Abstract
The skeletal system is a common site for neoplasia in dogs and cats, and primary bone tumors may develop from any of the mesenchymal tissues present in bone. Imaging and histopathology are routinely used in the diagnosis of bone tumors, and the 2 techniques are highly complementary. While imaging may be highly suggestive of a specific diagnosis and treatment may be instituted based on this, definitive diagnosis requires histopathology of either incisional or excisional biopsies or an amputation specimen. However, there are a number of diagnostic dilemmas when the pathologist interprets bone biopsy samples, such as distinguishing reactive bone and tumor bone, fracture callus and tumor bone, different benign fibro-osseous lesions, and different types of bone sarcoma. This review outlines the characteristic radiographic and histologic changes associated with these diagnostic problems to aid in resolving them. When a holistic approach is taken to evaluation of the signalment, history, and clinical, radiologic, and microscopic features, a diagnosis may be possible. The pathologist is greatly assisted in the interpretation of bone samples by having access to imaging and should routinely request either the images or the imaging reports if they are not received from submitting veterinarians.
Keywords
The skeletal system is a common site for neoplasia, and primary bone tumors may develop from any of the mesenchymal tissues present in bone. These tissues include bone, cartilage, adipose tissue, fibrous tissue, and blood vessels. 16 Importantly, the majority of primary bone tumors in dogs and cats are malignant. 50,51,64 In studies of neoplasia in insured dogs from the United Kingdom and Sweden, malignant bone tumors had a standardized incidence rate of 59/100 000 dogs/year and 55/100 000 dogs/year, respectively. 22,27 In the UK study, of all bone tumors reported, 73% were malignant, 0% were benign, and 27% were nonspecified. 22 Other papers have reported that 90% to 98% of primary bone tumors in dogs are malignant, 14,48,50 and 92% to 95% in cats. 51,64 Of these malignant tumors, osteosarcoma is by far the most common malignant bone tumor reported in both dogs and cats, with reported incidences of 67% to 87% 14,48,50 and 55% to 85% 51,64,87 of malignant bone tumors, respectively.
Imaging and histopathology are 2 highly complementary modalities commonly used in the diagnosis of skeletal disease, including bone tumors. Imaging may be plain radiographs; however, it is increasingly likely to involve more advanced imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI). While imaging may be suggestive of a specific diagnosis and treatment may be instituted based on this, definitive diagnosis requires either biopsy or amputation with histopathology. Cytology may also be a useful adjunct technique. In the authors’ experience, the interpretation of cytological smears and bone biopsies is greatly aided by the pathologist having access to radiographs or at a minimum a radiologist’s interpretation.
A common diagnostic decision for veterinary radiologists and pathologists is the question of whether the primary disease process affecting bone is inflammatory (reactive) or neoplastic and, then if the process is neoplastic, deciding whether the tumor is benign or malignant. This review will provide an overview of skeletal system imaging, and handling and processing of bone tumors, and then review the radiographic and histologic features of inflammatory bone diseases, benign and malignant neoplastic bone disease (osteosarcomas in particular), with the aim of assisting veterinary pathologists in answering common diagnostic dilemmas.
Diagnostic Imaging of the Skeletal System
Diagnostic imaging of the skeletal system in companion animals is frequently pursued to investigate lameness, wounds, palpable swelling, or neurologic abnormality. 97 Depending on initial findings, it is also used to assess treatment response, or for oncologic staging. 84 Accurate and early diagnosis is imperative to optimize patient outcome, and this requires correlation of imaging and pathology findings with clinical data. 40,48,55,97
Diagnostic imaging modalities used in companion animal practice include radiography, ultrasonography, nuclear scintigraphy, CT, and MRI. Radiography is readily accessible, relatively inexpensive, and the radiographic features of many skeletal diseases are well-reported. 48,50,51 Ultrasonography is also accessible but highly operator-dependent requiring experience of musculoskeletal ultrasound, and ultrasound systems in practice may not be optimized for the superficial nature of many bone neoplasms. Additionally, ultrasound utility is limited to evaluating soft tissues and the surface of the bone since shadowing at the bone-soft tissue interface precludes evaluation of the deeper structures within the bone, unless there is significant lysis. However, it may be useful to aid diagnostic sampling. 49 Nuclear scintigraphy with technetium-99m was once considered the standard of care for oncologic staging of bone tumors and metastases in many institutions; however, it demonstrates areas of active bone turnover and hyperemia, among other changes, and accordingly lacks specificity. 93 It has also been demonstrated to overestimate primary tumor margins. 47,84 Nuclear scintigraphy is also increasingly supplanted by more readily available imaging modalities such as CT or MRI, which have several beneficial features including higher spatial resolution. CT is increasingly available, with excellent spatial resolution and good contrast resolution, making it well suited to imaging of skeletal disease, metastatic screening, and biopsy or surgical planning. 6,49,93 Due to its tomographic nature, CT is not limited by tissue superimposition as is the case for radiology. MRI is excellent for identifying abnormalities of soft tissues as well as bone edema, and is considered the modality of choice in many orthopedic diseases including neoplasia in humans. 93 MRI is less frequently used as a first-line cross-sectional imaging modality in veterinary patients with suspected neoplasia of the appendicular skeleton: although the technique is very sensitive, the findings may not be specific, and the body area that can reasonably be imaged is smaller than that of CT, making it less useful as a screening modality. MRI is also the most expensive of these modalities, and usually requires general anesthesia in veterinary patients, which further restricts its utility. 49
An individual’s approach to diagnostic imaging assessment of osseous abnormalities may vary, but regardless of that approach, it requires a thorough evaluation of lesion location, extent, and morphology. While imaging findings in many disease processes overlap, these observations are interpreted in light of patient history and signalment, as well as any other imaging findings including those identified in imaging of the contralateral limb for comparison, as well as thoracic, abdominal, or draining lymph node imaging for metastasis in cases of suspect or confirmed neoplasia. 40,56,97 A prioritized differential diagnosis list can then be generated, with the goal of aiding clinical decision making. Given the divergence of treatment protocols and prognosis between inflammation of infectious or noninfectious causes and neoplastic disease, this review will focus on dividing reported imaging findings into these categories. Because the cytologic and histologic appearance of various bone tumors may overlap, we will then discuss ways that imaging can help differentiate benign from malignant tumors, and osteosarcoma from other malignant skeletal neoplasms.
Collection of Samples for Histopathology and Cytopathology
The ability to make a diagnosis of the cause of skeletal lesions is greatly enhanced by the pathologist receiving a good-quality sample. Advice is frequently sought from pathologists on how to obtain a good-quality bone biopsy from suspected tumors. Whether a bone biopsy is required should be carefully considered, as it may not be necessary if the signalment and clinical signs are suggestive of, and radiographic appearance is consistent with a primary bone tumor, particularly in countries where fungal osteomyelitis is uncommon. Similarly, a bone biopsy may not be indicated if the owner opts for either no treatment or palliative amputation regardless of the diagnosis, or if imaging suggests that a bone biopsy could precipitate pathologic fracture. 17 Bone biopsy is most useful if radiographic changes are not classical, or if a definitive diagnosis is required for determining future treatment options. 96 If limb-sparing surgery is to be performed, bone biopsy should be avoided or the position of the biopsy should be planned so that the biopsy tract can be removed en bloc at the time of surgery. 49 In human medicine, one study found that poorly planned biopsies resulted in unnecessary amputation in up to 10% of cases. 53 When positioning for the location of bone biopsies, clinicians should be guided by radiographs (2 views) and anatomic landmarks, or directly by CT or MRI, ultrasound or fluoroscopy. Post-biopsy radiographs are recommended to ensure correct placement of the biopsy needle. Alternatively, ultrasound- or CT-guided biopsy may assist the clinician in determining with confidence that the appropriate area of the tumor has been sampled. 46
Biopsy may not be necessary if a diagnosis is able to be obtained from fine needle aspiration and cytopathology. Cytopathology has the advantage of a rapid turnaround time with minimal risk of complications. It may be performed using ultrasound guidance to pass the needle through a region of cortical lysis. In one study, cytology of bone lesions in dogs was shown have an accuracy of 83%, similar to the 82% accuracy for histology of biopsies in distinguishing between benign and malignant bone disease (when compared with histologic examination of limb-sparing or amputation samples). 71 Furthermore, tumor type was successfully identified in 50% of cytopathology cases and 55% of histopathology cases. 71 Similarly, a study on ultrasound-guided fine needle aspiration of aggressive appendicular bone lesions found that cytopathology had a sensitivity of 97% and specificity of 100% for diagnosing a bone sarcoma. 13
Large specimens, such as from limb-sparing surgery or after amputation, should be sent whole and chilled to the diagnostic laboratory as soon as possible, rather than placing into fixative. They should not be frozen if it can be avoided. Amputations and large resections will not fix adequately in 10% neutral buffered formalin and need to be processed further as soon as received at the diagnostic laboratory. Rubin et al 69 and Mangham and Athanasou 52 provide excellent and very detailed protocols for specimen handling, examination, processing, and reporting of bone tumors in humans. Specimen radiographs may assist with sampling appropriate sites.
Is the Disease Process Reactive or Neoplastic?
