Abstract
Aspergillosis is the most commonly and widely reported fungal infection in birds. Disease development is often secondary to stressors that cause immunocompromise, and it is typically regarded as a disease of captivity. We retrospectively evaluated data from 133 birds diagnosed with aspergillosis at the Southeastern Cooperative Wildlife Disease Study from 2001–2023 to assess diversity and relative frequency across avian taxa, gross and histologic lesion patterns, and comorbidities. Of 10 taxonomic orders represented, Charadriiformes (shorebirds; n = 35) and Accipitriformes (raptors; n = 32) were most common. Among them, the laughing gull (Leucophaeus atricilla; n = 20) and bald eagle (Haliaeetus leucocephalus; n = 14) were infected most commonly. Gross lesions were most frequent in lung (n = 80), air sac (n = 71), or celomic cavity lining (n = 42). Four distinct gross lesion patterns were identified: 1) tan caseous plaques (n = 106), 2) hollow masses lined with mold (n = 26), 3) red pulmonary nodules (n = 15), and 4) necrotic brown plaques (n = 3). Histologically, fungal hyphae were most common in lung (n = 107) and air sac (n = 49). Comorbidities were diagnosed in 67 birds with a spectrum of viral (n = 19), bacterial (n = 11), parasitic (n = 6), other fungal (n = 4), and non-infectious (n = 50) causes. Six birds each were diagnosed with highly pathogenic avian influenza or salmonellosis. Twenty-two birds were emaciated. Free-ranging birds are susceptible to myriad stressors that can predispose them to the development of aspergillosis.
Aspergillosis is a fungal disease of birds, mammals, and invertebrates caused by Aspergillus spp. An environmentally ubiquitous fungus, Aspergillus has been documented globally in the environment. Infection, which is often subclinical, occurs via inhalation of spores from the environment; immunocompromised individuals that are infected opportunistically may develop nonspecific respiratory signs that can progress to systemic disease.8,57 Avian aspergillosis is often thought of as a disease of captivity, particularly in young poultry, where it is referred to as “brooder pneumonia”.8,57 In wild birds, aspergillosis reportedly occurs in rehabilitation settings and is generally not considered a population health concern, although mass mortality events occur and are often associated with a shared risk factor. 15
Among free-ranging birds, aspergillosis is most commonly reported in gulls, waterfowl, and raptors; however, available epidemiologic data are heavily fragmented and often limited to species of high research importance and public interest.8,15 Ecologic and environmental stressors in wildlife are numerous and can trigger acute and prolonged immunocompromise that facilitates disease development.25,32 Although specific risk factors for the development of aspergillosis are poorly documented, potential factors include those related to climate change, landscape fragmentation, alterations in food source availability, changes in population density, coinfections, and proximity to civilization.25,32 Due to the likelihood of exposure to stressors among wildlife populations globally, a better understanding of the significance of diseases such as aspergillosis and the relationship between such diseases and other causes or contributors to mortality is warranted.
To address these knowledge gaps, we describe here geodemographic data, species diversity, and lesions in wild birds from the eastern United States that were diagnosed with aspergillosis over 22 y. Additionally, we discuss the implications of comorbidities identified, trends within avian orders, and the potential impacts of aspergillosis in the context of a rapidly changing world that continually impacts animal, human, and environmental health.
Materials and methods
We reviewed all avian diagnostic cases submitted to the Southeastern Cooperative Wildlife Disease Study (SCWDS) at the University of Georgia (UGA; Athens, GA, USA) from January 2001 to May 2023 and selected those with a final diagnosis of aspergillosis. Cases were comprised primarily of carcasses and occasionally field-collected tissue samples from wild birds submitted by state wildlife agencies and the U.S. Fish and Wildlife Service after being found dead or after dispatch (i.e., cervical dislocation or gunshot) due to morbidity. For cases that included multiple birds (i.e., from the same location and timeframe), we excluded individuals for which aspergillosis was not listed as a final diagnosis. We reviewed the case history, spatiotemporal data, signalment, manner of death, gross and microscopic lesions, and laboratory test results for all cases.
Birds were subjectively aged by assessing feather type, color, anatomic distribution, and other morphologic traits (e.g., mass, beak shape, beak/leg skin coloration)12,53 and by gross and/or microscopic observation of thymus and/or cloacal bursa. Based on the above traits, age was classified as chick, juvenile, or adult. Chicks were predominantly covered in downy feathers with a grossly obvious thymus and/or cloacal bursa. Juveniles had mostly mature feathers, but some immature feathers may have persisted. For these, the thymus and/or cloacal bursa were decreased in size or absent grossly, with microscopic evidence of thymic involution and/or bursal atrophy. Adult birds had only mature feathers and lacked a thymus and cloacal bursa. Degree of autolysis was assessed based on markers of decomposition (e.g., tissue color, texture, softening or liquefaction) and other associated findings (e.g., insect larvae deposition, postmortem gas accumulation), and was classified subjectively as minimal, mild, moderate, or severe. Nutritional condition was assessed subjectively via evaluation of celomic adipose tissue and skeletal musculature, and was classified as poor (minimal to no celomic adipose tissue with grossly appreciable muscle atrophy), fair (mildly to moderately reduced celomic adipose tissue and adequate muscling), or good (adequate celomic adipose tissue and muscling).
Tissue samples for each case were collected at the discretion of the diagnostician but were typically representative of all major organ systems. Samples were fixed in 10% neutral-buffered formalin, processed routinely, and sections were stained with H&E. Aspergillosis was diagnosed via histologic observation of characteristic fungal hyphae (3–12-µm parallel walls, septate, and acute-angle dichotomous branching) 23 in tissues, which often corresponded grossly to nodules, plaque-like lesions, or masses containing gray-green mold within or adhered to visceral organs or the celomic wall. For 3 cases in which only formalin-fixed tissue samples were submitted, aspergillosis was diagnosed solely based on microscopic observation of characteristic hyphae. Histochemical special stains, including Grocott methenamine silver (GMS) stain or a periodic acid–Schiff (PAS) reaction, were used to highlight hyphae in select cases.
