU) coding region, as previously described. 7 Briefly, 50 µm thick sections were obtained from paraffin blocks and total DNA was extracted using the BiOstic Formalin-Fixed Paraffin-Embedded (FFPE) DNA Isolation Kit (MoBio, Carlsbad, CA), and fungal DNA was amplified by PCR. All PCR products were separated on a 2% agarose gel and bands were excised and purified using the EZNA kit (Omega Bio-Tek, Norcross, GA). DNA sequences obtained from Eton Bioscience Inc. (San Diego, CA) were queried against the GenBank database (NCBI) using the basic local alignment search tool (BLAST), and identities of the sequences were claimed based on 99% to 100% identity matches. 1
A summary of clinical, pathologic, and microbiology results is in Table 1. The cases ranged in age from 2 to 13 years. Three dogs were Labrador Retrievers, 1 was a Vizsla, and 1 was a Standard Poodle. There were 2 intact males, 1 intact female, and 2 spayed females. Infections involved the central nervous system, musculoskeletal system, and lymphatic system. Clinical progression in the 3 cases involving the musculoskeletal and lymphatic systems typically involved several months of worsening clinical signs relevant to the affected system prior to a final diagnosis. However, the 2 cases involving the central nervous system experienced a much shorter duration of 3 days to 1 week. Complete blood counts (CBCs) and chemistry panels were largely unremarkable in all dogs except for some instances of elevated urea and creatinine, which were referable to the progression of clinical disease or to the administration of amphotericin B in 1 dog. A definitive diagnosis was obtained in all cases with biopsy and subsequent panfungal PCR in correlation with histologic fungal characteristics. The majority of cases were treated with a combination of an azole antifungal, terbinafine, and anti-inflammatory medications. Three of the 5 dogs had no disease progression at the