Abstract
A Rock Alpine doe (Capra aegagrus hircus) was presented to the Colorado State University Veterinary Teaching Hospital because of scaling and ulceration over the withers, coronary bands, and dewclaws. The doe was euthanized because of poor prognosis associated with a radiographically identified cranial mediastinal mass, increased respiratory effort, and discomfort. Autopsy revealed a cranial mediastinal mass, and scaling-to-ulcerative lesions affecting the dorsum, ventrum, pinna, neck, teats, coronary bands, and dewclaws. Histologically, the mediastinal mass was an epithelial neoplasm with admixed non-neoplastic T lymphocytes, consistent with a lymphoepithelial (mixed) thymoma. Sections of affected skin were characterized by hyperkeratotic cell-rich interface dermatitis with transepidermal and follicular apoptosis. Thymoma-associated exfoliative dermatitis has been recognized in cats and a rabbit, but has not been reported previously in a goat, to our knowledge. Given that thymomas are not uncommon in goats, thymoma-associated exfoliative dermatitis should be considered a clinical differential in goats with dermatologic disease.
Thymomas arise from the epithelial component of the thymus. Traditionally, classification of thymomas has included epithelial, lymphocytic, and lymphoepithelial (mixed) subtypes, dependent on the degree of infiltration by non-neoplastic lymphocytes. 14 An additional classification of thymic epithelial neoplasms is based on the World Health Organization guidelines, described below. Histologic phenotypes are classified as type A (composed of spindle- or oval-shaped cells), type B (composed of epithelioid cells), and type AB (a mixture of both subtypes). 14 Although rare in most species, dairy goats may be predisposed to thymomas. 5 Thymomas were the third most common tumor in a survey of 102 tumors in goats. 7 Biologic behavior is most commonly benign, and affected goats may lack clinical signs. However, there is a single report of thymic carcinoma with metastases to the lung and spleen in a goat. 8 When present, clinical signs are frequently associated with mass effect, and include respiratory distress and congestive heart failure.6,9,10
Paraneoplastic syndromes are non-metastatic systemic disorders that often result from abnormal immune responses to neoplasia and can also result from non-immune mechanisms such as the humoral hypercalcemia of malignancy. Thymoma-associated exfoliative dermatitis is a paraneoplastic syndrome that is well-established in cats and has also been described in a rabbit.1,3,4,11,12 Additional paraneoplastic syndromes associated with thymomas include myasthenia gravis, polymyositis, erythema multiforme, and granulocytopenia.11,13 The lesion in thymoma-associated exfoliative dermatitis is proposed to be the result of autoreactive T lymphocytes that target keratinocytes. 4 The paraneoplastic nature of the lesion is supported by reports of regression of dermatologic signs post-thymectomy.2,11
Histologic features of thymoma-associated exfoliative dermatitis include transepidermal apoptosis, follicular apoptosis, and hyperkeratosis. Severity of transepidermal apoptosis is variable, although it is typically most severe in the basal cell layer. A lymphohistiocytic interface dermatitis is also variable and can be characterized as either cell-rich or cell-poor based on the degree of cellularity. Given the histologic similarities, erythema multiforme is considered a differential diagnosis for thymoma-associated exfoliative dermatitis, which may represent a milder form of erythema multiforme. Similarly, autoreactive T lymphocytes are also posited as the cause of keratinocyte apoptosis in erythema multiforme. When apoptosis is particularly mild and restricted to the basal cell layer but interface dermatitis is prominent, lupus erythematosus is considered an additional differential.
A 9-y-old, female Rock Alpine goat (Capra aegagrus hircus) was treated by the referring veterinarian because of a 3-d history of hyporexia and lethargy. The referring veterinarian diagnosed the doe with louse infestation, endoparasitism, and a heart murmur. No other goats on the farm were affected. Hematologic findings included regenerative anemia, stress leukogram, and hypoproteinemia. Despite treatment with ivermectin, the goat progressed to lateral recumbency and anorexia and was referred to the Colorado State University Veterinary Teaching Hospital (CSU-VTH). Physical examination demonstrated dermatitis with alopecia and a grade 4 of 6 systolic heart murmur, in addition to louse infestation. Additional blood parameters included hyperglycemia, and elevated creatinine, creatine kinase, and aspartate aminotransferase levels. Fecal flotation revealed a heavy burden of coccidia, strongyles, and Trichuris spp. The goat was hospitalized for 9 d and was treated with intravenous fluids, thiamine, fenbendazole, sulfadimethoxine, oxytetracycline, bismuth subsalicylate (Kaopectate; Chattem, Chattanooga, TN), and multiple blood transfusions. Clinical signs and clinicopathologic findings improved significantly, and the dermatitis resolved partially; therefore, the doe was discharged. Clinical signs were attributed to endoparasitism and louse infestation. Treatment for lice was continued by the owner after discharge.
