Abstract
Spontaneous migration of placental trophoblasts into maternal blood vessels and embolization to other organs (ie, lung, adrenal gland, spleen, and liver) occurs in women and certain animals with hemochorial placentation. Although considered incidental in most species, increased incidence and numbers of trophoblast emboli are reported in women with gestational diseases with arterial hypertension (pre-eclampsia and eclampsia). To the best of our knowledge, trophoblast emboli have not been reported in lagomorphs. This case report describes the identification of trophoblast emboli in the lung of a wild snowshoe hare (Lepus americanus). Death of this hare was attributed to pulmonary hemorrhages and hemothorax, but a definitive cause for the hemorrhages was not determined. It is unclear whether trophoblast embolism normally occurs in this species and represents an incidental finding, or whether it possibly contributed to rupture of pulmonary or thoracic blood vessels leading to hemorrhage.
Trophoblast embolism (also known as trophoblast dissemination or deportation) occurs when trophoblasts migrate from the placenta into maternal circulation and disseminate to other organs (ie, lung, adrenal gland, spleen, and liver).1,3,5,10,13,16 Trophoblast embolism was first described by Schmorl in 1893, who identified numerous emboli of fragmented multinucleated cells resembling trophoblasts in the lungs of pregnant women who died of eclampsia.12,19 Since then, spontaneous trophoblast embolism has also been documented in normal pregnancies and in certain animals with hemochorial placentation, specifically hamsters, chinchillas, gerbils, and cotton rats.1–3,5,13 Incidental presence of giant, epithelioid, trophoblast-like cells was also reported in the adrenal glands, liver, spleen, or lung of 4 female porcupines. 10 In women, pulmonary trophoblast emboli may be detected as early as 6 weeks of gestation and increase in frequency and numbers with gestational age. 1 Although the exact role of trophoblast emboli in hypertensive disorders of pregnancy in women (eg, eclampsia and pre-eclampsia) is still debated, aberrant quality and quantity of deported trophoblasts seems to play a role in these diseases. Particularly, the shedding of necrotic instead of apoptotic trophoblasts into maternal circulation with consequent endothelial cell activation and release of pro-inflammatory cytokines is suspected to play a role in their pathogenesis.1,7,16 To the best of our knowledge, despite having hemodichorial placentation, trophoblast emboli have not been documented in lagomorphs.5,8,14,17 In one study, Maximov (1898) examined the lungs of 40 rabbits during normal pregnancy and found no evidence of trophoblast emboli.5,14,17
A wild, pregnant, adult, female snowshoe hare (Lepus americanus) was found dead by one of the authors (A.J.V.W.) in the Wasatch Mountain range of northern Utah and was submitted to the Utah Veterinary Diagnostic Laboratory, Utah State University. The hare body was still warm when found and was located in a mountain valley 2 miles away from the nearest road. The snow around the body was undisturbed and there was no evidence of animal tracks other than the hare’s tracks. On postmortem examination, the hare was in a good body condition and weighed 1.5 kg. A small amount of blood was present around the nostrils and an estimated 5 to 10 ml of partially clotted blood was within the thoracic cavity (hemothorax) (Figs. 1, 2). There was no evidence of blunt force trauma or ruptured large blood vessels in the thoracic cavity. Rare cestode larval cysts morphologically consistent with cysticerci were present on the intestinal peritoneum. The uterus was gravid with 4 embryos with a 10-mm crown-to-rump length and recognizable limb buds but no discernible digits.
