Abstract
Five adult Saanen goats received a single oral dose of Heterophyllaea pustulata containing 42.25 μg/kg rubiadin (anthraquinone) and 3 adult goats were untreated controls. All goats were exposed to sunlight and sequential ear skin biopsies were collected before treatment and at 32 hours, 3 days, 8 days, and 15 days after treatment. Changes at 32 hours after dosing included epidermal spongiosis, single cell death and acantholysis, an increased BAX/BCL-2 protein ratio, and dermal edema. Lesions at day 3 included epidermal and adnexal necrosis, crust formation, and acanthosis. Acanthosis, hyperkeratosis, and dermal fibrosis and neovascularization were present at day 15. The pro-apoptotic (BAX)/anti-apoptotic (BCL-2) protein ratio increased at 32 hours, whereas epidermal and dermal PCNA immunolabeling increased between days 8 and 15 after treatment. The cutaneous lesions were consistent with sunlight-induced damage, and the occurrence in treated but not control goats indicates photosensitization.
Keywords
Photosensitization dermatitis is a frequent disorder in animals and humans that occurs when photodynamic pigments are deposited in sunlight-exposed skin. 10 Numerous studies of solar radiation have focused on the direct ultraviolet (UV) effects on the skin. 2,15 Photosensitizing substances absorb UV or visible light, exciting their electrons. As electrons return to a less excited state, there is electron transfer to different substrates of the medium, generating superoxide anion first and then other oxygen radicals and/or a release of energy; this process produces highly reactive singlet oxygen. These reactive oxygen species in turn react and damage lipid membranes, proteins, or nucleic acids, causing cell injury and death. 9,10,16
Anthraquinones with photosensitizing activity in natural and experimental cases in animals are found in certain plants such as Heterophyllaea pustulata (cegadera), Galium spp., and Alternanthera philoxeroides. 1,12 –14
Most dermatopathological studies addressing photosensitization dermatitis involve natural cases 12,17 ; however, no experimental studies or reports have explored the sequence of changes that occur during the pathogenesis of cutaneous lesions induced by photosensitizing substances. The present study aimed at investigating the histopathological characteristics induced by photosensitization by performing a sequential analysis of lesion development.
The study protocol was approved by the Committee for Care and Use of Laboratory Animals of the School of Veterinary Sciences, National University of La Plata, Ref. Number 62-3-17T. Requirements of the Argentine Policy for Animal Protection (Ley 14346) and the European Union Directive 2010/63 on protection of experimental animals were fulfilled. The study was conducted at the experimental research farm of the Estación Experimental Agropecuaria, Salta, National Agricultural Technology Institute.
The experiment involved eight 2- to 3-year-old female Saanen goats (average weight 50 kg). This breed was selected because it has nonpigmented ears virtually devoid of hair. The experimental group (n = 5) received a single dose (5 g/kg body weight) of dried leaves of H. pustulata, mixed with water, at room temperature. Total anthraquinone concentration in the crushed plant samples was 0.845 × 10−4% (w/w), expressed as concentration of rubiadin, the most important anthraquinone present in H. pustulata. 14,15 Therefore, the final dose of rubiadin provided to each animal was 42.25 μg/kg. The control group (n = 3) received 5 g/kg body weight of milled lucerne (alfalfa) hay. In all cases, doses were administered using an esophageal tube. All animals of both groups were exposed to sunlight for 7 days, 7 hours a day divided into 2 periods—7:00 to 11:00 and 15:00 to 19:00—to avoid day hours of maximal sunlight. The experiment was carried out in August, when the sunlight is high and no precipitations occur in the study area. The UV index varied between 6 and 11 during the sunlight exposure hours throughout the study period (Radiometer UV-S-E-T Kipp & Zonen). Animals had ad libitum access to feed (lucerne hay and commercial goat feed pellets) and water. Animals were examined every day and incisional skin biopsies were collected from the margins of the pinna (using local anesthesia) at 0 hours, 32 hours, 3 days, 8 days, and 15 days from the time of dosing.
