Abstract
A neurologic disease characterized by ataxia, hypermetria, hyperesthesia, and muscle tremors of the head and neck was observed for 2 years in a flock of 28 Anglo-Nubian and Saanen goats on a farm with 5 ha of pasture. Six newborns died during the first week of life, and five abortions were recorded. The predominant plant in the pasture was Sida carpinifolia. The disease was reproduced experimentally in two goats by administration of this plant. Three goats with spontaneous disease and the two experimental animals were euthanatized and necropsied. No significant gross lesions were observed. Fragments of several organs, including the central nervous system, were processed for histopathology. Small fragments of the cerebellar cortex, liver, and pancreas of two spontaneously poisoned goats and two experimentally poisoned goats were processed for electron microscopy. Multiple cytoplasm vacuoles in hepatocytes, acinar pancreatic cells, and neurons, especially Purkinje cells, were the most striking microscopic lesions in the five animals. Ultrastructural changes included membrane-bound vacuoles in hepatocytes, Kupffer cells, acinar pancreatic cells, Purkinje cells, and the small neurons of the granular cell layer of the cerebellum. Paraffin-embedded sections of the cerebellum and pancreas were submitted for lectin histochemical analysis. The vacuoles in different cerebellar and acinar pancreatic cells reacted strongly to the following lectins: Concanavalia ensiformis, Triticum vulgaris, and succinylated Triticum vulgaris. The pattern of staining, analyzed in Purkinje cells and acinar pancreatic cells coincides with results reported for both swainsonine toxicosis and inherited mannosidosis.
Keywords
A wide variety of lysosomal storage diseases has been reported in humans and in domestic animals. Some of them are of veterinary importance; others are important in comparative pathology as models of human diseases. 5 , 14 , 21 , 28 Induced α-mannosidosis, a lysosomal storage disease resulting from the ingestion of plants of the genera Swainsona, Oxytropis, Astragalus, and Ipomoea, has been studied in herbivores in Australia, the United States, and Mozambique. 9 , 11 , 17–19, 25 , 33 Oxytropis and Astragalus are known as locoweeds, and the disease in horses, cattle, and sheep is locoism. 18 The disease is caused by the indolizidine alkaloid swainsonine, which inhibits the lysosomal enzyme α-mannosidase, inducing the storage of mannose-contained oligosaccharides in lysosomes of several cells, particularly neurons, hepatocytes, and acinar pancreatic cells. 6 , 8 , 26 Swainsonine also inhibits Golgi mannosidase II, a key enzyme in the glycosylation of many glycoproteins. 32 , 34 Conventional light microscopic and ultrastructural analyses of tissues serve only to confirm the presence of a lysosomal storage disease and do not generally yield a more specific diagnosis. 13 , 14 , 27
Lectin histochemical analysis of paraffin-embedded sections may be used to identify specific sugars and hence aid in diagnosis of glycoprotein and glycolipid storage diseases. 2 , 3 , 21 , 29 Lectins are carbohydrate binding proteins of nonimmune origin that agglutinate cells and/or precipitate glycoconjugates 15 , 16 and, when bound to visulants, allow localization of complementary carbohydrates. 4 , 7
Here, we describe an acquired lysosomal storage disease in goats in the state of Rio Grande do Sul, Brazil Epidemiologic data indicate that the ingestion of Sida carpinifolia is involved in the etiology. The purposes of this study were to characterize the lesions observed at histologic and ultrastructural levels, investigate experimentally the hypothesis that S. carpinifolia is the etiological agent, and compare the lectin staining pattern observed in tissues with lesions in both natural and experimental poisoning cases.
Materials and Methods
A neurologic disease was observed for 2 years in a flock of 28 Anglo-Nubian and Saanen goats grazing in a 5-ha pasture. Abortions (seven in a period of 45 days) and stillbirths were frequent in goats on this farm. The predominant plant in the pasture was Sida carpinifolia L.f., which is a member of the Malvaceae family and is know also by the synonyms S. acuta Burm., S. ulmifolia Mill., S. acuta var. carpinifolia (L.f.) K. Schum, and S. frutescens Cav. The plant is an erected perennial scrub 30–70 cm high. The leaves are alternate and short-peciolate, with an upper glabrous surface and a lower surface with sparse hair over the veins. The flowers are distributed singly or in small clusters and are yellow with seven or eight carpels. 22 , 24 Samples were identified by Dr. Olinda Leites Bueno, Fundaçao Zoobot∘nica do Rio Grande do Sul, Porto Alegre.
