Abstract
Although pyloric and duodenal adenomas occasionally occur in cats, limited information is currently available on their phenotypes and molecular features. The present study investigated the pathological features of these tumors and the mechanisms underlying their tumorigenesis. Biopsy samples from 8 cats diagnosed with pyloric or duodenal adenomas were examined by histopathology and immunohistochemistry. Normal pyloric and duodenal tissues of cats were assessed for comparison. All cases showed a papillary growth of cuboidal to columnar cells with eosinophilic, ground-glass cytoplasm. Mucin in tumor cells was positive for periodic acid–Schiff and paradoxical concanavalin-A staining, but was negative for Alcian blue. Immunohistochemically, tumor cells were positive for cytokeratin (CK) 19 in 8/8 cases and for CK20 in 5/8 cases, and weakly positive for CD10 in 4/8 cases, CK7 in 3/8 cases, and β-catenin in 2/8 cases. Nuclear accumulation of p53 was not detected in any case. DNA sequencing analysis identified no KRAS or GNAS mutations in the 4/8 cases and 5/8 cases for which the KRAS and GNAS genes could be amplified. The histological and immunohistochemical features of tumor cells were similar to those of mucous neck cells and the pyloric gland of normal feline tissue. The morphology of feline pyloric and duodenal adenomas was consistent with that of pyloric gland adenoma in humans; however, its molecular pathogenesis may differ given the lack of KRAS and GNAS mutations in the feline tumors.
The gastrointestinal tract is the third most frequent organ affected by neoplastic diseases in cats, with more than 80% being malignant. 10 Benign epithelial tumors, such as adenomatous polyps or adenomas of the pyloric and duodenal area, have been reported. 6,18 The gastric and duodenal segment is composed of various types of epithelial cells: foveolar, fundic gland, pyloric gland, intestinal epithelium, intestinal submucosal gland, and pancreatobiliary duct cells (duodenal papilla). However, few histopathological studies have been conducted on pyloric and duodenal adenomas in the cat, 6 and the phenotypes of these neoplastic epithelial cells have not been examined in detail.
In humans, adenomas in the stomach and duodenum are broadly classified into gastric and intestinal types based on the phenotypes of tumor cells. 8,15 These 2 types of adenoma differ not only in their histological features but also in their molecular pathogenesis. Activating mutations in KRAS are common in both the gastric and intestinal types, whereas GNAS mutations are found only in the gastric type, particularly in pyloric gland adenoma. 11,12,19 However, limited information is currently available on the genetic features, including the KRAS and GNAS mutation status, of feline gastrointestinal tumors, 33 and it has not yet been clarified whether feline pyloric and duodenal adenomas correspond to human gastric- or intestinal-type adenomas.
In the present study, we examined the phenotypes of neoplastic cells in feline pyloric and duodenal adenomas by comparing their morphological, histochemical, and immunohistochemical features with normal epithelial cells comprising the feline gastrointestinal mucosa. The molecular features of tumor tissue, including KRAS and GNAS alterations, were analyzed. The mechanisms underlying the tumorigenesis of feline pyloric and duodenal adenomas are also discussed in comparison with those of human gastrointestinal adenoma.
Materials and Methods
The records of feline gastric and/or duodenal biopsy cases (681 cases) that were submitted to the Laboratory of Veterinary Pathology, the University of Tokyo, between 2009 and 2020 were reviewed. Eight cats histopathologically diagnosed with pyloric and/or duodenal adenoma were selected for further histopathological examinations. Tissue specimens were obtained through endoscopic biopsy or surgical resection. Follow-up information on clinical improvement and survival days were collected through local veterinarians.
All tissues were fixed in 10% neutral buffered formalin and routinely embedded in paraffin wax. Consecutive sections (thickness of 4 μm) were stained with hematoxylin and eosin (HE), Alcian blue (pH 2.5)-periodic acid–Schiff (AB-PAS), and paradoxical concanavalin-A staining (PCS) (which identifies class III mucins characterized by GlcNAcα1 → 4Galβ residue), and immunohistochemistry was performed. The gastric and duodenal tissues of normal cats were also stained with AB-PAS, PCS, and immunohistochemistry for comparison.
For immunohistochemistry, endogenous peroxidase was blocked by a treatment with 3% hydrogen peroxide in methanol for 5 minutes. To unmask antigens, sections were then treated with citrate buffer (pH 6.0) by heating in an autoclave (121 °C) for 10 minutes or were incubated with 125 µg/ml proteinase K (Wako) solution at room temperature for 30 minutes. After washing with Tris-buffered saline (TBS), slides were treated with 8% skimmed milk in TBS. The primary antibodies used are listed in Table 1. After an incubation with primary antibody at 4 °C overnight, immunolabeled antigens were visualized using the Dako Envision+ System (Dako) and reacted with 0.05% 3′,3-diaminobenzidine plus 0.03% hydrogen peroxide in Tris-hydrochloric acid buffer. Sections were then counterstained with hematoxylin. The immunoreactivity of neoplastic cells was scored semiquantitatively as follows: − (negative), <1% positive cells; + (weakly positive), 1% to 30% positive cells; ++ (moderately positive), 31% to 60% positive cells; +++ (strongly positive), >60% positive cells. Nuclear immunoreactivity was interpreted as positive for β-catenin, CDX2, and p53, whereas membranous or cytoplasmic reactivity was interpreted as positive for CD10, CK7, and CK20.