While the radiographic appearance of primary and metastatic bone tumors has been well described in both cats and dogs, CT and MRI findings are more recently reported, and a wide range of imaging findings are recorded for both neoplastic and inflammatory conditions. 1,36,38,48,50,51,73,77,97 It is not surprising that the imaging findings for many osseous disease processes overlap, given the finite ways in which bone can react to disease. 26,40,93 Despite this overlap, the imaging findings associated with some diseases are characteristic when combined with history and signalment, and index of suspicion for many other diseases can be narrowed depending on these factors. Imaging may also be used to assess the need for biopsy, or assist in guiding the biopsy as described above. 97
Imaging assessment of osseous abnormalities requires thorough evaluation of lesion location, extent, and morphology. These observations can be used to classify any finding as an aggressive or benign bone lesion from an imaging perspective. 26 This information is then interpreted in light of patient history and signalment, as well as any other imaging findings such as thoracic or contralateral limb studies. The differentiation of inflammation of infectious or noninfectious causes or neoplastic disease can be made with a varying degree of confidence depending on these factors, although very few lesions can be given a single definitive diagnosis on the basis of imaging. 97
Lesion morphology is used to assess how biologically aggressive a lesion appears. Simply put, if the appearance is ill-defined or poorly delineated it generally represents more rapid change, and is therefore more likely to be associated with aggressive disease—be that malignant neoplasia or osteomyelitis. This is true regardless of what facet of a lesion is being assessed—whether bone production or loss. Conversely, if the margins of any new bone or bone loss are well-defined or well-delineated, the osseous change has generally been a slower process, and is usually toward the benign end of the spectrum—for example, a bone cyst, fracture callus, or benign tumor. This generalization applies to periosteal reaction, medullary or cortical osteolysis, and the zone of transition between normal and abnormal bone. The length of the zone of transition also varies, and a short zone of transition is usually associated with benign disease, while a long zone is indicative of a more aggressive process. 93,97
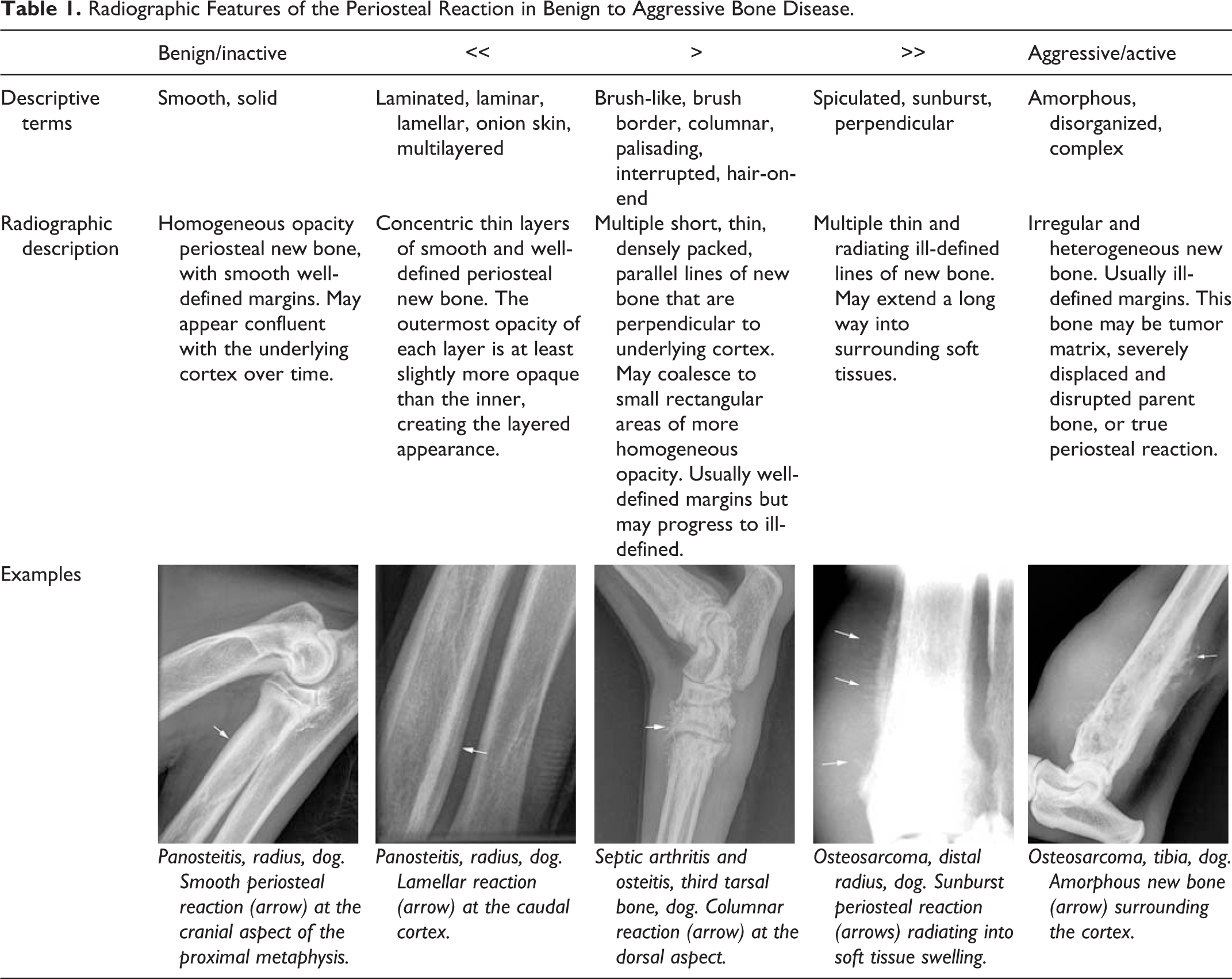
Exact terminology used to describe morphology of osteolysis or new bone production has varied over time and between authors. 26,93,97 Regardless of terminology, bone production or destruction occurs along a spectrum, varying with lesion duration, rate of change, and cause—whether neoplastic or inflammatory. Tables 1 and 2 explain some descriptive terms used in the veterinary and human medical literature, along a sliding scale of benign to aggressive appearance. 26,76,93,97 Where periosteal reaction is concerned, this scale can also be termed “inactive” to “active.” 93,97 The tables include representative images in an effort to reduce ambiguity in description. It is important to note that while multiple different morphologies of new bone or of osteolysis may be present within a single lesion, from an imaging perspective, the most aggressive characteristic is used to determine the lesion ranking (Table 3). A range of synonymous terms are included to aid in interpreting radiology reports, particularly for instances when access to the diagnostic imaging study is not possible.
Radiographic Features of the Periosteal Reaction in Benign to Aggressive Bone Disease.
Radiographic Features of Bone Loss in Benign to Aggressive Bone Disease.
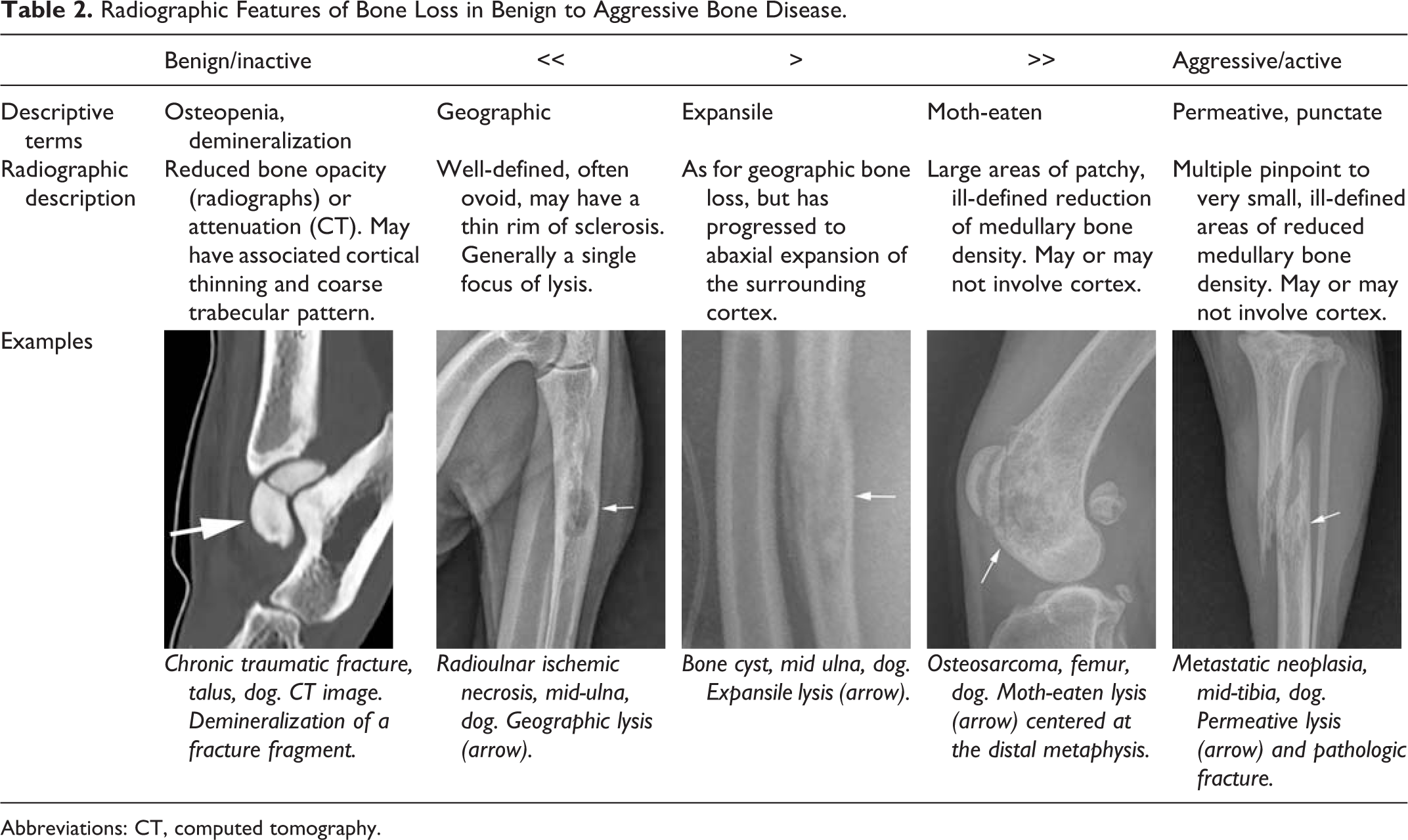
Abbreviations: CT, computed tomography.