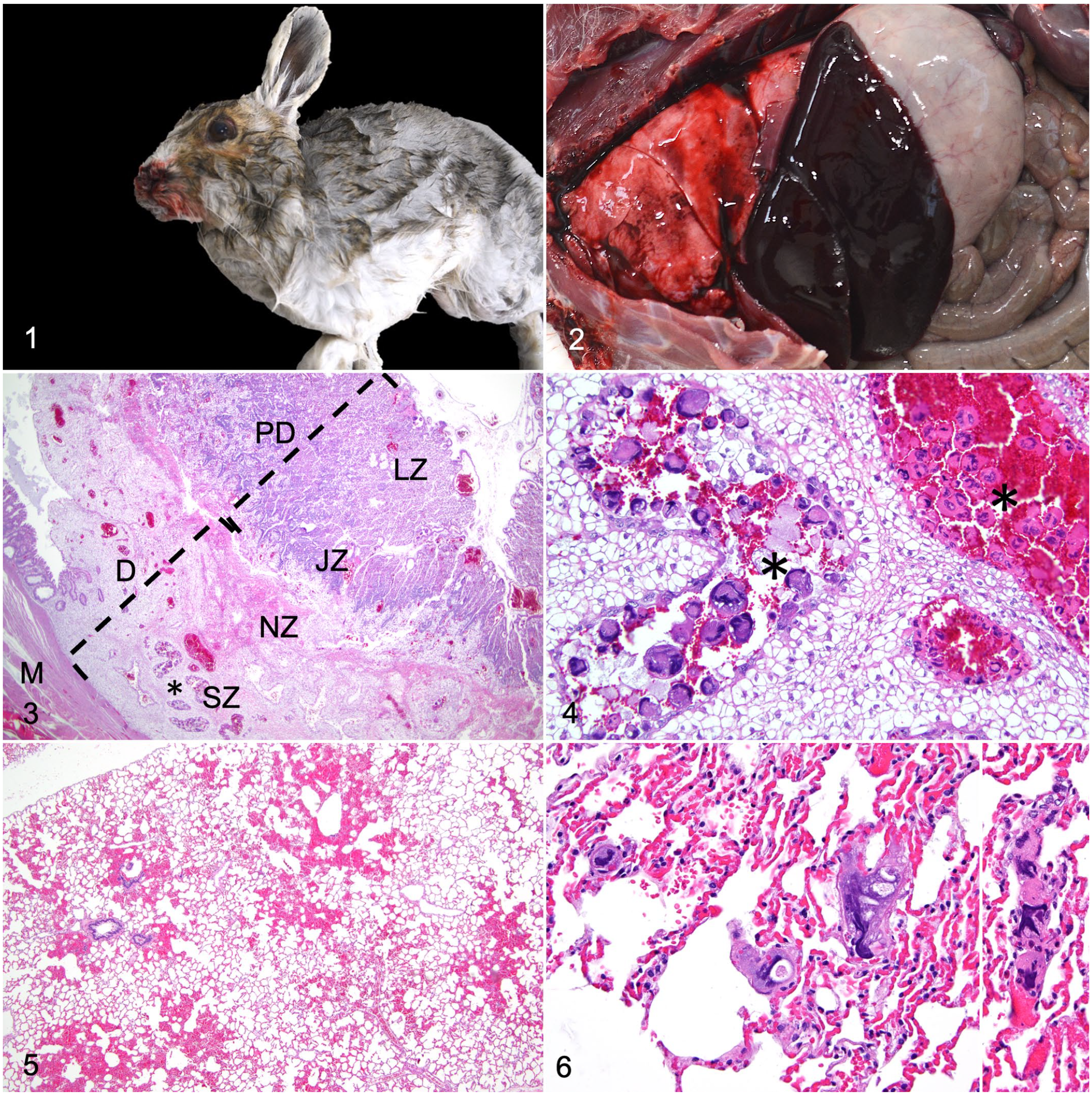
Snowshoe hare.
Samples from the dam’s esophagus, stomach, small and large intestine, trachea, lung, liver, gallbladder, spleen, pancreas, kidney, adrenal gland, heart, uterus, ovary, and brain and whole fetuses were fixed in 10% neutral buffered formalin for 24 hours. Tissue sections were processed routinely and embedded in paraffin wax. Sections (3–5 µm) were stained with hematoxylin and eosin and evaluated by light microscopy. Microscopically, the fetuses had nucleated red blood cells, closed lens vesicle, a prominent liver with hepatoblasts not forming hepatic cords, and intestinal loops. No histologic lesions were detected in the fetuses. Fig. 3 shows the microscopic anatomy of the gravid uterus with hemodichorial placentation. In the dam, multiple hemorrhages filling alveolar spaces and expanding bordering alveolar septa affected from 10% to 40% of the lung tissue in the examined sections (Fig. 5). A small number of scattered alveolar capillaries were filled with large, up to 60 μm in diameter, multinucleated cells with streaming nuclei which morphologically resembled trophoblasts in the placenta (Figs. 4, 6). In addition, a cystic larval cestode with an invaginated scolex and a 5-mm-diameter bladder surrounded by a fibrous capsule (cysticercus) and one 100-µm-diameter adiaspore morphologically consistent with Emmonsia sp. were observed in a histologic section of lung. The cysticercus was focally surrounded by minimal lymphocytic inflammation. The adiaspore was surrounded by 20 cell layers of lymphocytes, plasma cells, and fewer macrophages. Adiaspores, cysticerci, or other organisms were not observed in another 10 lung sections examined. No significant histologic abnormalities were detected in the other evaluated organs. Endothelial cell hypertrophy, infarction, thrombosis, erythrophagocytosis, or fibrin accumulation were not detected in the lung or other organs.