time of publication, over 12 months from the time of their initial presentations. One dog was lost to follow-up after a palliative hemipelvectomy, and another was euthanized several days after presentation due to rapid progression of clinical signs and underwent a full necropsy, which was unremarkable. All dogs were considered to be of normal immune status.
Summary of clinical, pathologic, and microbiology results in 5 dogs with Talaromyces spp. infections.
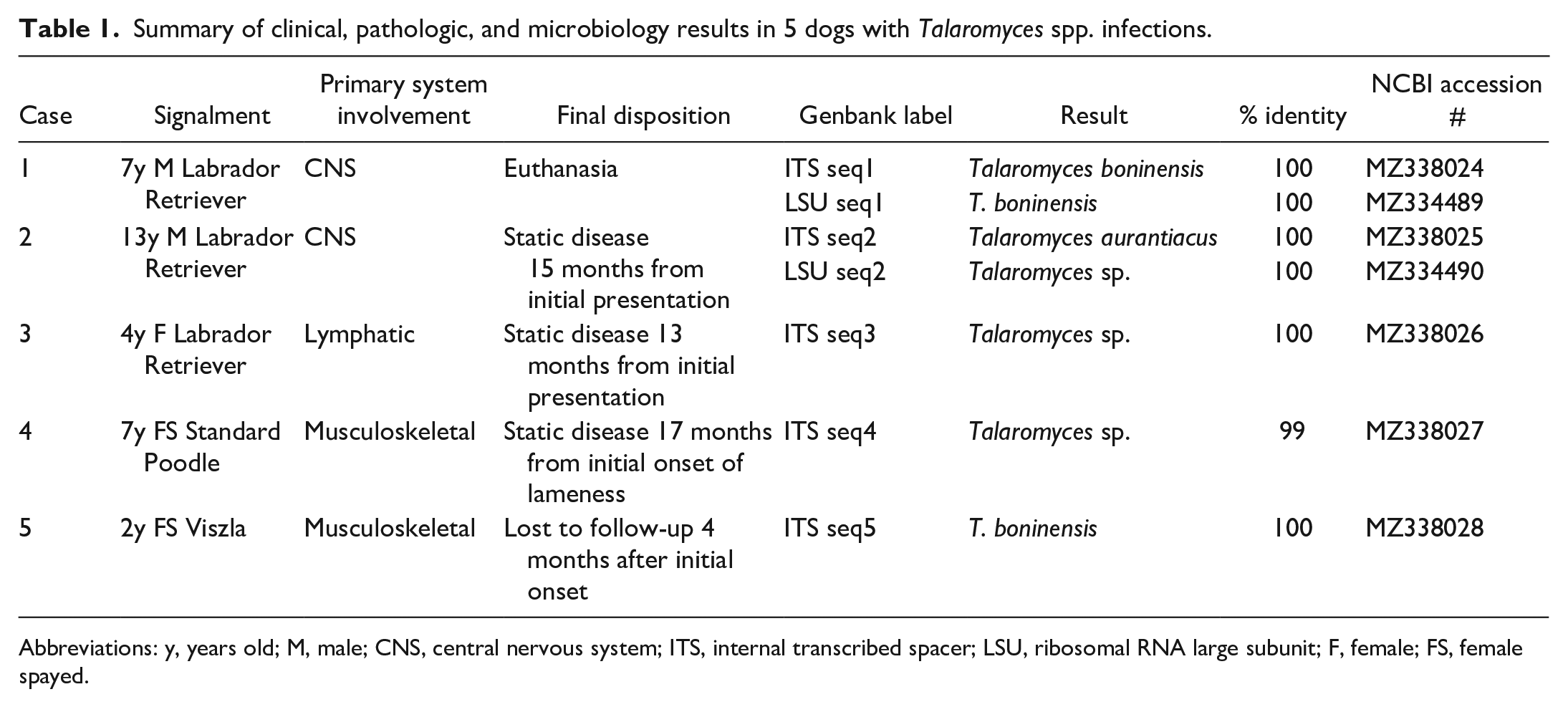
Abbreviations: y, years old; M, male; CNS, central nervous system; ITS, internal transcribed spacer; LSU, ribosomal RNA large subunit; F, female; FS, female spayed.
Radiographs and computed topography of affected bones in cases with skeletal involvement showed primarily a moderate periosteal reaction, with mild bony lysis in more advanced lesions. Magnetic resonance imaging in 1 case involving the cerebrum revealed a T2 hyperintensity of the caudal, medial, and dorsal aspects of the frontal lobes (Fig. 1). Histopathologic changes in all cases involved infiltration to marked effacement of the affected system by numerous epithelioid macrophages, multinucleated giant cells, lymphocytes, and plasma cells which frequently coalesced to form discrete granulomas. Within the cytoplasm of multinucleated giant cells were variable numbers of 5 to 25 µm, negatively staining to lightly basophilic, GMS-positive, fungal hyphae with irregular septation, nonparallel walls, acute angle branching, and occasional bulbous swellings up to 10 µm diameter (Figs. 2–6). The fungus less frequently formed small round structures resembling yeast and pseudohyphae. Similar structures have been described in other Talaromyces spp. infections and may represent irregular cross-sections of the bulbous projections.5,11 Other histologic findings were referrable to degeneration and necrosis of resident cellular populations in the affected tissue.