General information for the study animals is summarized in Table 1. Clinical examination was performed on three naturally and two experimentally poisoned goats. Goat No. 2 was removed from the pasture and maintained for 30 days in a stall. During this period it was fed with commercial ration and had free access to water. The other two naturally poisoned goats were euthanatized 2 weeks after removal from the contaminated pasture.
Clinical data for goats with natural or experimental Sida carpinifolia poisoning.
∗ M = male; F = female.
† + = mild; + + = moderate; + + + = severe.
One 36-kg female goat (No. 4) from a farm free of S. carpinifolia was force fed with 200 g of the plant daily for 15 days and subsequently with 400 g daily for 45 days (15 g/kg). A 6-month-old 26-kg male goat from the same farm (goat No. 5) was force fed with 400 g of S. carpinifolia daily for 90 days. Both animals received commercial ration and water ad libitum and were clinically monitored on a daily basis throughout the experiment. The ground leaves were mixed with water and administered intraruminally with a flexible tube.
Three adult female goats affected with the spontaneous disease and the two experimental animals were euthanatized at the end of the experiment and necropsied. Fragments of several organs, including central nervous system, were fixed in neutral formalin and embedded in paraffin. Tissue samples from two unaffected goats were processed as controls. Sections were stained with hematoxylin and eosin. Small fragments of cerebellar cortex, liver, and pancreas from four goats (Nos. 2–5) were fixed by immersion in cacodylate-buffered 2% glutaraldehyde–2% paraformaldeyde, postfixed in osmium tetroxide, and embedded in Epon. Thick sections were stained with methylene blue, and selected thin sections were stained with uranyl acetate and lead citrate and examined in a transmission electron microscope.
Representative sections of the cerebellum and pancreas were submitted for lectin histochemical examination. Samples were taken from both natural and experimental poisoning cases. Tissues from two unaffected goats were included as controls. Nine lectins of different specificities (E-Y Laboratories, San Mateo, CA) were employed (Table 1). After deparaffination, the sections were incubated in 0.3% hydrogen peroxide in methanol for 30 minutes at room temperature, rinsed several times in 0.01 M phosphate-buffered saline (PBS), pH 7.2, and treated with 0.1 bovine serum albumin in PBS for 15 minutes. They were then incubated with biotinylated lectins for 1 hour, followed by incubation with avidin–biotin–peroxidase complex (Vector Laboratories, Burlingame, CA) for 45 minutes. The horseradish peroxidase was activated by incubation for 4–10 minutes with buffered 0.05 M Tris-HCl, pH 7.6, containing 0.02% diaminobenzidine and 0.05% H2O2. All sections were counterstained with Mayer's hematoxylin.
Each lectin (Table 2) was used at a dilution of 30 µg/ml in PBS, except for Arachis hypogaea (PNA), which was applied at a concentration of 10 µg/ml. As controls for lectin histochemical procedure, the lectins were omitted or were blocked by incubating them with their blocking sugars (0.1 to 0.2 in PBS) for 1 hour at room temperature before application to the sections. 23
Type of lectins used in the histochemical studies of lysosomal storage disease associated with Sida carpinifolia poisoning in goats.

∗ Fuc = Fucose; Gal = Galactose; GalNac = N-acetyl-galactosamine; Glc = Glucose; GlcNac = N-acetyl-glucosamine; Man = Mannose; NeuNac = N-acetyl-neuraminic acid. 15
Results
Clinical findings
The severity of the clinical signs is summarized in Table 1. The neurologic disorder in spontaneous cases was characterized by ataxia, hypermetria, hyperesthesia, and muscle tremors in the head and neck. When the neck was forced backwards, the affected goats exhibited epileptiform seizures, sudden falls, nystagmus, opisthotonus, and tetany. This procedure is known as the HR test. 30
Goat No. 3 was recumbent for approximately 1 week. The disease was more frequent in primiparous goats and in animals >6 months of age. Affected goats kept their appetites. Six newborns died during the first week of life. Two abortions were observed in goats with clinical signs and three occurred in goats without clinical signs. No clinical signs were observed in animals <6 months of age. Of the two experimentally poisoned goats, goat No. 4 exhibited mild muscle tremors at the end of the experiment and fell to the ground when the neck was forced backwards and freed suddenly, but it recovered quickly in a few minutes. Goat No. 5 showed no clinical signs during the 90 days of the experiment.