Based on the presence of hemorrhages in the thoracic cavity and lung, a liquid chromatography-mass spectrometry (LC-MS) anticoagulant rodenticide assay (Texas Veterinary Medical Diagnostic Laboratory, College Station, TX) and rabbit hemorrhagic disease virus 2 (RHDV2) qPCR assay (Utah Veterinary Diagnostic Laboratory, Logan, UT) were performed on fresh liver samples. The anticoagulant rodenticides LC-MS assay targeted coumarins (brodifacoum, bromadiolone, coumatetralyl, difethialone, and warfarin) and indanediones (chlorophacinone and diphacinone) and was performed as previously described.15,20 Limits of detection of the anticoagulants included in the screen were 5 ppb for brodifacoum, bromadiolone, chlorophacinone, diphacinone, and warfarin; 10 ppb for coumatetralyl and difethialone. Real-time reverse-transcription polymerase chain reaction (RT-qPCR) for RHDV 2 in the liver was performed following the procedure provided by the National Veterinary Services Laboratories—Foreign Animal Disease Diagnostic Laboratory (NVSL—FADDL). Nucleic acid extraction was performed using Applied Biosystems MagMAX Pathogen RNA/DNA Isolation Kit (Thermofisher Cat. No. 4462359) following manufacturer recommendation. Two microliters of VetMAX Xeno Internal Positive Control RNA (Thermofisher Cat. No. A29763 or A29761) was added to the sample and negative extraction control. The 25 µl of PCR master mix consisted of 6.25 µl Taqman Fast Virus 1-Step Master Mix Kit (Thermofisher Cat. No. 4444432), 1 μM of forward primer (5′-TGG AAC TTG GCT TGA GTG TTG A-3′) and reverse primer (5′-ACA AGC GTG CTT GTG GAC GG-3′), 0.2 μM of Taqman probe (5′-6-FAM-TGT CAG AAC TTG TTG ACA TCC GCC C-Iowa Black FQ-3′), 5 µl of extracted nucleic acids, 1 µl of VetMAX Xeno Internal Positive Control—Liz Assay (Thermofisher Cat. No. A29766 or A29768), and 7.25 µl of nuclease-free water. The PCR was performed in an ABI 7500 fast Real-time PCR System (Applied Biosystems) for 1 cycle of 50°C for 5 minutes, 1 cycle of 95°C for 20 seconds, and 45 cycles of 95°C for 3 seconds, and 60°C for 30 seconds. Controls included an internal extraction control (VetMAX Xeno), a negative extraction control (VetMAX Xeno and DMEM), positive amplification control (purified DNA from a confirmed RHDV2 positive liver), and negative amplification control (no template control).
Anticoagulant rodenticides and RHDV2 were not detected and no hepatic lesions consistent with RHDV2 infection were observed histologically.
To confirm the identity of the intravascular multinucleated cells in the lung, immunohistochemistry with CD31, CKAE1/AE3, CK8/18, and CK7 primary mouse monoclonal antibodies was performed on sections of lung and placenta. CD31 and CKAE1/AE3 immunohistochemistry was performed at the Utah Veterinary Diagnostic Laboratory; CK8/18 and CK7 were performed at the University of Minnesota Veterinary Diagnostic Laboratory. Immunohistochemical methods are summarized in Table 1. Trophoblasts in the labyrinth zone, junctional zone, as well as within maternal decidual blood vessels were evaluated. Positive and negative controls from canine tissues performed as expected.