Fungal identification via culture and molecular methods was performed in a subset of cases at the Athens Veterinary Diagnostic Laboratory (AVDL; UGA). Submitted tissue samples were comprised either of nodular or plaque-like lesions or samples of tissue in which hyphae were observed histologically. Samples were cultured on Sabouraud dextrose agar, with and without chloramphenicol (BD BBL), and incubated at 25°C for ≥7 d. Colonies suggestive of Aspergillus were mounted on slides and stained with lactophenol aniline blue for microscopic analysis (40× magnification). In some cases, gene sequencing was performed to confirm genus and species; we extracted DNA from lesioned tissue isolates grown on Sabouraud dextrose agar (ZR fungal/bacterial DNA MiniPrep; Zymo Research) for samples processed until 2020. From 2020, DNA was extracted using the EZ1&2 DNA kit (Qiagen). Conventional pan-fungal PCR was performed targeting the internal transcribed spacer (ITS) region and the D1/D2 region of the large subunit of the 28S ribosomal RNA gene.18,36 Aspergillus niger ATCC 16404 was used as a positive control. PCR products from both of the ITS and 28S ribosomal RNA genes were purified and sequenced based on the Sanger method at the AVDL (SeqStudio genetic analyzer; Thermo Scientific). BLAST analysis (https://blast.ncbi.nlm.nih.gov/Blast.cgi) and the CBS-KNAW fungal database (https://wi.knaw.nl/fungal_table) were used on each sequence to identify related fungal sequences.
Additional ancillary testing was performed on a case-by-case basis at the discretion of the diagnostician and supervising board-certified veterinary pathologist. Tests were conducted either in a research laboratory using validated methodologies or in a diagnostic laboratory accredited by the American Association of Veterinary Laboratory Diagnosticians using protocols established by the Clinical and Laboratory Standards Institute (https://clsi.org). Virus isolation and PCR assays were performed in selected cases for West Nile virus (WNV)6,62 and eastern equine encephalitis virus (EEEV). For EEEV, ~0.5-cm3 pooled samples of heart, brain, and kidney were processed for virus isolation as described previously.35,49 For virus isolation samples that exhibited cytopathic effects, viral RNA was extracted (QIAamp viral RNA mini kit; Qiagen) and tested for EEEV via RT-PCR with modified thermocycler conditions.26,42 PCR was performed on fresh tissue samples or swabs in selected cases for Wellfleet Bay virus (WFBV), 9 lymphoproliferative disease virus (LPDV), 2 circovirus (Infectious Diseases Laboratory, College of Veterinary Medicine, UGA), highly pathogenic avian influenza A virus (HPAIV), 63 trichomonads, 3 and Atoxoplasma spp. 14 Avian pox and Macrorhabdus ornithogaster were identified via histopathology. 46 In some cases, endoparasites and ectoparasites were identified based on gross and/or histologic morphology. Bacterial culture for aerobes, anaerobes, and Salmonella spp. were performed in select cases using standard protocols at the AVDL. Organochlorine and heavy metal testing were performed in select cases using standard protocols at either the New Bolton Center Laboratory of Toxicology (University of Pennsylvania, Kennett Square, PA, USA) or the California Animal Health and Food Safety (CAHFS) Laboratory (University of California–Davis, Davis, CA, USA). Anticoagulant rodenticide testing, liquid chromatography–mass spectrometry (LC-MS), and gas chromatography–mass spectrometry (GC-MS) were performed in select cases at the CAHFS laboratory. Brevetoxin testing was performed in select cases via ELISA using validated methodology at the Ecosystem Assessment and Restoration Section (Florida Fish and Wildlife Conservation Commission, Tallahassee, FL, USA). 19
Results
Animals
We identified 116 diagnostic case reports of aspergillosis in wild birds from January 2001 to May 2023; 29 diagnostic case reports included multiple birds. For those, individual birds not diagnosed with aspergillosis were excluded from our study. In total, 133 individual birds met our inclusion criteria and were selected for our study.
