Abstract
Canine appendicular osteosarcoma is an aggressive bone neoplasm that imposes a short survival time. There are several published histologic grading systems for canine osteosarcoma but no universally accepted system. Location within the skeleton and therapy received are both correlated with survival time, but these factors were not always considered when the prognostic value of published grading systems was determined. Our objective was to compare 2 published histologic grading systems in a population of dogs with appendicular osteosarcoma treated with the standard of care for curative intent. Three evaluators graded 85 tumors using 2 histologic grading systems. The relationships between histologic grade as well as individual histologic features and outcome (survival time and disease-free interval) were evaluated using Kaplan-Meier survival functions and a univariate Cox proportional hazards model. Histologic grade, as assigned by any evaluator, did not correlate with outcome. Increased number of mitotic figures per 3 randomly selected 400× microscope fields, as assessed by 1 evaluator, was correlated with both survival time and disease-free interval; this was the only individual histologic feature that was significantly correlated with outcome for any evaluator. These findings cast doubt on the predictive value of routine histologic grading in dogs with appendicular osteosarcoma who receive amputation followed by adjuvant chemotherapy and highlight the need for better tools to predict outcome in canine appendicular osteosarcoma.
Keywords
Osteosarcoma is broadly categorized based on the anatomic location in which it arises. Appendicular, 5 axial (flat and irregular bones), 15,39 and extraskeletal 11,21 osteosarcoma are distinct in terms of their biology, clinical aggressiveness, and prognosis; in spite of this, they are frequently considered as a single disease. Osteosarcoma of the appendicular skeleton is the most common primary bone tumor of dogs. The standard of care for curative intent prescribed for these dogs is amputation of the affected limb followed by adjuvant chemotherapy, while palliative options (including amputation only) are offered to dogs who initially present with radiographic evidence of metastatic disease. 36 An increase in median survival time is consistently observed with the addition of adjuvant chemotherapy compared to amputation alone (range: 262–413 vs 134–175 days, respectively). 1 –4,16,23,27,37,41 Therapeutic intervention received and tumor location are prognostic factors for canine osteosarcoma, while serum alkaline phosphatase activity and histologic grade are commonly applied prognostic tools. 5,18,22,35
Although published histologic grading studies provide hazard ratios for both survival time and disease-free interval and have identified correlations with progression to metastatic disease for canine osteosarcoma, results vary with respect to the prognostic value of the individual histologic features and of the proposed grading systems (Supplemental Table S1). 12,15,18,20,22,26,27,31,33,39 It is well established that adjuvant chemotherapy significantly increases survival time for dogs with appendicular osteosarcoma, 28 yet treatment differences were not always considered as a potential source of bias in the development of published grading systems; either therapeutic data were not always available or dogs receiving different treatment were not considered separately. Additionally, previous grading studies have evaluated canine osteosarcoma without distinguishing between anatomic locations (appendicular vs axial vs extraskeletal). Finally, the majority of published canine osteosarcoma grading systems were developed using a single population and a single evaluator. 18,20,22,27,31,33,39 Based on these observations, evaluation of the established grading systems for canine osteosarcoma is warranted to assess their merit as a prognostic tool for diagnostic pathologists.
Recently, Kruse et al 20 evaluated individual histologic characteristics as well as histologic grade as predictors of survival in a relatively uniform group of 46 dogs diagnosed with osteosarcoma, assessing primary tumors arising in only the flat or irregular bones. This study found that neither histologic grade nor individual histologic features correlated with survival time. 20 This finding reinforces our suspicion that failure to control for tumor location and treatment received could be partially responsible for the prognostic significance assigned to histologic grade in canine osteosarcoma. Our objective was to evaluate the prognostic value of 2 published canine osteosarcoma grading systems for outcome in dogs with appendicular osteosarcoma, specifically assessing those receiving the standard of care, to determine whether histologic grade is correlated with survival time or disease-free interval and whether any individual histologic features are independent prognostic indicators.
Materials and Methods
Cases of canine appendicular osteosarcoma were identified in the Ontario Veterinary College teaching hospital database over a 17-year period (1998–2014). Cases considered for inclusion in this study did not receive treatment prior to amputation and had archived histology slides and/or formalin-fixed paraffin-embedded primary tumor available. Dogs were required to have undergone amputation of the affected limb followed by a minimum of 1 dose of chemotherapy (ie, commencement of the standard of care). In addition, dogs with radiographic evidence of metastatic disease at the time of diagnosis were excluded from the study. Medical records were reviewed to obtain the sex, breed, age at diagnosis, weight at diagnosis, presurgical serum alkaline phosphatase activity (ALP), primary tumor site, amputation date, histologic grade, histologic subtype, chemotherapy protocol, postchemotherapy treatment, metastatic disease status and location, date of confirmation of metastasis, date of euthanasia/death, and cause of death. ALP status (high or normal) prior to surgical intervention was determined based on the reference range of the laboratory that analyzed the serum. The diagnosis date was defined as the first instance where radiographical interpretation included a differential diagnosis of osteosarcoma. In cases where no presurgical radiographs were performed or where the imaging date was not known, the date when diagnostic samples were acquired (excisional/incisional biopsy or amputation of the affected limb) was used as the date of diagnosis. Disease-free interval (DFI) was defined as the number of days from diagnosis to the confirmation (radiographic or postmortem examination) or suspicion (radiographic or clinical signs) of metastatic disease. Survival time (ST) was defined as the number of days from diagnosis to euthanasia/death. For each dog, cause of death was either attributed to osteosarcoma or not. Cause of death was ascertained from the medical record; for dogs where no cause was listed, death was attributed to osteosarcoma only if metastatic disease was previously confirmed or suspected.
