Abstract
Obesity is a major health condition owing to its effects on chronic diseases and cancers in humans, but little information is available regarding the role of obesity in canine mammary cancer (CMC). In the present study, we performed immunohistochemistry to investigate the effect of obesity on CMC by analyzing the number of tumor-associated macrophages, intratumoral microvessel density (iMVD), and the expression of prognostic factors including epidermal growth factor receptor (EGFR), cyclooxygenase 2 (COX-2), and Ki67 in CMC specimens. These data were compared in CMC specimens from lean or ideal body weight (Group 1) versus overweight or obese (Group 2) female dogs (n = 60 for each group). Associations between obesity status and histologic characteristics, such as histologic subtype, grading, and lymphatic invasion, were also investigated. Compared with lean or ideal body weight dogs, TAM (tumor-associated macrophage) counts (P < .005) and iMVD (P < .001) were significantly higher in overweight or obese dogs. CMC specimens of dogs in the overweight or obese group also showed higher histologic grade (P < .001). In addition, although no association was found between obesity status and either COX-2 or EGFR expression, Ki67 expression was greater in CMC specimens of overweight or obese dogs (P < .005). The results of this study suggest that obesity may influence CMC development and progression, being associated with higher histologic grade, greater infiltration of TAMs, and increased tumor angiogenesis.
Keywords
Canine obesity is recognized as a major health condition, owing to its effects on chronic diseases, quality of life, and the lifespan of dogs. 14,44 The prevalence of obesity among dogs in various countries has been studied 21,22,30 ; one report suggested that companion dogs with obese owners were more likely to be overweight or obese as well. 26 Despite the importance of obesity in dogs, the effect of overweight or obese status on canine mammary cancer (CMC) is still controversial. One study suggested that obese status affects the development of CMCs, 39 whereas another reported no relationship between obesity and CMCs. 29
In contrast to reports in dogs, several human epidemiological studies have substantiated that obesity is a definite risk factor for breast cancer; this is particularly true in postmenopausal women where the risk is greater in obese than in nonobese women. 4,6,34,40 In studies of the relationship between body mass index, waist to hip ratio, or weight gain and breast cancer, obese women were diagnosed with higher grade tumors with more aggressive biological behavior and a higher frequency of metastasis. 3 In addition, overweight or obese status increased the risk of recurrence and mortality due to breast cancer, regardless of the menopause status. 8
The most acknowledged molecular link between excessively accumulated adipocytes and human breast cancer cells involves cellular interactions of hormones, such as leptin, insulin-like growth factor, and adiponectin. 19 In our previous study, we found no association between the expression of obesity-related hormones in CMCs and the overweight or obese status of the dogs, except for adiponectin. 18 However, others reported no relationship between adiponectin and the body condition score (BCS). 39 Therefore, further studies exploring other possible mechanisms of obesity-induced CMCs are required.
Another suggested role of obesity in mammary gland tumorigenesis is related to adipocyte-induced inflammation. Adipocytes mediate inflammation by releasing saturated fatty acids, promoting a chronic pro-inflammatory state. 23 Excessive adipocytes enhance the levels of immune mediators that contribute to breast carcinogenesis through dysregulation of the tissue microenvironment. 37 Recent studies have focused on the role of obesity-induced inflammation and tumor angiogenesis, including the role of macrophages in tumor growth. One recent study suggested that macrophages increase angiogenesis in mammary gland tumors in obese mice. 11 Moreover, another study revealed that adipocyte-induced inflammation enhances human breast cancer angiogenesis by secreting the peptide, angiopoietin-like 4. 15
CMC in obese female dogs has been poorly studied, and no study has investigated the relationship between CMC angiogenesis and obesity. The aim of this study was to investigate the effect of obesity on CMC by comparing the BCS to the expression of well-known prognostic factors including epidermal growth factor receptor (EGFR), cyclooxygenase 2 (COX-2), and Ki67. In addition, we sought to analyze intratumoral microvessel density (iMVD) and the number of tumor-associated macrophages (TAMs) in CMCs in relation to the BCS of dogs.
Materials and Methods
Study Population and Group Assignment
A total of 800 primary CMC specimens from the histopathologic database of the Department of Veterinary Pathology, Konkuk University Animal Teaching Hospital (Seoul, Korea), collected between 2017 and 2019, were analyzed. Specimens were selected that had all the basic clinical information recorded, including the breed, neuter status, age at diagnosis, body weight (BW), and BCS using a 5-point scale. The selected specimens were sorted and classified into 2 groups on the basis of their BCS: Group 1, BCS = 2 (lean) or 3 (ideal BW); Group 2, BCS = 4 (overweight) or 5 (obese). 38 Samples of the specimens were fixed in 10% neutral buffered formalin and embedded in paraffin wax for hematoxylin and eosin staining and immunohistochemistry (IHC).
Histopathology and Sample Selection
Histologic classification of CMC specimens was performed following the system established by Goldschmidt et al. 7 Subsequently, the grading system was applied as follows: grade I, well differentiated; grade II, moderately differentiated; and grade III, poorly differentiated. 28 Microscopic evidence of central necrosis and lymphatic or vascular invasion was also evaluated. In the cases of multiple tumors in one dog, the most aggressive and undifferentiated tumor was selected.