Key Features of a Radiographically Aggressive Bone Lesion.a

a It should be noted that the most aggressive feature evident is used to rank the lesion along the scale from benign to aggressive.
Radiographic findings in addition to those described in Tables 1 and 2 include Codman’s triangle, expansile bone lesions, and cortical defects. Codman’s triangle is a triangular area of solid periosteal reaction, generally seen at the margin of a lesion. Historically, Codman’s triangle was considered indicative of osteosarcoma; however, it is nonspecific and may occur with any process that results in rapid lifting of the periosteum, ranging from traumatic hemorrhage to osteosarcoma. 93,97 Expansile bone lesions are thought to arise by a combination of endosteal lysis or pressure resorption, and periosteal reaction resulting in abaxial cortical expansion. 93,97 Expansile bone lesions range from aneurysmal bone cysts to malignant neoplasia, 8,28,74,97 and the finding is therefore of low utility in differentiating benign from aggressive disease.
Cortical bone defects may occur due several reasons, including pathologic fracture, pressure resorption and atrophy, or osteolysis. While the morphology of cortical lysis can be categorized into the above-mentioned types of bone loss, identification of reduced cortical thickness raises concern for an aggressive bone lesion, and should be noted prior to biopsy in order to minimize the risk of pathologic fracture. It is similarly important to identify pathologic fracture prior to sampling, as biopsy of any callus developing as a result of the pathologic fracture may be nondiagnostic for the primary lesion. Additionally, fracture callus may share a number of microscopic characteristics with osteosarcoma making definitive diagnosis on a small biopsy challenging. As such, fracture callus may explain a concerning biopsy result in an otherwise benign-appearing lesion. For these reasons, identification of full-thickness cortical bone loss is crucial when viewing or reporting imaging findings. Cortical lysis originating from the periosteal surface in the absence of endosteal lysis occurs with regional invasion of extraskeletal soft tissue neoplasia, and parosteal and periosteal osteosarcoma. 97 Conversely, endosteal lysis is reported with primary malignant bone neoplasia, round cell neoplasia, and metastatic neoplasia, but may also be seen in cases of osteomyelitis 97 and is therefore a nonspecific but aggressive finding.
The rate and type of change of an osseous lesion may also be instrumental in reaching a diagnosis, or at least assessing biological aggressiveness. 26,93 Generally, a rapid rate of osseous change is indicative of aggressive disease, with probable diagnoses being malignant neoplasia or osteomyelitis. Characteristic patterns of progression may also yield a presumptive diagnosis: lesions of panosteitis and metaphyseal osteopathy may regress, whereas those of early malignant neoplasia may develop “aggressive” imaging characteristics (Table 3). 11,40,62 Identifying these changes over time can reduce the requirement for bone biopsy.
Lesion location within the body as well as within a bone is used to narrow and rank differential diagnoses based on reported predilection sites for both inflammatory and neoplastic diseases. For example, primary malignant bone neoplasia is usually centered at metaphyseal regions of long bones, whereas metastatic neoplasia is more frequently diaphyseal in location. Likewise, though the type of osteolysis and new bone production that occurs with fungal osteomyelitis may appear similar to osteosarcoma, it is more likely to be diaphyseal, polyostotic, and accompanied by tracheobronchial lymphadenopathy. 97 Interpretation of imaging findings must also be completed in light of patient signalment and history where available. Species, age, and breed incidences vary for many diseases with associated osseous change, and is well reported in some instances. 25,50,51,59,62,65,67,73,91,95 A wide variety of disease processes could produce a biopsy specimen of reactive bone depending on biopsy location, but when combined with complete signalment and history, these characteristics can be used to rank or even exclude initial differential diagnoses generated by either modality alone. For example, while fungal osteomyelitis and malignant neoplasia are the two most common differential diagnoses for an aggressive bone lesion, fungal osteomyelitis may be ruled out based on geographic location and travel history. It may alternatively be ranked higher than malignant neoplasia based on previous positive serology, urine antigen testing, or direct observation of the fungus in a biopsy sample.
Diagnostic Imaging Challenges in Determining Reactive From Neoplastic Disease
Defining a lesion as reactive versus neoplastic from an imaging perspective requires incorporation of all of the above-described factors in a logical order. Differential diagnoses can be categorized as inflammatory/infectious or neoplastic categories and it sometimes straightforward to rank the the morphology seen with initial imaging, but in other cases this is impossible. Such challenges frequently arise with early aggressive lesions and rapidly developing inflammatory or infectious lesions. Interpretation in light of history, signalment, and lesion location then becomes of heightened importance. Despite this combined approach, signalment, clinical signs, and lesion morphology may remain nonspecific in some cases. In this situation, a choice can be made between bone biopsy and radiographic surveillance. Though a definitive diagnosis requires biopsy, surveillance can be used as a noninvasive way to document progression or regression of aggressive imaging characteristics. 11 Surveillance can thereby be used to indicate positive response to a trial treatment (more likely with a true reactive process) versus progression of aggressive disease (eg, malignant neoplasia). Surveillance can likewise prove useful in cases where a biopsy results in a microscopic diagnosis of reactive bone, despite the presence of an aggressive or borderline-aggressive bone lesion from an imaging perspective.
The axial skeleton presents additional challenges in interpreting radiographic studies of bone lesions. For primary neoplasia such as osteosarcoma, the reported imaging characteristics vary more in the axial than the appendicular skeleton. 48 For example, osteosarcoma in the skull may have a smooth margin, and amorphous/productive tumor bone may not be recognized in osteosarcoma of the skull or vertebrae. Furthermore, there are additional location-specific lesions that may give the appearance of aggressive bone disease. One such example is monostotic or polyostotic aggressive lesions adjacent to intervertebral disc spaces, as these are most likely due to discospondylitis. 97 Additionally, radiographs of the axial skeleton are generally more difficult to interpret due to the anatomically complex structures of the skull and spine, and superimposition of thoracic and abdominal soft tissues on the spine in a dorsoventral plane. CT or MRI are therefore frequently utilized to increase sensitivity of detecting lesions in the axial skeleton, and these modalities may also identify factors that affect prognosis and treatment options such as intra-cranial or vertebral canal involvement.
Reactive Bone Versus “Tumor Bone”
The question of whether the bone in a biopsy is reactive or neoplastic is fundamental to the interpretation of bone samples, but also at times is a difficult question to answer. Reactive bone is formed by non-neoplastic osteoblasts, while tumor bone (sometimes also called tumor osteoid) is formed by neoplastic osteoblasts.
Bone responds to a number of different insults in a similar and often limited fashion, and this can make it challenging to determine the primary disease process, particularly on small biopsies. The clinician and pathologist must accept that definitive diagnosis from a small biopsy is sometimes not possible. However, when the biopsy is interpreted with a full history, clinical examination findings, and diagnostic imaging, it may be that a diagnosis can be made. Additionally, a diagnosis is only as good as the sample; it therefore requires that the biopsy is representative of the lesion as a whole. As discussed above, a common reason for obtaining a nondiagnostic sample is sampling only periosteal reactive bone rather than the medullary cavity.
The main difficulty in distinguishing between neoplasia and other disease processes in bone is that periosteal and endosteal new bone formation (reactive bone formation) is a common feature of many disease processes. Reactive bone may form as a result of physical elevation of the periosteum from a tumor or inflammatory exudate, mechanical weakness (ie, compensatory new bone formation), fracture, altered circulation such as periosteal hyperemia, or growth factors produced by tumors. If a bone biopsy consists solely of periosteal new bone, then a definitive diagnosis based on histopathology is not possible. But in practice, the more common question in the authors’ experience derives from this: “Is this reactive bone or so-called tumor bone produced by an osteosarcoma?”
The key microscopic feature of reactive bone formation is that of well-ordered maturation. Initially, there may be small spicules of woven bone that can appear similar to tumor bone in the early stages; however, the spicules of (reactive) woven bone are interconnected and become organized into thicker trabeculae. While in some cases the spaces between trabeculae may be loosely filled with undifferentiated mesenchymal cells, these cells mature to become a single layer of plump osteoblasts lining the bone trabeculae. There should not be large clusters of osteoblasts in reactive bone. The mesenchymal cells and obvious osteoblasts are relatively bland, and there are no atypical mitoses. As the bone trabeculae become thicker, osteocytes become encased in slit-like bone lacunae at regular intervals. One of the keys to recognizing this well-organized maturation is to observe that woven bone spicules are attached to mature lamellar bone; thus, the biopsy must be oriented correctly, or the biopsy needle must fully penetrate through the cortex instead of just obliquely sampling the cortex.