Two months later, the goat was presented again to the CSU-VTH because of progression of the dermatitis. There was severe scaling and ulceration affecting ~60% of the body, especially over the withers, dewclaws, and coronary bands. On physical examination, the doe was dull, depressed, febrile, tachypneic, and tachycardic. The doe also had shifting leg lameness, head pressing, and reluctance to lie down. A complete blood count revealed neutrophilia with no anemia. Serum chemistry revealed hypoalbuminemia, hyperglobulinemia, hyperglycemia, hypomagnesemia, mild hypokalemia, and mild hypochloremia. Thoracic radiographs were performed because of increased respiratory effort. A large soft tissue opaque mass with ill-defined margins was present within the cranial thorax and caused caudodorsal and lateral displacement of the left and right cranial lung lobes. The cranial aspect of the cardiac silhouette was obscured. Thoracic ultrasound was performed; however, imaging was difficult given the skin condition and pain of the doe during physical contact. Brief imaging showed a large cranial mediastinal mass that was resting on the heart base. Given the poor prognosis and quality of life, euthanasia was elected.
The doe was submitted for postmortem examination and was in good body condition. Approximately 60% of the skin was alopecic, dry, and thickened. Exfoliating epithelial crusts were entrapped within sparse hairs. The most severely affected ulcerated regions of the body included the skin over the back, ventral abdomen, coronary bands, dewclaws (Fig. 1), and teats. No ulceration or vesicles were found in the oral cavity. Vesicular diseases were ruled out given the generalized nature of the dermatologic lesion and lack of vesicles and ulcers in the oral cavity. Located in the