Abstract
A 12-year-old neutered female cat was referred with clinical signs referable to lower urinary tract disease. Clinical examination revealed a tense, painful urinary bladder, and proximal urethral thickening. Endoscopic studies showed a pedunculated mass with polypoid projections. Multiple full-thickness mucosal biopsies were obtained, and the mass was almost completely excised. The neoplasm was confined to the mucosa and consisted of epithelial cells arranged in anastomosing trabeculae and nests, growing downward into the lamina propria. Neoplastic cells showed minimal atypia and low mitotic activity. Histological findings were consistent with inverted urothelial papilloma. Feline papillomavirus DNA was not amplified from biopsies. One year later, the cat had no urological signs, and urinary bladder was normal at ultrasound. To the authors’ knowledge, this is the first report of a case of inverted urothelial papilloma in a cat and is a differential diagnosis for low-grade urothelial carcinoma and other lesions with inverted growth pattern.
A 12-year-old, neutered female, domestic shorthair cat was referred to the Veterinary University Hospital of the University of Bologna with a 3-day history of dysuria, hematuria, and anorexia. The cat had a long clinical history of recurrent bacterial cystitis treated with repeated antimicrobial therapies. Clinical examination only revealed a tense, painful urinary bladder, and proximal urethral tract thickening. The cat was moderately anemic with a hematocrit value of 20.4% (reference interval 32.0% to 48.0%) at a complete blood count. Serum chemistry abnormalities included increased liver enzymes (alanine transaminase 146 U/L; reference interval 20–72 U/L; aspartate transaminase 62 U/L, reference interval 9–40 U/L), azotemia (serum urea 73 mg/dL; reference interval 30–65 mg/dL), mild hypoproteinemia (total protein 5.49 g/dL; reference interval 6.50–8.80 g/dL), and hypoalbuminemia (albumin 2.34 g/dL; reference interval 2.60–4.00 g/dL). Urinalysis findings included hematuria, proteinuria, glycosuria, and abundant leukocytes and bacteria recorded on urine sediment examination. Urine culture was performed and yielded no bacterial growth.
Clinical signs were indicative of lower urinary tract disease; hence, urinary tract infection, feline idiopathic cystitis, urolithiasis, and neoplasms with partial ureteral obstruction were initially considered in the differential diagnosis. Radio-opaque material in the urinary bladder, consistent with uroliths, was detected in abdominal radiographs. Ultrasound examination showed a pedunculated, oval, 16 × 7.6 mm mass, in the trigone region of the bladder, also involving the proximal urethra, leading to partial obstruction of the urinary tract. The ultrasonographic pattern of the mass was homogeneous, and color Doppler showed a slight vascularization. Based on imaging results, differential diagnoses included inflammatory diseases (eg, polypoid and/or eosinophilic cystitis) and neoplasms (eg, papilloma, adenoma, urothelial carcinoma). A percutaneous antegrade cysto-urethroscopy was performed, 4 which revealed a 2-cm multilobulated, pedunculated mass in the dorsal aspect of the bladder trigone, just cranial to the ureteral openings, with multiple polypoid projections arising from the lateral borders of the mass. The mass appeared pale pink, except for the apex where tissue was erythematous. The remainder of the urinary tract was apparently normal (Fig. 1).