Antibodies used for immunohistochemistry.

Abbreviations: CK, cytokeratin; mAb, monoclonal antibody; pAb, polyclonal antibody.
Genomic DNA was extracted from formalin-fixed paraffin-embedded pyloric and/or duodenal tissues using the QIAamp DNA FFPE Tissue Kit (Qiagen) for gene mutation analysis. Purified DNA was used in subsequent polymerase chain reaction (PCR), in which amplification was conducted with specific primer pairs for the feline KRAS gene that amplifies a 286-bp fragment that incorporates exon 1, 5′-AGGTGAATTTGCGTTAAAAGGTAT-3′ (forward) and 5′-AAAGAATGGGCCTGCACAAA-3′ (reverse), and with specific primer pairs for the feline GNAS gene that amplifies a 441-bp fragment incorporating exons 8 and 9, 5′-CCCTTTCCAAGCTCGTCAGG-3′ (forward) and 5′-AGCAGAAGCAGTGTGGGTTA-3′ (reverse). PCR was performed as follows: for primers for the KRAS gene, 30 cycles at 95 °C for 30 seconds and 53 °C for 30 seconds; for primers for the GNAS gene, 30 cycles at 95 °C for 30 seconds and 61 °C for 30 seconds. PCR products were submitted for a sequence analysis in the forward direction (Eurofins Genomics Inc). Each chromatogram was visually inspected for any abnormalities. Unaffected feline tissue was also used to confirm the normal sequences of the feline KRAS and GNAS genes.
Results
Clinical and Endoscopic Findings
Among 681 feline gastric and/or duodenal biopsy cases, 8 (1.2%) were diagnosed as pyloric and/or duodenal adenoma. Information on each case is summarized in Table 2. Archived samples were obtained from 3 intact males, 3 spayed females and 2 intact males, aged 5 to 13 years (average: 10 years). Cases involved 4 mixed breed, 1 Abyssinian, 1 Birman, 1 Russian Blue, and 1 Scottish Fold. The most common clinical signs were anorexia (6/8 cases), vomiting (5/8 cases), diarrhea (2/8 cases), and weight loss (1/8 case). Intestinal T-cell lymphoma (small-cell type) and severe colitis were also present in 2/8 and 1/8 cases, respectively. Upper gastrointestinal endoscopy was performed on all cases and revealed smooth, round masses within the pylorus or proximal duodenum (Figs. 1, 2). Six cases had a single mass in gastroduodenal junction or duodenum, and the other 2 cases had 2 masses in the stomach and duodenum that were separated from each other (Figs. 1, 2). In case 6, the pyloric mass was pedunculated, smooth and larger than the duodenal mass, whereas the duodenal mass was sessile and multilobulated. In case 8, the pyloric and duodenal masses were both well-demarcated semi-pedunculated, and similar in size. Polyps were removed during endoscopy in 6/8 cases, while laparotomy and surgical resection were performed in 2/8 cases. Improvement of clinical signs was observed after endoscopic or surgical removal in 6/7 cases; however, case 7 developed T-cell lymphoma/leukemia and died 344 days after resection. Case 4 had intestinal lymphoma and died 21 days after the duodenal adenoma was diagnosed. The clinical outcome of case 2 was not available.
