Abstract
A femoral mass from a 15-year-old rhesus macaque was evaluated. Grossly, the mass consisted of a large, osteolytic focus in the distal femur, a gelatinous core of neoplastic tissue in the medullary cavity, and an invasive mass-obliterating musculature of the thigh. On histopathologic evaluation, three neoplastic mesenchymal cell populations, osteoblasts, fibroblasts, and primitive mesenchymal cells were identified. The mass was diagnosed as a combined type osteosarcoma. To our knowledge, this is the first osteosarcoma in a rhesus macaque with this subclassification.
Primary bone neoplasms can arise from a variety of mesenchymal tissues present in bone including osteoblasts, chondroblasts, fibroblasts, adipocytes, endothelial cells, or any hematopoietic element. 12 Malignant bone tumors are uncommon in most species with the exception of canine (osteosarcoma, chondrosarcoma, and liposarcoma), feline (osteosarcoma), and human (malignant myeloma, osteosarcoma). 4, 6, 11, 12
With a few exceptions, spontaneous malignant neoplasms are uncommonly reported in nonhuman primates. 1, 3, 5, 7, 9, 10, 13 In a 1973 study of 32 species of adult nonhuman primates, spontaneous neoplasia occurred in 2.8% of the animals (24 of 1,065). 9 This low number may be partially explained by the lack of aging studies before the early 1980s. The Wisconsin National Primate Research Center supports an aging colony of rhesus macaques (Macaca mulatta). During the past 5 years (1999–2003), 164 necropsies were performed on animals 15-years-of age or older, which died or were euthanatized for medical reasons. Thirty percent of these deaths were because of neoplasia, with 26% (43 of 164) diagnosed as enteric adenocarcinoma. The remaining 4% were individual cases of salivary adenocarcinoma, pheochromocytoma, malignant pancreatic tumor, oral squamous cell carcinoma, renal cell carcinoma, thyroid C-cell carcinoma, and multicentric lymphoma. With the exception of a few benign tumors (uterine leiomyomas, subcutaneous lipomas, adrenal cortical hemangiomas), spontaneous mesenchymal-derived tumors have been rarely reported in this species. Only five osteosarcomas have been described in rhesus macaques during the past 80 years. 9, 10 This case report describes an atypical presentation of osteosarcoma in an adult rhesus macaque.
A 16-year-old, male rhesus macaque from the Wisconsin National Primate Research Center's breeding colony was presented with a large palpable mass in the mid to distal left femoral region of unknown duration. The animal did not bear weight on the limb and exhibited no signs of discomfort. The physical examination was otherwise normal. All serum chemistries, including alkaline phosphatase, 189 U/liter (normal 28–221), were normal with the exception of a slightly elevated creatine kinase, 786 U/liter (normal 0–326). Radiographs of the affected femur revealed a large focus of osteolysis at the distal metaphysis extending into the diaphysis and a moderate periosteal reaction adjacent to lytic focus (Fig. 1). An ill-defined soft tissue mass extended from the medullary space into adjacent soft tissues. Multiple, irregular foci of mineralization were scattered throughout the mass. There was no radiographic evidence of metastasis seen in thoracic radiographs. After considering the unknown prognosis of osteosarcoma in macaques, the absence of clinical pain or metastasis, and the value of the adult, breeding-age rhesus macaques, a decision to amputate the affected limb, rather than to euthanatize, was made by the attending and clinical veterinarians.