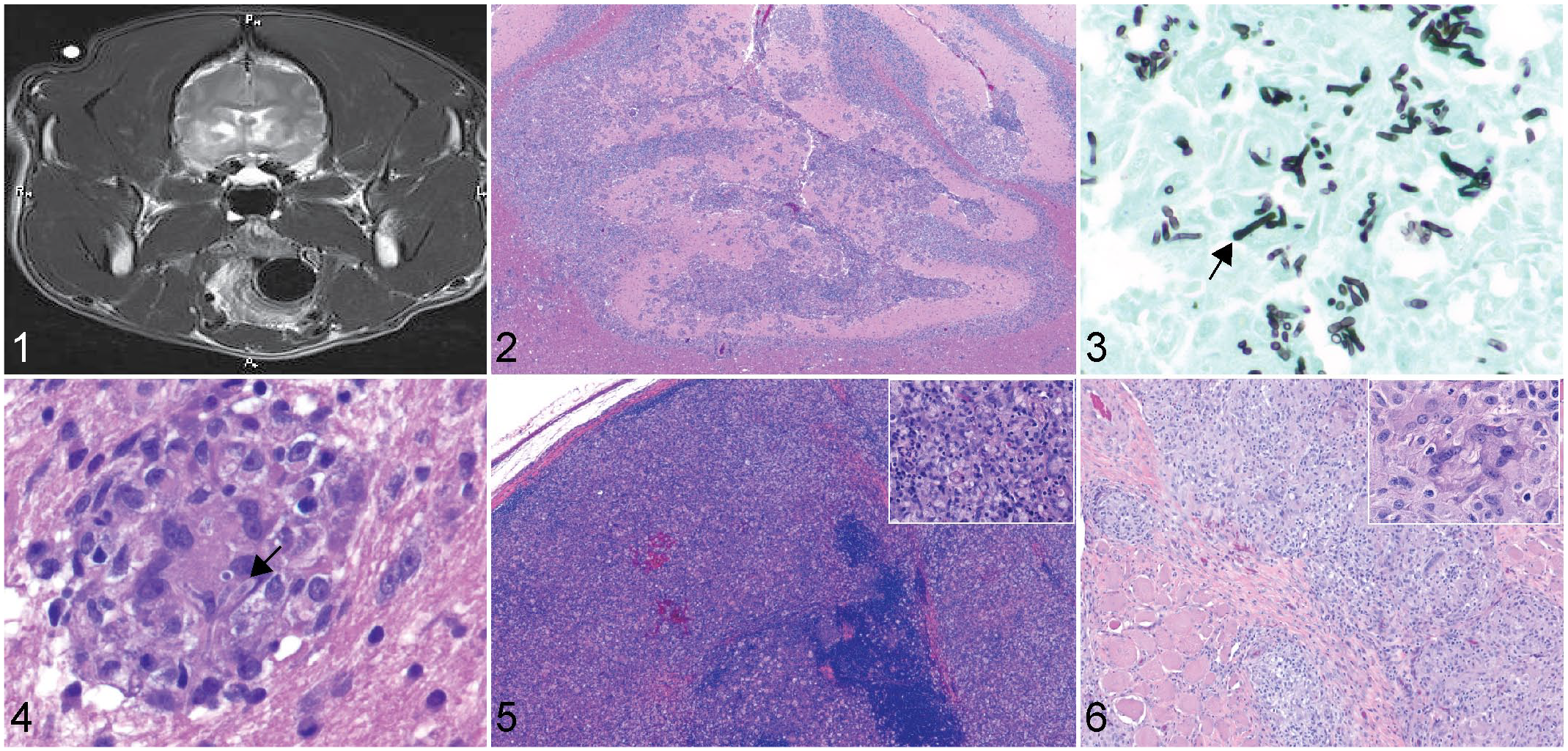
Talaromyces spp. infection, dogs.
Following panfungal PCR, 2 cases yielded 100% identity with Talaromyces boninensis, 2 yielded 100% identity with Talaromyces sp., and 1 case 100% identity with Talaromyces aurantiacus. No fresh tissue was available for fungal culture. As such, presumptive diagnosis was made based on panfungal PCR in conjunction with the fungal morphology and correlation with previous case reports.