cranial mediastinal space, a 12 × 8 × 8 cm white, multilobular, and cystic mass filled ~40% of the thoracic cavity (Fig. 2). An 8-cm diameter cystic cavity in the mass contained abundant translucent yellow, mucoid fluid. The pericardial cavity contained ~200 mL of serous fluid. Fibrin strands extended between the pleura, pericardial sac, and the mass. All other organ systems were grossly normal.
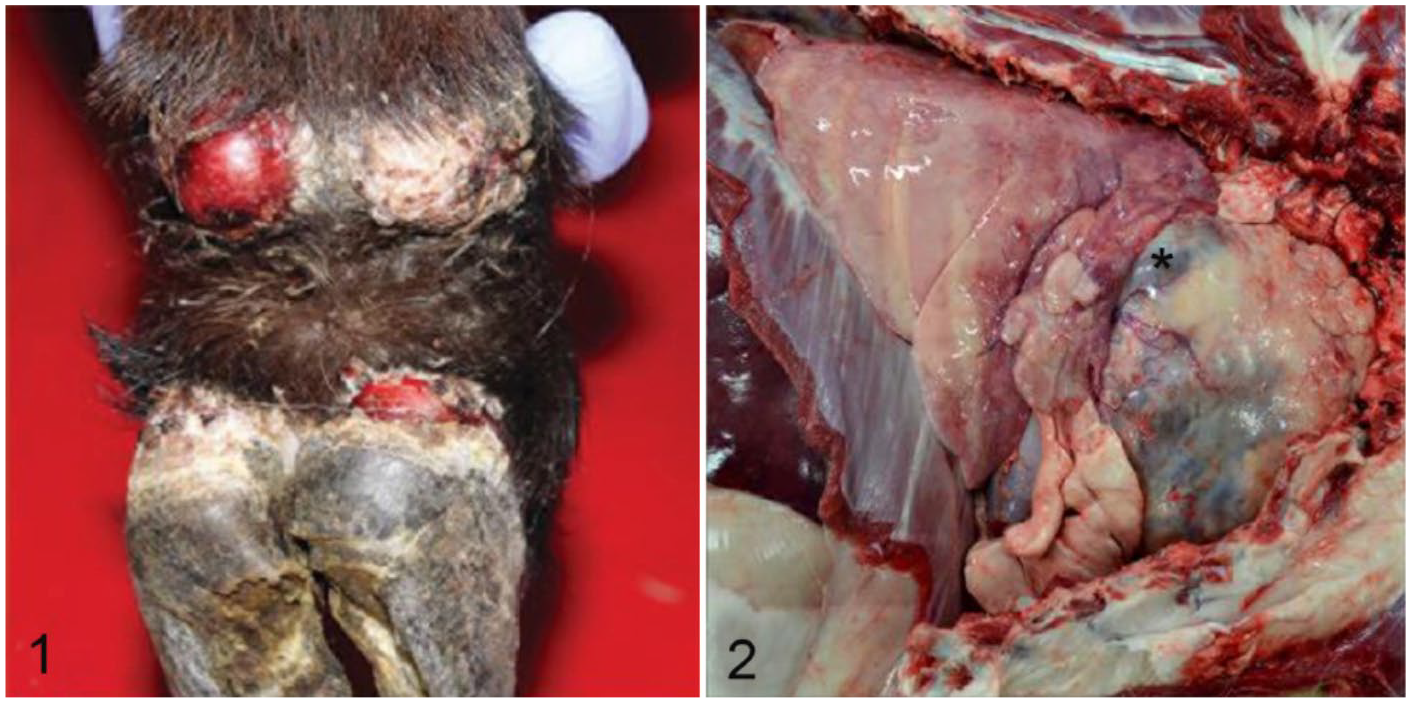
Thymoma-associated exfoliative dermatitis in a goat. Marked ulceration and sloughing of one dewclaw and coronary band with thickening of the non-ulcerated skin.
Histologically in sections of affected skin, the superficial dermis was infiltrated by a dense band of lymphocytes admixed with fewer plasma cells, macrophages, and rare neutrophils that multifocally obscured the dermal–epidermal junction (Fig. 3). A similar but milder inflammatory infiltrate was present in the deep dermis and perifollicular areas. There were regions of acanthosis with marked orthokeratotic and parakeratotic hyperkeratosis. Folliculosebaceous units were decreased in number and variably atrophied. Occasional keratinocytes of the stratum corneum and spinosum, basal cells, and rare follicular epithelial cells were apoptotic, and there was occasional satellitosis (Fig. 4). Rare sections contained greater amounts of apoptosis. Occasional basal cells were swollen, with vacuolated cytoplasm. The epidermis was multifocally ulcerated and covered with a thick crust of admixed degenerate neutrophils, hair shaft fragments, keratin flakes, and serum lakes. Rarely present within the crust were colonies of 2–3 µm diameter cocci and 2–4 µm long asymmetric, peanut-shaped budding yeasts.
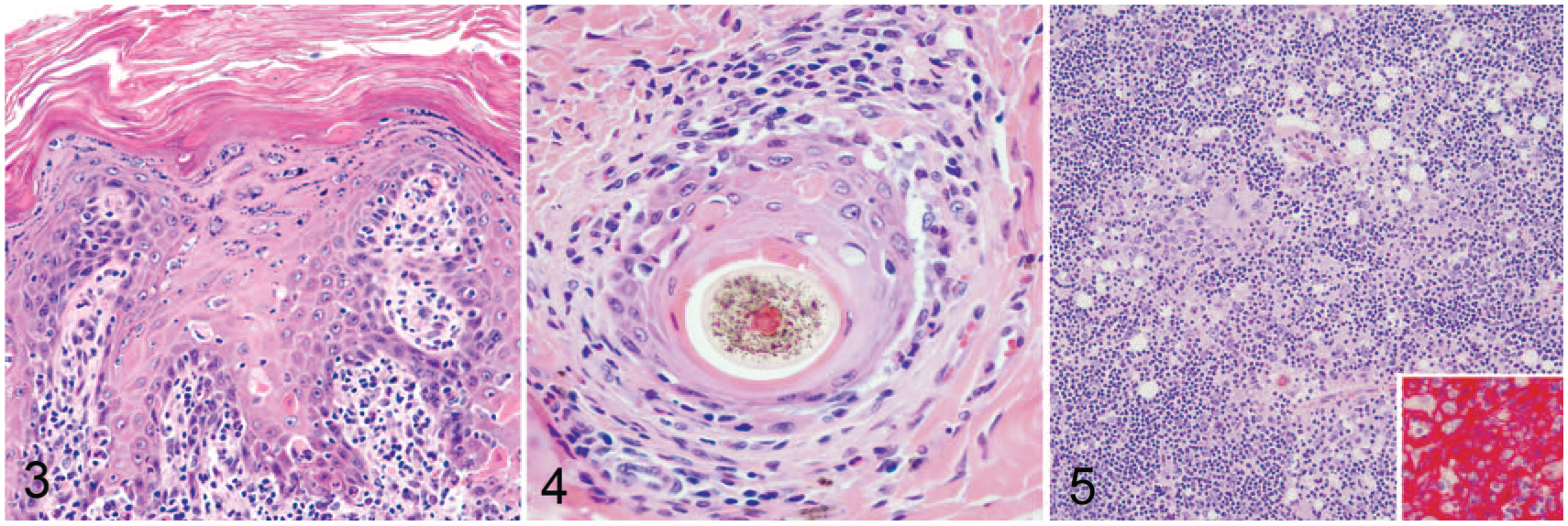
Thymoma-associated exfoliative dermatitis in a goat.