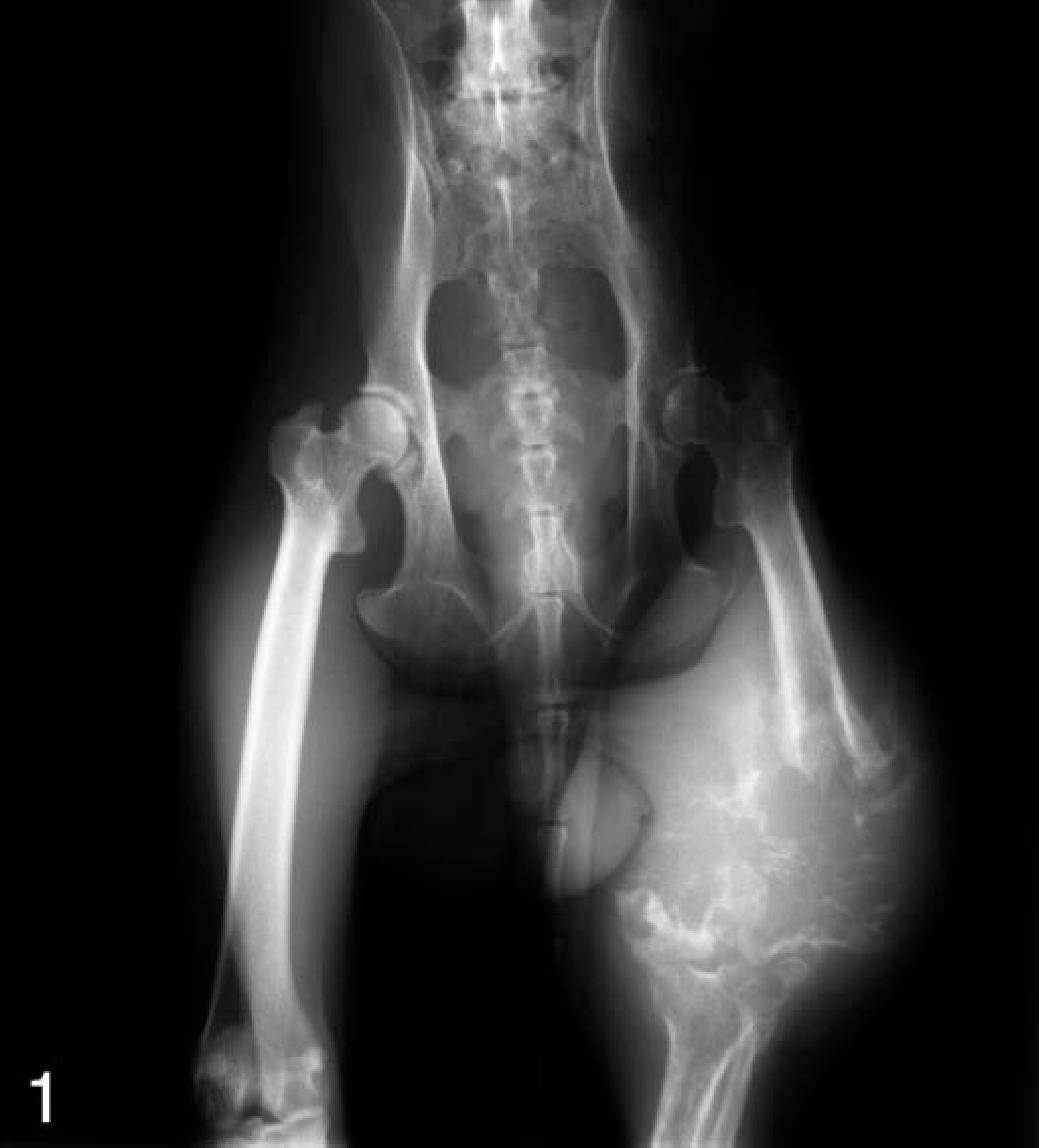
Pelvis, proximal hindlimbs, rhesus macaque. Anterior-posterior radiographic projection showing a large focus of osteolysis in the distal left femur surrounded by a large, poorly demarcated soft tissue mass.
The left leg was amputated at the coxofemoral joint and was submitted for pathologic examination. Ten centimeters distal to the femoral head, there was a large, approximately 20 × 10 × 12 cm, poorly demarcated expansile mass, which encircled the distal femur and infiltrated the adjacent musculature. On cut surface, the mass was mottled yellow-tan and red, soft and gelatinous, and exuded abundant mucoid material. Multiple, 1–3-cm, irregularly shaped, yellow-white, gritty to bony foci were randomly distributed throughout the soft tissue mass. At approximately 17 cm from the femoral head, the periosteal surface became red and roughened by numerous, small bony spicules (periosteal reaction). Two centimeters further distal, near the metaphysis, the bone was obliterated. The surface of the distal femur was also red and roughened by bony spicules. A 1.5-cm-diameter, yellow-tan, gelatinous core filled the proximal medullary cavity and extended through the focus of lytic bone, continuous to the medullary space of the distal femoral fragment. The neoplastic mass did not extend into the joint or across the joint space.