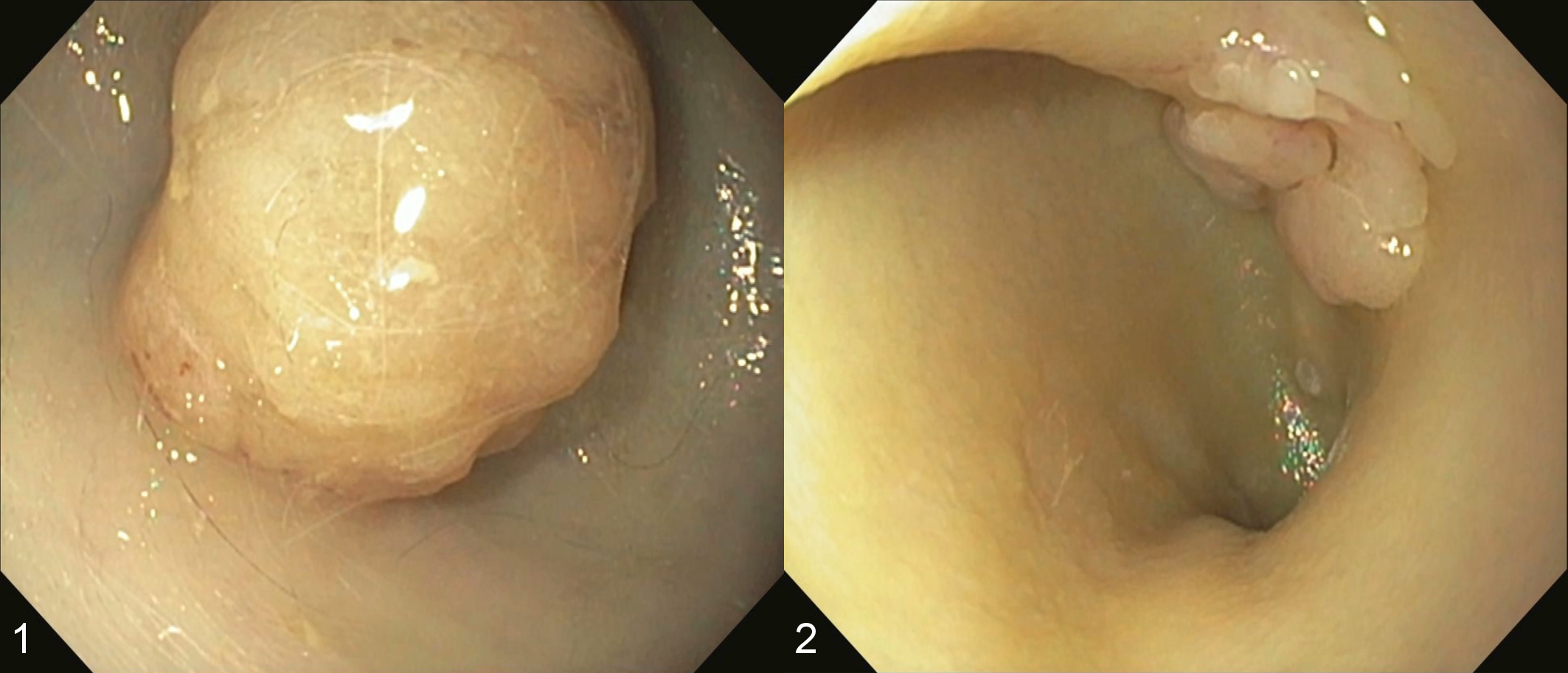
Pyloric and duodenal adenoma, cat, case 6.
Demographic and clinical information on feline pyloric and duodenal adenoma cases.
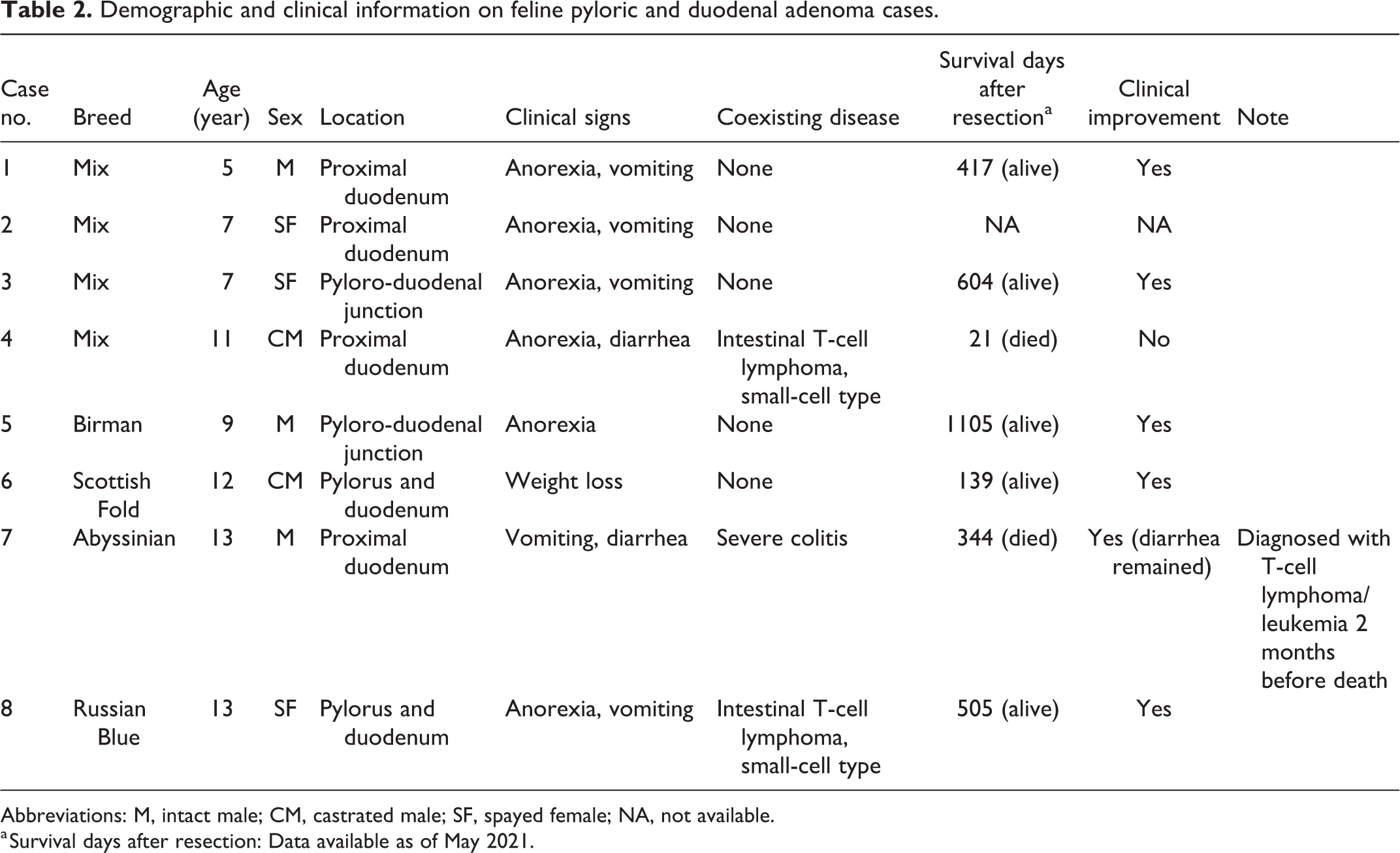
Abbreviations: M, intact male; CM, castrated male; SF, spayed female; NA, not available.
a Survival days after resection: Data available as of May 2021.
Histopathology
All tumors had similar histologic lesions irrespective of the affected site. Tumors were composed of a tubulopapillary arrangement of cuboidal to columnar cells, and tubules were often