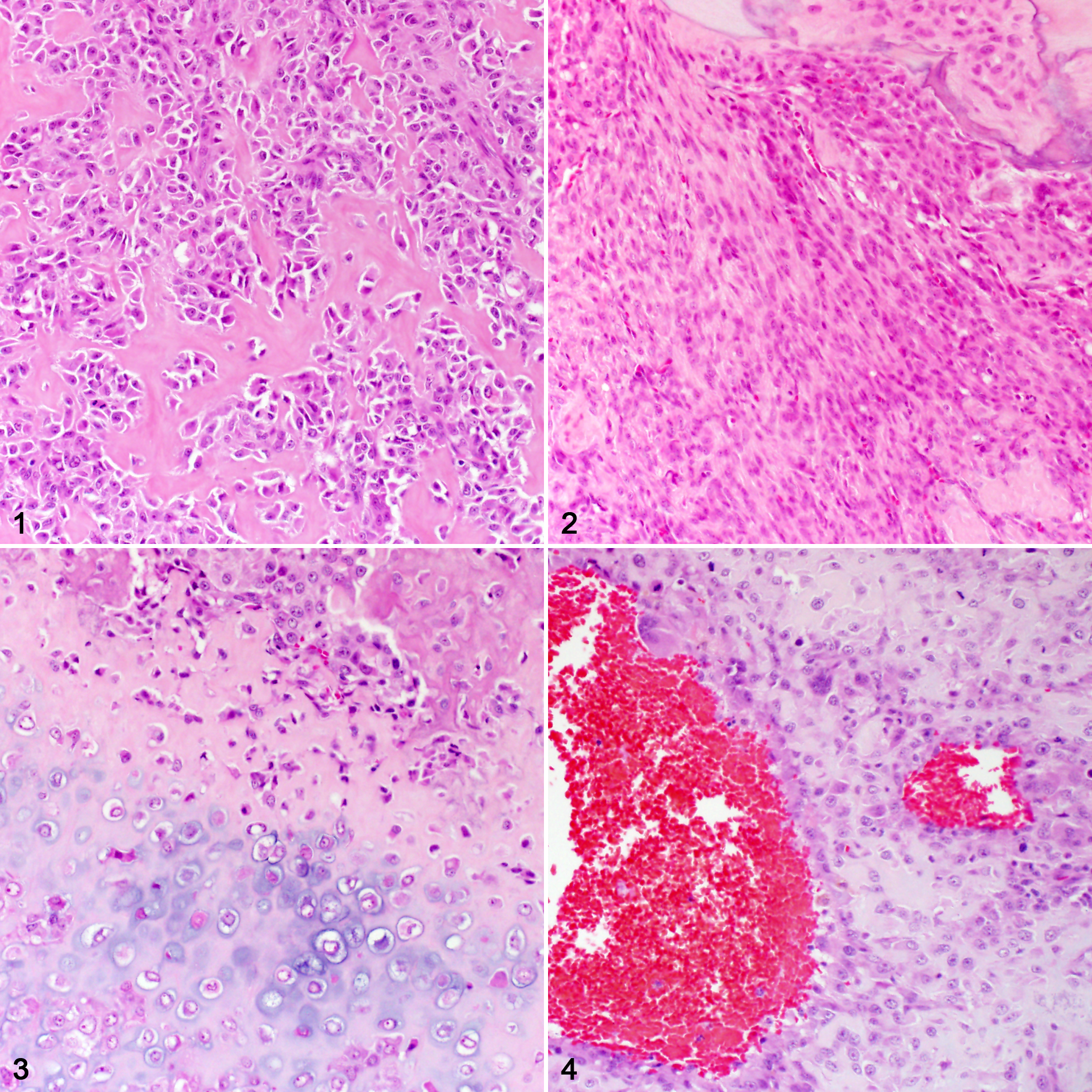
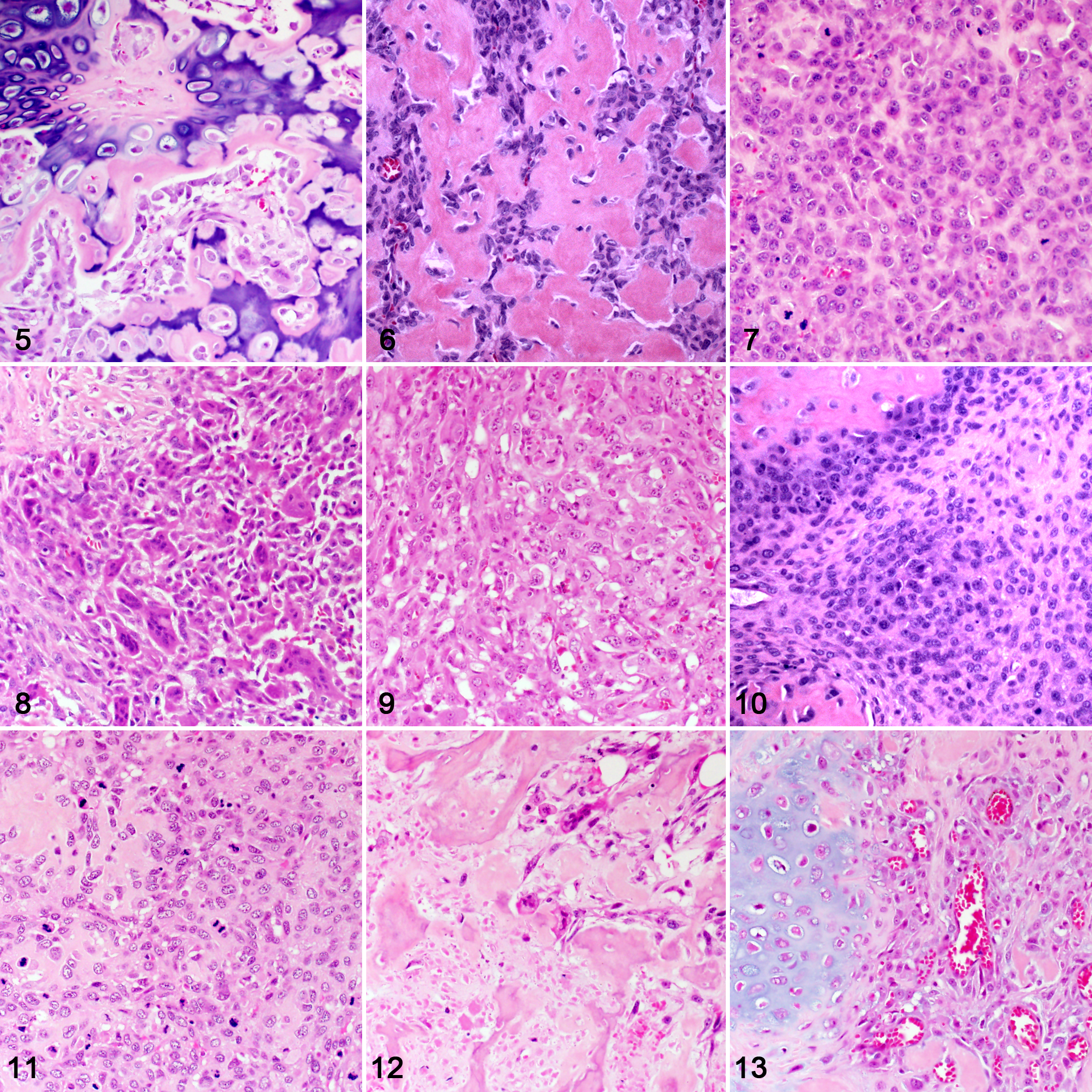
The routine H&E-stained histologic sections for each case were reviewed by a single evaluator (CRS) and when necessary a second evaluator (RAF) to confirm the diagnosis of osteosarcoma; conflicts were resolved by consensus. A single histologic section was selected by CS for each case in the study. The only case material provided to each evaluator was the slides. Histologic grades were assigned by 3 evaluators (A–C) using 2 established grading systems: Kirpensteijn et al 18 (2002 system, Table 1) and Loukopoulos and Robinson 22 (2007 system, Table 2). Each evaluating pathologist was provided with a copy of both published studies describing the respective grading systems as the only form of guidance to assign histologic grades. Neither methods for determining the final histologic grade nor for evaluating individual features were discussed between the evaluators, who were instructed to extract their methods from the original publications. For this study, the diameter of 1 high-power field (hpf) was 2.37 mm2 for all of the microscopes used. Some of the microscopic features considered during diagnosis, histologic grading, and histologic subtyping of canine osteosarcoma are illustrated in Figs. 1–13. Each grading system was also converted into a 2-tier grading system by combining either grades II-III (high) and comparing to I (low) or combining grades I-II (low) and comparing to III (high).