Biopsy samples were fixed in 10% neutral buffered formalin and processed routinely. Sections stained with hematoxylin and eosin (HE) and with Masson’s trichrome were independently examined by 2 pathologists blinded to the group of animals, following the criteria detailed in Supplementary Table S1. Immunohistochemistry (IHC) for proliferating cell nuclear antigen (PCNA; (monoclonal; dilution:1/100, Novocastra), BAX (pro-apoptotic protein; polyclonal; dilution:1/30, BioGenex), and BCL-2 (anti-apoptotic protein; polyclonal; dilution:1/100, Abcam) was performed as previously described. 5,9 Cell proliferation and apoptosis were measured using CellSens Dimension (v1.6. Olympus) and Image-Pro Plus 6.3 (Media Cybernetics) image analysis software. IHC was quantified by analyzing 5 images per biopsy at 20× objective magnification; the mean percentage of immunolabeled area was obtained. In the case of apoptosis, the BAX/BCL-2 expression ratio was calculated. Differences between the treated and the control groups were determined using a nonparametric ANOVA test. Results are shown as mean ± standard deviation (SD). Results were considered statistically significant at P < .05.
Clinically, the affected goats exhibited photophobia, dermatitis, and photosensitization. Skin lesions comprised edema, erythema, and crust formation, followed by severe hyperkeratosis. This lesion affected the face, the pinna of the ears, and back and loin. Details of the clinical and biochemical changes were published elsewhere. 12 Phylloerythrin was not detected in sera but a high serum concentration of rubiadin and soranjidiol (the 2 dominant anthraquinones in H. pustulata) was observed at 24 hours and 3 days after treatment, coinciding with the most evident clinical signs. Therefore, clinical findings were associated with the presence of these anthraquinones in serum. 11
In the control group, the skin of the pinnae was thin and no lesions were detected during the experiment. The epidermal pegs were short and thick, and the dermal papillae were inconspicuous. Hair follicles, apocrine glands, sebaceous glands, and blood vessels were present in the dermis. Dermal collagen, as identified by Masson trichrome stain, was present in homogeneous horizontally arranged bundles surrounding the adnexa.
In the goats that were dosed with H. pustulata (Experimental Group), the cutaneous histologic lesions were characterized as extensive necrotizing dermatitis (Table 1, Figs. 1-4). At the initial sampling times, 32 hours to 3 days after dosing, the epidermis had degenerative changes that primarily affected the stratum spinosum and included spongiosis, diffuse acantholysis, and formation of intra-epidermal vesicles. Acute lesions in the dermis were most severe at the 32 hours and day 3 sampling times, and included edema characterized by the presence of extracellular proteinaceous eosinophilic material and disorganization of collagen fibers, as well as associated hemorrhages, and inflammatory cell infiltration. Inflammatory cells (mostly represented by neutrophils) were first located at the dermo-epidermal junction, extending toward the epidermal layer, and forming small aggregates. Perivascular and periadnexal proteinaceous exudate was also observed at this stage.
Histopathologic Lesion Scores in 5 Goats at Different Time Points After Oral Dosing With Heterophyllaea pustulata.