Necropsy findings
There were no significant gross lesions except for emaciation and cutaneous ulcers due to recumbency that were observed in one spontaneously poisoned goat.
Microscopic and ultrastructural findings
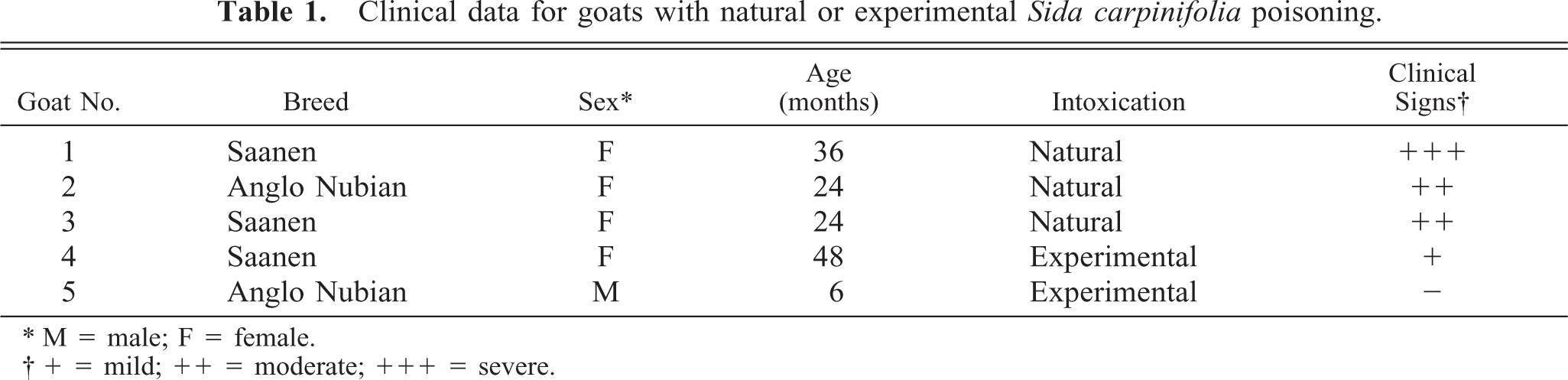
The histopathology and the ultrastructure of the central nervous system, liver, and pancreas was similar in both naturally and experimentally poisoned goats. There were various degrees of vacuolization in the cytoplasm of acinar pancreatic cells, hepatocytes, and neurons of the cerebellar cortex, thalamus, hypothalamus, pons, and spinal cord. The vacuolization was more marked in the cytoplasm of Purkinje cells, hepatocytes, and acinar pancreatic cells (Fig. 1).
Cerebellum; goat No. 1. Resin-embedded section showing “empty” vacuoles in a Purkinje cell. Methylene blue stain. Bar = 25 µm.