Canine Osteosarcoma Histologic Grading System From 2002, by Kirpensteijn et al.a

aFrom Kirpensteijn et al, 18 predetermined histologic classification schedule for tumor grade determination. hpf, high power field.
Canine Osteosarcoma Histologic Grading System From 2007, by Loukopoulos and Robinson.a
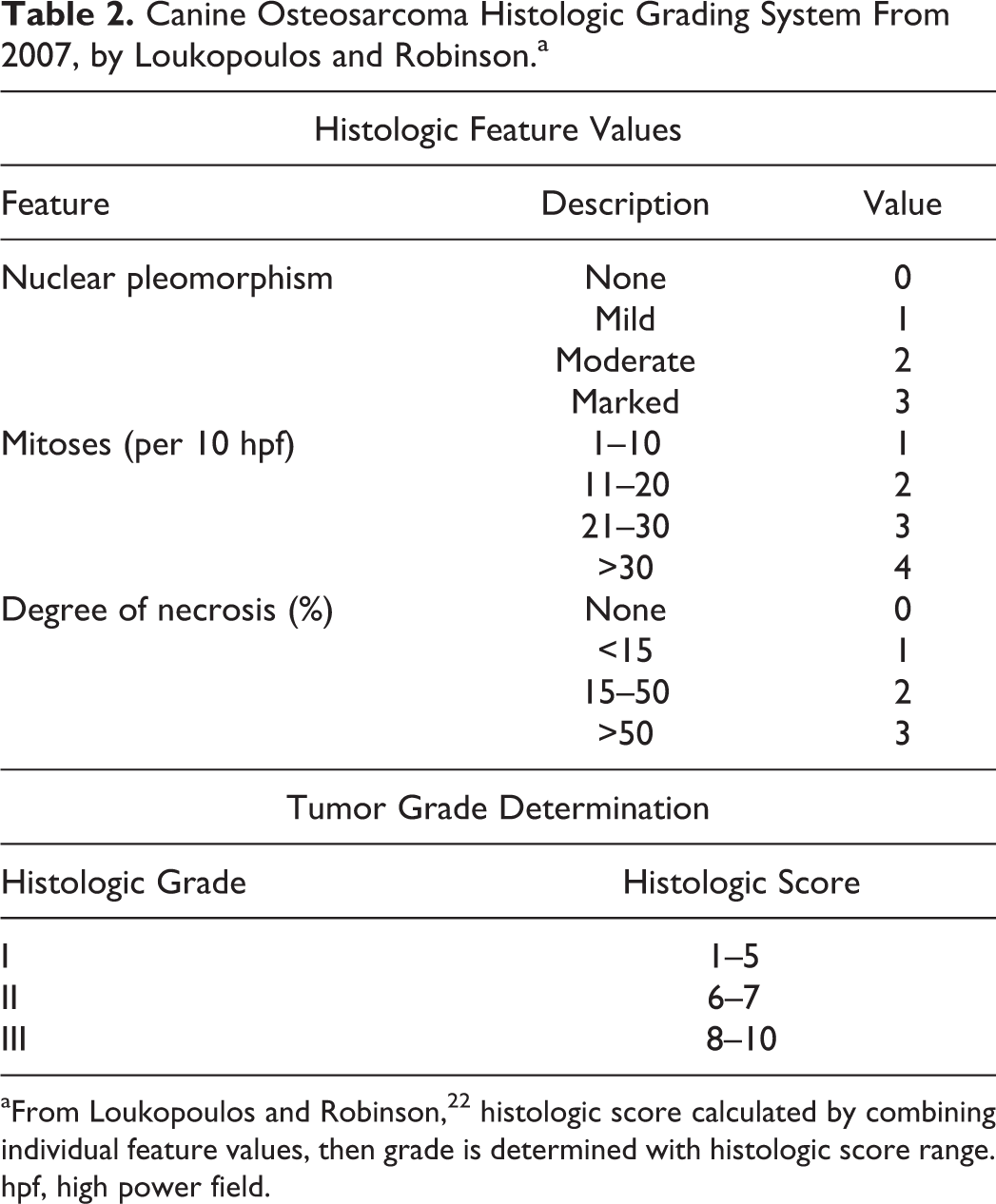
aFrom Loukopoulos and Robinson, 22 histologic score calculated by combining individual feature values, then grade is determined with histologic score range. hpf, high power field.
For continuous variables, univariate analyses were performed using a Cox proportional hazards model, and for categorical variables, Kaplan-Meier survival functions were compared using the logrank test to identify correlations with DFI and ST. Survival analyses were performed with R statistical analysis software 32 using the survival package, 40 and correlations were considered significant if P < .05. Dogs lost to follow-up (ie, euthanasia/death was never confirmed) or confirmed to be alive at the end of the study were censored during statistical analysis. Survival analyses were performed on the population twice, first for euthanasia/death by any cause (ie, all deaths considered to be events) and second for euthanasia/death attributed to osteosarcoma (ie, only deaths attributed to osteosarcoma considered to be events). Interobserver analyses were performed using R statistical analysis software 32 with the irr package. 14 Agreement and Fleiss’s kappa were employed for categorical variables, and intraclass correlation coefficient (ICC: 2-way, absolute agreement) was used for continuous variables. The data analyzed in this study can be made available upon request to the author.
Results
Case Selection and Clinical Data
From January 1998 to September 2014, 217 cases of appendicular osteosarcoma were identified; 85 met all inclusion criteria. Serum ALP was above the normal reference range for 20 of 84 dogs (24%); presurgical serum ALP was unavailable for 1 dog.