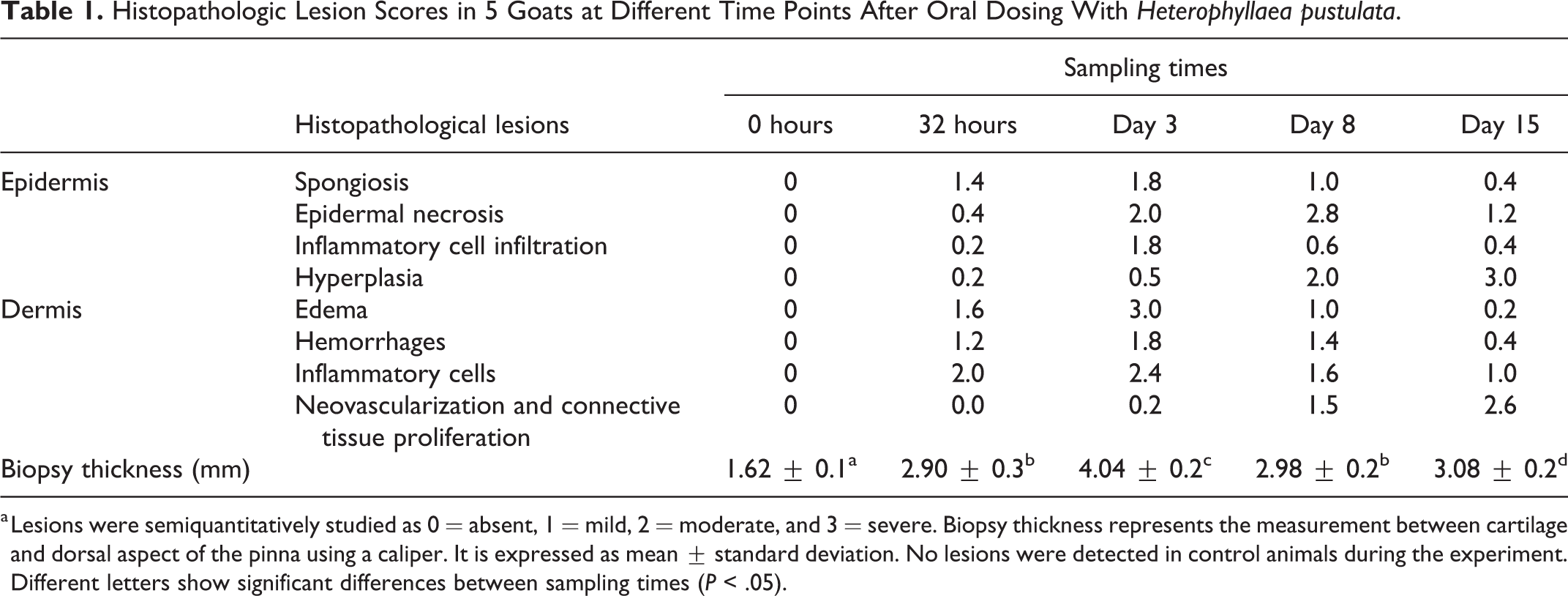
a Lesions were semiquantitatively studied as 0 = absent, 1 = mild, 2 = moderate, and 3 = severe. Biopsy thickness represents the measurement between cartilage and dorsal aspect of the pinna using a caliper. It is expressed as mean ± standard deviation. No lesions were detected in control animals during the experiment. Different letters show significant differences between sampling times (P < .05).
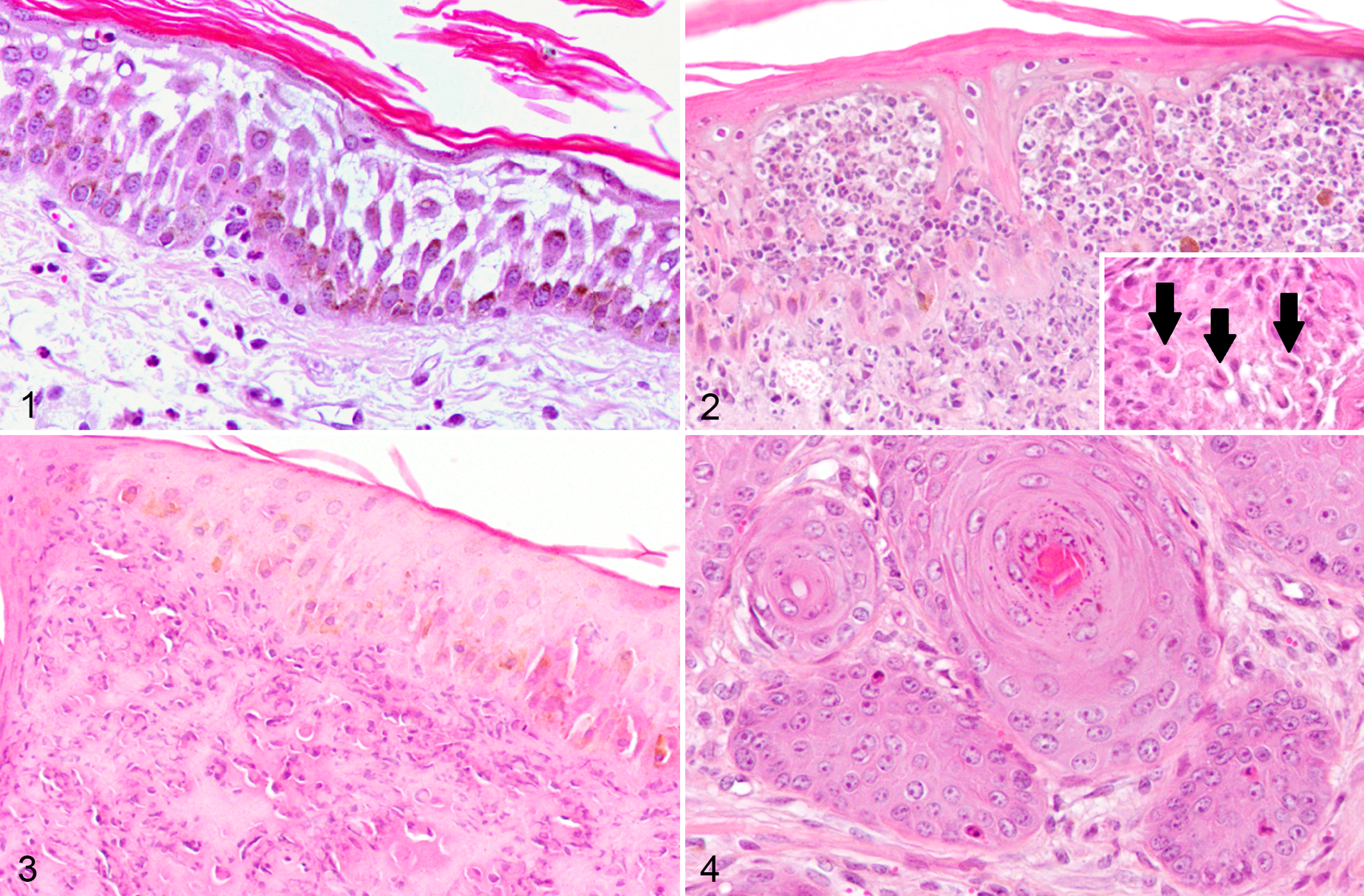
Heterophyllaea pustulata induced photosensitization, skin, goat. Hematoxylin and eosin (HE).
Between days 3 and 8, the stratum spinosum contained numerous single dead cells characterized by retraction of cell borders, hypereosinophilic cytoplasm, and pyknotic nuclei. Hyperplasia of the epidermis was present at this stage. At days 3 and 8, the most prevalent change was epidermal necrosis with crust formation. The necrotic tissue extended to the adnexa and was accompanied by hyperplasia of the stratum spinosum (acanthosis), mild increase of dermal collagen fibers, and vascular proliferation. At days 8 and 15, there was marked acanthosis with numerous mitotic figures in the basal layer, rete peg formation, and hyperkeratosis. Dermal changes at this stage were characterized by neovascularization, with formation of disorganized connective tissue. Endothelial cell enlargement and proliferation were observed throughout the study. Most of the above-described acute changes had almost disappeared by day 15 days after dosing.
The expression of PCNA, BCL-2, and BAX was evaluated by IHC in sequential skin biopsies (Figs. 5-11). All markers were expressed in the epidermis, whereas only PCNA was detected in the dermis. Compared to 0 hours, cellular proliferation increased significantly (P < .05) from 32 hours onwards. The highest values were recorded on days 3 and 15 after dosing. In the dermis, increased PCNA expression started at 24 hours, peaking on day 15. Expression of BCL-2 and BAX increased at 32 hours and day 3 as compared to day 0, with their values returning to basal levels at day 8. The BAX/BCL-2 ratio was highest at 32 hours after treatment, before epidermal necrosis was evident.