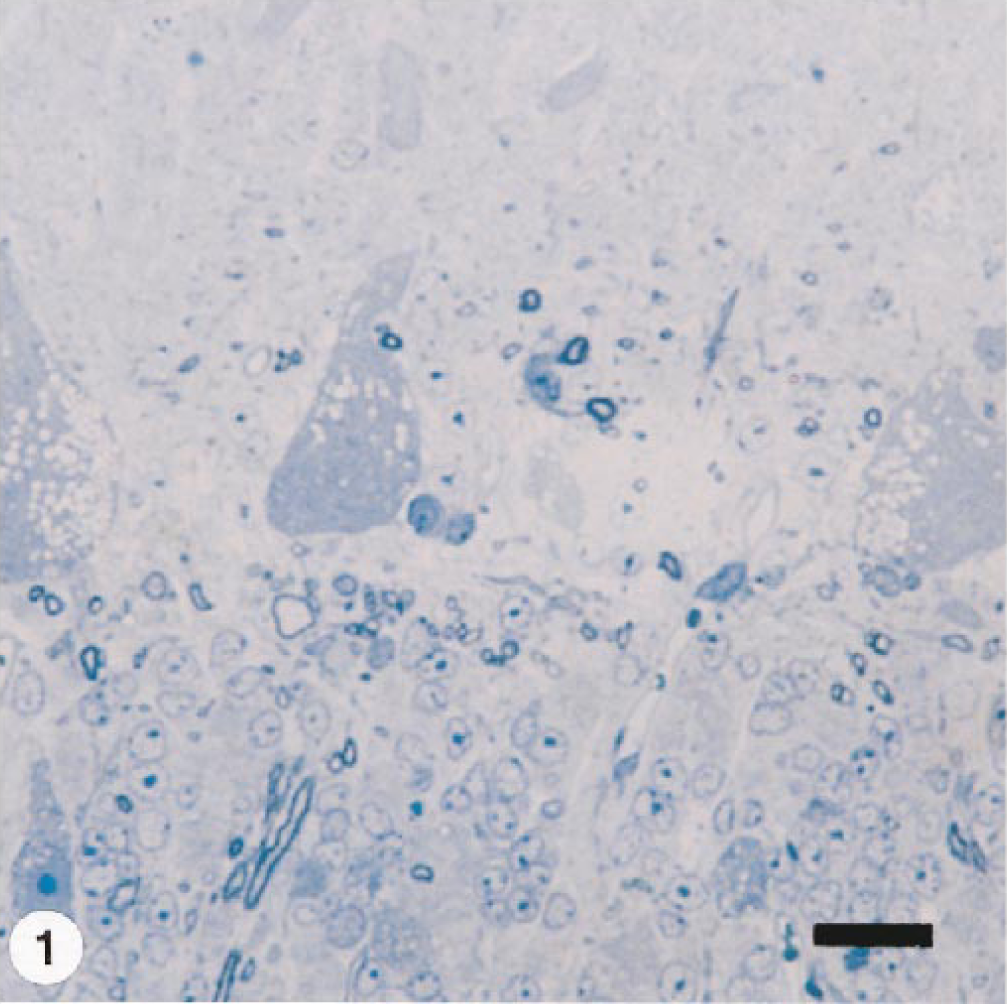
Ultrastructurally, there were membrane-bound vacuoles, some of which had finely granulated material and myelinoid figures in cytoplasm of hepatocytes, Kupffer cells, acinar pancreatic cells, neurons (mainly Purkinje cells), and small neurons of the granular layer of the cerebelar cortex (Fig. 2). Axonal spheroids, filled with dense bodies and tubulovesicular bodies, were also seen in this layer.
Electron micrograph. Cerebellum; goat No. 1. Purkinje cell pericaryon with numerous membrane-bound vacuoles. Bar = 1 µm.
Lectin histochemical findings
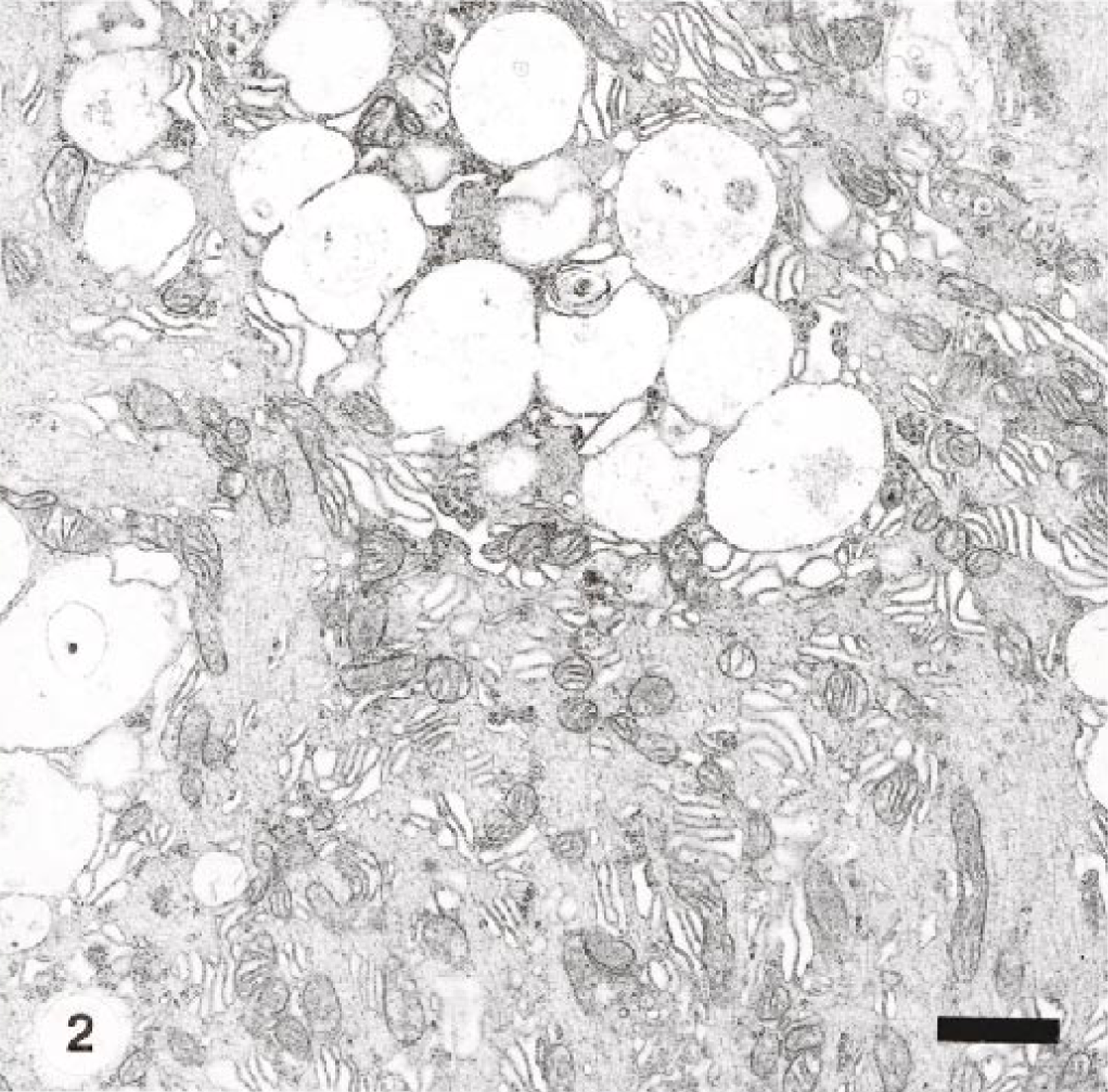
Results of the lectin binding patterns for affected and control goats are summarized in Table 3. Lectin reactivity was identical in both natural and experimentally intoxicated animals. The cytoplasm of Purkinje cells and cytoplasm of multiple cells of cerebellar granular and molecular layers of affected goats stained strongly with Concanaralia ensiformis (Con A), Triticum vulgaris (WGA), and succinyl-WGA (sWGA) (Fig. 3). These cells did not stain in corresponding slides from control animals. Lectin binding permitted the demonstration of stored material in numerous cells that appeared normal with the hematoxylin and eosin stain. Additionally, weak to clear reaction to Ulex europaers I (UEA-I), Glycine max (SBA), Dolickos bifloris (DBA), and Ricinus communis I (RCA-I) was observed in a few cells. The incubation with Bandeiria simplicifolia (BS-I) gave a negative reaction (Fig. 4). In the pancreas, three lectins (WGA, sWGA, and Con A) reacted strongly but variably with vacuoles observed in acinar cells. Other lectins, UEA-I, PNA, SBA, and DBA, showed an heterogeneous binding pattern (Table 3).
Intensity of lectin binding at sites on affected cells in cerebellum and pancreas of poisoned and normal goats. ∗

∗ Numbers indicate staining intensity on a subjective estimated scale from 0 = unreactive to 3 = most reactive. Control results from normal goats are provided in the parentheses.