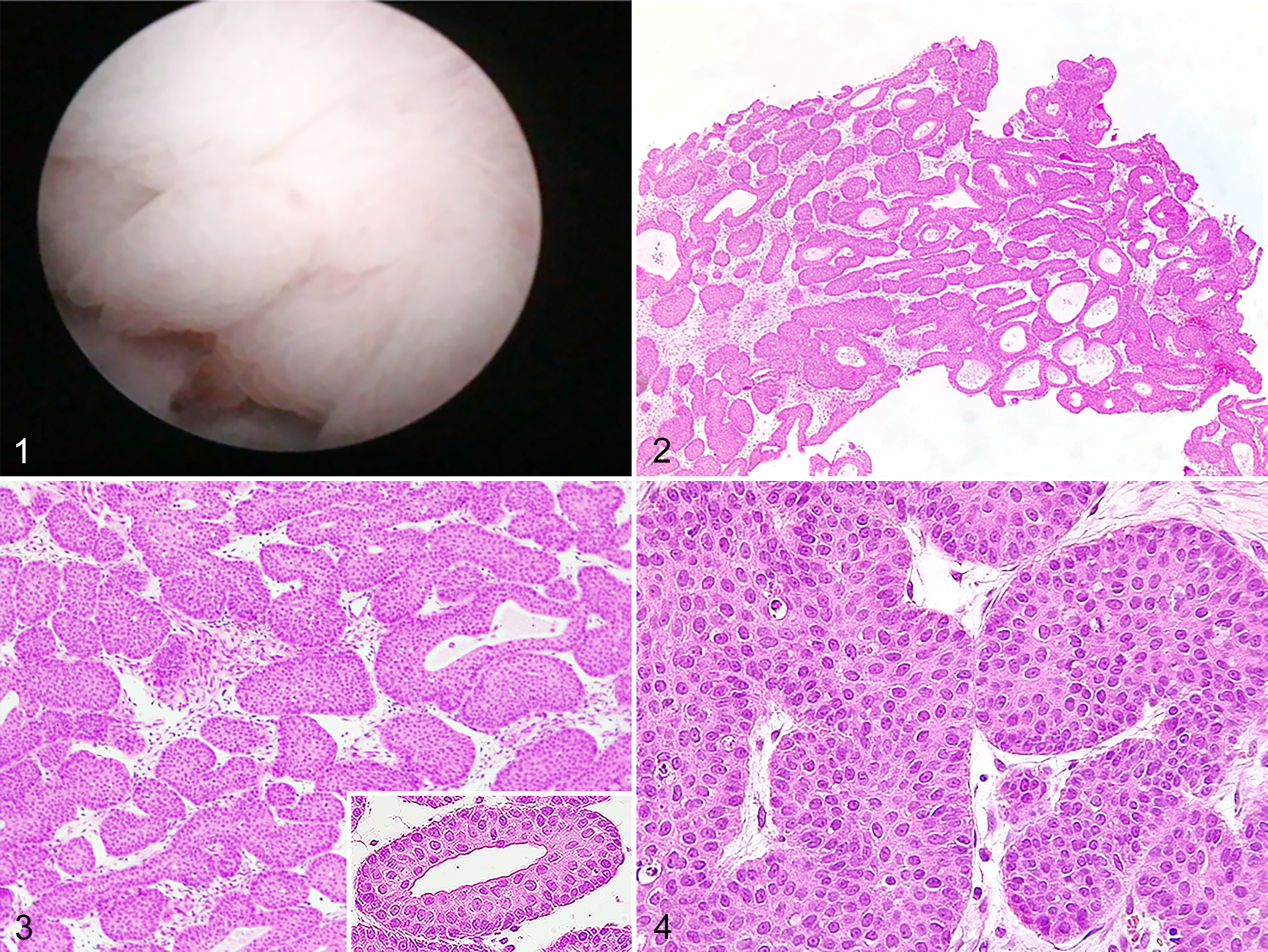
Inverted urothelial papilloma, urinary bladder, cat.
Multiple incisional, full-thickness mucosal biopsies were obtained; some were smeared on slides for cytology and others were fixed in 10% buffered formalin for further histopathological examination. The mass was almost completely excised during sampling procedures. The clinical condition of the cat progressively improved and at 14-day interval follow-up, hematocrit value increased to 27.1%; urinalysis and serum chemistry variables were within reference intervals. One year after diagnosis, the cat had no urological signs and the urinary bladder appeared normal at ultrasound.
In impression smears, stained using Romanowsky quick stain (Diff-Quick; Bio-Optica) small papillary clusters of polygonal epithelial cells were recorded, with distinct cell borders and abundant light blue cytoplasm. Nuclei were round, central, with finely reticulated chromatin and occasional distinct nucleoli. Anisocytosis and anisokaryosis were mild, mitoses were rare. Considering the mild nuclear atypia and the mild cellular pleomorphism, cytological findings were considered suggestive of a benign epithelial tumor of the bladder.