The entire distal femur as well as samples from the adjacent soft tissue mass were immersed in 10% neutral buffered formalin. Bone sections were decalcified in either 1.35 N HCl, 0.003 M ethylenediaminetetraacetic acid (EDTA) (Cal-EX® decalcifying solution, Fisher Scientific, Fairlawn, NJ) for 7–10 days or 10% EDTA in phosphate-buffered saline for 21–28 days. All specimens were then embedded in paraffin, sectioned at 4–6 μm, and stained with hematoxylin and eosin (HE). A few specimens were sectioned serially and stained with HE, Masson's trichrome, or Alcian blue, pH 2.5.
An aggressive, poorly demarcated, neoplastic mass of mesenchymal cells filled the marrow space of the femoral metaphysis and adjacent diaphysis and infiltrated the cortical bone into adjacent fascia and muscle. The neoplastic cells displayed mixed mesenchymal phenotypes including osteoid-producing cells (osteoblastic cells); fibroblastic cells; and large, sparsely cellular areas of more primitive, less differentiated, spindle cells embedded in abundant myxoid matrix.
Small sheets of neoplastic osteoblasts were observed within the marrow space along the endosteal surfaces (Fig. 2). The osteoblasts were mildly pleomorphic, round to occasionally polygonal, and contained various amounts of amorphous, fibrillar, brightly eosinophilic material (osteoid). A few thin ribbons of osteoid were in the interstitium between scattered small sheets of neoplastic osteoblasts.
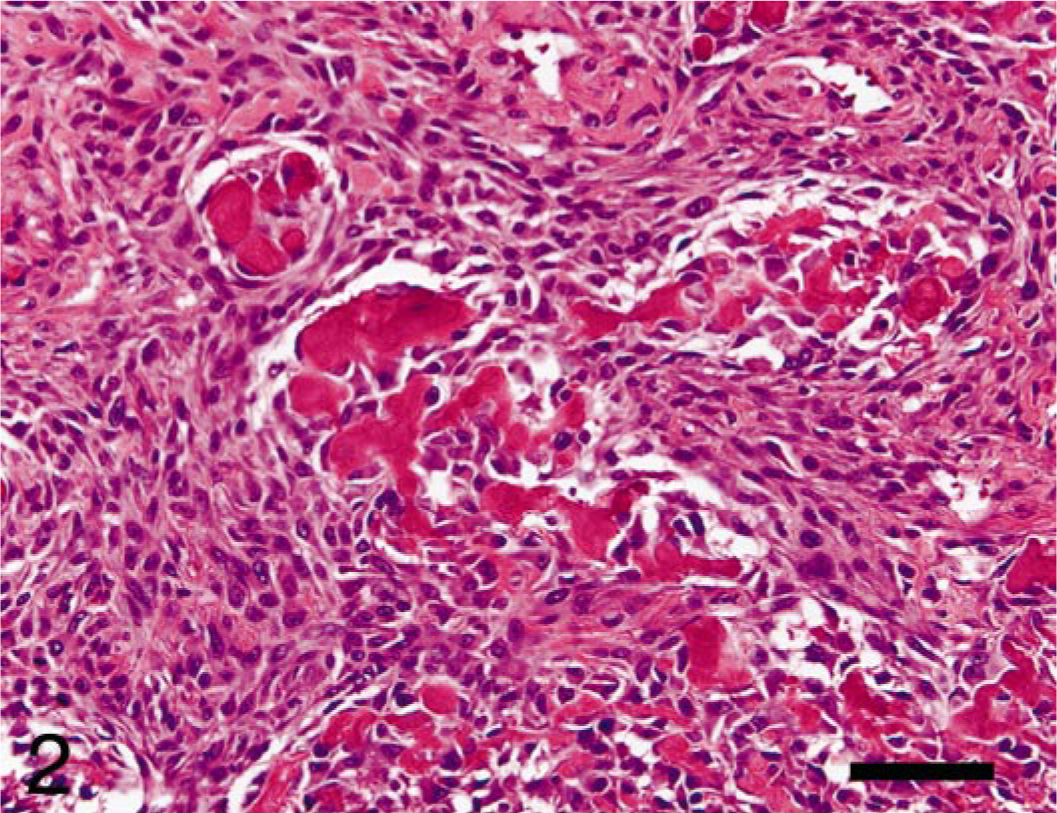
Femur, osteosarcoma, rhesus macaque. Intramedullary mass composed of scattered sheets of mildly pleomorphic, well-differentiated, osteoid-producing, neoplastic osteoblasts lying predominantly along endosteal surfaces. HE. Bar = 50 μm.