The study population was composed of 45 of 85 (53%) castrated males, 34 of 85 (40%) spayed females, 5 of 85 (6%) intact males, and 1 intact female. Of the 85 dogs, 64 (75%) were purebred, representing 26 breeds, and 21 (25%) were mixed-breed. Breeds represented by more than 2 dogs were: Golden Retriever (12), Rottweiler (9), Labrador Retriever (6), Greyhound (6), Doberman Pinscher (4), Great Dane (3), and Bullmastiff/Mastiff (3). At the time of diagnosis, body weight ranged from 14.8 to 70.2 kg (mean = 39.3 kg), and age ranged from 1.33 to 13.95 years (mean = 7.99 years) with only 7 dogs under 5 years of age.
Of the 85 primary tumors, 45 (53%) arose in a forelimb, and 40 (47%) arose in a hindlimb, with 45 (53%) located distally, 39 (46%) located proximally, and a single midshaft tumor. The most common site was the proximal humerus (22/85; 26%), followed by distal radius (20/85; 24%), distal tibia (13/85; 15%), proximal tibia (11/85; 13%), distal femur (10/85; 12%), proximal femur (6/85; 7%), distal ulna (2/85; 2%), and midshaft humerus (1/85; 1%). Of the 85 tumors, 47 (55%) were on the left, and 38 (45%) were on the right.
Histopathologic Findings
All 3 evaluators used osteoblastic, chondroblastic, fibroblastic, and telangiectatic histologic subtypes; in addition, evaluator B also assigned giant cell subtype (Supplemental Table S2). All 3 evaluators agreed in 57% of cases (Fleiss’s kappa = 0.393, fair agreement).
The level of pleomorphism assigned (as a categorical variable: none, mild, moderate, or severe) was identical for all evaluators in 23% of cases (Fleiss’s kappa = 0.115, slight agreement). Comparing all 3 evaluators, ICCs for the assigned percentages for pleomorphism, matrix, and necrosis were 0.342, 0.473, and 0.626, respectively (poor, fair, and good agreement, respectively).
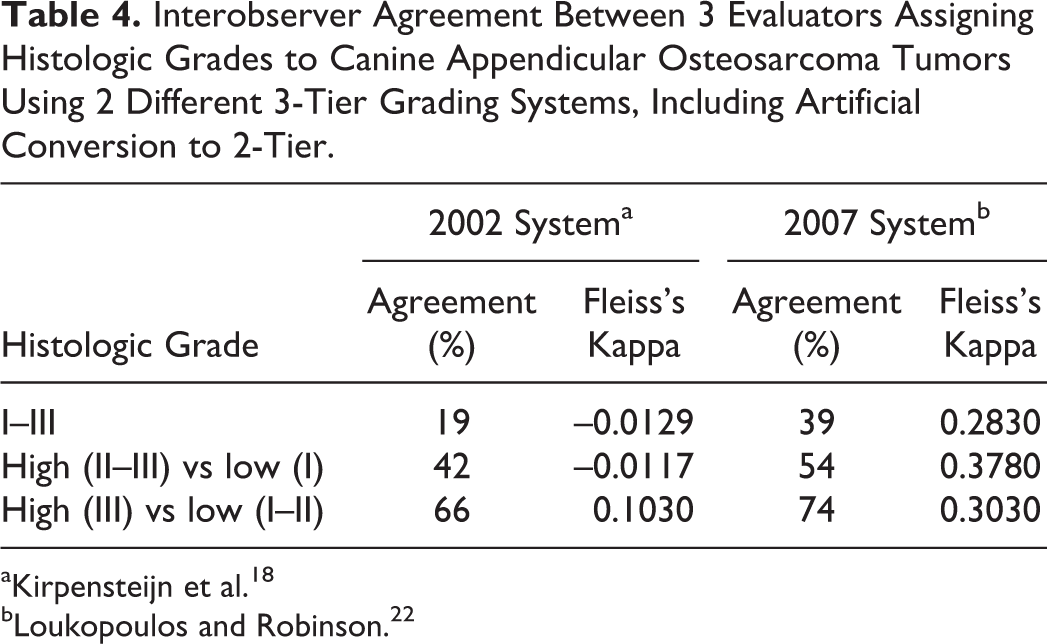
All tumors selected for histologic grading were amputation specimens; no core biopsies were included. The original histopathology report was available for all but 1 case. Evaluator B determined that the neoplastic tissue area from a single case was inadequate (<10 hpf) to be graded using the 2007 system. The final grades as assigned by each evaluator as well as the original grade (assigned at the time of diagnosis) are summarized in Table 3. The 2002 system yielded more grade II tumors overall, while the 2007 system yielded more grade I tumors (Table 3). The amount of agreement for histologic grade between the evaluators is summarized in Table 4.
Summary of Histologic Grades Assigned to Canine Appendicular Osteosarcoma Tumors by 3 Evaluators Using 2 Different Systems, With Comparison to the Original Histology Report and the Cases Published in the Original Studies.