dilated with a serous component. Tumor cells had abundant, eosinophilic, ground-glass cytoplasm, and round and basally located nuclei with small nucleoli (Figs. 3, 4). Mild anisokaryosis and pseudostratification with loss of polarity were observed (Fig. 4). Mitotic figures were rarely detected. The stroma was composed of fibrous tissue accompanied by mild edema and mild infiltration with lymphocytes and plasma cells. Invasive growth of tumor tissue was not detected in any case.
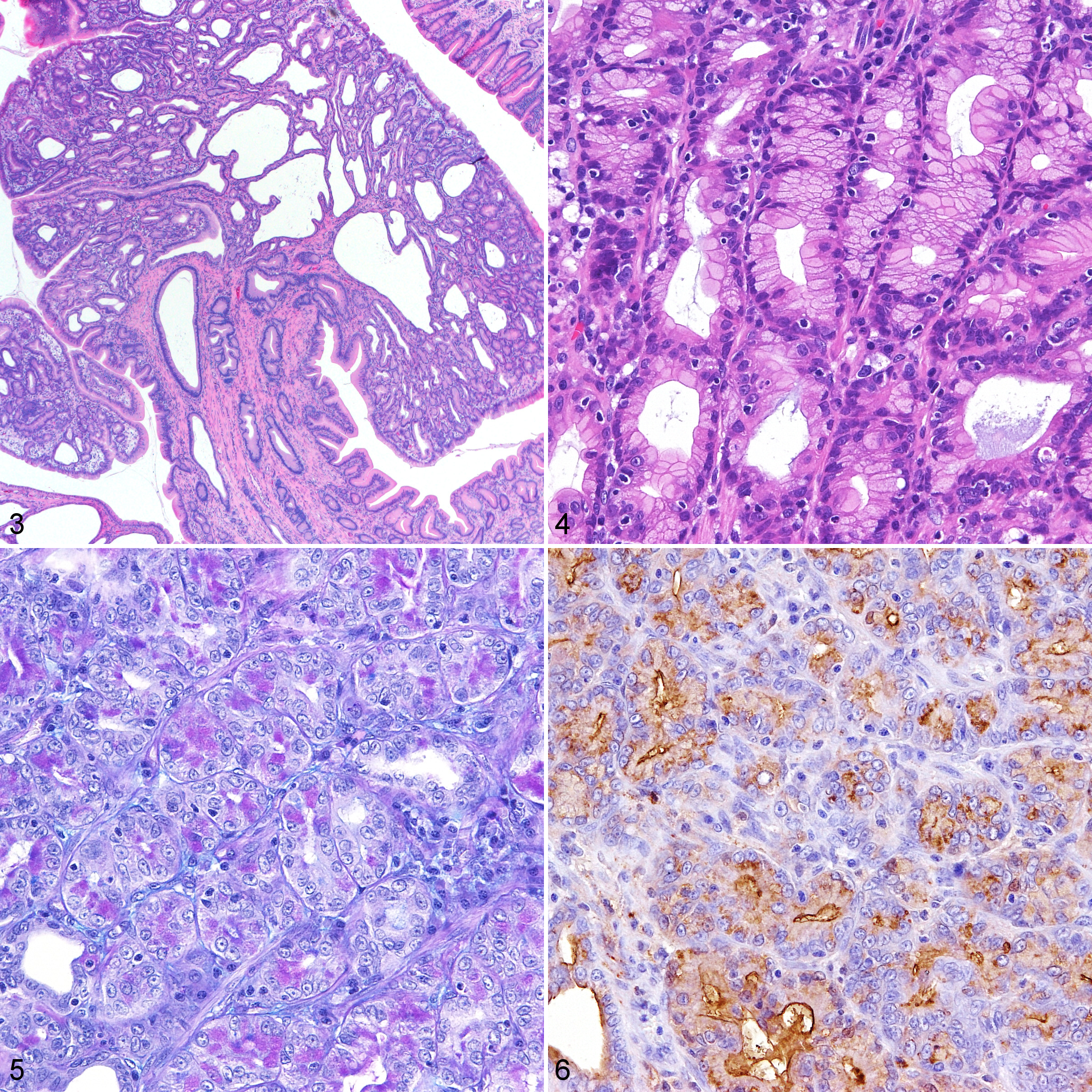
Pyloric adenoma, stomach, cat, case 6.