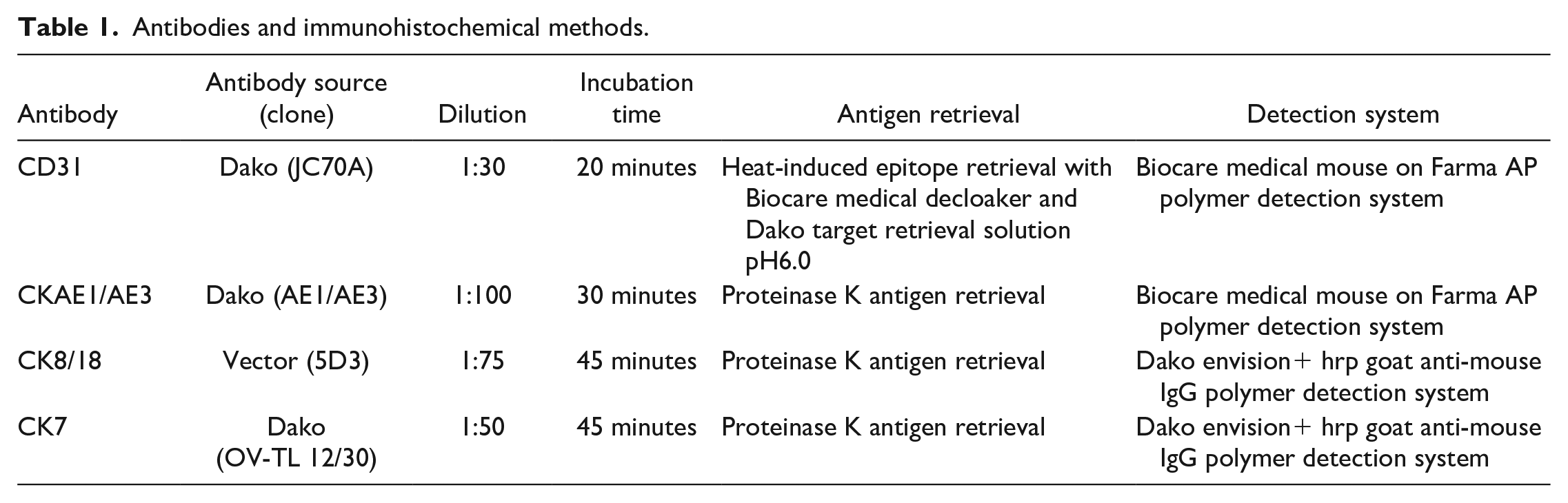
Antibodies and immunohistochemical methods.
Most placental trophoblasts and pulmonary intravascular multinucleated cells showed cytoplasmic immunoreactivity for CD31 (Figs. 7a, 8a). A few placental trophoblasts and pulmonary intravascular multinucleated cells showed cytoplasmic immunoreactivity for CKAE1/AE3 and CK8/18 (Figs. 7b, 7c and 8b, 8c). A few trophoblasts in the placental labyrinth, but no cells within maternal decidual vessels or pulmonary vessels, showed cytoplasmic immunoreactivity for CK7 (Figs. 7d, 8d).
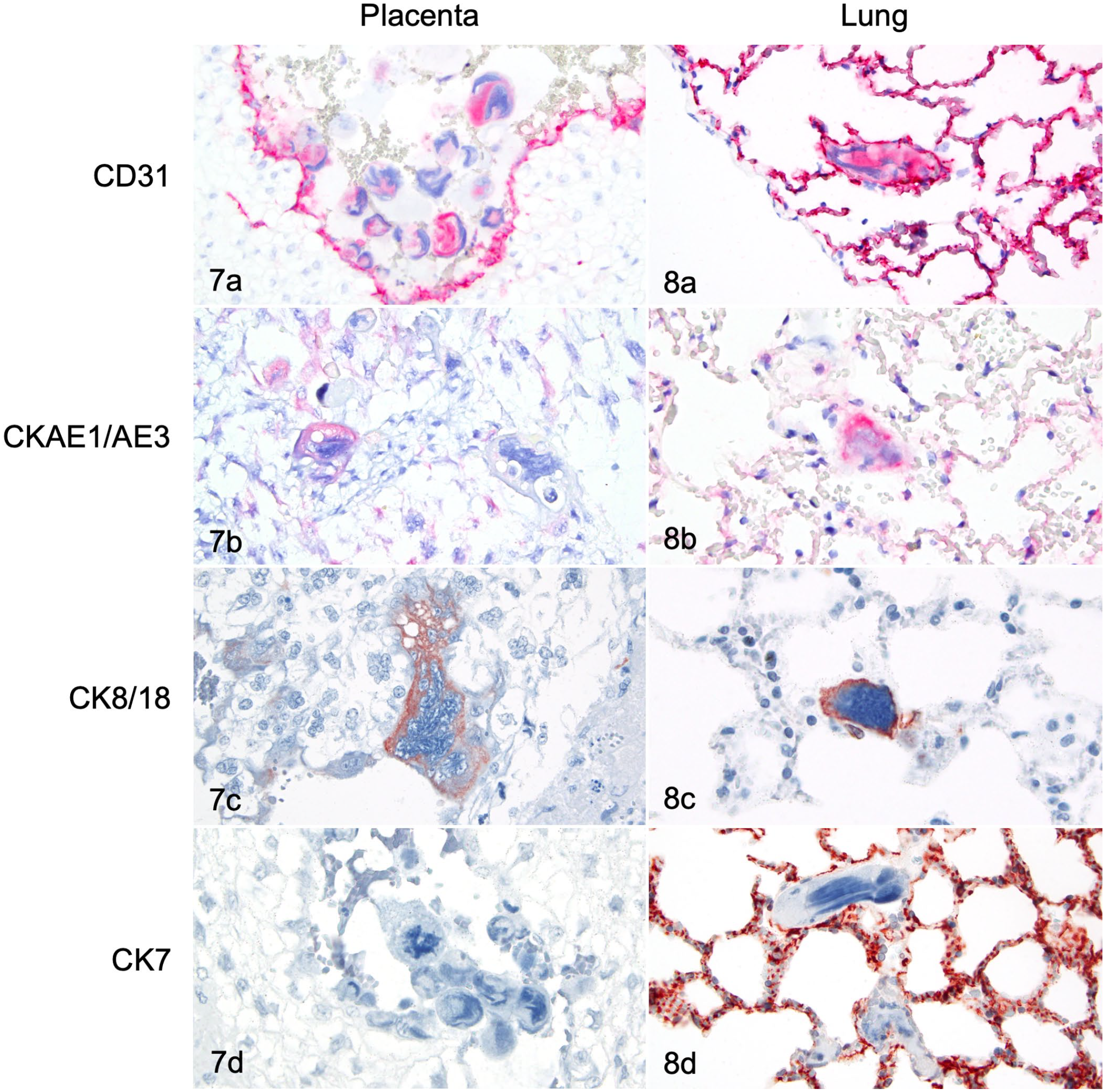
Placenta (Fig. 7) and lung (Fig. 8), snowshoe hare. Most placental trophoblasts and pulmonary intravascular cells resembling trophoblasts have cytoplasmic immunoreactivity for CD31. Placental and pulmonary endothelial cells are also immunoreactive for CD31 (Figs. 7a, 8a). A few placental trophoblasts and pulmonary intravascular cells resembling trophoblasts are immunoreactive for CKAE1/AE3 (Figs. 7b, 8b) and CK8/18 (Figs. 7c, 8c). No trophoblastic cells within maternal decidual vessels or pulmonary intravascular cells resembling trophoblasts show immunoreactivity for CK7 (Figs. 7d, 8d). The cytoplasm of the alveolar epithelium is immunoreactive for CK7 (Fig. 8d). Immunohistochemistry. Chromogens: Red AP (Figs. a, b); DAB (Figs. c, d).
In conclusion, this case describes spontaneous trophoblastic embolism in a wild snowshoe hare, a member of the order Lagomorpha. Trophoblastic origin of the intravascular multinucleated cells in the lung was supported by the similar morphology and immunoreactivity for CD31 and cytokeratins to the trophoblasts in the placenta. Megakaryocytes may resemble intravascular trophoblasts but are expected to have membranous immunoreactivity to CD31 and no immunoreactivity to cytokeratins.11,18