This study reviewed Talaromyces spp. infections in 5 cases from the Southern United States, confirmed via panfungal PCR in conjunction with histologic fungal morphology. Two cases involved T. boninensis and 1 involved T. aurantiacus, neither of which have been previously reported to cause disease in dogs. Similar to previously reported cases, several of the dogs presented with granulomatous lymphadenitis and osteomyelitis. However, 2 of the cases involved granulomatous meningoencephalitis, another novel finding in this series of cases.4,8,11,16,17,18,20 Taken in conjunction with the previously reported cases, Talaromyces spp. appear to be capable of infecting a wide range of tissues. Of the now reported cases, 7 of 13 had synovitis and/or osteomyelitis as the primary disease process. Of these, only 1 case involved the axial skeleton, indicating a predisposition for appendicular skeletal infection involving a single location or multiple bones. Only 1 of 13 cases was diagnosed with pneumonia without any of the other more commonly reported systems involved.4,5,8,11,16,17,18,20 Of the 13 reported canine cases of Talaromyces spp. infection, 7 were from the Southern United States, in environments which mimic those in the reported endemic countries, so environment should be considered a risk factor.4,5,8,11,16,17,18,20 The route of exposure in dogs remains unclear, although with the apparent predilection for disseminated lymph node infection and the potential for multifocal skeletal involvement, systemic spread after penetration of intact barriers seems most likely.2,14
While many cases of Talaromyces spp. infections in humans occur in immunocompromised patients, most canine cases have not involved immunocompromised animals. Only 2 of the previously published cases and none of the 5 cases presented here were obviously immunocompromised.5,17 In previous reports, a breed disposition for Labrador Retrievers was hypothesized.16,17 This breed accounts for 5 of 13 of all reported cases. The only other breed with more than one reported case is the German Shepherd Dog (2/13).4,5,8,11,16,17,18,20 More cases need to be documented before a breed disposition can be determined. Other epidemiologic factors such as diet and potential sources of environmental exposure also need to be explored.
Canine Talaromyces spp. infections appear to be increasing in frequency in dogs in the United States, although the recent higher rate of diagnosis may be due to increasing access to molecular diagnostic tools. Molecular diagnostics allow speciation of the causative agent, which can be difficult based on morphology alone and impossible if fungal cultures are unrewarding or unavailable. The gold standard for fungal identification should be fungal culture, and combining morphology with sequencing is considered ideal. Previous publications have noted the difficulty of speciating Talaromyces based on culture characteristics alone.4,16 Sequencing of the internal transcribed spacer (ITS) region has classically been used for speciation, but combination with sequencing of the beta-tubulin (BenA) gene has allowed more specific resolution of taxonomy and may be worth investigating in future clinical cases.
Treatment of the cases described here involved various combinations of empirically selected azole antifungals, terbinafine, and amphotericin B. In human cases of Talaromyces marneffei, itraconazole and voriconazole have reliably low mean inhibitory concentrations and are preferred over amphotericin B. Ideally, if fresh tissues are available, final treatment decisions in human and veterinary patients should rely on culture and sensitivity results. 15 In human cases, mortality rates are over 50% if left untreated and over 24% in cases that receive antifungal therapy, with better outcomes the more rapidly a case is diagnosed. 6 Many of the dogs in this study had stable disease at the time of publication, but others had more rapid, severe progression despite intensive management. No case described here had complete resolution of disease, and mortality rates seem to mirror those of human cases.
In conclusion, Talaromyces spp. need to be considered as potential differential diagnoses for localized or disseminated fungal disease in the dog. We described T. boninensis and T. aurantiacus as capable of causing disease and described meningoencephalitis as a possible sequela to infection, previously unknown facets of disease caused by this fungal genus in dogs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221075589 – Supplemental material for Talaromyces spp. infections in dogs from the Southern United States
Supplemental material, sj-pdf-1-vet-10.1177_03009858221075589 for Talaromyces spp. infections in dogs from the Southern United States by Rebecca L. Bacon, Stephanie A. Lovell, Aline Rodrigues Hoffman and Alycia P. Fratzke in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank all of the primary care and referring veterinarians involved in these cases and for the use of their records in the creation of this publication. We would also like to thank Dr Dominique Wiener for her assistance with the histologic images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