Histochemistry and Immunohistochemistry
The results of special stains and immunohistochemistry for normal and neoplastic feline gastric and duodenal tissues are summarized in Tables 3 and 4. In the normal gastric and duodenal tissue, AB−/PAS+ mucin was observed in foveolar, fundic gland, pyloric gland, and intestinal submucosal gland cells. AB+/PAS+ or AB+/PAS− mucin was only found in the intestinal epithelium and fundic gland cells, respectively. PCS-positive mucin was also detected in fundic, pyloric, and intestinal submucosal gland cells. Immunolabeling for CD10 and CDX2 was restricted to the intestinal epithelium. CK7 immunoreactivity was also limited to pancreatobiliary ducts, while CK19 immunoreactivity was observed in all epithelial cells, except for intestinal submucosal gland. CK20 immunolabeling was detected in all epithelial cells examined.
Histochemical and immunohistochemical results in normal feline stomach and duodenum.
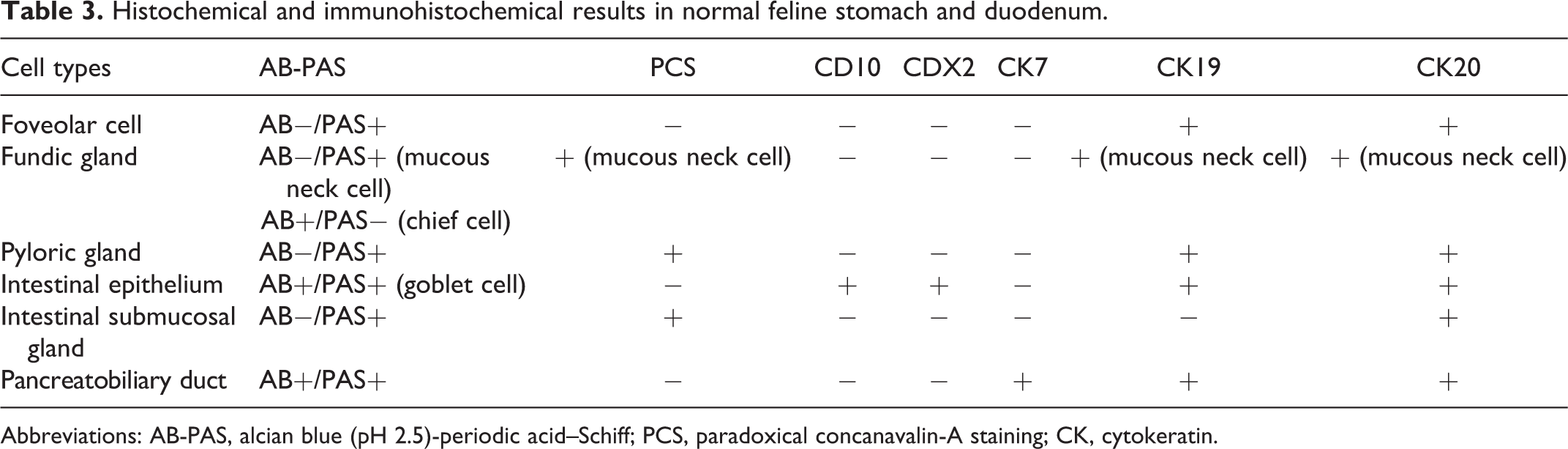
Abbreviations: AB-PAS, alcian blue (pH 2.5)-periodic acid–Schiff; PCS, paradoxical concanavalin-A staining; CK, cytokeratin.