The fibroblastic cells filled the marrow space and also infiltrated through cortical bone and into periosteal soft tissue. The fibroblastic cells were densely packed, formed interlacing bundles, and were minimally pleomorphic with mild anisokaryosis and one to numerous nucleoli (Fig. 3). These cells produced small amounts of eosinophilic, fibrillar matrix, which stained positively with Masson's trichrome (collagen).
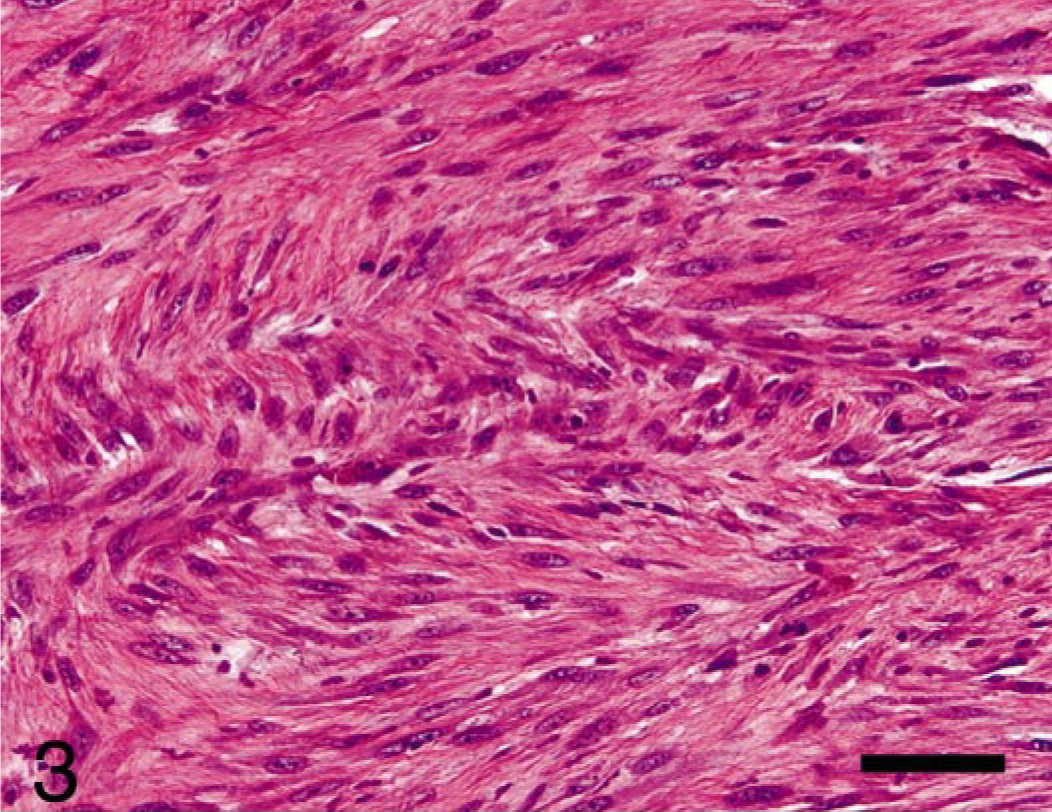
Femur, osteosarcoma, rhesus macaque. The second population of cells arising in intramedullary mass and invading cortical bone were mildly pleomorphic, neoplastic fibroblasts forming interlacing fascicles and producing discrete amounts of collagen. HE. Bar = 50 μm.
The primitive spindle cells were scattered in small foci in the marrow space in the lytic focus but were the predominant phenotype in both the medullary space in the proximal femur and in the soft tissue around the bone. The muscles of the thigh were almost completely obliterated by these cells. The poorly differentiated neoplastic mesenchymal cells were often haphazardly arranged in swirling patterns and produced abundant matrix that stained positively with Alcian blue (mucopolysaccharides). The spindle-shaped cells were markedly pleomorphic, with marked anisokaryosis and occasional karyomegaly. The hyperchromatic nuclei ranged from fusiform to irregularly ovoid and contained one to multiple nucleoli. The scant cytoplasm was lightly basophilic. There were occasional scattered binucleate cells (Fig. 4).