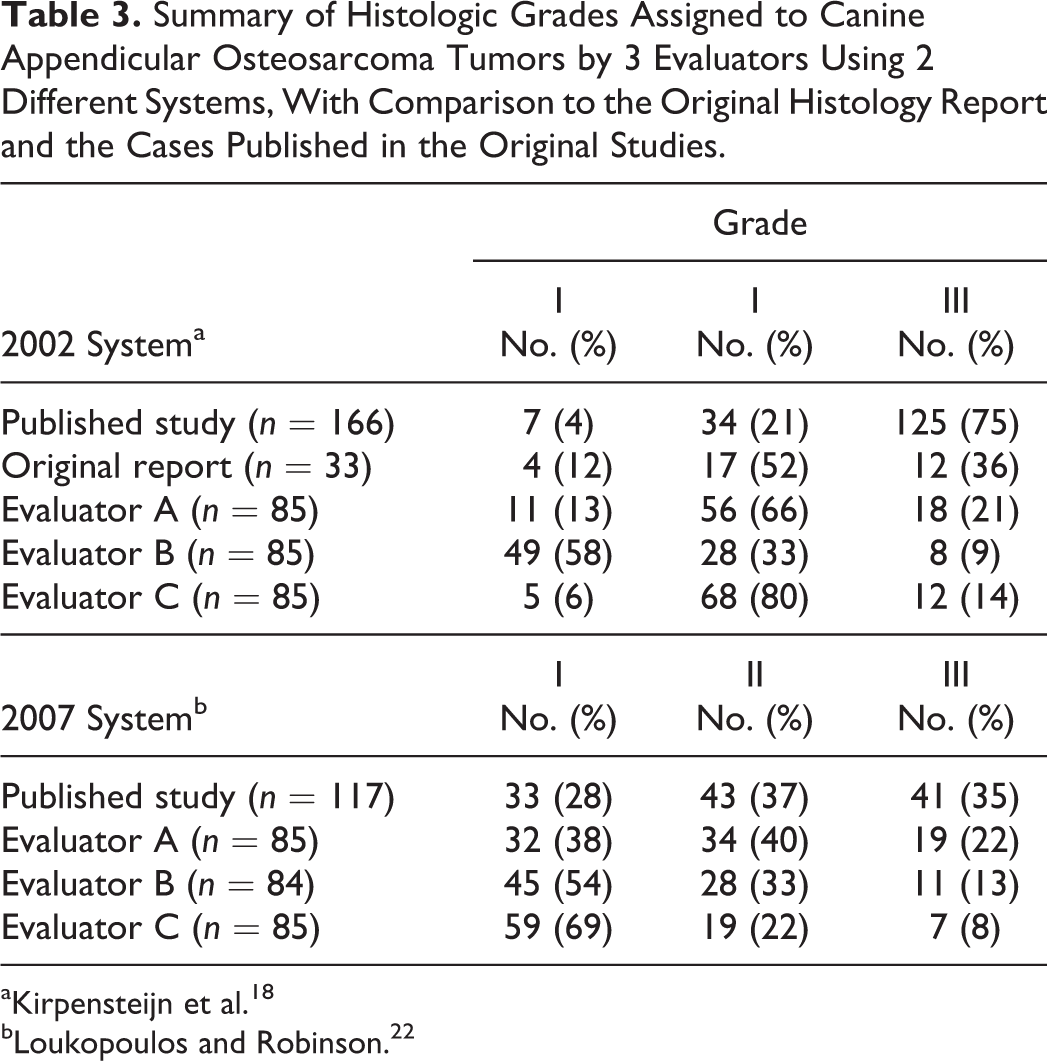
aKirpensteijn et al. 18
bLoukopoulos and Robinson. 22
Interobserver Agreement Between 3 Evaluators Assigning Histologic Grades to Canine Appendicular Osteosarcoma Tumors Using 2 Different 3-Tier Grading Systems, Including Artificial Conversion to 2-Tier.
aKirpensteijn et al. 18
bLoukopoulos and Robinson. 22
Treatment
Dogs received 1 to 9 doses of chemotherapy, with the majority receiving 4 to 6 doses (66/85; 78%). Typically, dogs received single-agent chemotherapy with either carboplatin (68/85; 80%) or doxorubicin (9/85; 11%). The remainder received multiple agents (8/85; 9%), including alternating doses of the aforementioned drugs, and 1 dog also received a dose of epirubicin. Of the 16 of 85 dogs (19%) who received ≤3 doses of chemotherapy, discontinuation of treatment was due to the diagnosis of metastatic disease in 11 of 16 cases (68%), adverse reaction to the drug in 4 of 16 cases (25%), and was unknown for a single case. A total of 30 of 85 dogs (35%) received additional treatment after completing chemotherapy, including cyclophosphamide, toceranib, or pamidronate plus radiation therapy.
Metastases
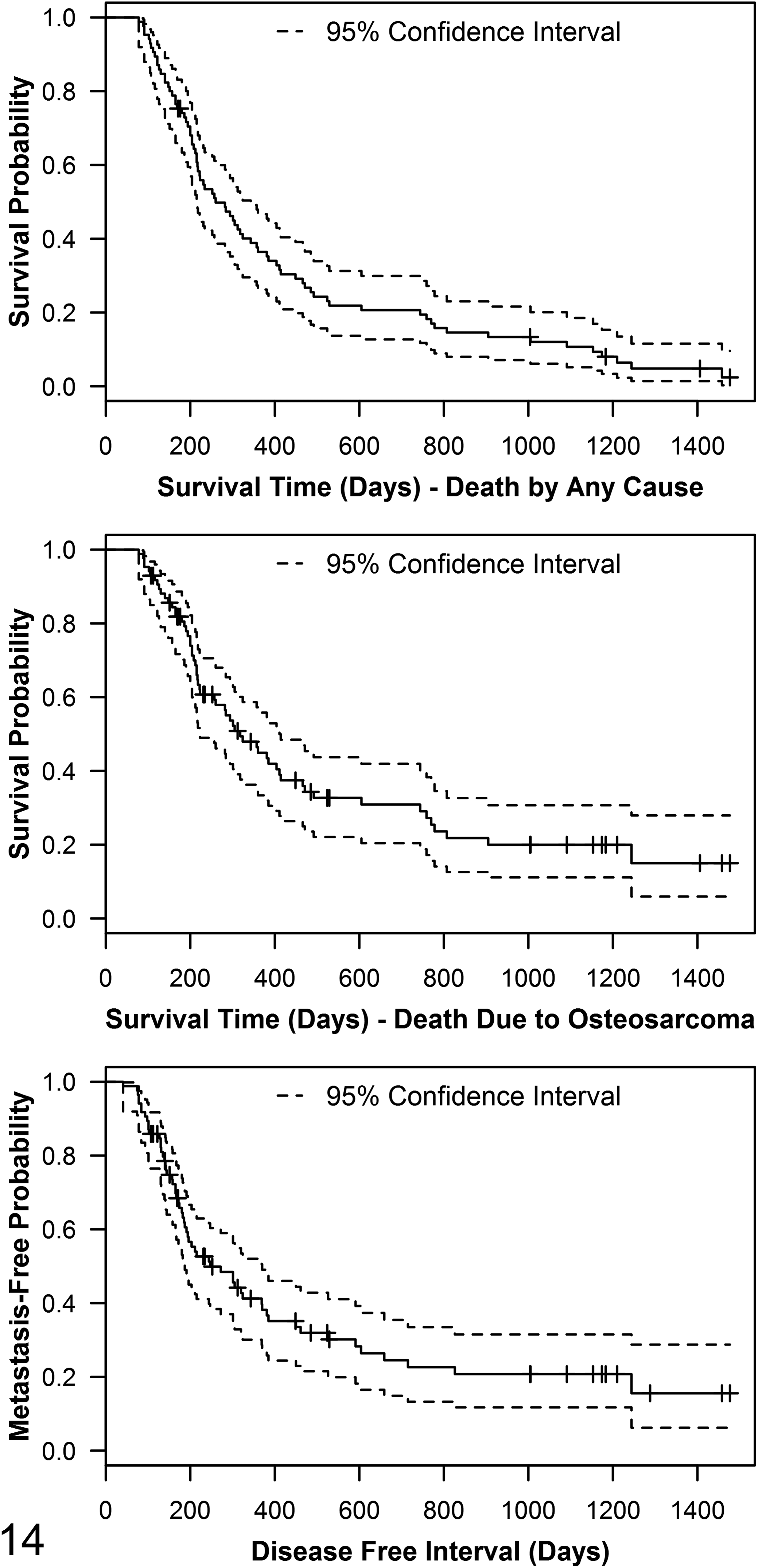
Metastatic disease was absent in 13 of 85 dogs (15%) at the time of their death, as well as in 2 of 85 dogs (2%) that were alive at the end of the study (December 2016). Of the 85 dogs, 10 (12%) had an unknown metastatic disease status either because they were lost to follow-up or euthanized by the referring veterinarian and the information could not be obtained. Metastatic disease was confirmed in 54 of 85 dogs (64%) based on medical imaging or postmortem examination, and a further 6 of 85 dogs (7%) were suspected to have developed metastatic disease based on clinical history or medical imaging, but the diagnosis was never confirmed; a single dog with confirmed metastatic disease was also diagnosed with histiocytic sarcoma and was censored from DFI survival analyses as the origin of the metastases was not confirmed. The median DFI was 247 days (Population size [events] = N [n] = 85 (59), 95% CI, 187–370; range, 41–1477 days, Fig. 14). Metastases were confirmed in the lung of 45 of 85 dogs (53%), bone of 14 of 85 dogs (16%), sternal lymph node of a single dog, and other sites of 13 of 85 dogs (15%), including: liver (3), kidney (5), spleen (2), heart (3), skin (4), subcutis (3), urinary bladder (1), abdominal fat (5), skeletal muscle (1), and adrenal gland (1). A total of 16 of 85 dogs (19%) had metastases confirmed in multiple tissues.