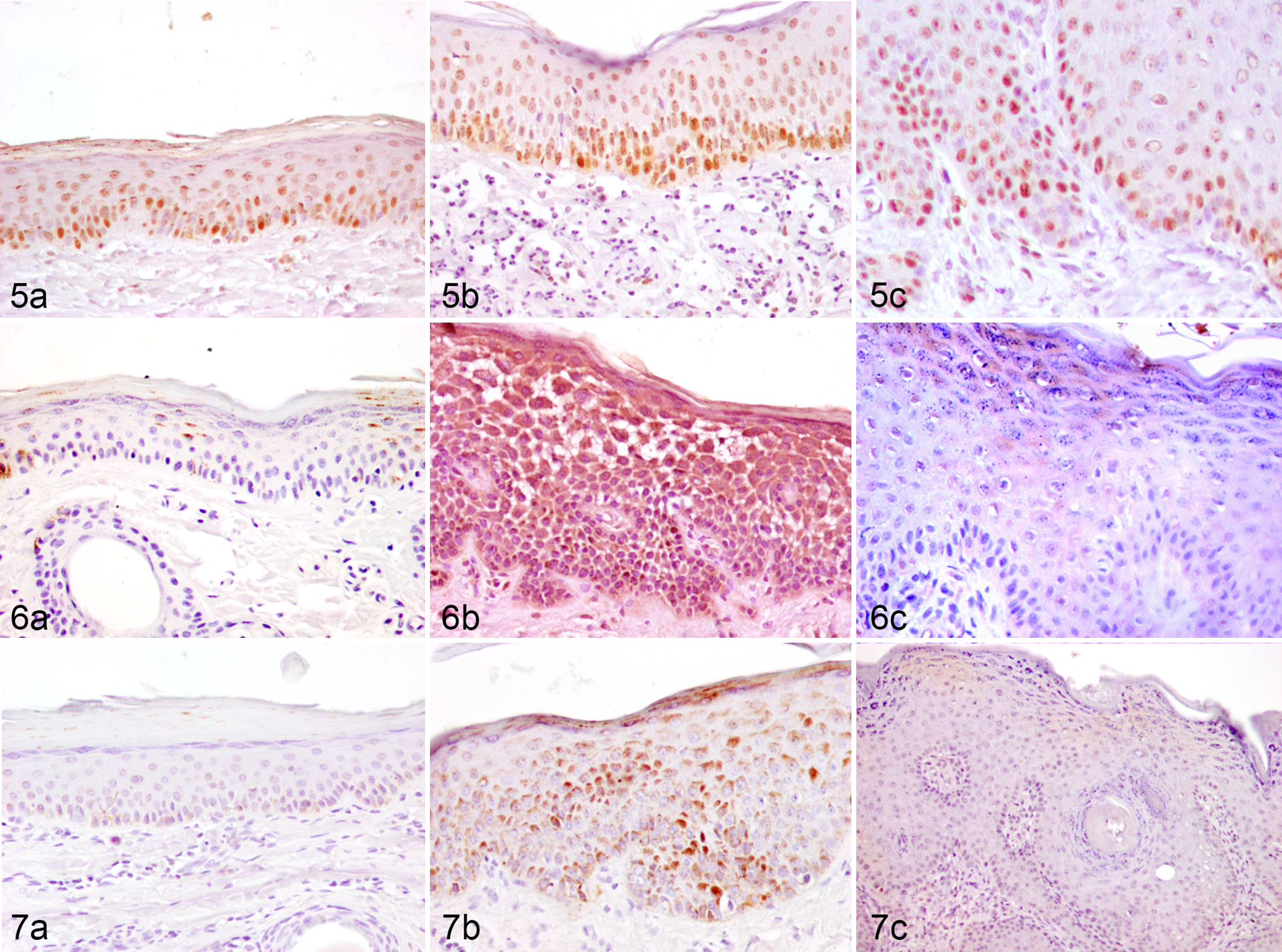
PCN, BAX, and BCL-2 expression following Heterophyllaea pustulata induced photosensitization, skin, goat.