Histochemical and immunohistochemical results in feline pyloric and duodenal adenomas.
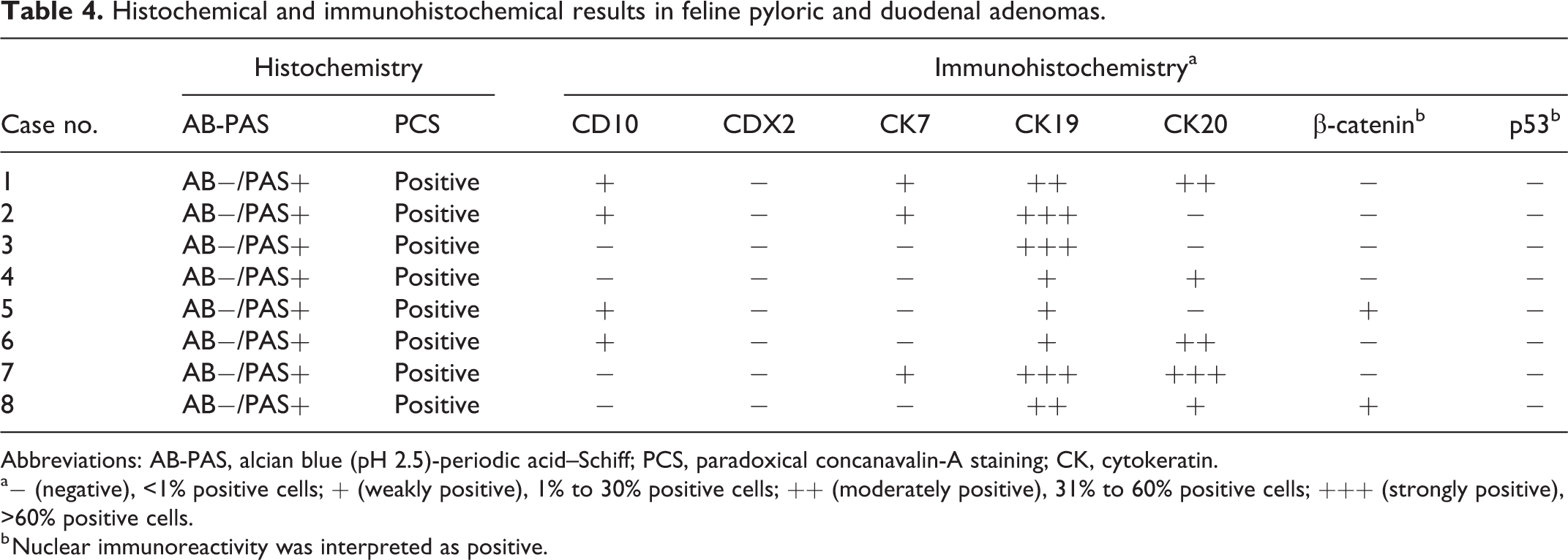
Abbreviations: AB-PAS, alcian blue (pH 2.5)-periodic acid–Schiff; PCS, paradoxical concanavalin-A staining; CK, cytokeratin.
a− (negative), <1% positive cells; + (weakly positive), 1% to 30% positive cells; ++ (moderately positive), 31% to 60% positive cells; +++ (strongly positive), >60% positive cells.
b Nuclear immunoreactivity was interpreted as positive.
Neoplastic cells contained AB-/PAS+ mucin in all cases (Fig. 5). The intracellular mucus and serous components in neoplastic ducts were also predominantly positive for PCS (Fig. 6).
Immunohistochemically, neoplastic cells in all cases were weakly or strongly positive for CK19 (Fig. 7). Tumor cells in 5/8 cases were also positive for CK20 to various degrees (Fig. 8). In 4/8 and 3/8 cases, neoplastic cells were weakly positive for CD10 and CK7, respectively (Figs. 9, 10). Weak nuclear immunolabeling for β-catenin was observed in 2/8 cases (Fig. 11). The nuclear accumulation of p53 was not detected in any case (Fig. 12).