Tumor bone in an osteosarcoma, like reactive bone, is composed of woven bone. However, the key feature of tumor bone is that the trabeculae lack the orderly maturation that occurs with reactive bone. Trabeculae do not interconnect in a well-structured manner and are often not connected to preexisting lamellar bone. Trabeculae of tumor bone are not lined by a single layer of plump osteoblasts. Instead, the spaces between trabeculae of preexisting mature lamellar bone are filled with pleomorphic mesenchymal cells, between which there may be fine ribbons of osteoid or larger unorganized islands and clumps of osteoid that show no evidence of appropriate maturation. The color of tumor osteoid may vary from the glassy pale eosinophilic color seen particularly with fine strands of osteoid, to more basophilic immature trabeculae and islands of woven bone. The basophilia may be globular to more diffuse and is thought to be associated with bone matrix that is less mature and contains a greater amount of noncollagenous bone proteins such as proteoglycans and osteocalcin. 92 Neoplastic osteoblasts or less well-differentiated mesenchymal cells may be highly variable, with frequent and often atypical mitoses.
Periosteal and endosteal reactive bone formation differ in appearance depending on the rate at which the new bone is formed. Endosteally, if a tumor or inflammatory lesion is slow growing, then lamellar bone is deposited on preexisting trabecular surfaces resulting in thickening of the trabeculae. 5 Alternatively, if a tumor or inflammatory lesion is fast growing, then woven bone may be deposited perpendicular to trabecular surfaces forming a web of woven bone in the space between trabeculae. 5 Periosteal reactive bone forms perpendicular to the surface of the cortex and the new bone trabeculae are thickest close to the cortex and thinnest at the leading edge directly beneath the periosteum. If the lesion is slow growing, the periosteal reaction is continuous along the cortex, and the cortex may appear thickened when thick interconnecting trabeculae predominate and merge with the cortex. Alternatively, if the disease process is rapid, then the periosteal reaction is interrupted, and long, thin spicules of woven bone predominate.
In osteosarcoma, both reactive and tumor bone are likely to be present, so the presence of identifiable reactive bone does not rule out the concurrent presence of tumor bone. To add additional complexity to the decision, if the osteosarcoma has breached the cortex, then tumor cells with tumor bone may be present between trabeculae of reactive bone.
The confidence with which reactive bone and tumor bone can be distinguished is greatly enhanced by the pathologist having access to radiographs showing the site of planned biopsies, or even better radiographs taken after the biopsy. However, not all pathologists have sufficient experience examining radiographs, and as such greater interdisciplinary interactions during both pathology and imaging residencies would be advantageous. As a minimum, this would assist each discipline in understanding the limitations of each technique.
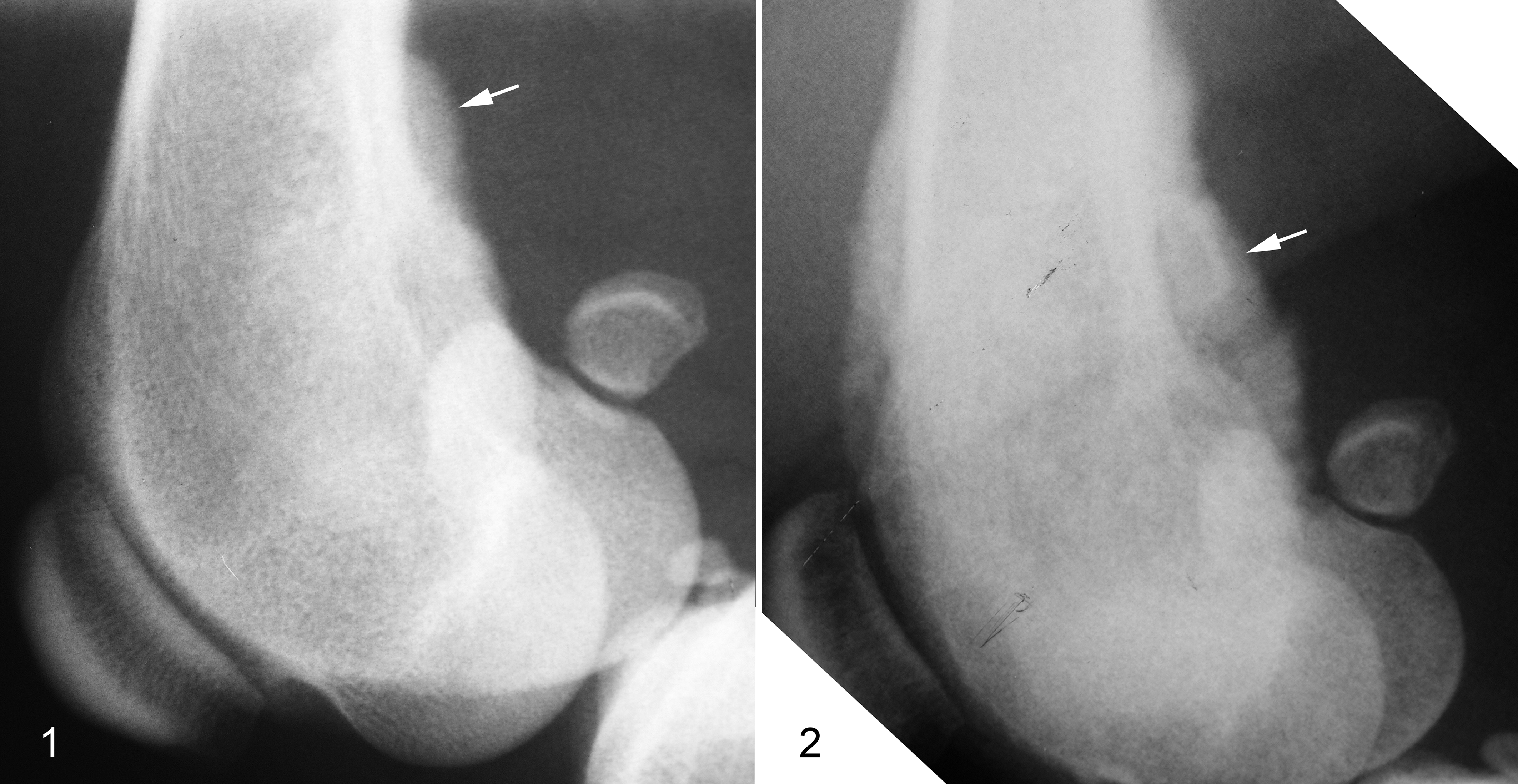
These issues are illustrated by a case involving a 5-year-old German Shepherd dog with a several-week history of left hindlimb lameness (Figs. 1 –8). Radiographs taken 1 month apart showed the utility of repeated radiographs to identify the rapid progression of an aggressive bone lesion (Figs. 1, 2). One biopsy sample from the distal femur consisted predominantly of necrotic bone (Fig. 3). A second sample consisted solely of reactive bone with interconnected spicules of woven bone and organization into thicker trabeculae (Fig. 4). However, between some mature trabeculae of bone were lacy eosinophilic strands of osteoid consistent with tumor bone (Fig. 3). A presumptive diagnosis of osteosarcoma was based on the presence of this osteoid, together with the radiographic identification of a rapidly progressive and aggressive bone lesion. Cytopathology of the limb at necropsy was highly suggestive of osteosarcoma in this case, based on the presence of mesenchymal cells with evidence of malignancy, as well as alkaline phosphatase activity in the neoplastic cells based on 4-chloro,3-indolyl phosphate/nitroblue tetrazolium (NPT/BCIP) staining (Fig. 5). Histopathology of the affected limb after necropsy identified both reactive endosteal bone and malignant mesenchymal cells at the invading front of the tumor (Figs. 6, 7). In some areas, even though neoplastic cells were present, the adjacent trabeculae were clearly lined by a single layer of plump reactive osteoblasts, confirming them to be reactive bone (Figs. 6, 7). However, in another area, atypical osteoblasts produced fine glassy spicules of tumor bone, consistent with osteosarcoma (Fig. 8).
Osteosarcoma, distal femur, dog (5-year-old German Shepherd).
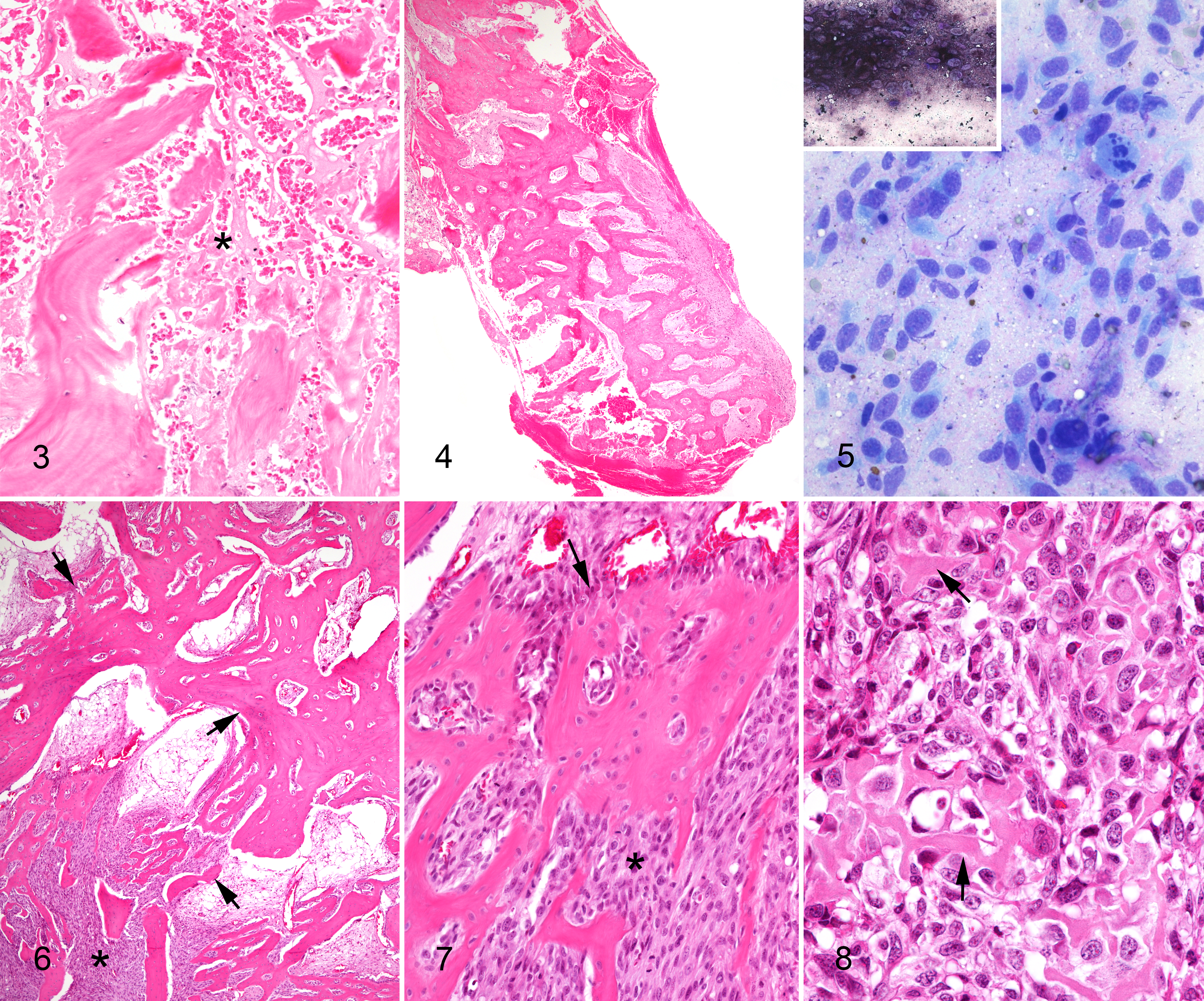
Osteosarcoma, distal femur dog.