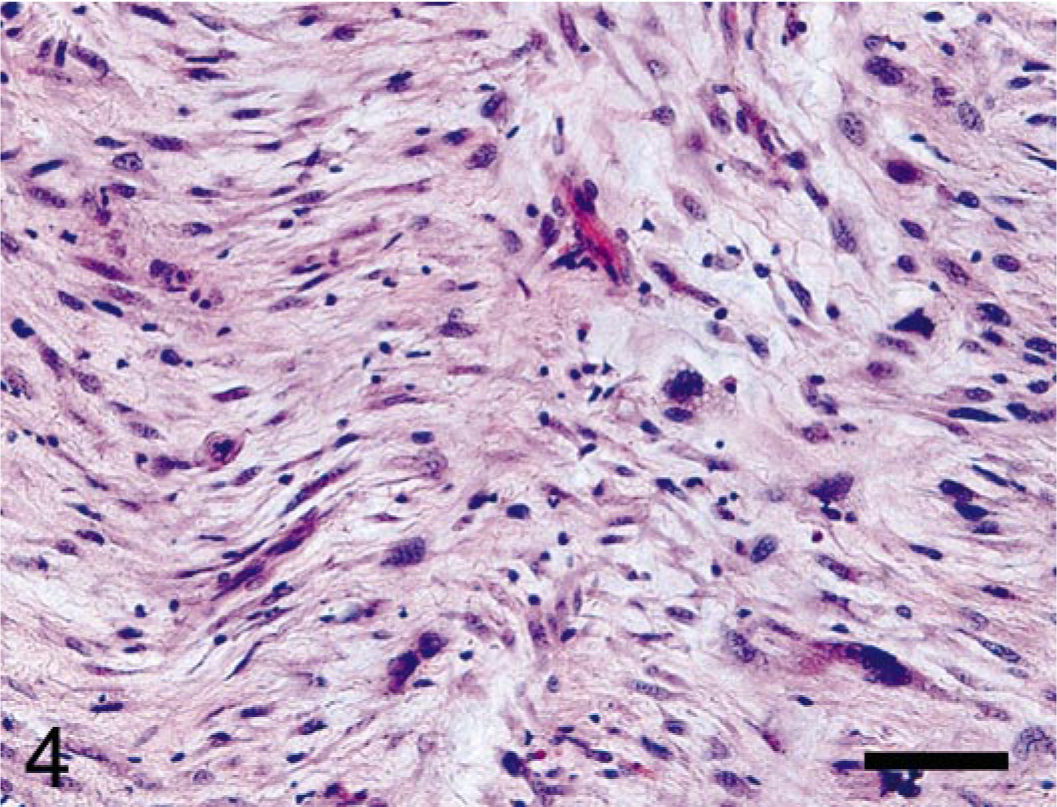
Femur, rhesus macaque. Markedly pleomorphic, neoplastic, primitive mesenchymal cells embedded in myxoid matrix were scattered within the medullary space with cortical invasion and effacement of femoral musculature. HE. Bar = 50 μm.
Within the osteoblastic and fibroblastic populations, there were rare mitotic figures. However, there was one mitotic figure per two 40× fields in the primitive spindle cells areas. Several mitotic figures were atypical. Scattered within the medullary cavity and in the osteolytic foci were numerous clusters of multinucleated cells. Throughout the mass, there were a few small foci of necrosis. No vascular invasion was seen. At the periphery of the lytic zone, there was an irregular chondro-osseous callus and also woven, new bone (periosteal reaction).
Bone tumor classification is based on morphologic characteristics of neoplastic cells, type of matrix produced, and anatomic location (or point of origin) of the tumor. 12 Diagnosis of osteosarcoma rests on identifying osteoid production by neoplastic mesenchymal cells. 6, 12 The World Health Organization (WHO) produces classification systems for people and also domestic animals, which subdivide osteosarcomas on the basis of the predominant cell type or matrix (Table 1). 4, 8, 11 Subdivision is attempted in humans because there may be prognostic significance. The majority of human and canine osteosarcomas are of the osteoblastic type. The presence of another predominant cell type or multiple different cell lines in bone tumors is less commonly reported.
Comparative chart of World Health Organization, human and veterinary, and Mirra human osteosarcoma classification schemes.