Histologically, the neoplasm consisted of large, polygonal epithelial neoplastic cells arranged in anastomosing trabeculae and nests, arising from the surface urothelium and growing downward into the lamina propria, supported by a scant amount of connective stroma (Fig. 2). Trabeculae and nests were limited by a well-defined basement membrane and composed of multiple layers of cells (3–10 cell layers) resembling mature urothelium, sometimes with a palisading basal cell layer (Fig. 3). Neoplastic cells had large, round nuclei with indistinct nucleolus, abundant pale eosinophilic cytoplasm and distinct cell borders. Microcystic formations (Fig. 3 inset) and scattered apoptotic cells were observed throughout the neoplasm, as well as lymphocytes and plasma cells in the stroma. Nuclear atypia and pleomorphism were mild (Fig. 4), and mitoses were 3 in 10 high-power fields (2.37 mm2). The mucosal surface of the neoplasm was lined by normal urothelium.
Histopathological findings were consistent with inverted urothelial papilloma. 5,9,13,14,16
Formalin-fixed, paraffin-embedded tissue scroll of the samples was submitted for papillomavirus (PV) DNA amplification. Total DNA was extracted using the NucleoSpin DNA FFPE XS kit (Macherey Nagel), according to manufacturer’s instructions. DNA integrity was checked by PCR (polymerase chain reaction) amplification of 119 bp of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) gene using the primers dogGAPDHF 5′ GGCGTGAACCACGAGAAGTATAA 3′ and dogGAPDHR 5′ CCCTCCACGATGCCAAGT 3′. To detect PV DNA, 3 primer pairs were used, targeting the L1 ORF of the Felis catus papillomavirus types 1, 2, 3, 4, and Bovine PV type 14, namely, FPV12F/R, FPV34F/R, and jmpSA F/R. 1,12 A no template control and positive control, consisting of bidistilled water and a DNA sample previously sequenced, respectively, were also performed with each reaction. Papillomavirus DNA was not amplified in biopsies analyzed; thus, a viral cause of inverted urothelial papilloma (IUP) was ruled out.
This study documents the occurrence of inverted urothelial papilloma of the bladder in a cat. In veterinary pathology, inverted urothelial papilloma has only been described in cattle as a rare bladder neoplasm sharing histological features of urothelial papilloma, except for the endophytic pattern of growth of papillary projections. 5 In the case described here, cytological examination was not reliable for diagnosing IUP as it is possible that the cells observed on cytology were exfoliated from the normal urothelium covering the neoplasm; thus, histopathology was required to reach a definitive diagnosis.
Histologically, IUP needs to be differentiated from other lesions and neoplasms of the bladder with an inverted growth pattern, including polyp with Brunn’s nests, cystitis cystica, cystitis glandularis, and low-grade urothelial carcinoma, as well as with an exophytic growth pattern (ie, polypoid cystitis, urothelial papilloma). The histological features that allow to differentiate IUP from other bladder lesions are reported in Table 1.
Main histological features that allow differentiation of inverted urothelial papilloma from other neoplastic and non-neoplastic lesions.