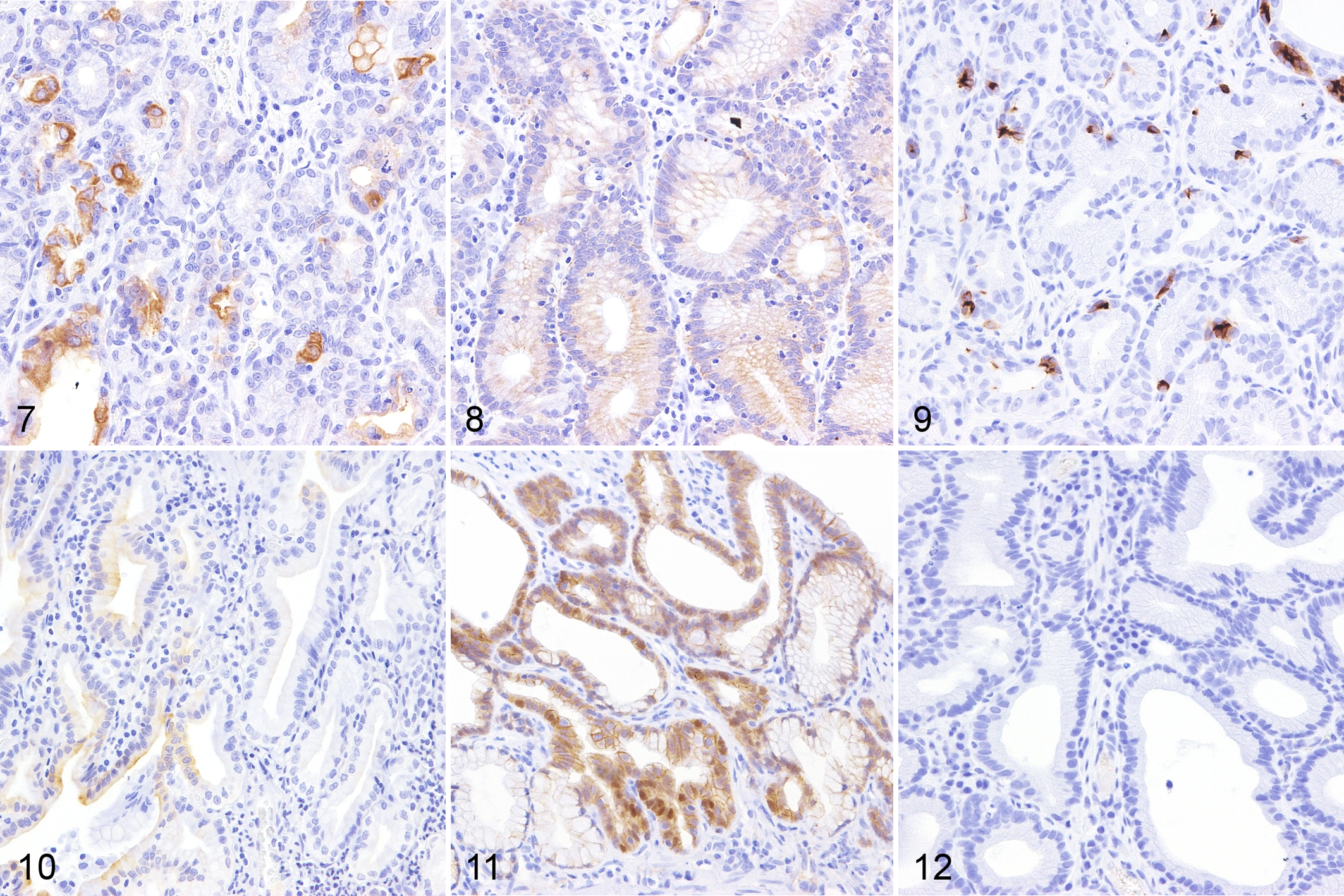
Pyloric and duodenal adenoma, cat. Immunohistochemistry.
Mutation Analysis
Extracted genomic DNA from 4 (cases 1, 4, 6, and 8) and 5 (cases 1, 3, 4, 6, and 8) out of the 8 cases were successfully amplified with the primers for KRAS and GNAS, respectively; DNA of sufficient quality for analysis was not obtained from the other cases. The DNA sequencing analysis identified no KRAS or GNAS mutations, including KRAS codons 12 and 13 and GNAS codons 775 and 801, in any examined case.
Discussion
In the present study, pyloric and duodenal adenomas developed in elderly cats with a mean age of 10 years, which is consistent with previous findings. 6,18 No sex or breed predisposition was observed. Similar to previous studies, anorexia and vomiting frequently occurred. 6,18 In the present study, these clinical signs improved after polyp resection in 6/7 cases; therefore, anorexia and vomiting appear to be associated with pyloric and duodenal adenomas in cats. However, improvement of clinical signs was not observed in case 4, and intestinal lymphoma likely contributed to the signs in this case. Given the match of location, age and clinical signs, feline pyloric, and duodenal adenoma is presumably the same condition as adenomatous polyp of the feline pylorus and duodenum previously reported. 6,18
The distributions of AB-PAS, PCS, CD10, CDX2, CK7, CK19, and CK20 in normal pyloric and duodenal tissue were similar to those reported in previous human and feline studies with minor variations. 4,7,9,14,20,21,26 –30 In the present study, immunolabeling for CK19, which has been detected in intestinal submucosal gland in humans, 29 was not observed in intestinal submucosal gland in cats.