Survival
For overall survival analyses (ie, death by any cause), a total of 6 of 85 dogs (7%) were censored from the population, including 2 that were still alive and 4 that were lost to follow-up at the end of the study (December 2016); 2 of the dogs were lost to follow-up after 1000 days. The median survival time (MST) was 260 days (N [n] = 85 [79], 95% CI, 215–343; range, 78–1477 days, Fig. 14).
Of the dogs confirmed to have died during the study period (79/85; 93%), 58 of 79 deaths (73%) were considered to be directly due to osteosarcoma. Of these 58 dogs, 48 (83%) died or were euthanized due to progression of osteosarcoma as per the medical record, either with clinical signs that the clinician attributed to metastatic disease (including coughing, respiratory distress, lethargy, and pain associated with the skeleton) or with a radiographic diagnosis of metastasis (ie, owners elected for euthanasia as soon as metastatic disease was identified). For the remaining 10 of 58 (17%), no specific cause of death was listed in the medical record, but the deaths were attributed to osteosarcoma because metastatic disease was either confirmed (by previous medical imaging or seen at postmortem examination, 7/10; 70%) or suspected (clinically or radiographically, 3/10; 30%).
For the 21 of 79 (27%) dogs whose deaths were not attributed to osteosarcoma, cause of death/euthanasia for 14 of 21 (67%) was confirmed to be unrelated to osteosarcoma (reasons for euthanasia provided in the medical record in these cases included: 6/14 [43%] “old age,” 5/14 [36%] an alternate neoplasm, 2/14 [14%] pneumonia, and 1/14 [7%] bilateral cruciate rupture), and for 7 of 21 (33%), the cause could not be definitively determined based on the medical history. After censoring these 21 dogs, the MST for death due to osteosarcoma was 318 days (N [n] = 85 [58], 95% CI, 223–411; range, 78–1477 days, Fig. 14).
Survival Analysis
All continuous variables included in the univariate analyses met the proportional hazards assumption. Results from the univariate analyses for ST and DFI are shown in Supplemental Tables S3–S5. The number of mitoses per 3 randomly selected hpf was the only factor that showed statistical significance, for ST (death due to osteosarcoma; N [n] = 85 [58], HR = 1.04, 95% CI, 1.00–1.08; P < .05, Supplemental Table S4) and DFI (N [n] = 85 [58], HR = 1.05, 95% CI, 1.01–1.09; P < .05, Supplemental Table S5), as counted by only 1 of the 3 evaluators (B).
There were no significant differences between the Kaplan-Meier survival functions for ALP status (normal vs high) or sex for ST or DFI (data not shown). The MSTs for death by any cause for the most common tumor locations were 208 days (N [n] = 22 [20], 95% CI, 186–318) for proximal humerus, 488 days (N [n] = 20 [19], 95% CI, 252–807) for distal radius, 301 days (N [n] = 13 [12], 95% CI, 194–n/a) for distal tibia, 204 days (N [n] = 11 [11], 95% CI, 157–n/a) for proximal tibia, 287 days (N [n] = 10 [9], 95% CI, 126–n/a] for distal femur, and 466 days (N [n] = 6 [5], 95% CI, 234–n/a) for proximal femur. Dogs with distal radial tumors had a statistically significant survival advantage over dogs with proximal humeral tumors (Supplemental Fig. S1). There were significant differences between ST and/or DFI Kaplan-Meier survival functions for right versus left and for distal versus proximal tumor location (Supplemental Fig. S1).