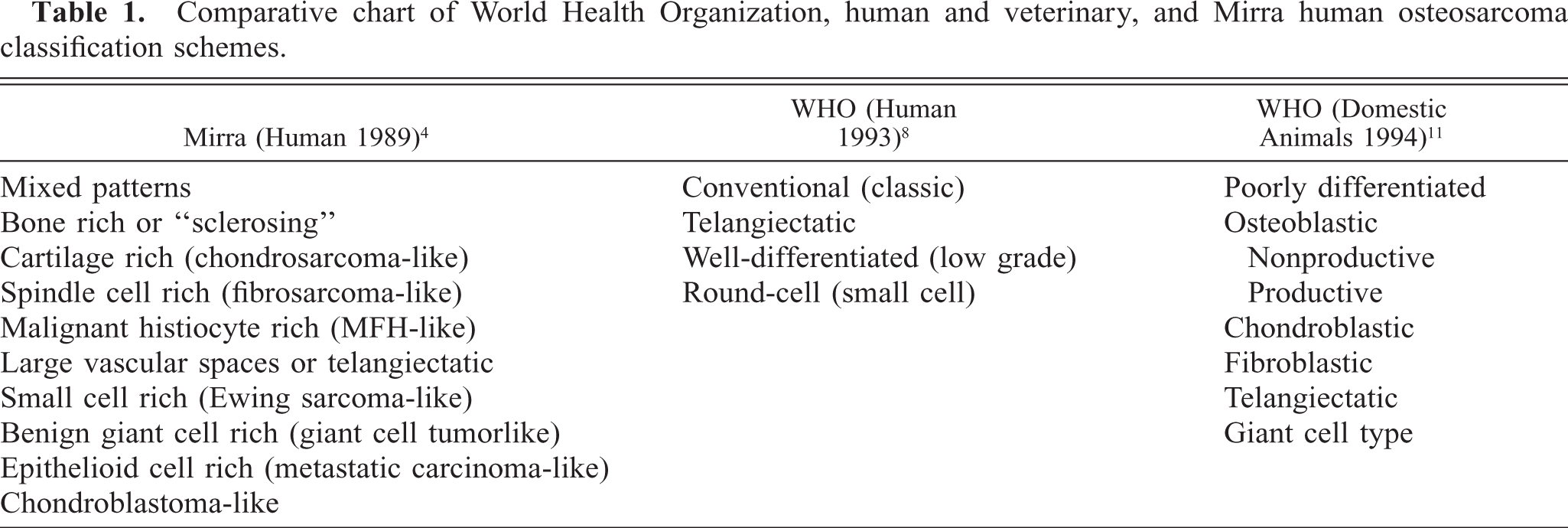
Differentials for this tumor in the rhesus included osteosarcoma or malignant mesenchymoma. Malignant mesenchymoma is a sarcoma consisting of two or more differentiated mesenchymal elements (other than fibrosarcoma). 2 This tumor contained two differentiated elements, osteosarcoma and fibrosarcoma, and the undifferentiated primitive cells; therefore, the diagnosis of malignant mesenchymoma was excluded. Although, in this tumor, the smallest neoplastic cell population was osteoblasts, we made the diagnosis of osteosarcoma on the basis of the osteoid production. This bone tumor does not easily fit into either (human or veterinary) WHO classification scheme. 8, 11 An older, human classification by Mirra would be osteosarcoma, mixed cell type. 4 Thompson and Poole describe a “combined type osteosarcoma,” which would more closely fit with the tumor seen in this study. 12
With the paucity of cases of osteosarcoma in nonhuman primates, especially rhesus macaques, prognosis is difficult. Adjuvant chemotherapy was not used in this animal partially because of interference with the animal's assigned function of breeding. The animal has been closely monitored for 10 months since diagnosis. There has been no clinical evidence of recurrence, metastasis, or pain. Thoracic radiographs and alkaline phosphatase remain within normal limits.
Footnotes
Acknowledgements
This work was supported by the Wisconsin National Primate Research Center with funding provided by NIH base grant 5P51 RR 000167. I thank the assistance of Dr. Amy Usborne. Also, thanks to Jessica Krawczyk for her excellent technical support and Drs. Iris Bolton and Michelle Harke, the veterinary technicians, and the laboratory animal technicians for their clinical contributions and animal care.