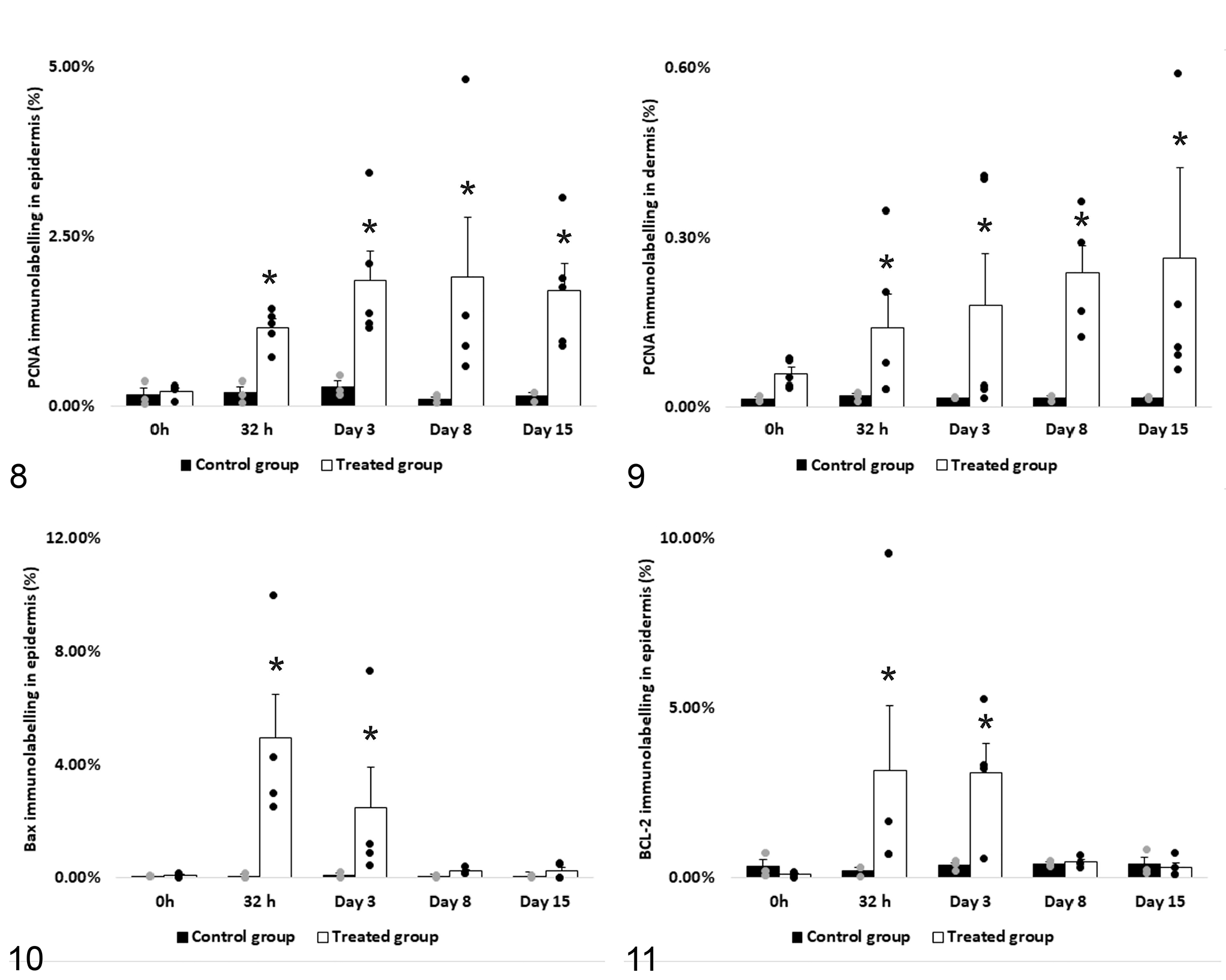
PCNA, and BCL-2 and BAX antigen expression determined by immunohistochemistry in skin biopsies. PCNA significantly increased in epidermis (a) and dermis (b), whereas BAX (c) and BCL-2 (d) only increased in the epidermis at 32 hours and day 3 post intake. Asterisks on the bars show significant differences between sampling times (P < .05).
Photosensitization in domestic animals is a common condition that has not been sequentially studied microscopically to our knowledge. In this work, we describe these changes in goats during a 15-day period. The lesion can be described as a severe, necrotic interface dermatitis, similar to that previously reported 12,16 and includes initial inflammatory and necrotic changes, followed by epidermal hyperplasia and neovascularization, and later connective tissue proliferation. The increase in single dead cells in the epidermis (day 3) was detected before the evidence of epidermal necrosis (day 8).