Fracture Callus Versus Tumor
Another possible diagnosis for a biopsy containing mesenchymal cells producing different types of matrix is a fracture callus, in particular an immature fracture callus. Depending on the stage of fracture healing, the predominant cell type may be fibroblastic, chondroblastic, or osteoblastic, and therefore fibrosarcoma, chondrosarcoma, and osteosarcoma could all be differential diagnoses. This diagnostic conundrum is further complicated by the fact that pathological fracture is common in bone tumors that have obliterated normal bone structure.
As with reactive bone, the major feature of a healing fracture is the organized maturation of matrix produced by mesenchymal cells. In early lesions (approximately 1 week post-fracture), a healing fracture consists predominantly of hemorrhage, necrosis, inflammation, and granulation tissue. 20,40 In laboratory animal models, small tendrils of woven bone may start to be seen as early as 1 week post-fracture, cartilaginous matrix may be seen by 10 days post-fracture, and distinct trabeculae of woven bone are present by 2 weeks post-fracture. 20,40 After 2 weeks post-fracture, the cartilage matrix is sequentially replaced by woven bone in a zonal manner starting from the outside of the bone and moving inwards, 20 and it is this maturation sequence that is key to distinguishing the immature, mitotically active mesenchymal cells in a fracture callus from neoplastic cells.
This description of fracture repair applies to the healing of experimentally induced fractures in animal models. While the overall general process of repair can be applied, it is unlikely to be directly analogous to that seen in cats and dogs in veterinary practice. Excessive movement at the fracture site, necrosis of comminuted bone fragments, and infection are common in veterinary patients, complicating the process of fracture repair. Additionally, in a small biopsy, it may not be possible to appreciate the diagnostic indicators of fracture repair: the transition from immature cartilage matrix and bone to wider trabeculae of woven bone. Therefore, other features suggestive of a fracture callus include reactive osteoblasts with abundant cytoplasm and mild anisokaryosis; the cells in a fracture callus do not show marked pleomorphism as would occur in osteosarcoma. Additionally, while osteoblasts in a fracture callus are mitotically active, atypical mitoses are not a feature as they would be in osteosarcoma. Trabeculae of woven bone should be lined with a single layer of osteoblasts, and while the spaces between trabeculae may be filled with mesenchymal cells, the tissue is a mix of vascular and fibroblastic cells consistent with granulation tissue, but this tissue is not highly cellular and does not contain a fine stream of woven bone as would occur in an osteosarcoma. 40 Muscle and fat may be present, trapped within the fracture callus. Necrotic cortical and trabecular lamellar bone may also be surrounded by granulation tissue, but this can also occur in osteosarcoma, when preexisting lamellar bone is imprisoned by the infiltrating neoplastic mesenchymal cells. However, in a repairing fracture, there should be remodeling of necrotic bone as evidenced by the presence of osteoclasts or resorption lacunae, and woven bone may be laid down on top of the pre-existing trabeculae. 40
The distinction between fracture healing and osteosarcoma is illustrated by a case involving a 6-year-old Greyhound dog that presented with a pathologic fracture of the proximal humerus resulting from osteosarcoma (Figs. 9 –12). New trabeculae of woven bone were present that were consistent with reactive bone, because they were lined by a single layer of mesenchymal cells, and osteocytes were being incorporated into osteoid in a regular manner (Figs. 9, 10). Increased numbers of mesenchymal cells were present between the bone trabeculae, but they were loosely arranged, with capillaries and hemorrhage present. In contrast, another area of the same bone contained an osteosarcoma. In this area, trabeculae of woven tumor bone were not lined by a single layer of osteoblasts, and the spaces between the tumor bone were filled with large numbers of atypical mesenchymal cells with numerous mitoses (ie, neoplastic cells; Figs. 11, 12).
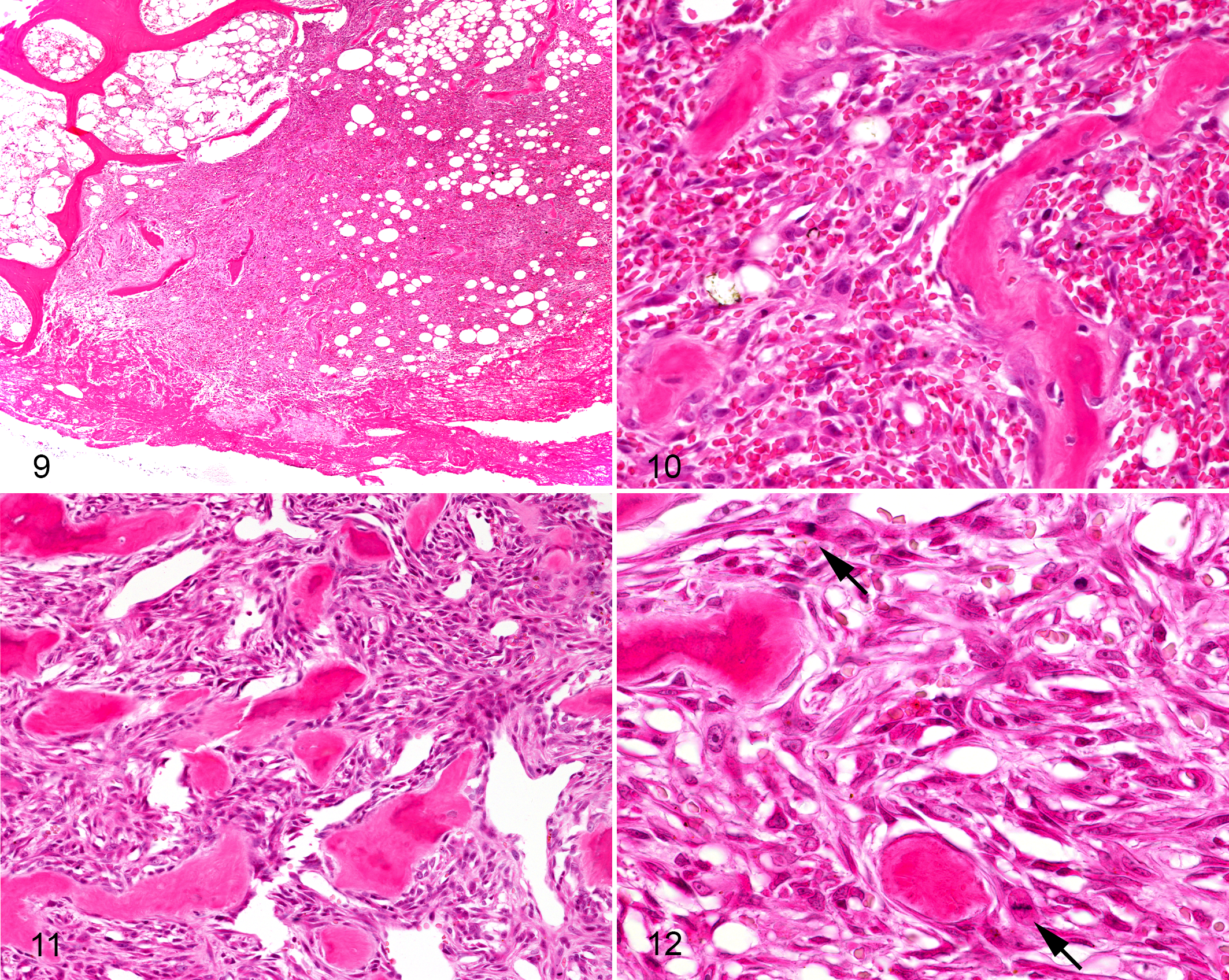
Osteosarcoma with pathologic fracture, proximal humerus, dog (6-year-old Greyhound).