In women, trophoblast emboli may be detected as early as 6 weeks of gestation and increase in frequency and number with gestational age. 1 In a case study on cotton rats, pulmonary trophoblastic emboli were detected between 8 and 18 days of the 27-day gestation period and were most numerous between 10 and 14 days. 13 The gestation in snowshoe hares is 34 to 37 days and the embryos in this case were estimated to be approximately 12 to 14 days old according to their crown to rump length (10–12 mm long), external appearance, and stage of development of internal organs on microscopic examination.4,6,9
A specific cause for the pulmonary hemorrhage and hemothorax was not determined in this snowshoe hare. There was no gross evidence of blunt force trauma, predation, or ruptured blood vessels, or gross or microscopic lesions indicative of a significant infectious disease. Anticoagulant rodenticides and RHDV2 were not detected. Increased frequency and quantity of trophoblast emboli are reported in women with hypertensive disorders of pregnancy (eg, pre-eclampsia and eclampsia). 1 In the case study on cotton rats, high numbers of pulmonary trophoblastic emboli were often associated with vascular changes such as pulmonary edema, hemorrhage, endothelial hypertrophy, fibrinoid vascular necrosis, and presence of hemosiderophages and erythrophagocytosis. 13 In this hare, similar changes were not detected in the lung or other organs and the pulmonary hemorrhages appeared to be acute or peracute. These findings suggest that the trophoblastic dissemination may have been an acute or an incidental event in this animal. Based on this sole report, it is unclear whether trophoblast dissemination normally occurs in snowshoe hares, or whether it represents an abnormal event contributing to acute pulmonary or intrathoracic hemorrhage.
Footnotes
Acknowledgements
We would like to thank Sheri Iodice and Amanda Wilheim from the Utah Veterinary Diagnostic Laboratory, and the staff of the University of Minnesota Veterinary Diagnostic Laboratory and Texas Veterinary Medical Diagnostic Laboratory for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