Signalment and taxonomy
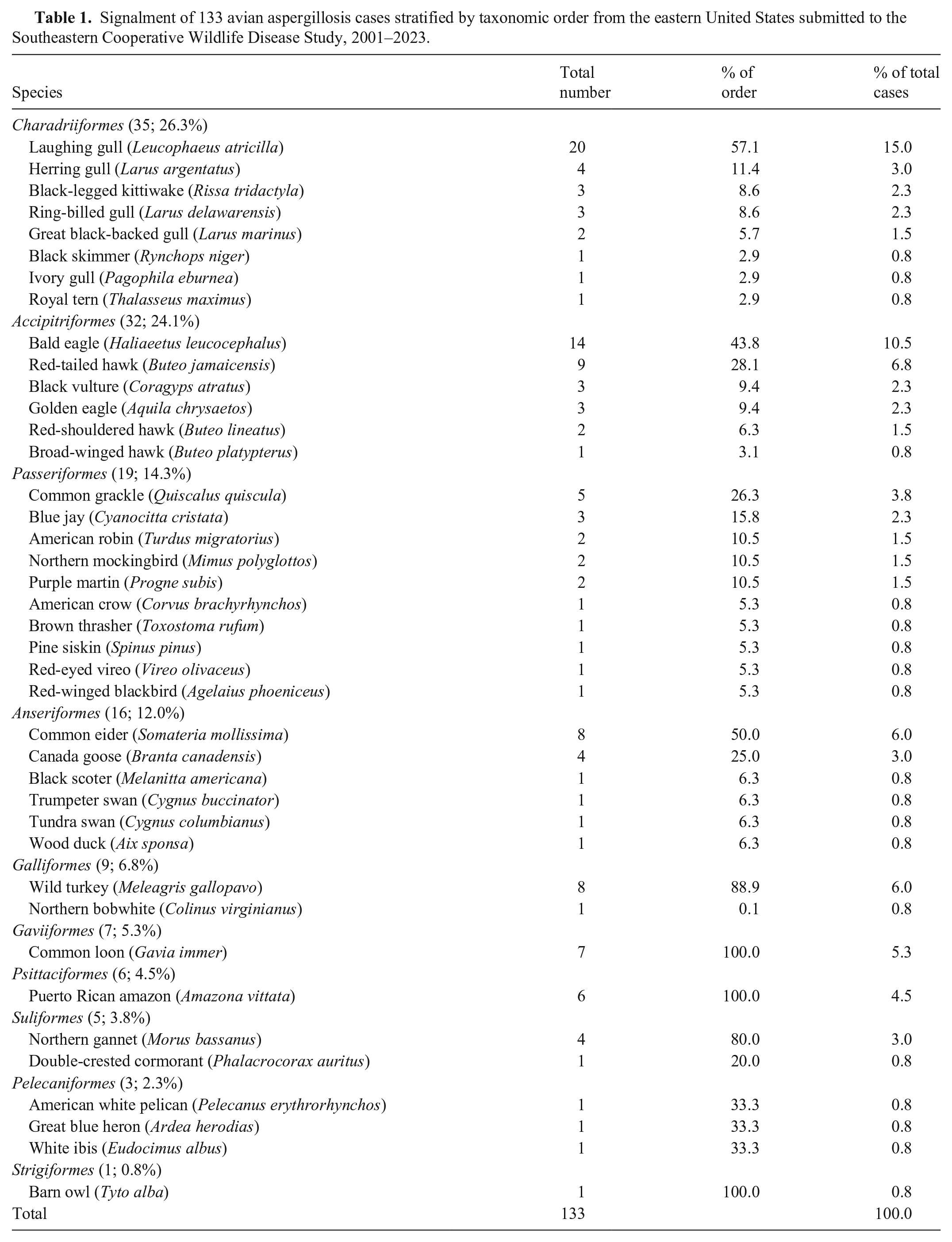
For the 133 birds with aspergillosis, 46 (34.6%) were identified grossly and/or histologically as female, 71 (53.4%) as male, and 16 (12%) as unknown. Life stage estimates were reported for all but 1 bird and included 71 (53.4%) adults, 55 (41.4%) juveniles, and 6 (4.5%) chicks. Forty-one species across 10 taxonomic orders were reported (Table 1), with the most frequent being the laughing gull (Leucophaeus atricilla; n = 20, 15%) and the bald eagle (Haliaeetus leucocephalus; n = 14, 10.5%). Additionally, 53 (39.8%) birds were part of mortality events. At the time of case submission, 13 (9.8%) birds had been held for ≤24–48 h within a rehabilitation setting (e.g., wildlife rehabilitator or veterinary hospital).
Signalment of 133 avian aspergillosis cases stratified by taxonomic order from the eastern United States submitted to the Southeastern Cooperative Wildlife Disease Study, 2001–2023.
Spatiotemporal distribution
The median number of avian aspergillosis cases diagnosed yearly from 2001 to 2021 was 5 (min. = 1; max. = 11; mean = 5.0). An increased number of wild birds submitted to SCWDS occurred in 2022, as well as an increased number of aspergillosis cases (n = 24), which coincided with an HPAIV outbreak in wild birds and poultry in North America. 22
Cases originated from 16 states and 1 U.S. territory. Most originated from southern states (Alabama, Arkansas, Florida, Georgia, Kentucky, Louisiana, Mississippi, North Carolina, South Carolina, Virginia, and West Virginia; n = 114). A small subset of cases originated from northeastern states (Maine, Massachusetts, Nebraska, and Pennsylvania; n = 12), a midwestern state (Missouri; n = 1), and Puerto Rico (n = 6).
Gross pathology
Gross findings were reported for 130 (97.7%) birds. The remaining 3 birds were submitted as formalin-fixed tissue sets. Lesions attributed to aspergillosis were described in 115 (88.5%) birds. Four distinct gross lesion patterns were described. Single or multiple dry, round, yellow-to-white plaques or masses (Fig. 1A) were reported most commonly (106; 92.2%). Firm, tan-to-white, hollow masses lined by gray-green mold on cut surface (Fig. 1B) were the second most commonly reported (26; 22.6%). Less common presentations included dark-brown plaques with a necrotic core (3, 2.6%; Fig. 1C) and regionally extensive dark-red pulmonary nodules (15, 13.0%; Fig. 1D). Typically, only 1 pattern was present in an individual bird; however, in a subset (33; 28.7%), 2 or rarely 3 patterns were described within a single bird. Gross lesions were described in 21 tissues (Table 2). Lung (80; 69.7%), air sac (71; 61.7%), celomic cavity wall (42; 36.5%), and heart (40; 34.8%) were most common.