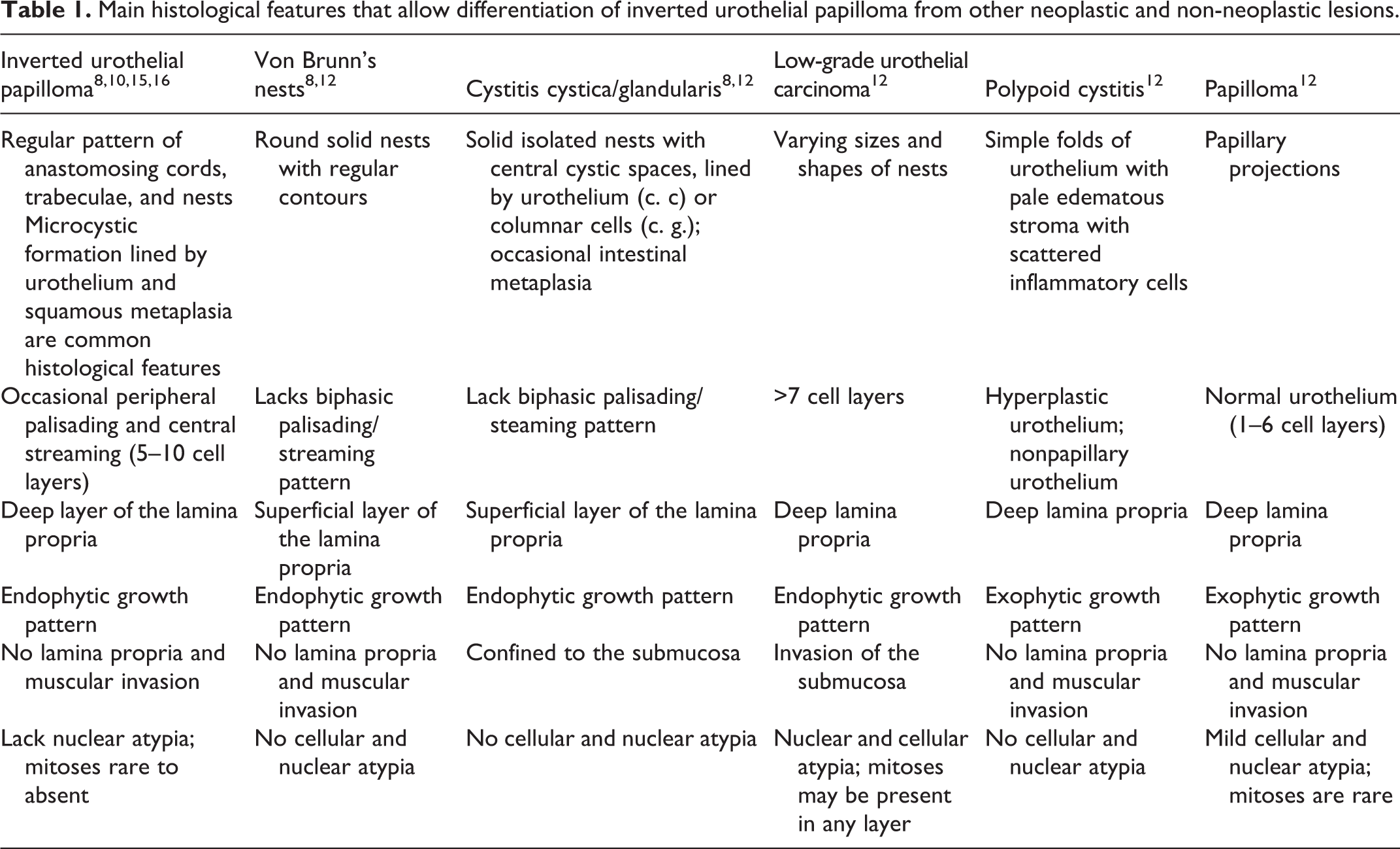
We favored the diagnosis of papilloma because the growth of the cells was in nests and trabeculae. Exuberant proliferation of von Brunn’s nests can mimic IUP; however, nests are separated, and a complex trabecular architecture is absent. 7,11 Cystic and glandular cystitis were excluded due to the trabecular pattern of growth that characterized IUP, and because the cysts are not lined by cuboidal or columnar epithelium typical of cystitis glandularis. 11 Due to the overlapping morphology, the distinction between IUP and endophytic urothelial carcinoma can be challenging, in particular in low-grade urothelial carcinomas. Distinctive histological features that allowed us to differentiate IUP here described from low-grade urothelial carcinoma included the following: low cellular atypia and mitotic activity, lack of invasion into muscularis propria, mild stromal inflammation, and microcystic formation. Unlike urothelial carcinoma, human IUP typically shows no evidence of nuclear atypia and cellular pleomorphism, and mitoses are usually rare to absent. 14,16 Additionally, invasion into the muscularis propria does not occur in IUP, confirming its benign biological behavior and low recurrence rate reported in human patients. 14,16 In human medicine, immunohistochemistry and molecular methods may be useful in distinguishing an IUP from a low-grade urothelial carcinoma. In particular, low Ki-67 proliferation rate (<1%) and negative staining for CK20 are generally associated with IUP. 14 Also, chromosomal aberrations detected with fluorescence in situ hybridization method, are common in urothelial carcinoma, but rare in IUP. 10
In humans, IUP has been described as a rare noninvasive endophytic neoplasm, accounting for 1% to 2% of all urothelial neoplasms. 13 The cause is unknown, but some authors suggested that IUP arises from reaction to inflammation, chronic infection, smoking, or hyperplasia of von Brunn’s nests. 16 A viral cause has been ruled out in human patients, as human papillomavirus has never been detected by immunohistochemistry or in situ hybridization and recent studies strongly suggest that IUP of the bladder may arise following chronic cystitis and infection. 2 Also in the case here described, feline papillomavirus DNA was not amplified from biopsy specimens, but a larger case series would be necessary to exclude a viral cause in cats.