Acute changes described in this experiment at 32 hours and day 3 after dosing are similar to those observed in a severe acute solar dermatitis. 10,18 The appearance of single cell death of keratinocytes, corroborated by IHC methods, was compatible with sunburn cells and was very frequent at this stage. 17 The photosensitization agent was able to exacerbate the effect of solar radiation since control animals, under the same sun exposure conditions, did not show any histologic lesions in the skin during the experiment.
Most lesions observed at 8 and 15 days in this work were compatible with those observed in solar elastosis and/or solar keratosis. Solar elastosis is characterized by disorganization of dermal components with the appearance of scattered or agglomerated, thick, irregular, basophilic degenerated elastic fibers. 6 Solar keratosis is characterized by hyperplasia, spongiosis, acute dermal inflammation, and focal necrotic keratinocytes, 10 and is considered a common precancerous skin lesion in humans, cats, and horses. In the sections stained with HE and Masson’s trichrome, an increase in the amount of collagen fibers was observed at days 8 and 15, and disorganization and thickening of the subepidermal collagen was identified with Masson’s trichrome stain. Additionally, there was loss of many hair follicles and sebaceous glands during this period.
No vascular lesions other than endothelial hypertrophy and hyperplasia were observed in this study. However, a previous report refers to fibrinoid degeneration of blood vessels and thrombosis in the superficial dermis as a characteristic finding of photosensitization. 16 Our results suggest that vascular damage is not observed in this form of photosensitization dermatitis. The reported vascular lesions 16 might be dependent on the chronicity of the process, species-specific differences, or by of the microanatomic structures targeted by the photosensitizing compounds.
Several molecular and cellular events may occur in the skin as a result of UV irradiation, including changes in cell cycle activity, cellular repair, cell proliferation, and apoptosis. 3,4,15,17,18 In our work, the expression of PCNA, a nuclear protein marker of cell proliferation, was significantly increased in the epidermis during all time points in the treated group, a feature that was accompanied by epidermal hyperplasia. Mice irradiated with UV light also showed an increase of cell proliferation in the epidermis. 17 The proteins BCL-2 and BAX regulate activation of the caspase cascade in early apoptosis. BAX is an activator and BCL-2 is an inhibitor of caspase cascade activation. The increase of the BAX/BCL-2 expression ratio results in activation of the caspase cascade and consequently in apoptosis progression. 7 –9 It is known that patterns of apoptosis at the epidermal layer are modified by UV radiation, 3,15,18 but these changes have not been previously described in cases of photosensitization. We detected apoptotic cell death within 32 hours after the intake of the photosensitization agent, that is, immediately before the evidence of tissue necrosis. The time of exposure possibly determines different outcomes regarding cell death: apoptosis may occur initially, and prolonged insult may cause necrosis.
In conclusion, the present study demonstrates the changes in goat skin resulting from oral dosing with a photosensitizing agent followed by solar exposure. The lesions were similar to those occurring in natural cases. Early changes at 32 hours to 3 days included spongiosis, single cell death of epidermal cells, acantholysis, and dermal edema, with increased epidermal expression of BCL-2 and BAX at these times. Lesions were more evident after 3 days of exposure including epidermal and adnexal necrosis, crust formation, acanthosis, and dermal fibroplasia. Acanthosis, hyperkeratosis, and dermal fibrosis and neovascularization were present at day 15.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985821999322 - Photosensitization Induced by Heterophyllaea pustulata in Goats: Sequential Development of Skin Lesions
Supplemental Material, sj-pdf-1-vet-10.1177_0300985821999322 for Photosensitization Induced by Heterophyllaea pustulata in Goats: Sequential Development of Skin Lesions by Juan F. Micheloud, Lluís Luján, Luis A. Colque-Caro, Susana C. Núñez-Montoya, Claudio G. Barbeito, Enrique L. Portiansky and Eduardo J. Gimeno in Veterinary Pathology
Footnotes
Acknowledgement
We thank Dr Victor Suarez for his contribution to statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SCNM, ELP, CGB, and EJG are members of the Research Career of Consejo Nacional de Investigaciones Científicas y Técnicas.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Instituto Nacional de Tecnología Agropecuaria, Consejo de Investigación de la Universidad Católica de Salta (Res 1494/16) and PICT 2011/1379 (FONCyT).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