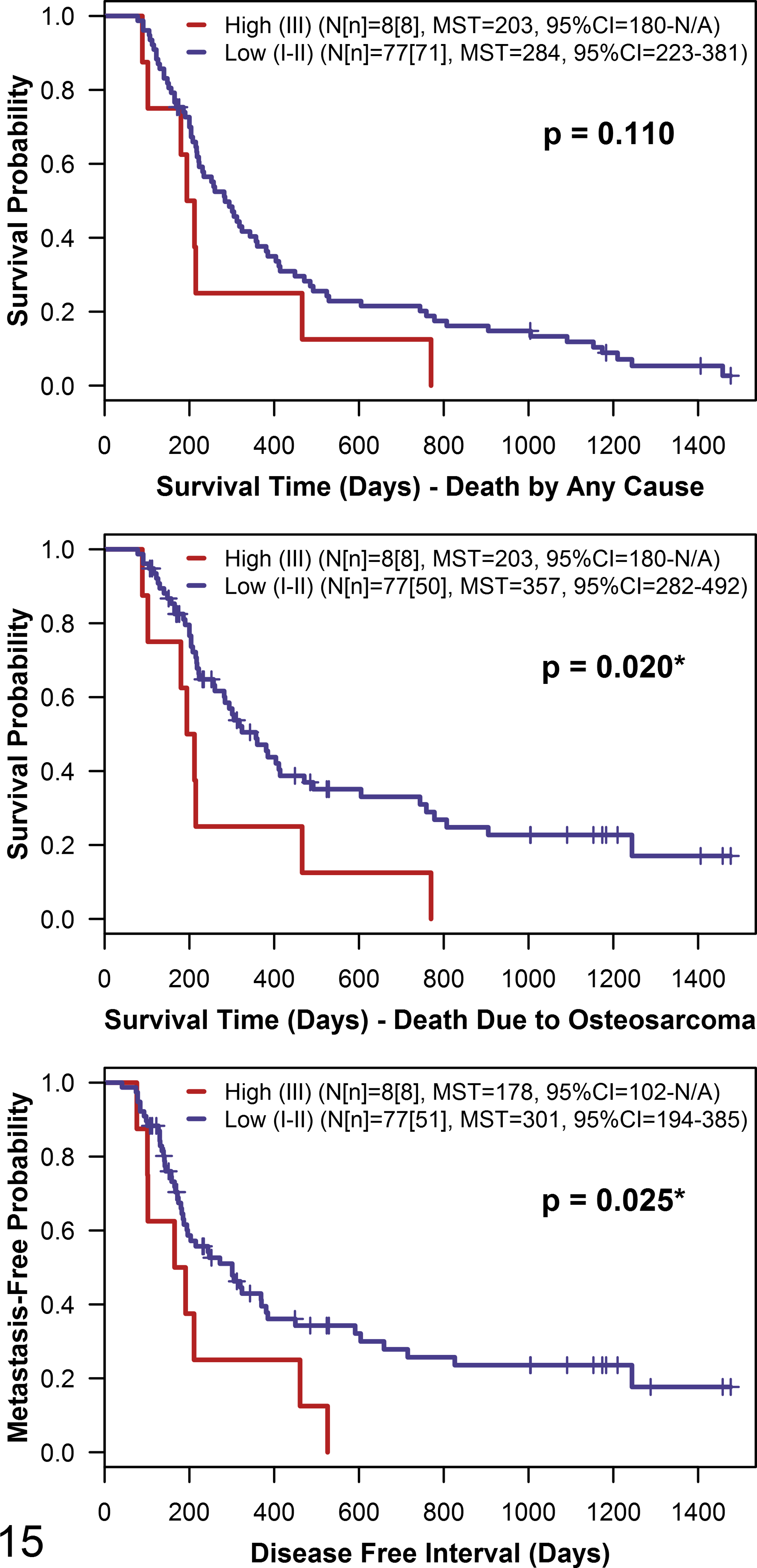
There were no statistically significant differences between Kaplan-Meier survival functions stratified by histologic grade as assigned by any evaluator for either 3-tier grading system for either ST or DFI (Supplemental Figs. S2, S3). Grade III tumors as assigned by evaluators A and B using the 2007 system tended to have decreased ST and DFI; for the 2002 system, the same trend was only seen for evaluator B (Supplemental Figs. S2, S3). With the 2002 system converted to a 2-tier grading system, for a single evaluator (B), dogs with low (I–II) grade tumors lived significantly longer and had a significantly longer DFI than dogs with high (III) grade tumors (Fig. 15; data not shown for evaluators A and C); less than 10% of tumors were designated as high grade after conversion to a 2-tier system (Fig. 15). There were no significant differences between the Kaplan-Meier survival functions for histologic subtypes as assigned by any evaluator for ST or DFI (data not shown).
Discussion
In veterinary pathology, histologic grading systems are often developed using a single study population with tumors evaluated by a single pathologist 10,25,29 ; rarely have studies employed multiple grading systems, 24 multiple evaluators, 19 or both. 34 Our objective was to evaluate the prognostic value of histologic grading retrospectively in dogs who received the standard of care for canine appendicular osteosarcoma by employing 3 evaluators using 2 different published grading systems. In this study, no correlations were observed between grade and either median survival time or time to development of metastatic disease. In addition, the amount of interobserver agreement for grade assignment was poor for both grading systems evaluated. There is variation in the prognostic utility of histologic grading systems in canine osteosarcoma, and some published grading systems were developed without controlling for anatomic site or treatment received (Supplemental Table S1).
Most grading systems for canine osteosarcoma employ almost identical histologic criteria, yet not all studies applying these systems identify a correlation with disease outcome (Supplemental Table S1). This may result from variation in the designated number of hpf for counting mitoses (3 18 vs 10 22 ), variation in the histologic score ranges, and whether or not lymph node status and/or cellularity are incorporated into the grading system. Additionally, a pathologist’s level of experience with examining bone tumors could also have an impact. One major difference between the 2002 and 2007 systems is how the individual histologic features are used to determine the final grade. The 2007 system uses overall histologic score tallied from individual histologic feature values to assign the final grade (Table 2). The 2002 system provides a “classification schedule” (Table 1) to determine the final grade; however, no specific instructions are provided for how to use the schedule. 18 All 3 evaluators in the current study determined final grade using a slightly different method for the 2002 system, which may explain why agreement between pathologists in our study was better for the 2007 system (Table 4). Concordance among pathologists should be required for widely adopted classification systems and grading systems. Agreement between the 3 evaluators in this study improved when each of the 3-tier systems was converted to a 2-tier system (Table 4), particularly when low grade was determined by combing grades I and II and high grade included only grade III tumors, indicating that agreement was highest for grade III tumors. However, per Fleiss’s kappa statistic, there was still only slight to fair agreement after conversion to 2-tier (Table 4). Based on the low concordance between evaluators in this study, neither histologic grading system is appropriate for widespread adoption.