Clinical signs of IUP in the current report were similar to those described in human cases. In particular, hematuria and dysuria are the most commonly described clinical abnormalities, with partial or complete urinary tract obstruction due to growing neoplasms. 16 However, a high percentage of IUP cases are asymptomatic and incidentally found during unrelated clinical examinations. 16 Imaging studies reveal nonspecific findings, and a combination of noninvasive cross-sectional imaging, cystoscopy, and histopathological examination is mandatory for detection and diagnosis of IUPs. Based on imaging and cystoscopy findings, other differential diagnoses were considered in the case here described, including chronic proliferative inflammation and tumors. Endoscopic findings, in particular, were unusual and difficult to interpret due to the lack of previously described cases, and were not suggestive of a specific disease.
In human pathology, diagnostic criteria for IUP include the following: inverted growth pattern; uniform epithelial morphology, rare to absent mitotic figures, occasional microcyst formation, squamous metaplasia; lack of lamina propria invasion; and mucosal surface of the neoplasm covered by normal urothelium. 3 Based on cell differentiation and pattern of growth, inverted papillomas are further classified into trabecular, glandular, and mixed subtypes. 9,10 In the trabecular subtype, urothelial cells have an anastomosing and trabecular arrangement, with occasional peripheral palisading of the cells. Microcystic spaces lined by flattened urothelial cells are commonly reported. 10 The glandular subtype is characterized by islands of urothelial cells, with pseudoglandular and true glandular structures, lined by stratified urothelium and mucus-secreting columnar epithelium, respectively. 10
The IUP described shares histological features with the trabecular subtype described in human pathology with widely distributed microcystic formations lined by mature urothelial cells. Biphasic palisading/streaming pattern is lacking in our case; however, it does not represent a mandatory diagnostic criterion in diagnosing IUP in humans. 3 The different histological subtype reflects the different origin of the neoplasm. While the trabecular subtype arises from proliferation of the basal cell layers, the glandular subtype develops from cystitis cystica et glandularis, considered to be preneoplastic lesions in human pathology, with a high ratio of coexistence with bladder neoplasms. 5,6,10,15 Conclusively, although mitoses are usually rare to absent in IUPs in humans, 14,16 the neoplasm described here showed low mitotic activity.
A case of IUP of the bladder in cat is reported. Practicing veterinarians and pathologists should be aware of the possibility of occurrence of IUP in cats and include this entity in the differential diagnosis of urothelial lesions. Although IUP is considered benign in humans, it does appear to be a risk factor for urothelial carcinoma development. 14 Therefore, careful monitoring of the urinary tract in patients diagnosed with inverted papilloma is recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