Cerebellum; goat. Brown staining indicates lectin binding to (β-(1-4)-
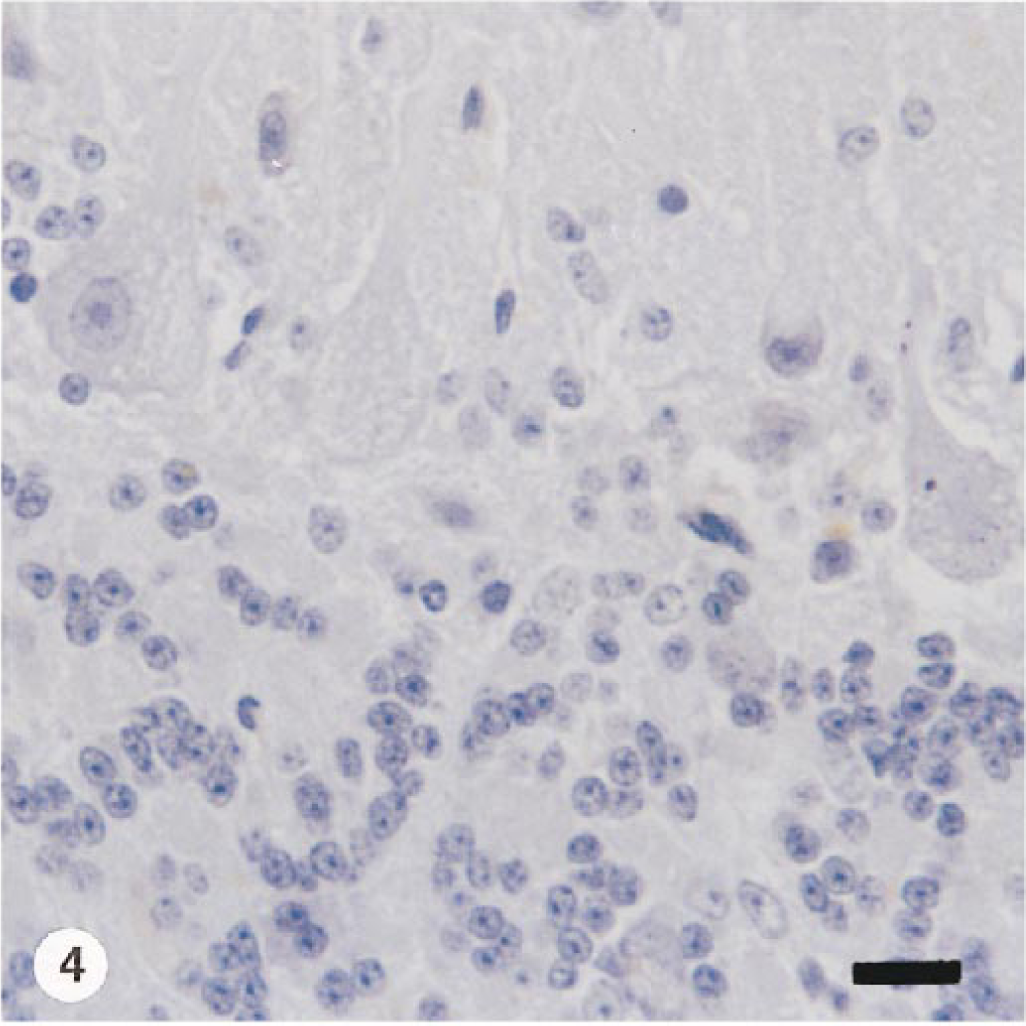
Cerebellum; goat. In this affected goat, no cytoplasmic staining is evident. Section of formalin-fixed and paraffin-embedded incubated with Bandeirea simplicifolia (BS-I) and counterstained with Mayer's hematoxylin. Bar = 25 µm.
Control sections for the lectin histochemical procedure were negative.