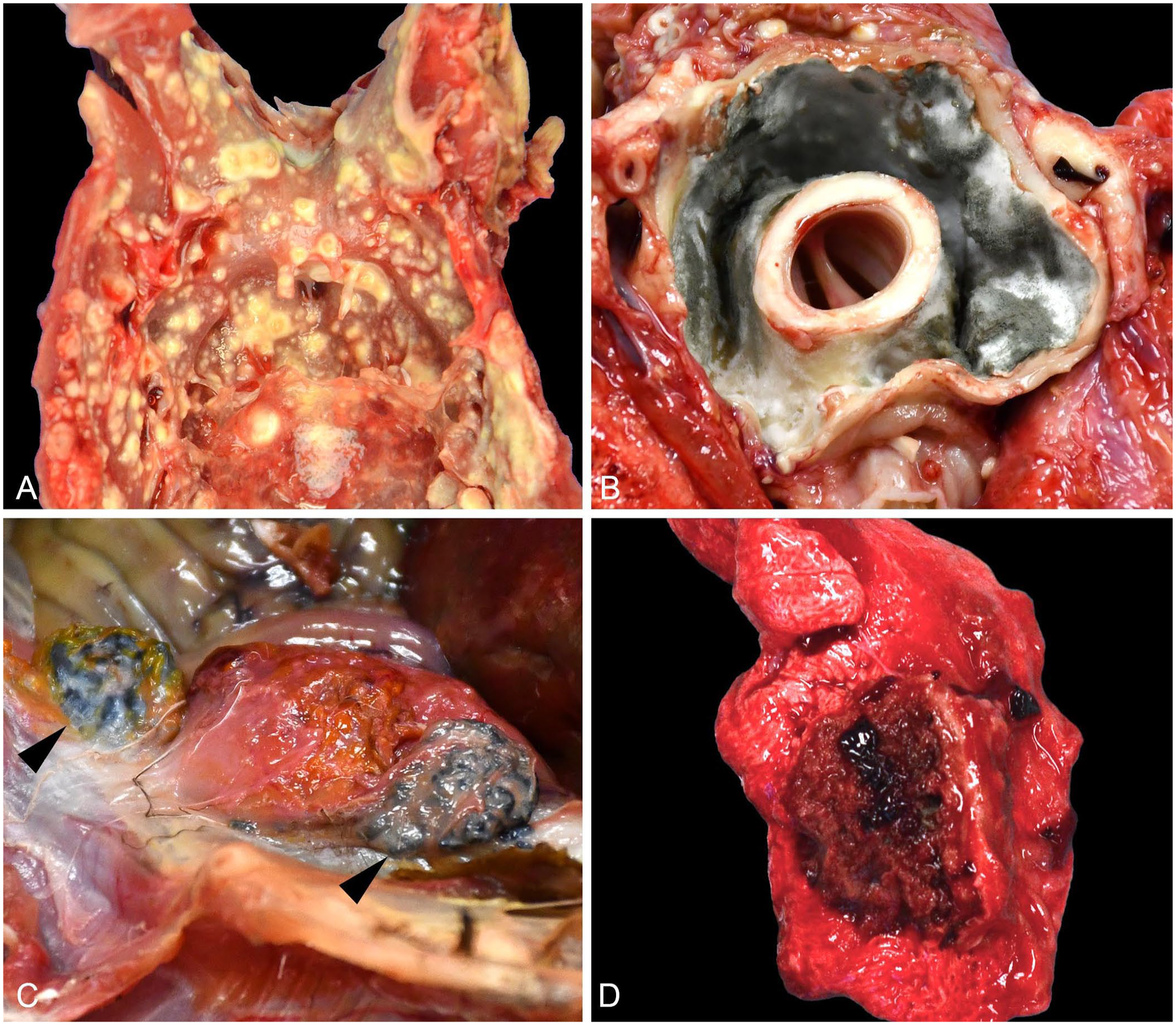
Gross lesions attributed to avian aspergillosis.
Anatomic distribution of gross lesions attributed to aspergillosis and histologic distribution of Aspergillus spp. fungal hyphae among 133 birds from the eastern United States submitted to the Southeastern Cooperative Wildlife Disease Study, 2001–2023. In some cases, limited autopsies were performed, and thus, all organs were not evaluated histologically in all cases.
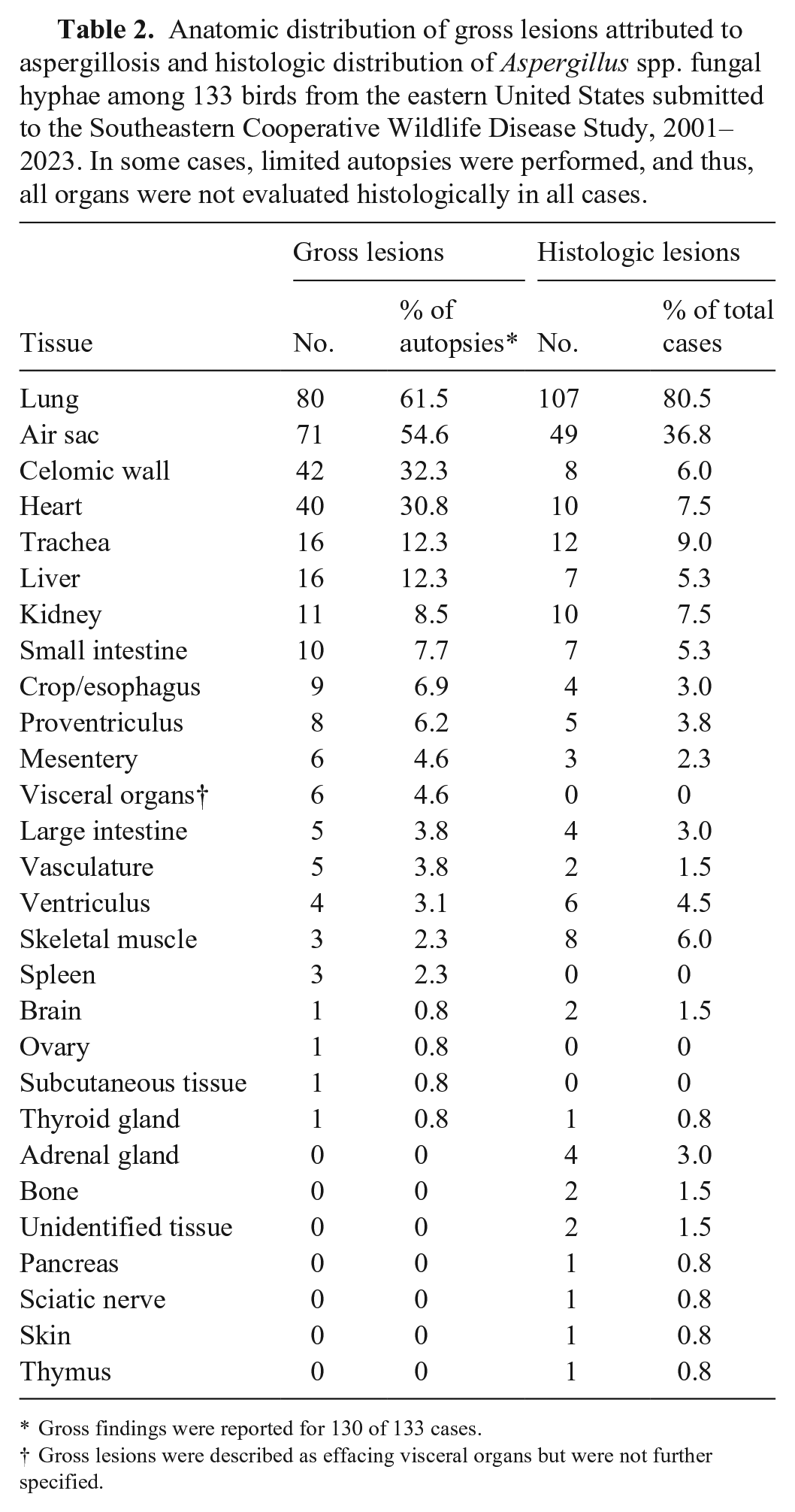
Gross findings were reported for 130 of 133 cases.