In diagnostic imaging, fracture callus and malignant bone neoplasia can be differentiated by their benign and aggressive characteristics, respectively (Table 1). Access to imaging studies or reports should therefore greatly assist the pathologist in making this distinction. Additionally, the diagnostic imaging findings should alert the pathologist to the possibility of pathological fracture in an osteosarcoma, making it slightly easier to interpret such cases when both disease processes occur concurrently (Fig. 13). The presence of any full-thickness cortical lysis or gross bone displacement on radiographs, CT, or MRI is indicative of pathologic fracture, and a biopsy sample consistent with fracture callus may be obtained in these cases.
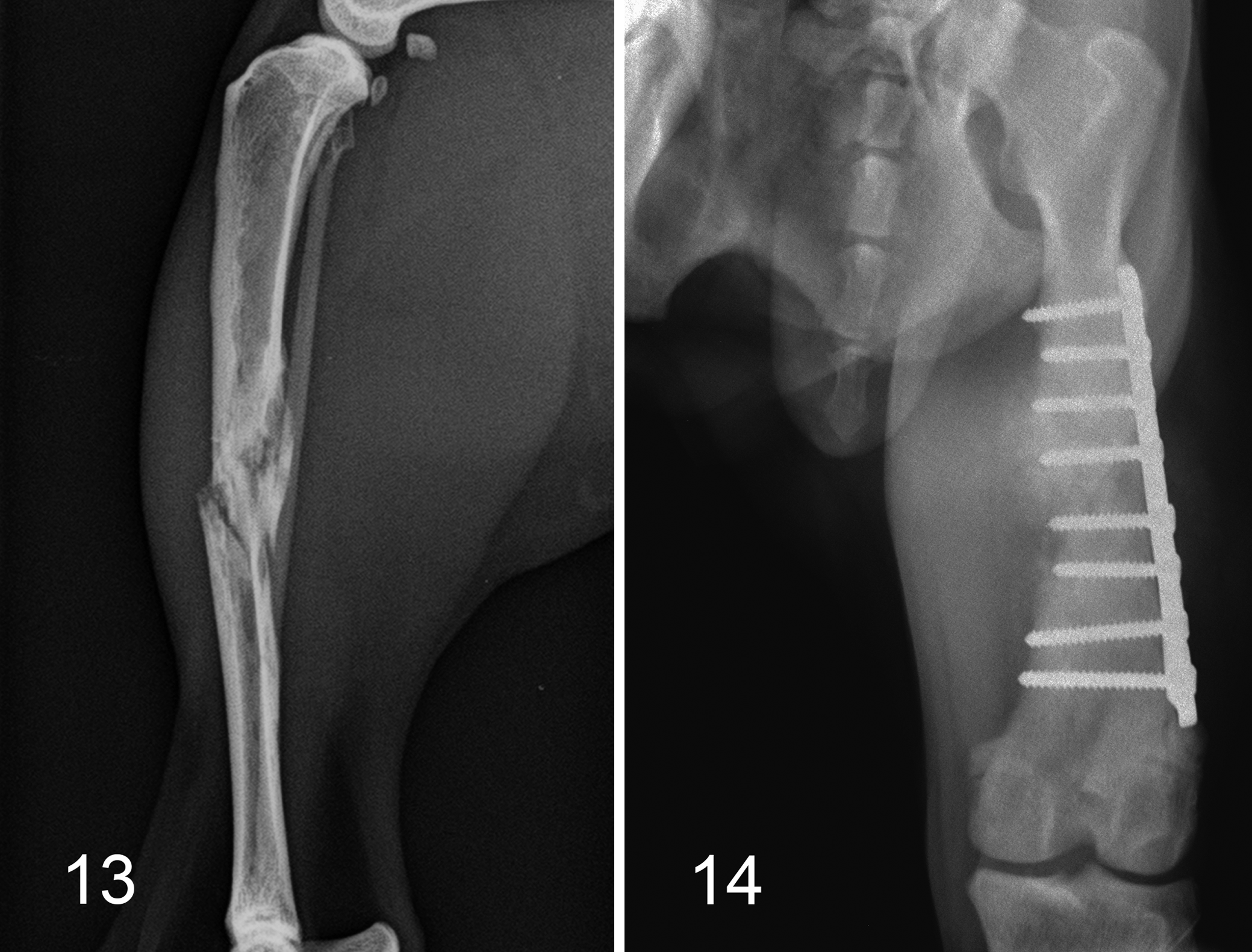
Osteosarcoma and pathologic fracture, tibia, cat. Several irregular fracture lines with associated ill-defined lysis, surrounding sclerosis, and a long zone of transition at the mid-diaphysis.
Reactive Bone Versus Fibro-Osseous Bone Lesions
Fibro-osseous bone lesions, such as osteoma, ossifying fibroma, and fibrous dysplasia, can be challenging to microscopically distinguish from each other and from reactive bone. Low-grade osteosarcoma is a differential diagnosis for fibro-osseous bone lesions in humans, and one paper has described 3 cases of low-grade central osteosarcoma in dogs. 80 However, the paper included only 3 cases, one which was doing well 8 months after diagnosis, one with progressive disease 2 months after excision, and one which was lost to follow up. 80 Therefore, in the opinion of the authors, more published evidence is required to be sure this entity exists in dogs and to determine both the prognostic features of the tumor and the expected outcome. The features of osteoma, ossifying fibroma, and fibrous dysplasia are well described in textbooks. 16,86 However, there can be significant overlap between these tumors microscopically, such that they do not fit cleanly into any category. In which case, it may be that they cannot be classified further than a “proliferative fibro-osseous bone lesion.”
Though the diagnostic imaging of these fibro-osseous lesions is usually distinct in humans, 88 it is more variable as reported in small cases series of cats and dogs. 32,41,90 Osteomas are reported to appear radiographically as amorphous to solid masses having the opacity of bone, with well-defined margins and no evident osteolysis. Ossifying fibromas are reported to have a similar appearance, but with variable associated bone lysis. Both are reported to be expansile in some cases. 80,90 The CT findings of osteoma in cats diverged into well-defined solid dense lesions described as “compact,” and a lower density “cancellous” form that was less clearly demarcated and expansile. 32 Though rare in veterinary patients, fibrous dysplasia is usually reported in juvenile dogs rather than adults. Reports describe osteolytic or expansile lesions with soft tissue opacity and ill-defined margins. 80,94 This is in contrast to cases in humans where fibrous dysplasia produces a radiolucent expansile lesion, leading to thinning of the cortex but with a surrounding sclerotic rim. 88 Thus, interpretation of biopsies with both signalment and diagnostic imaging enhances the ability of the pathologist to make a diagnosis, though limitations remain due to relatively low numbers of reported veterinary cases and an inability to directly extrapolate from human literature with surety.
Is the Tumor Benign or Malignant?
From an imaging perspective, this decision is based on how aggressive a mass or discrete swelling appears according to the above-described criteria (Table 1). After inflammatory lesions are ruled out or deemed unlikely based on the combination of imaging, signalment, and history, imaging characteristics are used to categorize a lesion along the spectrum from benign to malignant. In most cases, this correlates well to biological behavior; however, exceptions do occur. These exceptions include very early stage malignant neoplasia, and malignant transformation of a benign condition. In the early stages of any malignant neoplasm, aggressive imaging characteristics may not be radiographically visible. If doubt exists at initial assessment, repeating the radiographic study may be recommended and can be completed as early as 10 days after initial screening, by which time aggressive characteristics are likely to be visible (Figs. 1, 2). 97 Alternatively, advanced imaging modalities could be utilized where clinical suspicion is high, or microscopic results are nondiagnostic or equivocal. In particular, CT is more sensitive for osseous change, so that subtle periosteal reaction or osteolysis is expected to be identifiable at an earlier stage.
Malignant transformation of a benign condition is another situation where imaging characteristics may be initially misleading. This may occur when a primary bone tumor develops at the site of previous orthopedic surgery, fracture, bone infarct, or at a previously benign mass such as an osteochondroma or aneurysmal bone cyst. 8,37,54,75,81 Benign abnormalities such as fracture callus, orthopedic implants (Fig. 14), or dystrophic mineralization can obscure early osteolysis or active periosteal reaction. 97 However, these same features can ultimately be used to support a pathological diagnosis of malignancy in cases that would otherwise be anomalous. For example, a primary osteosarcoma in an atypically diaphyseal location can be explained by the presence of an orthopedic implant or healed fracture.
The expected histologic appearance of bone tumors are described in a number of texts, 16,86 so the following sections concentrate on common diagnostic dilemmas.
Parosteal Versus Periosteal Versus Central Osteosarcoma
The classification of an osteosarcoma as central or peripheral may have implications for prognosis. Most data on how the location within bone is relevant to prognosis comes from human medicine and is extrapolated to animals. As such, it might be misleading or erroneous. Of osteosarcomas in humans, central and high-grade surface osteosarcomas are considered to have the worst prognosis, periosteal an intermediate prognosis, and parosteal the best prognosis. 88 Both periosteal and parosteal osteosarcoma exhibit slow, progressive growth in humans, with eventual invasion of the cortex and extension into the medullary cavity. In humans, lung metastases occur later in the course of disease, and in the case of parosteal osteosarcoma are associated with dedifferentiation. 10,43,88
While there are little data in animals on periosteal and parosteal osteosarcomas, the data that exist on parosteal osteosarcomas do appear to suggest a better prognosis. Studies that have reported parosteal osteosarcomas indicate that metastasis is rare and in some cases complete surgical excision has been curative, but as with humans, invasion of the cortex and medullary cavity does occur late in the disease process. 35,50,51,80 Interestingly, the majority of parosteal osteosarcomas reported in animals have affected the skull, particularly the zygomatic arch, in contrast to humans where the long bones are predominantly affected. 35,50,51,80,88 Relatively few reports describe the radiographic features of periosteal and parosteal osteosarcomas in dogs and cats due to the rarity of these tumors. 58,85,87 A range of radiographic findings are reported, varying from “well-ordered bony spicules parallel to the axis of the diaphysis” to “solid active periosteal reaction” and “faint particulate areas of mineralization” within soft tissue swelling. The most common characteristic among these reports is the radiographic appearance of an intact or minimally lytic underlying bone, consistent with the expected biologic behavior. Having access to radiographs is critical for the pathologist in being able to distinguish periosteal and parosteal osteosarcomas from central osteosarcoma, as radiographs give the best evidence of the location of the tumor with the bone (Figs. 15 –17).