Histological subtyping was also performed based on the proposed histologic classification by Goldschmidt et al. 7 Among the samples, CMC specimens with the 5 most prevalent histologic subtypes were selected, which were complex carcinoma, simple tubular carcinoma, carcinoma arising in a benign mixed tumor, simple tubulopapillary carcinoma, and solid carcinoma. Finally, 60 specimens from each group were randomly selected as follows: Group 1, BCS = 2 (n = 13) or 3 (n = 47); Group 2, BCS = 4 (n = 51) or 5 (n = 9). The evaluation and interpretation of clinical and histopathologic parameters were performed by 3 pathologists (HY-L, BJ-S, and MK-B) and rechecked by one pathologist (JH-S).
Immunohistochemistry
Serially sectioned 4-µm-thick formalin-fixed and paraffin-embedded tissue samples were deparaffinized, rehydrated in graded ethanol, washed in phosphate-buffered saline (PBS; pH 7.4), and incubated in 3% hydrogen peroxide for 20 minutes at room temperature to block endogenous peroxidase activity. Heat-induced antigen retrieval with citric acid (pH 6.0) or Tris-EDTA (pH 9.0) was performed in a pressure chamber for the detection of myeloid/histiocyte antigen (MHA; MAC387), COX-2, Ki67, and von Willebrand factor (vWF). Antigen retrieval for EGFR was performed using Proteinase K (Dako) for 15 minutes at room temperature. After washing 3 times in PBS, the sections were blocked by incubating with 5% normal goat serum for 30 minutes and then primary antibodies were applied to the slides. All the antibodies used in this study were validated to have specificity to canine tissue. 2,18,24,35 Detailed information about the antibodies and the protocol conditions are summarized in Table 1. After washing 3 times in PBS, secondary antibodies (Dako REAL EnVision kit; Dako) were applied for 40 minutes. After washing 4 times in PBS, the slides were stained with 3,3′-diaminobenzidine (Dako REAL EnVision kit; Dako), and counterstained with Gill’s hematoxylin.
Primary antibodies and immunohistochemistry protocols.
Abbreviations: MHA, myeloid/histiocyte antigen; EGFR, epidermal growth factor receptor; COX-2, cyclooxygenase 2; vWF, von Willebrand factor; RT, room temperature; ON, overnight.
A positive and negative control slide was included in each immunohistochemical assay. Tissue samples used as positive controls were as follows: MHA, normal canine lymph node; COX-2, canine colorectal cancer; EGFR, Ki67, and vWF, adjacent normal skin tissues. For negative controls, the appropriate antibody was replaced by either an iso-type matched immunoglobulin or PBS. All control tissues were selected from our database.
Evaluation of IHC and Macrophage Quantification
Cells were considered as positive for MHA when cytoplasmic labeling was observed in cells with macrophage morphology. Anti-COX-2 immunoreactivity was evaluated and semiquantified using ImageJ v.1.8, a free software downloaded from the National Institutes of Health website. 36 To determine the cytoplasmic expression of COX-2, 5 “hot spots” from each section were captured at 100× magnification, and the mean integrated density divided by the area of the tissue was calculated. EGFR expression was considered as positive when tumor cells had cytoplasmic and/or membranous labeling; the labeling was semiquantified as described in the EGFR PharmDx recommendation (Dako). To determine the percentage of Ki67-positive tumor cells, CMC cells that showed nuclear immunolabeling were counted in 5 “hot spots” at 200× magnification and the percentage calculated by an automated application. 41 Microvessel density in tumors was obtained by counting vWF-positive microvessels within “hot spots” captured in ten 200× fields in each CMC specimen. 43 All digital images were acquired using an Olympus BX51 microscope with an ocular FN 22 mm objective and image transfer software (Olympus, Tokyo, Japan).
To count MHA-positive macrophages (TAMs), CMC specimens were observed at a low magnification to identify the “hot spots” and then 5 areas were acquired at 400× magnification. Areas exhibiting necrosis or severe bleeding were excluded. The number of macrophages was counted manually. The evaluation of IHC was performed by 3 pathologists (HY-L, SH-C, and SH-K) and rechecked by 1 pathologist (JH-S).
Statistical Analyses
Pearson’s χ2 test was used for analyzing the significance of categorical variables or EGFR score between groups. To compare ages, average TAM counts, iMVD, and expression of COX-2 and Ki67 between groups, Student’s t test or Mann-Whitney U test was used when appropriate. P < .05 was considered significant. Data were analyzed using Prism 9 (GraphPad) and IBM SPSS Statistics for Windows, version 21 (IBM Company).