Gross lesions were described as effacing visceral organs but were not further specified.
Two birds had gross presentations unique from those mentioned previously. A wild turkey (Meleagris gallopavo) had a large subcutaneous inguinal mass that was surrounded by ulcerated skin, contained yellow-to-green caseous material on cut surface, and extended deep into the body-wall musculature. An American robin (Turdus migratorius) had a diffusely dark-red left cerebral hemisphere, in addition to firm, yellow nodules in thoracic air sacs.
The degree of autolysis was reported for 90 (67.7%) birds and consisted of minimal (6; 6.7%), mild (27; 30.0%), mild-moderate (6; 6.7%), moderate (34; 37.8%), moderate-severe (5; 5.6%), and severe (12; 13.3%). Nutritional condition was reported for 122 (91.7%) birds and consisted of poor (77; 63.1%), poor-fair (4; 3.3%), fair (27; 22.1%), fair-good (2; 1.6%), and good (12; 9.8%).
Histopathology
Among 133 birds, fungal hyphae were histologically observed in 25 tissues. Lung (107; 80.5%) and air sac (49; 36.8%) were affected most commonly. Hyphae were generally described as 3–10-µm diameter, septate, parallel-walled, exhibiting dichotomous acute-angle (“y-shaped”) branching, and having variably bulbous ends. Conidiophores were observed in 29 (21.8%) birds and were described as having hemispherical or flask-shaped vesicles, elongate phialides, and small round conidia (Fig. 2A). Histochemical special stains were applied for 32 (24.1%) birds. Hyphae stained black with a GMS stain (21, 65.6%; Fig. 2B) and magenta with a PAS reaction (11; 34.4%).
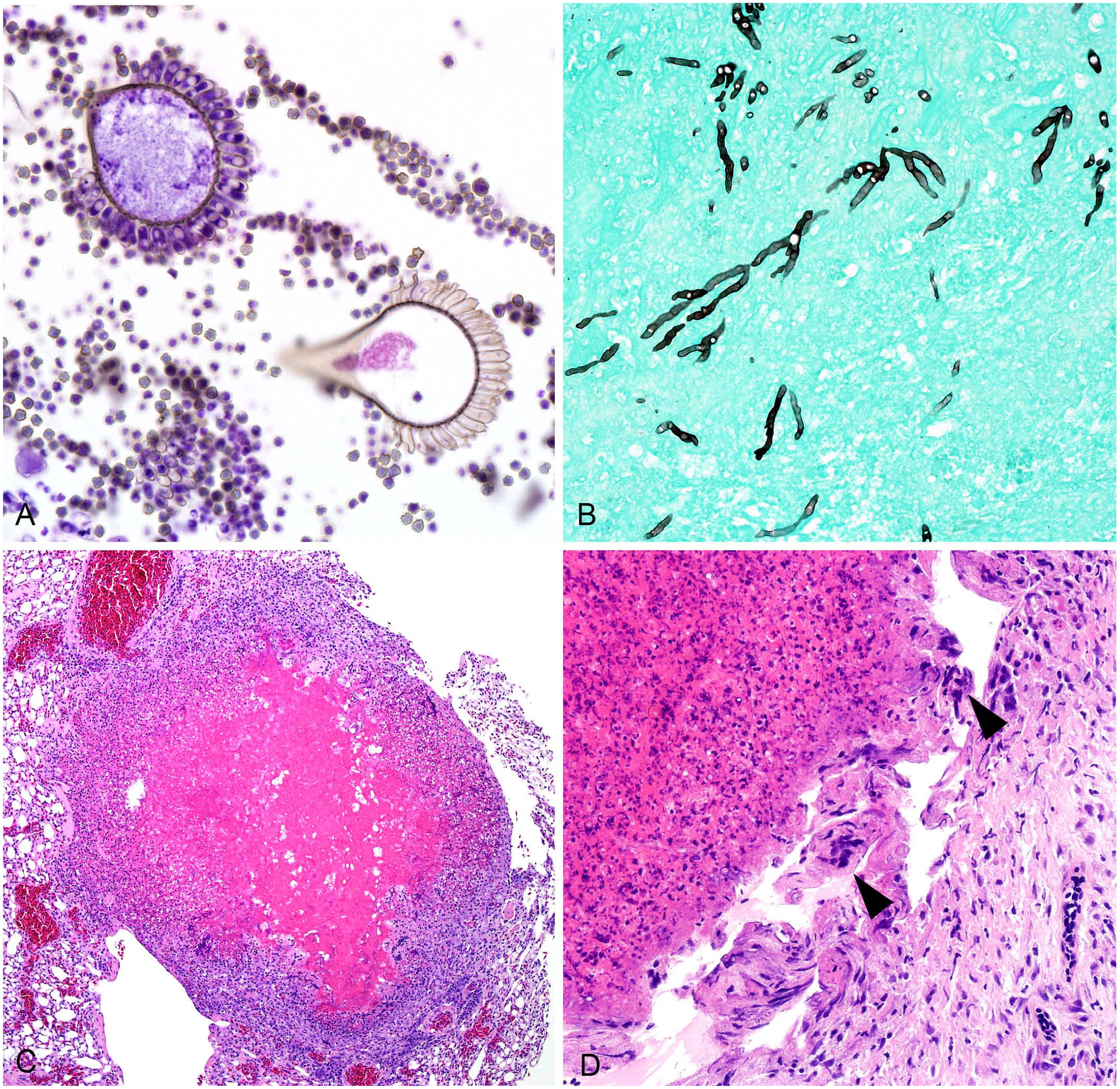
Heterophilic granulomas associated with avian aspergillosis.