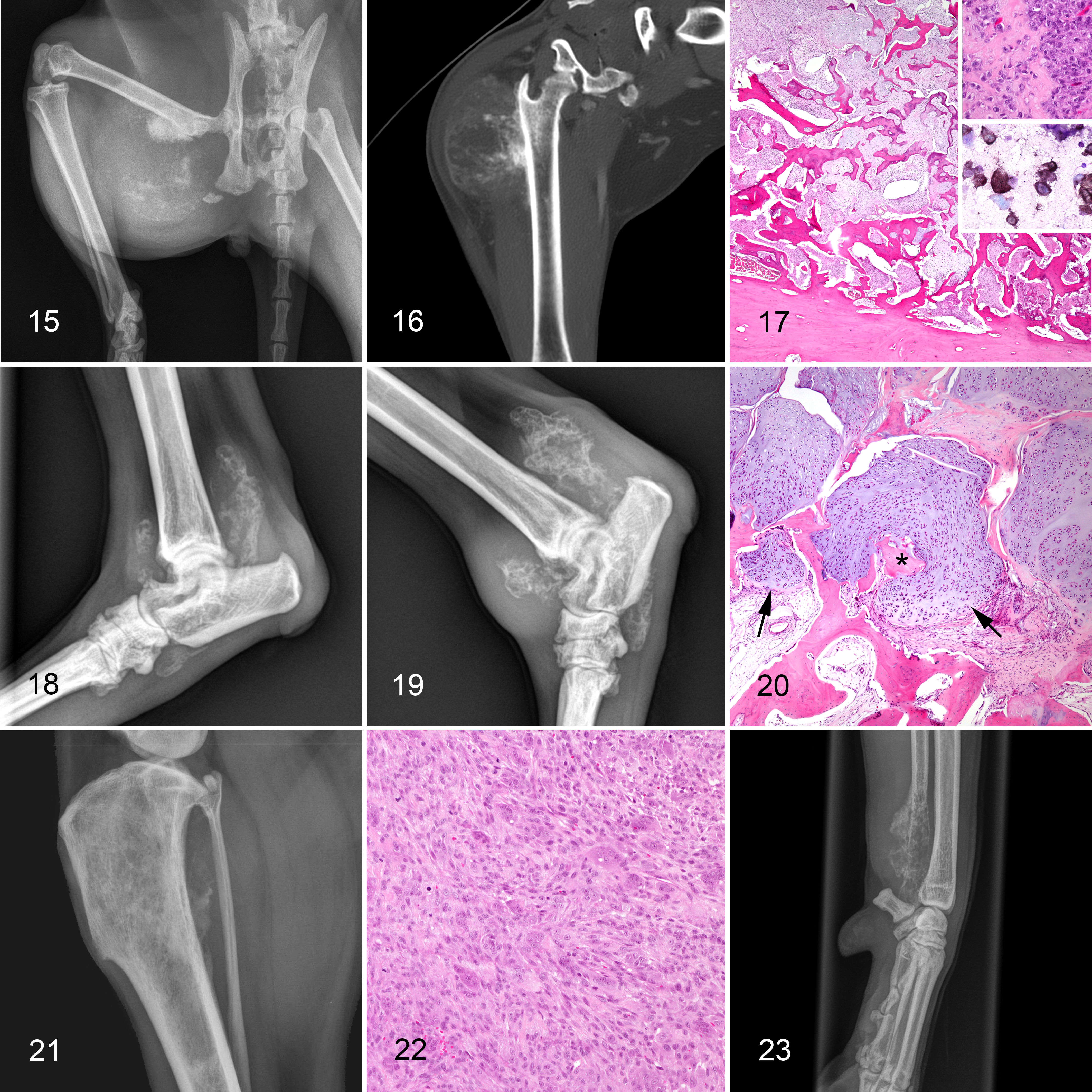
Periosteal chondroblastic osteosarcoma, femur, cat (7-year-old domestic short hair).
There are some evidence that different subtypes of canine osteosarcoma may have different prognoses. 4,57 Classification into osteoblastic, fibroblastic, or chondroblastic subtypes is based on the predominant type of extracellular matrix produced by the tumor (based on the matrix comprising greater than 50% of the tumor area). If no matrix type predominates then the tumor is classified as a combined-type osteosarcoma. 86 Tumor matrix can vary markedly between locations in the same osteosarcoma; as such, subtype classification on small biopsy samples may be misleading and is most reliable after examination of multiple sections from amputation or en bloc resection specimens. While some studies have failed to show a significant effect of tumor subtype on survival time, 42,45,83 others have found that fibroblastic osteosarcoma has an improved survival time compared with osteoblastic and chondroblastic osteosarcoma. 4,57 Similarly, telangiectatic osteosarcoma has been associated with a shorter survival time in some studies but not others. 34,42,78,83 The problem with many studies examining tumor subtype and survival time is the small number of each subtype, making it challenging to draw well-supported conclusions.
Diagnostic imaging is imprecise for differentiating between subtypes of central osteosarcoma in dogs and cats due to the range and overlap of observed features. 97 Some reports have stated that telangiectatic osteosarcoma is characteristically osteolytic compared to the mixed osteolytic and productive appearance of other osteosarcoma subtypes, as was historically reported with human cases. 89 However, low numbers of canine and feline cases are reported, many have scant descriptions of the diagnostic imaging findings, and at least one published image appeared to show marked periosteal reaction or amorphous bone. 1,12,34 Additionally, more recent reports of the disease in humans have identified subtle matrix mineralization contrary to earlier literature, making differentiation of telangiectatic osteosarcoma on the basis of imaging characteristics alone ill-advised. Diagnostic imaging can be used to identify the geographic center of a lesion, as well as the predominant aspect of any lysis—periosteal versus endosteal, for example—and may therefore be used to characterize osteosarcoma as parosteal, periosteal or central. 97 As pathological classification becomes more standardized, and more information becomes available regarding prognosis and metastatic patterns, further imaging studies using CT and MRI may allow more precise categorization of subtypes.
Osteosarcoma Versus Other Sarcomas of Bone
The decision as to whether a bone tumor is an osteosarcoma or another sarcoma of bone is important for prognosis. Because, like osteosarcoma, other sarcomas of bone are generally aggressive, the diagnostic imaging of these tumors may be similar to osteosarcoma. Nonetheless, there are some specific features that, together with the histopathology, suggest one type of sarcoma may be more likely than the other. The frequent diagnostic challenges discussed in this section are to differentiate osteosarcoma from other bone sarcomas in the absence of osteoid; distinguish osteosarcoma and chondrosarcoma when cartilaginous matrix is present; identify multilobular tumor of bone in small biopsies; distinguish between an aneurysmal bone cyst, hemangiosarcoma, and telangiectatic osteosarcoma; and differentiate giant cell osteosarcoma and giant cell tumor of bone.
A primary diagnostic issue with small bone biopsies is to differentiate osteosarcoma from other types of bone sarcoma when tumor matrix is present but there is little or no osteoid. For example, a predominance of cartilaginous matrix could be consistent with a chondrosarcoma or with chondroblastic osteosarcoma. Similarly, a predominance of pleomorphic spindle cells and collagen could be consistent with a fibrosarcoma or a fibroblastic osteosarcoma. As such, when receiving bone biopsies consisting of mesenchymal cells and cartilaginous or fibrous matrix, osteosarcoma cannot be ruled out.
An additional complication may occur in distinguishing chondrosarcoma and osteosarcoma, as the endochondral ossification present in some chondrosarcomas may be difficult to differentiate from tumor osteoid. The key to identifying endochondral ossification is the sudden transition from hypertrophic chondrocytes to bone matrix. In humans, endochondral ossification is a particular feature of mesenchymal chondrosarcoma, whereby the layers of the physis may be recapitulated, with progression from the resting zone to the proliferative and hypertrophic zones, followed by apoptosis and bone production. 31 In comparison, tumor matrix in an osteosarcoma is directly produced by neoplastic osteoblasts without an intermediary cartilage step.