In humans, gastric- and intestinal-type tumors in the alimentary tract are characterized by the presence of neutral mucin or goblet/Paneth cells, respectively. 16 Gastric-type adenomas have been further divided into foveolar-, pyloric gland-, and oxyntic gland-type adenoma based on histopathology, mucin production, and peptide profiles. 8 In pyloric gland adenoma, the neoplastic cells have a ground-glass appearance and positive staining for PCS, similar to the of pyloric gland-type mucin that is generally produced by the pyloric gland, mucous neck cells, and intestinal submucosal gland. 8 In the present study, the majority of tumor cells in feline pyloric and duodenal adenomas had ground-glass cytoplasm, and histochemical and immunohistochemical phenotypes were similar to those of mucous neck cells and cells of the pyloric gland. In humans, pyloric gland adenoma commonly develops in the stomach and duodenum. 2,3,34 The present results demonstrated that the location, histopathology, and histochemical and immunohistochemical profiles of feline pyloric and duodenal adenomas were similar to those of human pyloric gland adenoma, suggesting that feline pyloric and duodenal adenomas are the counterpart of human pyloric gland adenoma.
Immunolabeling for β-catenin is normally detected at the cell membrane, and nuclear β-catenin localization indicates the activation of Wnt/β-catenin signaling. 17 The aberrant activation of Wnt/β-catenin signaling contributes to tumorigenesis in many organs, 5 and the nuclear accumulation of β-catenin is frequently observed in human gastrointestinal tumors. 25,31 In the present study, weak nuclear accumulation of β-catenin was observed in 2/8 cases, which is a lower incidence than the incidence of 60% previously reported in feline colorectal tumors. 33 This result is consistent with human duodenal adenoma, in which nuclear β-catenin is less frequently detected in the gastric type than in the intestinal type. 24 Therefore, excessive Wnt/β-catenin signaling may not be involved in the pathogenesis of feline pyloric and duodenal adenomas, in contrast to duodenal/intestinal adenomas in humans.
Immunohistochemical staining for p53 is used as a surrogate for a mutational analysis of TP53, and the strong and diffuse immunostaining of p53 suggests TP53 gene mutations. 22 The P53 protein has been detected in various feline cancers by immunohistochemistry; however, diffuse immunolabeling for p53 has rarely been observed in feline colorectal tumors. 23,33 In humans, a previous study showed that approximately 70% of pyloric gland adenomas were immunopositive for p53 and the incidence of p53 expression was higher in cases with transition to adenocarcinoma. 35 In accordance with previous findings on feline colorectal cancer, neoplastic cells in feline pyloric and duodenal adenoma were negative for p53 in the present study. This result suggests that a p53 malfunction does not play a critical role in the tumorigenesis of feline pyloric and duodenal adenomas, in contrast to humans.
The KRAS gene regulates many crucial cellular processes, such as cell proliferation, differentiation, and survival in response to extracellular stimuli through the Ras signaling pathway. KRAS mutations, particularly at codons 12 and 13, are frequently involved in human gastrointestinal tumors, including pyloric gland adenoma. 1,3,19,32,34 KRAS mutations at codon 13 have also been found in feline colorectal adenocarcinoma and are suspected to be involved in tumor development. 33 However, in the present study, there were no KRAS mutations at this region in the 4 examined cases, and its role to the pathogenesis of feline pyloric and duodenal adenomas was not supported.
The GNAS gene encodes the guanine nucleotide-binding protein G α-subunit, and its activating mutations have been detected in tumors of various organs in humans, such as the pancreas, pituitary gland, bone, and gastrointestinal tract. 13 In the human gastrointestinal tract, GNAS mutations are specifically found in pyloric gland adenoma, but not in other types of tumors, and are considered to be characteristic of pyloric gland adenoma. 19,34 More than 90% of mutations in the human GNAS gene involve codons 844 and 870, which correspond to codons 201 and 227 in the isoform Gnas-1. 32 Protein sequence alignment showed >99% similarity between the human GNAS isoform Gnas-1 and the feline GNAS isoform; therefore, mutational hotspots in the human GNAS gene are equivalent to codons 775 and 801 in the feline GNAS gene. 36 However, no GNAS mutations were detected in feline pyloric and duodenal adenomas in the present study. Based on the absence of detectable KRAS mutations in the examined cases, feline pyloric and duodenal adenoma appears to have a different molecular pathogenesis than that of pyloric gland adenoma in humans. However, the mechanisms underlying the tumorigenesis of feline pyloric and duodenal adenoma were not clarified in the present study.
In conclusion, the present study revealed the histological and immunohistochemical features of feline pyloric and duodenal adenoma. Tumor cells were characterized by eosinophilic, ground-glass cytoplasm and were histochemically and immunohistochemically similar to mucous neck cells or pyloric gland cells. The nuclear accumulation of β-catenin was limited, and p53 immunolabeling was not detected. Neither KRAS nor GNAS mutations were found. Feline pyloric and duodenal adenomas were histologically similar to human pyloric gland adenomas, whereas its molecular features were not. Further investigations are needed to elucidate the pathogenesis of feline pyloric and duodenal adenoma.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