Results
Clinical and Histopathological Characteristics
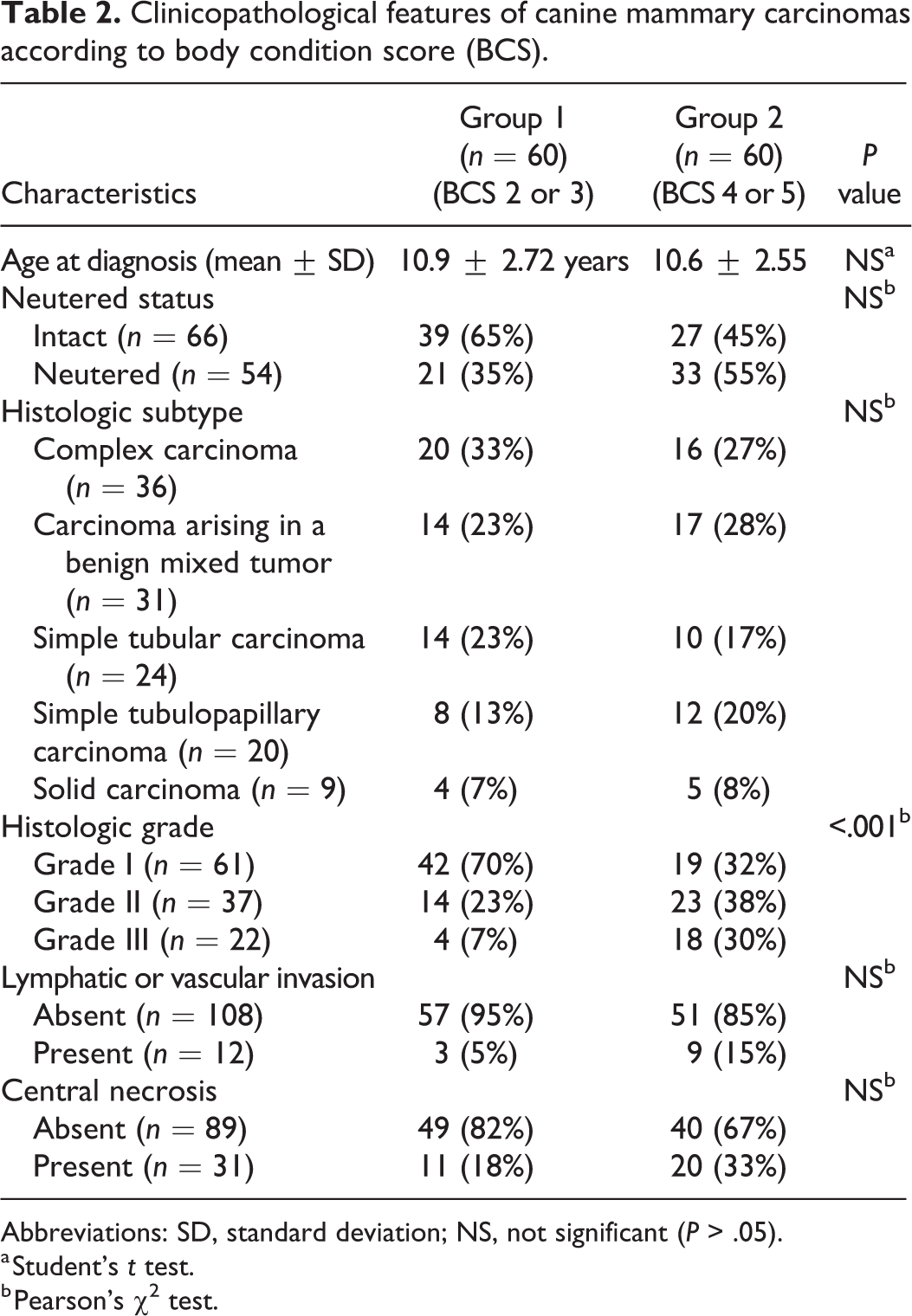
Table 2 summarizes the clinical and histological characteristics of the specimens used in this study. The mean age of the dogs (n = 120) was 10.7 ± 2.6 years (range, 3–18 years), and there was no significant difference between groups. The breeds included Maltese (n = 40), Shih Tzu (n = 22), Poodle (n = 10), Yorkshire Terrier (n = 10), Cocker Spaniel (n = 8), Pomeranian (n = 3), Miniature Pinscher (n = 3), Spitz (n = 2), Miniature Schnauzer (n = 2), Jindo (n = 2), and mixed breeds (n = 15). The breeds of the remaining 3 dogs were Beagle, Dachshund, and Pekingese.
Clinicopathological features of canine mammary carcinomas according to body condition score (BCS).
Abbreviations: SD, standard deviation; NS, not significant (P > .05).
a Student’s t test.
b Pearson’s χ2 test.
The histologic subtypes of the 120 CMC specimens were as follows: complex carcinoma (n = 36), carcinoma arising in a benign mixed tumor (n = 31), simple tubular carcinoma (n = 24), simple tubulopapillary carcinoma (n = 20), and solid carcinoma (n = 9). Tumor grade assessment included 61 (50.8%) grade I, 37 (30.8%) grade II, and 22 (18.35%) grade III CMCs. Thirty-one tumors (25.8%) had central necrosis in the mass, and lymphatic or vascular infiltration of neoplastic cells was present in 12 (10.0%) cases.
Immunohistochemistry
MHA was expressed in macrophages infiltrating within and around tumors with a granular or diffuse cytoplasmic labeling pattern (Figs. 1, 2). The cytoplasmic immunolabeling for COX-2 and EGFR, nuclear labeling for Ki-67, and labeling of vWF-positive microvessels are shown in Figures 4–6.