A variety of histopathologic findings associated with fungal hyphae were described. Briefly, tissues were typically effaced by single or numerous heterophilic granulomas with central necrotic cores containing fungal hyphae and surrounded by concentric layers of necrotic heterophils, epithelioid macrophages, and sometimes multinucleate giant cells, with or without an outer fibrous capsule (Fig. 2C, 2D). In the lungs, these granulomas were frequently centered around, adjacent to, or occasionally invaded blood vessels. Heterophilic granulomas in non-respiratory tissues typically adhered to the serosal or capsular surface of the affected organ or to the lining of the celomic cavity. Depending on the affected tissue, other lesions, including edema, hemorrhage, mineralization, and thrombosis, were variably described in association with or adjacent to fungal lesions.
Fungal identification
Aspergillus spp. were identified in 45 of 54 (83.3%) birds for which fungal culture was completed. Sequencing for species identification was performed for 16 isolates. A. fumigatus was the most common isolate (13; 81.3%) followed by A. niger (3; 18.8%). Both A. fumigatus and A. niger were sequenced from the air sac of a red-tailed hawk (Buteo jamaicensis). In another red-tailed hawk and a bald eagle, both A. fumigatus and Mucor spp. were sequenced. In a common grackle (Quiscalus quiscula), Mucor spp. was sequenced. However, because Mucor spp. hyphae are traditionally considered morphologically distinct from those of Aspergillus spp., a false-negative result was suspected. In that case, both aspergillosis and mucormycosis were reported as final diagnoses, with the former diagnosis based on histopathologic findings. For 8 cases, fungal culture was attempted but resulted in no fungal growth, contamination, or growth of sterile mycelia.
Comorbidities
Comorbidities were diagnosed in 67 (50.4%) birds. Among them, 20 (29.9%) were diagnosed with multiple comorbidities. Both infectious (41; 61.2%) and non-infectious (50; 74.6%) etiologies were reported (Table 3). Infectious comorbidities included a spectrum of viral (19; 28.4%), bacterial (11; 16.4%), parasitic (6; 9.0%), and non–Aspergillus spp. fungal (4; 6.0%) agents. HPAIV was detected in 7 birds, all from 2022, mirroring the start and continuation of an outbreak in North America. The most common non-infectious comorbidities included emaciation (22; 32.8%), heavy metal or chemical exposure (11; 16.4%), and trauma (10; 14.9%).
Comorbidities and coinfections diagnosed among 133 cases of avian aspergillosis submitted to the Southeastern Cooperative Wildlife Disease Study, 2001–2023.

LPDV = lymphoproliferative disease virus.
Discussion
Aspergillosis has been reported in a wide variety of bird species but historically is designated principally as a disease of captive birds.5,8,24 Here we described 133 cases of aspergillosis in free-ranging birds representing 41 species across 10 taxonomic orders, each with their own unique geographic range, habitat, biology, and behaviors. The following taxonomic orders have been reported as most susceptible to developing aspergillosis: Anseriformes (waterfowl), Accipitriformes (eagles, hawks, vultures), Charadriiformes (shorebirds, gulls), Galliformes (landfowl), and Passeriformes (songbirds).8,15,50,52 These 5 orders were also the most frequent in our study, with Charadriiformes followed by Accipitriformes represented most commonly. Differences in susceptibility to aspergillosis among individual species or species-groups have been rarely described; however, data are heavily fragmented, and the incidence of disease in many species, especially among free-ranging birds, has not been characterized.8,40,41,47 Regardless, the vast diversity of species in our study firmly establishes aspergillosis as a general disease of birds, even in free-ranging populations.