Discussion
This spontaneous storage disease of goats is similar to the acquired α-mannosidosis secondary to the ingestion by herbivores of plants of the genera Swainsona, Oxytropis, Astragalus, and Ipomoea. This acquired storage disease mimics the inherited α-mannosidosis of domestic animals described principally in Angus cattle in New Zealand, Australia, Scotland, North America, and Argentina and due to a deficiency of lysosomal α-mannosidase, an enzyme that normally catabolizes the various oligosaccharide moieties of glycoproteins. 21 The animals intoxicated after ingestion of Sida carpinifolia showed clinical signs similar to those observed in cases of swainsonine, locoweed, and Ipomoea toxicosis, manifested as chronic neurologic disease, abortion, and stillbirths. 9–11, 19 , 26 , 32
The mechanism involved in α-mannosidosis is a deficiency of lysosomal α-mannosidase, an enzyme that catabolizes the various moieties of glycoproteins. As a consequence, a variety of mannose-containing oligosaccharides derived from high-mannose, complex, and hybrid oligosaccharides accumulate in lysosomes. 8 , 21 , 31 Plant-induced α-mannosidosis seems to be very similar to the genetic form, although the disease is not identical. In the induced disease, the indolizidine alkaloid-1,2,8-triol (i.e., swainsonine) inhibits lysosomal α-mannosidase but also Golgi mannosidase II, an enzyme associated with the processing of oligosaccharides during the glycosilation of proteins. The consequence is again the accumulation of mannose-containing oligosaccharides in the lysosomes. 10 , 12 , 26
The neuronal and axonal lesions are responsible for the severity of the neurologic disorders observed in all spontaneous cases. The animals probably grazed the S. carpinifolia because it was the predominant plant in the pasture. The experimental reproduction of the condition incriminated S. carpinifolia as the causative agent. Goat No. 4 received plant material at a dosage of 5.5 g/kg of body weight during 15 days and additionally 13.8 g/kg of body weight for the subsequent 45 days and showed mild clinical signs and vacuoles in the neurons. No clinical signs were observed in goat No. 5, but the vacuolization of the neurons was evident; this animal received 15 g/kg of body weight for 90 days. This discrepancy in the appearance of clinical signs probably reflects different susceptibility to the poison.
The electron micrographs showed that the vacuoles are membrane bound and some contain reticuloparticulate material and myelinoid figures. The presence of spheroids filled with tubulovesicular and dense bodies could be due to a partial impairment of some overloaded lysosomes to degrade lipids, which were then conducted by axonal flux to a distal position. 36 The observed ultrastructural changes agree with α-mannosidosis, 21 , 27 but lysosomes of the same or very similar morphology have been found in animals with other lysosomal storage diseases. 13 Like most lysosomal storage diseases, 33 neurons were the most consistently affected cells.
The pattern of lectin staining observed in Purkinje cells partially agrees with the results reported for locoweed and swainsonine toxicosis and for mannosidosis in humans, cats, and calves.
1
In feline mannosidosis, WGA and Con A recognized the undegraded glycoproteins and oligosaccharides stored in lysosomes of affected cells, as was the case in S. carpinifolia intoxication.
5
The reaction was clear to sWGA and WGA, which indicates the expression of β-
The exocrine acinar cells in pancreas of affected goats were positive for sWGA, WGA, and Con A. The adult exocrine acinar cells of mouse, rats, guinea pigs, and rabbits expressed at a more or less high rate binding of DBA, PNA, Con A, UEA-I, RCA-I, and WGA. 24 , 28 The lectin binding properties revealed extensive and specific heterogeneity of acinar cell population; a population which appears homogeneous by classical light and electron microscopic preparations. 20 The partial staining obtained in the present study with UEA-I, PNA, RCA-I, SBA, and DBA is part of the lectin binding pattern of normal acinar cells in goats, as was corroborated by the lectin reactivity of control tissues.
The discovery of a novel induced α-mannosidosis opens a wide array of possibilities for further studies. The disease, unrecognized until now, should be better characterized epidemiologically, e.g., the distribution of the plant, its significance for other domestic animals, the relation of toxicity and vegetative period, and the effects of other Sida species. This caprine acquired α-mannosidosis could be a useful model in comparative pathology and biomedical research. It remains to be determined whether the mechanisms involved in this disease are similar to those demonstrated in Swainsona, Oxytropis, and Astragalus toxicosis.
Footnotes
Acknowledgements
We thank Mrs. Carolina Aralda and Mrs. Laura Paoli for their technical assistance. The language correction of Silvina Viviana Macnie is also acknowledged. Financial support was provided in part by a grant from the Conselho Nacional para o Desenvolvimento Científico e Tecnológico (CNPq-PRONEX), Brazil, and the Consejo Nacional de Investigaciones Científica y Tícnicas (CONICET), Argentina. DD is a Research Fellow of CNPq, EJG is a Research Career Member of CONICET, and SSB is a Research Fellow of the Fundaçâo de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), Brazil. Part of this information was presented as a poster at the 16th Congress of the European Society of Veterinary Pathology, Lillehammer, Norway, 23–26 September 1998.