The cranial mediastinal mass was an encapsulated, highly cellular neoplasm. The neoplasm was composed of cords of polygonal epithelial cells supported by a delicate fibrovascular stroma (Fig. 5). Cells had distinct cell borders and abundant eosinophilic cytoplasm. Cell nuclei were ovoid with finely stippled chromatin and indistinct nucleoli. Anisocytosis and anisokaryosis were marked. Mitotic figures were rare. The neoplasm was admixed with small mature lymphocytes. These findings are consistent with a lymphoepithelial thymoma.
Immunohistochemistry for cytokeratin AE1/AE3 (Fig. 5, inset; PA0909; Leica Microsystems, Buffalo Grove, IL), CD3 (Leica Biosystems Newcastle, Newcastle Upon Tyne, UK), and PAX5 (610863; BD Biosciences, San Diego, CA) were performed on sections of the cranial mediastinal mass using an automated staining system (Bond-Max, Leica Biosystems Newcastle). Epithelial cells within the neoplasm had strong, diffuse cytoplasmic immunoreactivity for cytokeratin AE1/AE3. Approximately 95% of non-neoplastic lymphocytes had strong cytoplasmic and membranous CD3 immunoreactivity. Rare admixed lymphocytes had strong cytoplasmic PAX5 immunoreactivity. Additional special stains highlighted organisms in the superficial serocellular crusts overlying the skin. Gomori methenamine silver stain highlighted argyrophilic asymmetric 2–4 µm long, peanut-shaped yeasts consistent with Malassezia spp. Gram stain highlighted 2–3 µm diameter gram-positive cocci.
Thymoma-associated exfoliative dermatitis has not been reported previously in a goat, to our knowledge. Gross lesions were remarkable, with extensive alopecia, ulceration, and exfoliation on the dorsum, ventrum, teats, coronary bands, and dewclaws. In classic feline cutaneous lesions, interface dermatitis is described as either cell-rich or cell-poor depending on the degree of infiltrating inflammatory cells. 11 In our goat and a case series in cats, there was significant cell-rich inflammation, characterized by bands of inflammation at the dermal–epidermal junction. 11 In addition, there were superficial cocci and yeasts, consistent with Malassezia spp. These concomitant bacterial and yeast infections likely exacerbated the lesions.
Histologically, findings in the skin of our goat were similar to erythema multiforme and lupus erythematosus, which have transepidermal apoptosis and interface dermatitis as their respective prominent lesions. Although severe transepidermal apoptosis is associated with erythema multiforme, differentiation of (feline) thymoma-associated exfoliative dermatitis and erythema multiforme is considered to be artificial given that the lesions are similar and the pathogenesis is assumed to be the same. 4 In our case, the transepidermal apoptosis was primarily mild-to-moderate, with severe apoptosis present in the minority of sampled sections. Importantly, diagnosis of thymoma-associated exfoliative dermatitis hinges on the clinical and histologic diagnosis of a thymoma, which was present in our doe. Thymomas in goats can occur without associated clinical signs.6,10 Therefore, the recognition of thymoma-associated exfoliative dermatitis in goats is important, as it should be considered a clinical differential diagnosis in goats with dermatologic disease.
Footnotes
Acknowledgements
We thank the staff of the histopathology laboratory of the Colorado State University Veterinary Diagnostic Laboratories, especially Todd Bass, for technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