Raptors are considered highly susceptible to respiratory fungal infections. Among this group, golden eagles (Aquila chrysaetos), goshawks (Astur atricapillus), gyrfalcons (Falco rusticolus), ospreys (Pandion haliaetus), red-tailed hawks, rough-legged hawks (Buteo lagopus), and snowy owls (Bubo scandiacus) are reportedly at high risk for aspergillosis.8,30 In our study, red-tailed hawks and golden eagles were diagnosed with aspergillosis, but among Accipitriformes, bald eagles were the most common, with approximately twice as many cases compared to red-tailed hawks and 5 times as many as golden eagles. However, this finding is likely biased by preferential submission of bald eagles to SCWDS for postmortem examination as a species of conservation concern. 45 A study in free-ranging swans reported that fungal lesions associated with aspergillosis were more common in males and in juveniles. 54 Swans in our study were poorly represented, with only one tundra swan (Cygnus columbianus) and one trumpeter swan (Cygnus buccinator) included, preventing assessment of demographic trends for this species-group. However, among all birds in our study, aspergillosis was diagnosed in similar numbers of males and females, and mostly occurred in adults and juveniles. The lack of chicks in our sample may be due to submission bias as the total number of chicks submitted to SCWDS tends to be substantially lower than the number of juvenile and adult birds submitted, possibly due to the inherent difficulties of identifying and recovering small carcasses of chicks. 45
Gross patterns of aspergillosis were consistent with those described previously 13 ; however, we found no clear association between a specific gross pattern and a particular species or taxonomic order. Additionally, we observed a fourth distinct pattern not reported previously, that of regionally extensive, dark-red pulmonary nodules. These gross lesions corresponded microscopically to the invasion of blood vessels by fungal hyphae. Angioinvasion is a common microscopic feature of Aspergillus spp., which can spread hematogenously or via macrophages.10,11 The variety of gross patterns, whether occurring singly or in combination, is also described in domestic poultry and is thought to be influenced by inoculation route, dose, and disease severity.7,11
Despite being classically considered a respiratory disease, lesions of aspergillosis can develop in any tissue.8,11 Among the wild birds in our study, gross lesions were observed in 17 different tissues, with a unique pattern in 2 cases. A wild turkey had a subcutaneous inguinal mass filled with yellow-green caseous material that corresponded microscopically to a fungal granuloma. Dermatologic lesions associated with Aspergillus spp. infection have been described sporadically in birds and include blepharitis and facial dermatitis in a peregrine falcon–gyrfalcon hybrid, 1 epidermal cysts within the comb of a silky bantam chicken (Gallus domesticus), 56 and subcutaneous fungal plaques within the head, neck, and wings of a Desi chicken. 4 The impetus for development of the subcutaneous mass in this wild turkey was not determined, although one potential cause may be that the mass developed secondary to a penetrating injury by something contaminated with environmental debris. In an American robin, an indentation in the skull overlaid a diffusely dark-red cerebral hemisphere that microscopically corresponded to a focus of neuroparenchymal necrosis effaced by myriad intra- and extra-vascular fungal hyphae. Cases of avian aspergillosis with brain lesions have been reported, but are rare.8,29 Blunt-force trauma to the head was suspected in this robin, followed by possible inoculation of the skull cavity with fungi either during the inciting trauma or through subsequent environmental contamination of the open wound.
Microscopic patterns attributed to aspergillosis in birds have been described, categorized, and/or graded in a variety of ways. Three patterns of pulmonary aspergillosis have been described: 1) acute hemorrhagic pneumonia, 2) subacute caseous fungal granulomas, or 3) chronic pneumonia with hepatization and complete loss of tissue architecture due to widespread granuloma formation. 39 A grading system was described for pulmonary lesions in which fungal hyphae surrounded by poorly circumscribed aggregates of macrophages and multinucleate giant cells were considered grade I; the more classic heterophilic granulomas were grade II. 47 Differentiation between respiratory and non-respiratory patterns are less common. Aerated tissues, such as lung, have been described as having superficial diffuse lesions, and non-aerated tissues, such as parenchymal organs, as developing deep nodular lesions. 13 Nevertheless, as demonstrated within our sample, microscopic lesion patterns can be highly variable and are likely influenced by factors such as infection chronicity, tissues affected, dose and route of inoculation, comorbidities, and species-specific anatomic variations, among others.
No histologic lesion patterns of aspergillosis were evident among tissue types or avian taxonomic orders within our sample population. Heterophilic granulomas were the most consistent histologic lesion and were routinely described in both respiratory and non-respiratory tissues. Other histopathologic findings, such as vascular invasion, edema, or mineralization, can be attributed to the pathologic behavior of Aspergillus spp. or to the typical response to tissue injury in birds. 34 Hemorrhage and thrombosis are recognized sequelae to angioinvasion,10,31 along with edema and mineralization as common responses to tissue damage.
In most of our cases, aspergillosis was diagnosed based on histologic observation of characteristic hyphae. Historically, this was considered sufficient to make a diagnosis.37,48 However, the accuracy of histopathology for identification of fungal organisms has been questioned.23,37,48,51 Misidentification based on histologic observation has been reported for fungi that are morphologically similar to Aspergillus spp., such as Fusarium spp. and Scedosporium spp. 51 In domestic mammals, mycoses attributed to these 2 fungi are grouped with various other nonpigmented fungi under the broad term hyalohyphomycosis and are sparsely reported in the literature.16,48 We retrieved no reported cases of hyalohyphomycosis via our search of Google Scholar, PubMed, CAB Direct, Web of Science, and Scopus using the search terms “hyalohyphomycosis”, “fusariosis”, or “scedosporiosis” in conjunction with “wild bird” or “free-ranging bird”, suggesting that this condition has not been reported in wild birds.
Misidentification based on histologic observation has also been reported in mammals for fungi traditionally considered to be morphologically distinct from Aspergillus spp., such as Mucor spp. 51 In a common grackle, for which only Mucor spp. were sequenced from culture, the hyphae observed histologically were septate, parallel-walled, and exhibited dichotomous acute-angle branching. Conversely, Mucor spp. hyphae are pauci-septate, non–parallel-walled, and exhibit irregular branching.21,48 Accordingly, a mixed condition of aspergillosis and mucormycosis was diagnosed and it was theorized that the lack of Aspergillus spp. growth in culture was a false-negative. Like Aspergillus spp. fungi, Mucor spp. are ubiquitous in nature and rarely associated with opportunistic infections in humans and animals. 43 Cases with mixed fungal infections can pose an even greater diagnostic challenge when using histopathology. 23 In our study, fungal culture and sequencing of isolates only revealed 3 cases of mixed infections: 1 with 2 Aspergillus spp. and 2 with A. fumigatus and Mucor spp. However, fungal culture and sequencing were inconsistently performed, and thus, the true prevalence of mixed infections in our sample is unknown.