Differentiation of osteosarcoma from chondrosarcoma by diagnostic imaging relies on the site of the lesion more than the morphology of the osseous change, as primary malignant bone tumors cannot be differentiated by imaging alone. Chondrosarcomas are most frequent in flat bones of the ribs, skull, pelvis, and scapula, compared to the higher incidence of osteosarcoma in long bones (particularly the metaphyses of the proximal and distal femur and tibia, proximal humerus, and distal radius). 97 Accordingly, an aggressive bone lesion centered at a costochondral junction is more likely to be a chondrosarcoma. Chondrosarcoma has been reported to develop via malignant transformation of synovial chondromatosis or osteochondromatosis (Figs. 18 –20), and these cases are joint-centered aggressive lesions affecting the bones on either side of the joint. 2,19 This is contrary to the usual monostotic metaphyseal presentation with osteosarcoma. 48,51 Similarly, development of chondrosarcoma from multiple cartilaginous exostoses or osteochondroma is reported, although infrequent. Rapid expansion of such lesions may warrant radiographic surveillance to identify development of aggressive features as described above. 23 This is particularly relevant to long-standing mass lesions that have become painful or undergone a period of rapid growth. However, osteosarcoma is the other neoplasm reported to occur by malignant transformation at sites of multiple cartilaginous exostoses, and remains a differential diagnosis for such a lesion from an imaging perspective. 37,61
Multilobular tumor of bone can be challenging to differentiate from chondrosarcoma or osteosarcoma in biopsy specimens, because the characteristic lobular pattern of the tumor may not be apparent in these small samples. In such cases, correlation of the biopsy and radiography findings will greatly enhance the likelihood of obtaining the correct diagnosis. The radiographic appearance of multilobular tumor of bone is frequently characterized by a finely to coarsely granular mass with the opacity of bone originating on the skull. 15,18,29,82 This appearance was historically described as “popcorn”-like; however, subsequent reports show greater variation in appearance.
Another diagnostic difficulty is distinguishing between aneurysmal bone cyst, hemangiosarcoma, and telangiectatic osteosarcoma. Diagnostic imaging is of assistance in diagnosing an aneurysmal bone cyst as these lesions are usually metaphyseal in location, expansile, and primarily lytic, with no periosteal reaction. Cortical thinning may occur but the cortex should not appear lytic, and osseous septa may be present. The zone of transition should be short and well-defined. 24,68,97 Additionally, while the mesenchymal cells in an aneurysmal bone cyst may have a high mitotic rate, mitoses should not be atypical, and the cells show minimal pleomorphism. This contrasts with hemangiosarcoma and telangiectatic osteosarcoma, where marked pleomorphism and atypical mitoses are a feature.
On histopathology both hemangiosarcoma and telangiectatic osteosarcoma feature mesenchymal cells lining blood-filled spaces. Differentiating between the two predominantly relies on identifying the presence (telangiectatic osteosarcoma) or absence (hemangiosarcoma) of tumor bone. In a small biopsy, however, it may not be possible to distinguish between hemangiosarcoma and telangiectatic osteosarcoma without additional cytochemistry or immunohistochemistry. In fact, a recent study found that when immunohistochemistry for factor VIII–related antigen/von Willebrand factor (FVIII-RAg/vWF) was used on tumors previously diagnosed as telangiectatic osteosarcoma, 14% (6/43) were reclassified from telangiectatic osteosarcoma to hemangiosarcoma, and 1 out of 43 reclassified from hemangiosarcoma to telangiectatic osteosarcoma. It should be noted that when obvious osteoid was present in histology samples, it allowed definitive diagnosis of telangiectatic osteosarcoma; 33 a selection of those that did not have obvious osteoid (5/8 cases) were the tumors that were reclassified using immunohistochemistry. The obvious issues with distinguishing between hemangiosarcoma and telangiectatic osteosarcoma may also perhaps explain some of the discrepancies in survival times for telangiectatic osteosarcoma between different papers.
Differentiating a biopsy specimen of hemangiosarcoma from a vascular region of osteosarcoma may be aided by ancillary diagnostic imaging. Hemangiosarcoma of bone was originally reported to be osteolytic, extensive, and intramedullary, without associated new bone production. However, subsequent reports suggest a more variable appearance including amorphous bone within surrounding soft tissues, and marked periosteal reaction (Fig. 21). 28,48,51,63 It is likely that the emergence of digital radiography and advanced imaging modalities such as CT have allowed identification of lesions with lesser degrees of new bone formation, due to higher contrast resolution and lack of superimposition among other factors. In a recent multi-institutional case series of 70 dogs, no significant difference was found between the imaging characteristics of hemangiosarcoma and telangiectatic osteosarcoma, including osteolysis, periosteal reaction, and less frequently osseous proliferation and pathologic fracture. 34 However, lesion distribution was significantly different between these groups, with 78% of primary appendicular hemangiosarcoma occurring in the hindlimb, compared to only 31% of telangiectatic osteosarcoma. Additionally, half of the primary appendicular hemangiosarcoma cases occurred in the tibia, and these findings may help rank differential diagnoses prior to immunohistochemistry. 34 Disease staging due to presence of an aggressive bone lesion usually includes thoracic and abdominal screening by radiography, ultrasonography or CT. Presence of pleural or peritoneal effusion, splenomegaly, splenic mass, right atrial mass, or pericardial effusion raise concern for metastatic hemangiosarcoma, whereas these findings are expected much less frequently with metastatic osteosarcoma or coincidental comorbidities. Pulmonary parenchymal lesions due to metastatic hemangiosarcoma may be nodular or miliary and only 27% are expected to be well-defined, compared to metastatic osteosarcoma, which is more frequently well-defined and nodular. 39 Alternatively, metastases of hemangiosarcoma may have larger areas of soft tissue opacity or attenuation due to parenchymal hemorrhage, 39 but this is unlikely with metastatic osteosarcoma.
Differentiating giant cell osteosarcoma and giant cell tumor of bone can also be challenging, particularly if little or no osteoid is present in a biopsy. Three types of cells are typically present in a giant cell tumor of bone: fibroblastic spindle cells (the putative neoplastic cell, likely of osteoblastic lineage), mononuclear cells (CD68+ monocyte/macrophage lineage), and multinucleated giant cells recruited to the tumor. 72,79 There are few reports of giant cell tumor of bone in domestic animals, so much of the information in the literature is extrapolated from humans. Microscopically, the key to differentiating the two is that a giant cell tumor of bone has no or minimal osteoid; if substantial osteoid is present, then the tumor is a giant cell osteosarcoma. (Figs. 22, 23). However, in some giant cell tumors of bone, particularly in association with pathologic fracture or occasionally at the edge of the tumor, spicules of reactive bone may be present and should not be interpreted as “tumor bone.” Additionally, in humans, the majority of giant cell tumors of bone are benign, and while the mitotic rate is high, mitotic figures are not atypical, and the fibroblastic and mononuclear cells show no evidence of pleomorphism. 88 In comparison, atypical mitoses and other features of malignancy are common in osteosarcoma.
Examining the diagnostic imaging may be of assistance, though reports in domestic animals are rare. Radiographically, giant cell tumor of bone is reported to be predominantly radiolucent with minimal to no periosteal reaction and a short zone of transition that lacks a sclerotic margin. 79 New tumor bone is not expected to be visible in a giant cell tumor of bone even via CT, which may help differentiate it from giant cell osteosarcoma where tumor bone may be visible radiographically (Fig. 22). Additionally, in comparison to osteosarcoma that rarely crosses the joint, giant cell tumor of bone is frequently epiphyseal in location and may erode the articular cartilage and then spread into the joint. 21,88 In humans, giant cell tumor of bone is associated with mutations in the histone 3.3 (H3F3A) gene. 9,66 Other sarcomas may have similar mutations but at a much-decreased frequency compared with giant cell tumor of bone. 44,66
The differentiation of osteosarcoma versus other bone sarcomas is greatly aided by application of NPT/BCIP to indicate alkaline phosphatase (ALP) activity by osteoblasts. This reagent may be used either prior to or after staining of cytology preparations with Romanowsky-type stains. Demonstrating ALP activity in mesenchymal cells showing features of malignancy has excellent sensitivity (reported values 88% to 100%) and specificity (89% to 94%) for osteosarcoma (Figs. 5 and 17 insets). 7,60,70 Other tumors reported to occasionally express ALP on cytology smears include chondrosarcoma and melanoma. 7,70
Immunohistochemistry is advantageous in assisting the pathologist to distinguish between fibrosarcoma and fibroblastic osteosarcoma, and between hemangiosarcoma and telangiectatic osteosarcoma, but does not allow the pathologist to discriminate between chondrosarcoma and chondroblastic osteosarcoma. In humans, osteocalcin has been shown to have excellent specificity (100%) for osteosarcoma, with no chondrosarcomas (7 cases) showing immunoreactivity to osteocalcin. 30 However, the sole study examining osteocalcin immunoreactivity in canine bone tumors other than osteosarcoma found that while it had good sensitivity (83%) for the diagnosis of osteosarcoma, all 4 chondrosarcomas in the study were immunopositive for osteocalcin, which limited its specificity (71%). 92 However, none of the fibrosarcomas (4 cases), histiocytic sarcomas (4 cases), or hemangiosarcomas (2 cases) in the study were immunoreactive for osteocalcin, suggesting it may have utility in distinguishing between these tumors, and osteosarcoma or chondrosarcoma. ALP immunoreactivity may also be used to diagnose osteosarcoma; however, to the authors’ knowledge there are no published studies of ALP immunoreactivity in other bone sarcomas. 3 The effect of different decalcifiers on immunoreactivity must be taken into consideration. Studies on canine osteosarcoma samples showed that osteocalcin and osteonectin immunoreactivity were greatest in samples decalcified with 15% EDTA, followed by 10% formic acid, and least in samples decalcified with 10% hydrochloric acid. 92
Conclusion
A definitive diagnosis of the type of bone tumor affecting an animal assists in determining the likely prognosis for the animal. However, definitive diagnosis can be challenging, both in distinguishing between reactive and neoplastic disease processes, and between different types of bone sarcomas. Nonetheless, when a holistic approach is taken with evaluation of the signalment, history, clinical features, radiologic features, and microscopic features, a presumptive diagnosis may be possible. The pathologist is greatly assisted in their interpretation of bone samples by having access to the imaging findings. Pathologists should routinely request the imaging if it is not received from submitting veterinarians.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