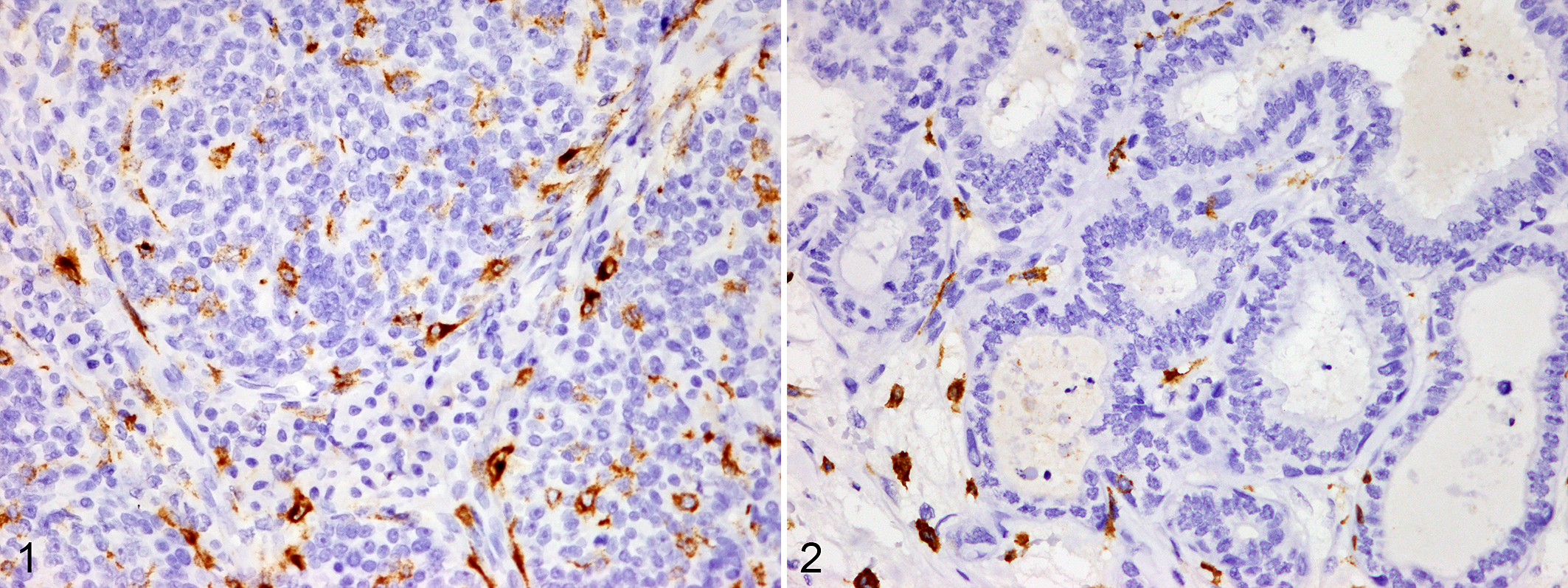
Mammary gland, dog. Myeloid/histiocyte antigen (MAC387) is expressed in the cytoplasm of macrophages infiltrating within and around tumors. Immunohistochemistry, DAB.
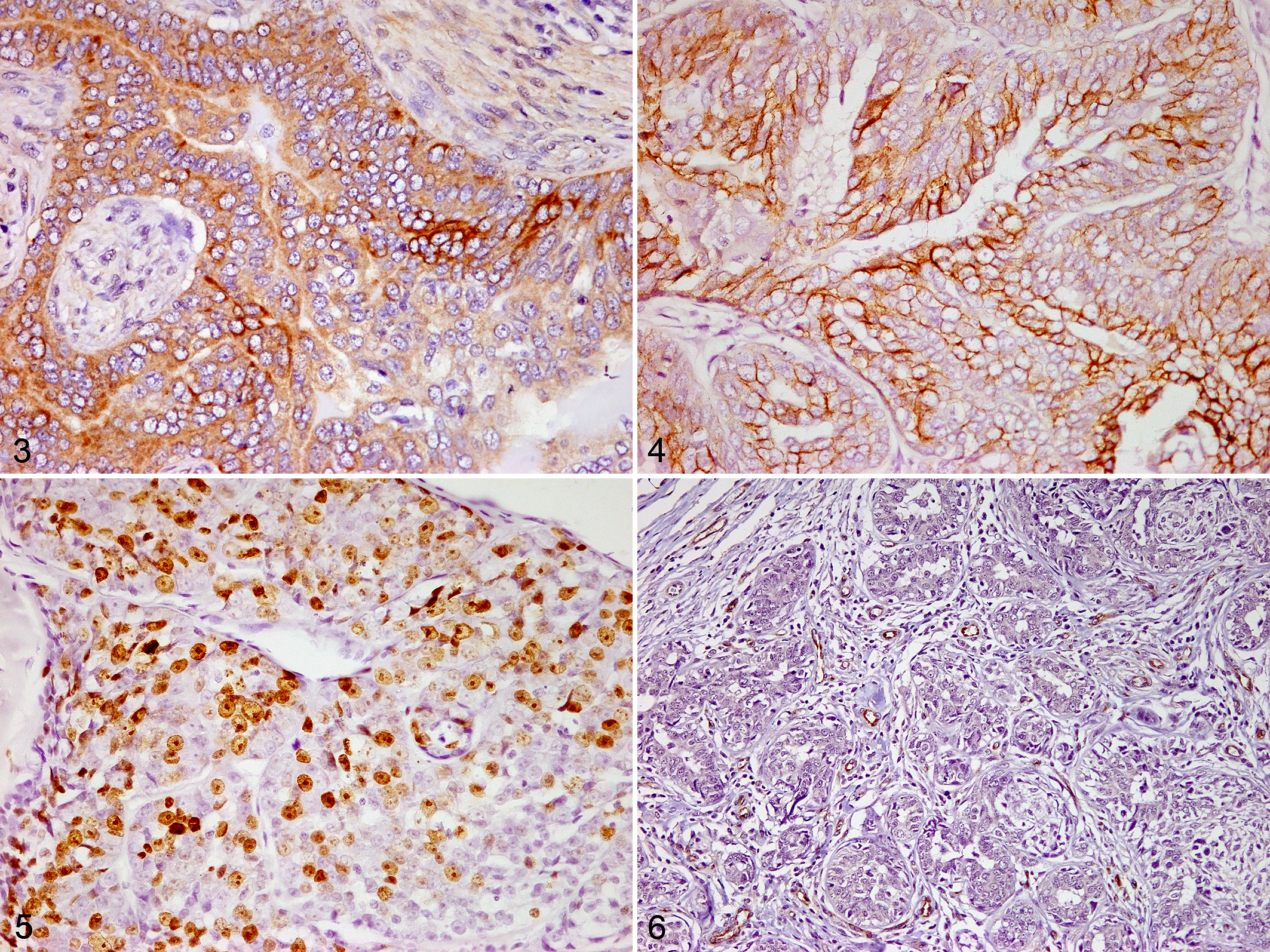
Mammary gland, dog. Immunohistochemistry, DAB.