In addition to aspergillosis, we diagnosed comorbidities in ~50% of the cases in our dataset. Specific diagnoses varied and most were reported in only one or a few cases, making comparisons within and among taxonomic orders challenging. This suggests that a broad array of disease entities or immunosuppressive factors (e.g., viral infection, stress) likely predispose wild birds to developing aspergillosis. Similarly, in domestic poultry, factors such as enclosure hygiene, stocking density, air renewal, and nutrition have been implicated as key factors in development of aspergillosis. 7 Emaciation was the most commonly diagnosed comorbidity, occurring either alone or in addition to other comorbidities, and was reported in at least 1 bird from 7 of the 10 taxonomic orders in our sample. This is a common diagnosis in free-ranging wildlife and tends to disproportionately impact immature individuals of some species. 33 However, many factors, including progressive fungal disease, can contribute to declining nutritional status and vice versa, and thus pathogenesis can be difficult to elucidate.
Traumatic injury concurrent with aspergillosis was reported in at least 1 individual in 5 of the taxonomic orders in our study but was most prevalent among Accipitriformes. Trauma is a common diagnosis among free-ranging raptors with many causes, including collisions with stationary objects (e.g., buildings), objects in motion (e.g., vehicles), or gunshot wounds. 44 Further, aspergillosis in wild birds coinciding with traumatic injury has been described, 8 but, similar to emaciation, determination of whether one predisposed to the other often is unknown. In 6 of our cases, it was thought that aspergillosis precluded traumatic injury due to the severity and chronicity of the fungal infection relative to the traumatic injuries, which occurred around the time of death. In a common loon (Gavia immer) in our study, aspergillosis developed subsequent to a puncture wound in the body wall around the site of injury.
Infectious comorbidities in birds with aspergillosis most often consisted of viral etiologies, which were most common in Accipitriformes, although this taxonomic order was disproportionately represented in our sample population. Coinfections with aspergillosis in birds have been described for HPAIV, 27 WNV, 60 EEEV, 59 avian pox virus, 28 WFBV, 9 and circovirus. 58 We retrieved no reported cases of Aspergillus and LPDV coinfection via our search of Google Scholar, PubMed, CAB Direct, Web of Science, and Scopus using the search terms “lymphoproliferative disease virus” and “Aspergillus” in conjunction with “wild bird” or “free-ranging bird”, suggesting that this association has not been reported in wild birds. Both birds infected with LPDV lacked lesions consistent with lymphoproliferative disease, and death was attributed to aspergillosis. However, the possibility of LPDV-induced immunosuppression is recognized, 2 and could predispose to other infections, including with Aspergillus spp. Bacterial disease was the next most common type of infectious comorbidity, and Salmonella spp. were most frequently implicated. In the 1970s, concurrent aspergillosis and Salmonella spp. infection (but lacking evidence of fulminant salmonellosis) occurred in common loons during an oil spill along both Florida coasts. 61
Elevated lead levels were detected in several bald eagles, a golden eagle, and a red-tailed hawk in our study. Lead exposure is commonly observed in free-ranging raptors, mainly occurring through ingestion of bullet fragments in prey, carcasses, or field-discarded offal, and is associated with metabolic and immunologic derangements.17,20 Cases of aspergillosis coinciding with lead toxicosis have been reported sporadically in Anseriformes and an Andean condor.38,54 However, paradoxically, swans with aspergillosis and lead toxicosis were less likely to develop aspergillosis than non–lead-poisoned swans, possibly due to rapid lead-associated death. 54
Our study limitations and biases are among those commonly encountered in retrospective studies utilizing diagnostic data. 55 Cases submitted for diagnostic evaluation were motivated, and thus biased, by public and state wildlife agency personnel interests at the time. Availability and ease of access to laboratory tests evolved throughout the study period. Likewise, the diagnosticians and pathologists changed over time and may have varied in their approaches and/or interpretations of cases. Additionally, with free-ranging wildlife, postmortem intervals are often lengthened due to the logistical challenges of carcass recovery, transport, storage, and submission to a diagnostic laboratory. Finally, the low sample size for many species and some taxonomic orders limited our ability to make comparisons within and between groups.
Footnotes
Acknowledgements
We thank SCWDS member states and U.S. Fish & Wildlife Service for their continued support, membership, and case submissions. Notably, the states of Alabama, Arkansas, Florida, Georgia, Kentucky, Louisiana, Massachusetts, Maine, Mississippi, Missouri, Nebraska, North Carolina, Pennsylvania, South Carolina, Virginia, and West Virginia as well as the U.S. territory Puerto Rico provided the case submissions included in our study. We also thank SCWDS staff, students, laboratorians, diagnosticians, and pathologists for their contributions to case completion, including Charlie Bahnson, Jennifer Ballard, Jeanenne Brewton, Justin Brown, John Bryan II, Caitlin Burrell, Katy Callaghan, Martha Frances Dalton, William Davidson, Brian Dugovich, John Fischer, Richard Gerhold, Chloe Goodwin, Nicole Gottdenker, Samantha Gibbs, Lisa Last, Brandon Munk, Rebecca Radisic, Mark Ruder, Aidan O’Reilly, Prachi Sharma, Nancy Stedman, Xuan Hui Teo, and Michelle Willis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by SCWDS member state wildlife agencies through the Federal Aid to Wildlife Restoration Act (50 Stat. 917), the U.S. Fish & Wildlife Service National Wildlife Refuge System, and U.S. Geological Survey Ecosystems Mission Area.