The signalment and tumor characteristics of our study population are consistent with the common features observed in dogs diagnosed with appendicular osteosarcoma. Overall, the study population had more males than females, which is commonly reported, and the mean age at diagnosis was in the appropriate range of 7 to 9 years. 28 Dogs diagnosed with osteosarcoma rarely weigh <15 kg 44 ; fittingly, only a single dog in our study had a weight below this cut-point. Proximal humerus and distal radius were the most common tumor sites, and similar to other studies, dogs with proximal humeral tumors had a shorter ST and DFI than dogs with distal radial tumors (Supplemental Fig. S1). 44 The statistically significant survival advantage in dogs with distally located tumors (regardless of which bone was involved) in our study also reflects this common feature of the disease (Supplemental Fig. S1). Additionally, although the proximal humeral tumors were evenly divided between right and left, most distal radial tumors were on the left side, which may partially explain the significantly longer ST when tumors were located on the left (Supplemental Fig. S1). Asymmetry has been reported in various human neoplasms with the left side of the body being more prone to mammary carcinoma and melanoma while lung, testicular, and ovarian tumors are more common on the right; these asymmetries are not typically associated with outcome. 43 In colorectal cancer, there is increased risk of death when the tumor is located on the right versus the left, but this more accurately reflects proximal versus distal location rather than asymmetry. 30 The MST in our population, for either death attributed to osteosarcoma (318 days) or for any reason (260 days), is similar to other studies where the dogs received the standard of care. 23,31,33
Both carboplatin and doxorubicin are accepted chemotherapeutics for the treatment of appendicular osteosarcoma; the specific chemotherapeutic agent and number of doses for the standard of care are not universally defined. Diagnosis of metastatic disease was the main reason for early discontinuation of chemotherapy (≤3 doses) in our study, and therefore omitting these dogs from survival analyses would have resulted in exclusion of the most aggressive tumors. Most dogs in this study received a minimum of 4 doses of single-agent carboplatin or doxorubicin following amputation, which is in line with the standard of care; however, some dogs received alternating treatments with both drugs. A large study of 470 dogs evaluating various single-agent or alternating doxorubicin and carboplatin protocols revealed no significant differences among treatment protocols in DFI or ST of dogs with appendicular osteosarcoma. 36 Although the chemotherapy regimen varied slightly across our study population, the protocols were all in line with the standard of care; therefore, treatment effect is unlikely to have impacted our patient outcome.
Numerous risk factors and prognostic indicators have been proposed for canine appendicular osteosarcoma. Two meta-analyses identified ALP and proximal humeral location as 2 of the main negative prognostic indicators. 5,35 In our study, proximal humeral location was similarly correlated with a negative outcome, namely, both a shorter ST and DFI (Supplemental Fig. S1). Although serum ALP is commonly used as a prognostic indicator, similar to our study, other investigators have also been unable to find a correlation with ST. 9,33 One study found that absolute tumor burden affects bone ALP, raising the question of whether tumor burden is the true negative prognostic indicator rather than ALP level itself. 38
Several limitations exist in the current study. Most dogs were diagnosed and all dogs were treated at the same tertiary care facility; therefore, the study population may not be representative of the total population of dogs diagnosed with and treated for appendicular osteosarcoma. In addition, cases were not further stratified for the treatment received after completion of the standard of care. Finally, as this was a retrospective study, retrieval of all relevant outcome data was not possible for all dogs; some medical records from referral clinics had been destroyed by the time data were collected.
Although it is frequently stated in the veterinary literature that histologic grade is prognostically significant in human osteosarcoma, 18,20,22,26 to our knowledge, no specific histologic grading system for treatment-naïve human osteosarcoma is described in the literature; however, it appears that a 2-tier modified Broders system (originally designed for epithelial tumors) is commonly applied and evaluates similar features to the canine grading systems (cellularity, nuclear features, mitotic rate, and necrosis). 6,13,42 High grade intramedullary osteosarcoma is termed “conventional” osteosarcoma in human patients, while low grade tumors in the same location are categorized as “low grade central” osteosarcoma; these diagnoses are distinct and are classified separately under the World Health Organization’s Classification of Bone Tumours. 13 This is in contrast to canine osteosarcoma where all cases of appendicular osteosarcoma are managed similarly regardless of grade. In 1 human study, 8 expert orthopedic pathologists graded 12 different osteosarcoma tumors from various anatomic locations. 17 The 8 evaluators each used a grading system of their choosing, ranging from 2 to 4 tiers, all of which were converted to a 2-tier system (high vs low grade) for comparison. 17 Agreement by all pathologists on histologic grade was reached in 50% of cases with a kappa coefficient of 0.547 (fair to moderate agreement). Thus, interobserver variability in histologic grade assignment exists among human orthopedic pathologists, just as it did with the veterinary pathologists in our study. One factor that separates this human study from our canine study is that the grading system used was at the pathologist’s discretion; one could speculate that concordance between the human pathologists may have improved with the use of a common system. In the end, Kilpatrick et al 17 concluded that a universal grading system incorporating more than just histological features (eg, subtype and clinicoradiological information) could be attempted by identifying practical as well as reproducible histologic and clinical criteria for its development. At present, the 2 most common osteosarcoma staging systems used by human orthopedic pathologists, the MSTS (Enneking) system for malignant musculoskeletal neoplasms and the American Joint Committee on Cancer (AJCC) system for bone sarcomas, both incorporate histologic grade. 7
With the objective of identifying histologic features associated with prognosis in human osteosarcoma, 1 group assessed tumors from 165 patients, evaluating vascular invasion, mitotic rate, nuclear pleomorphism, tumor viability, tumor necrosis, amount of osteoid, and histologic subtype in treatment-naïve initial biopsies; none of the individual histologic features achieved statistical significance. 8 During the assembly of the current study, various combinations of features and ranges for histologic evaluation/scoring were combined in an attempt to retrospectively develop a prognostically significant histologic grading system, to no avail (data not shown).
The results of this study highlight the importance of employing more than a single evaluator during the development of histologic grading systems for the purpose of prognostication in veterinary oncology; use of more than 1 study population is also desirable. Unfortunately, the value of routine histologic grading of canine appendicular osteosarcoma amputation samples is called into question by the lack of correlation between individual histologic features or histologic grades and outcome in dogs receiving the standard of care. This is disappointing as there are few prognostic tools available for canine osteosarcoma, and histologic grading is a relatively simple and inexpensive test that requires no additional sample collection beyond that already collected for diagnostic purposes. For routine histologic grading of canine appendicular osteosarcoma to provide meaningful prognostic or treatment guidance to clinicians and owners, a clearly outlined well-defined histological review process that results in reproducible categorization is required. The question remains whether such a system is attainable for canine appendicular osteosarcoma or whether efforts would be better directed toward the pursuit of molecular markers of prognosis. The findings of the current study call into question the value of histologic grading as a routine test for prognostication in canine appendicular osteosarcoma. Moving forward, the larger veterinary oncology community should take the opportunity to collaborate across institutions in multicenter, multi-pathologist studies to develop, refine, and confirm the validity of tumor grading systems to predict disease outcome.
Footnotes
Supplementary material for this article is available online.
Acknowledgements
We thank the Animal Health Laboratory (AHL), especially the histotechnology team, for assistance with retrieval of archived blocks and slides and for the provision of re-cut slides when necessary. We also thank Dr Murray Hazlett of the AHL for his assistance with the retrieval of cases diagnosed prior to 2008 and the OVC HSC Client Logistical Services team for filling our many requests for patient records.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: OVC Fellowship Program and Ontario Graduate Scholarship Program to C.R.S., and OVC Pet Trust funded the research and publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