Association of Obesity Status With Clinicopathological Parameters and IHC Results
The clinical and histopathological features of the 120 CMC specimens are summarized according to BCS in Table 2. Histologic grade was significantly different between groups. The number of grade III CMC specimens was higher in overweight or obese dogs (Group 2, n = 18, 30.0%) than in lean or ideal body weight dogs (Group 1, n = 8, 6.7%; P < .001). There were no significant differences between groups in the average age and neutered status at diagnosis, lymphatic or vascular invasion, or central necrosis.
The average TAM count was significantly higher in overweight or obese dogs (Group 1, 53.57 ± 67.71; Group 2, 72.33 ± 46.21; P < .005; Fig. 7). There were no differences between groups for COX-2 expression or EGFR scores (Table 3). The percentage of CMC cells showing nuclear Ki67 expression was higher in overweight or obese dogs (Group 1, 5.62 ± 4.57%; Group 2, 9.70 ± 5.96%; P < .005; Fig. 8). The iMVD was greater in overweight or obese dogs (Group 1, 102.05 ± 36.97; Group 2, 135.38 ± 39.25; P < .001; Fig. 9).
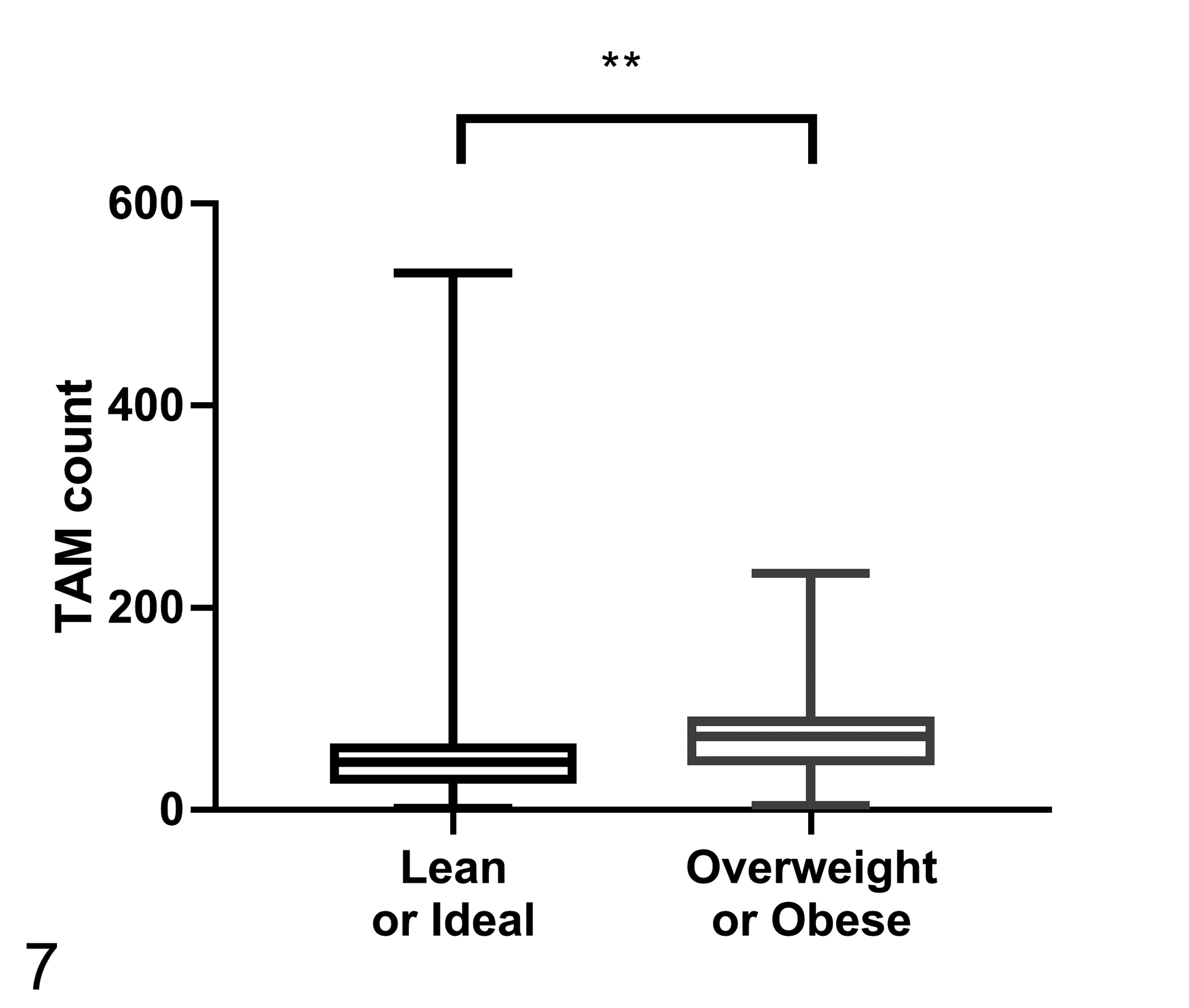
The number of tumor-associated macrophages (TAMs) within malignant canine mammary tumors according to body condition score (n = 60 per group). The TAM count is the number of MAC387-positive cells in 5 selected 400× microscopic fields. The TAM count is significantly higher in overweight or obese dogs. Mann-Whitney U test, **P < .005.
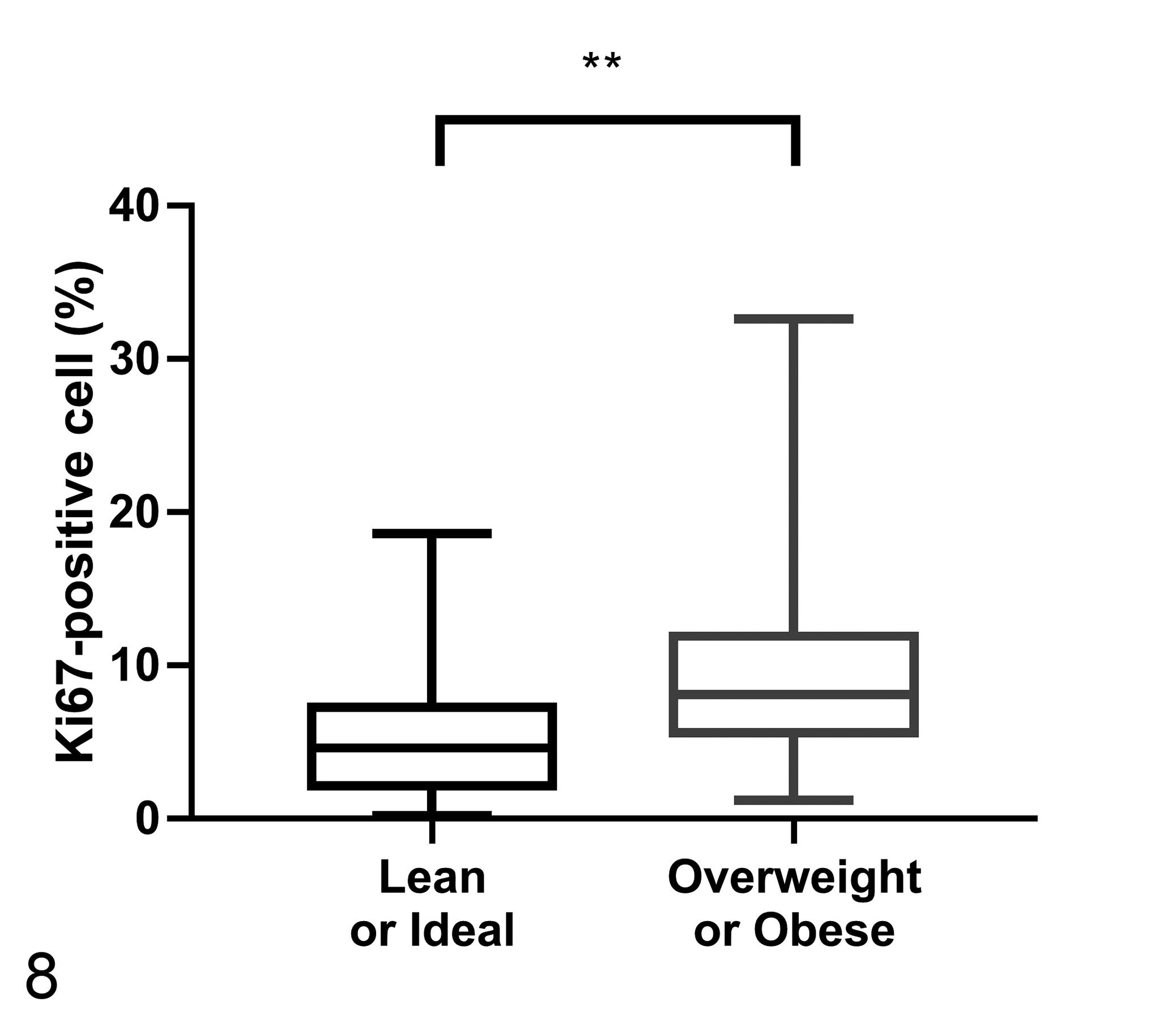
Percentage of Ki67-positive neoplastic cells in malignant canine mammary tumors according to body condition score (n = 60 per group).The percentage of Ki67-positive neoplastic cells is significantly higher in overweight or obese dogs. Mann-Whitney U test, **P < .005.
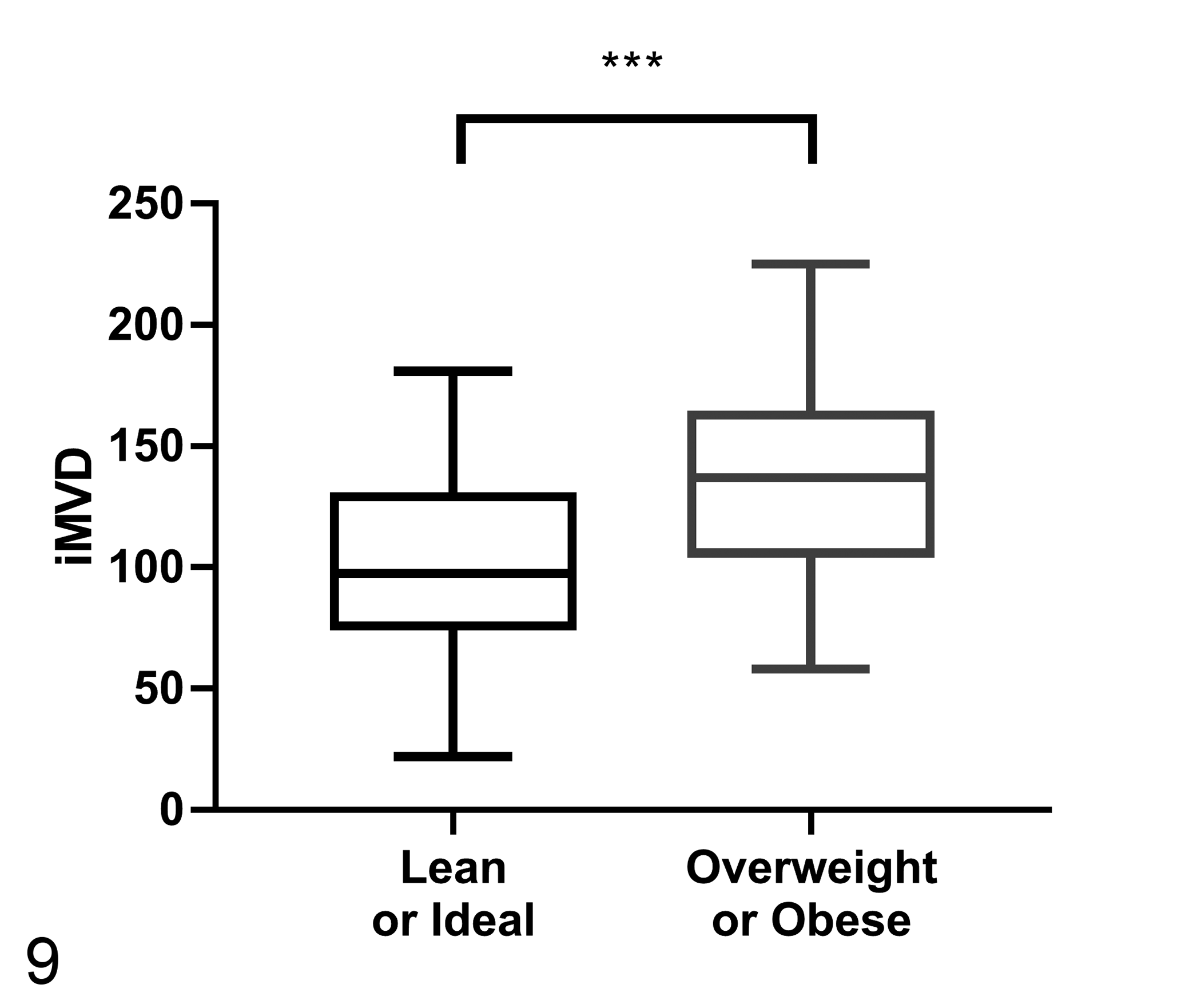
Intratumoral microvessel density (iMVD) in canine mammary cancers according to body condition score (n = 60 per group). The iMVD, measured as the number of vWF-positive microvessels within 10 selected 200× fields, is significantly higher in overweight or obese dogs. Student’s t test, ***P < .001.
Association between groups for expression of cyclooxygenase 2 (COX-2) and epidermal growth factor receptor (EGFR).
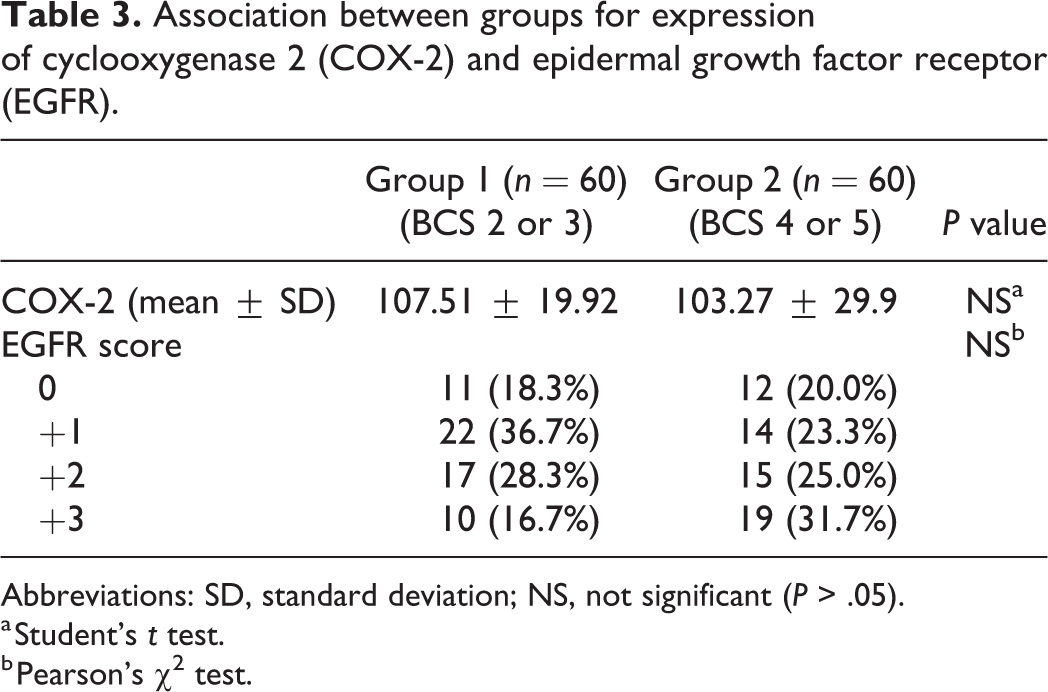
Abbreviations: SD, standard deviation; NS, not significant (P > .05).
a Student’s t test.
b Pearson’s χ2 test.
Discussion
The results of this study demonstrate that CMCs in overweight or obese female dogs have a higher histologic grade, which is consistent with earlier studies. 20,39 In addition, Ki67, considered as a reliable prognostic marker of tumor cell proliferation in CMC, 45 had higher expression in overweight or obese dogs than in lean or ideal body weight dogs. This tends to support the idea that obesity influences CMC malignancy, and could also be a candidate clinical prognostic factor in dogs.
The prevalence of TAMs is higher in malignant tumors than in benign tumors in mammary tumors of humans and dogs, which implies that the number of macrophages in mammary gland tumors is a characteristic of tumor malignancy. 13,32 Furthermore, a higher TAM count in specimens with aggressive histologic subtypes of CMC has also been reported. 25 We observed higher TAM counts in CMC specimens from overweight or obese dogs, which is consistent with our previous study. 18 These results raise the possibility that obesity may influence CMC progression by promoting the inflammatory status, such as via obesity-induced macrophage infiltration.
In addition, macrophages can stimulate tumor angiogenesis, and infiltration of TAMs in CMC is significantly associated with the expression of vascular endothelial growth factor, an established marker of angiogenesis. 33 Saturated fatty acids from adipocytes stimulate TAMs to express pro-inflammatory genes, such as COX-2, interleukin-1β, and tumor necrosis factor-α. 12 Among these inflammatory mediators, COX-2 overexpression is associated with vascular endothelial growth factor expression and angiogenesis in CMC. 5,31 Epidermal growth factor is one of the growth factors secreted by TAMs and the EGFR ligand/receptor system affects progression of tumors by promoting cell proliferation, metastasis, and angiogenesis. 10,20 We did not observe a significant association between EGFR scores or COX-2 expression and BCS. We were not able to determine whether there was an association between COX-2 expression and obesity in CMC because we only analyzed the level of COX-2 in neoplastic cells and not in TAMs. It is noteworthy to investigate the potential role of COX-2, TAM, and obesity in CMC because COX-2 plays an important role in carcinogenesis through the COX-2/prostaglandin E2 pathway, and COX-2 inhibition suppresses macrophage polarization to M2 phenotype. 9,27 Therefore, further studies using other experimental methods are needed to evaluate the relationship between obesity status and the expression of COX-2 in TAMs.
We did, however, observe a statistically significant relationship between obesity status and both iMVD and TAM counts, which have been previously studied in humans and mice. 1,15 Angiogenic factors derived from adipose tissue include leptin, vascular endothelial growth factor, basic fibroblast growth factor, hepatocyte growth factor, heparin-binding epidermal growth factor-like growth factor, tumor necrosis factor-α, and interleukin 6. 42 Angiogenesis is important for tumor progression and metastasis both in human breast cancer and CMC. 16,17 Several authors correlate the obesity and TAM with angiogenesis in human breast cancer and in a murine animal model. 1,11 Since dogs are considered as spontaneous animal models for human breast cancer, these factors are likely to be potential molecular mediators of obesity-induced tumor angiogenesis in dogs as well.
In conclusion, our results suggest that obesity is associated with enhanced infiltration of TAMs and tumor angiogenesis in CMCs, and CMC specimens of overweight or obese female dogs had higher histologic grade than those of lean or ideal BW dogs. Further prospective studies should be conducted to clarify the specific mechanism by which obesity enhances infiltration of TAMs and tumor angiogenesis in CMC.
Footnotes
Acknowledgements
We thank Ms E.-M. Yu for her excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by Konkuk University Researcher Fund in 2020.
