Abstract
Urinary bladder diseases are common in dogs and cats; however, there is little published work on urinary bladder disease in Australian pets. We identified pathology records of Australian dogs and cats with urinary bladder tissue submitted to the University of Queensland Veterinary Laboratory Service during 1994–2016 (n = 320). We described the proportion of bladder diseases in dogs and cats, and applied the less-commonly used logistic regression procedure to quantify associations between signalment variables and disease diagnosis that were evident using descriptive statistics alone. After preliminary analysis, both species were combined because of similar results. Spayed/castrated animals were 74% less likely to be diagnosed with cystitis compared with intact animals. Animals 4–11 y old were also at lower risk of being diagnosed with cystitis compared with younger or older animals. Male animals were at increased risk of neoplasia compared to females, which contrasts with reports from North America and Europe. There was increased risk for developing neoplasia with progressive age, with up to 20 times higher odds in the > 11-y age group. Logistic regression modeling provided unique insight into proportionate morbidity of urinary bladder diseases in Australian dogs and cats.
Introduction
Bladder diseases may occur in up to 8% of cats evaluated at veterinary teaching hospitals or clinics in the United States 20 ; ~14% of North American dogs are afflicted with a urinary tract infection (UTI) during their lifetime. 21 Urinary bladder neoplasms account for 1.5–2% of all canine neoplasms; the proportionate morbidity (or prevalence) of bladder neoplasia in cats is much lower. 8
Importantly, qualitative and quantitative data analysis techniques have not been applied previously to companion animal pathology or urinary bladder diseases in an Australian context, to our knowledge. Logistic regression is a statistical method used to explain the relationship between data categories and outcomes. 11 Logistic regression has been applied occasionally in veterinary pathology5,13 but such methodology is not comparable to our study. In addition, information on Australian dogs and cats will provide a useful comparison to studies from other countries and will enhance awareness by Australian veterinarians and pet owners of potential risk factors for bladder wall diseases.
Urinary bladder disorders can be grouped into 4 broad categories: neoplastic, infectious, and cystitis with or without uroliths. 8 Animal factors may be associated with the major categories of bladder disorders in dogs and cats; bitches aged > 7 y old 30 and queens > 10 y old are at higher risk for bacterial UTIs. 23 Non-infectious cystitis (feline idiopathic cystitis, FIC) has been associated with young-to-middle aged cats, 20 and male cats are overrepresented. 3 Bladder neoplasia is more common in older male cats 33 and in older spayed bitches, with Scottish Terriers overrepresented. 19
Our first aim was to determine the proportion of urinary bladder diseases in cats and dogs that had urinary bladder tissue submitted to the University of Queensland Veterinary Laboratory Service (UQ-VLS) between January 1994 and March 2016. Our second aim was to quantify the associations of species, breed, age, sex, and neuter status as putative risk factors for urinary bladder diseases in dogs and cats.
Methods
Data collection and case definitions
In our retrospective medical records review, we extracted data from the UQ-VLS pathology archive for all dogs and cats with urinary bladder tissue submitted between January 1994 and March 2016. Case records were obtained via a search inquiry of the UQ-VLS pathology database for canine and feline pathology reports containing at least one of the following terms: bladder, cystitis, transitional cell, urinary, or urothelial (Fig. 1). The terms “bladder” and “urinary” alone were deemed insufficient given that UTI is often termed “cystitis” and bladder neoplasia may be recorded as transitional cell carcinoma or urothelial (cell) carcinoma without mention of “bladder” or “urinary”. Pathology records were excluded if they only mentioned gallbladder (not urinary bladder) or if the animal was < 6 mo old; only mature bladder wall tissue was included. Depending on age, immature bladder wall can have umbilical artery or urachal remnants. 24 Records were also excluded when urinary bladder had been discussed in the gross section of the pathology report, but no histology had been taken, and when no diagnosis could be made as a result of poor sample size or quality. Tissue collection had been undertaken for a variety of reasons (Suppl. Table 1).
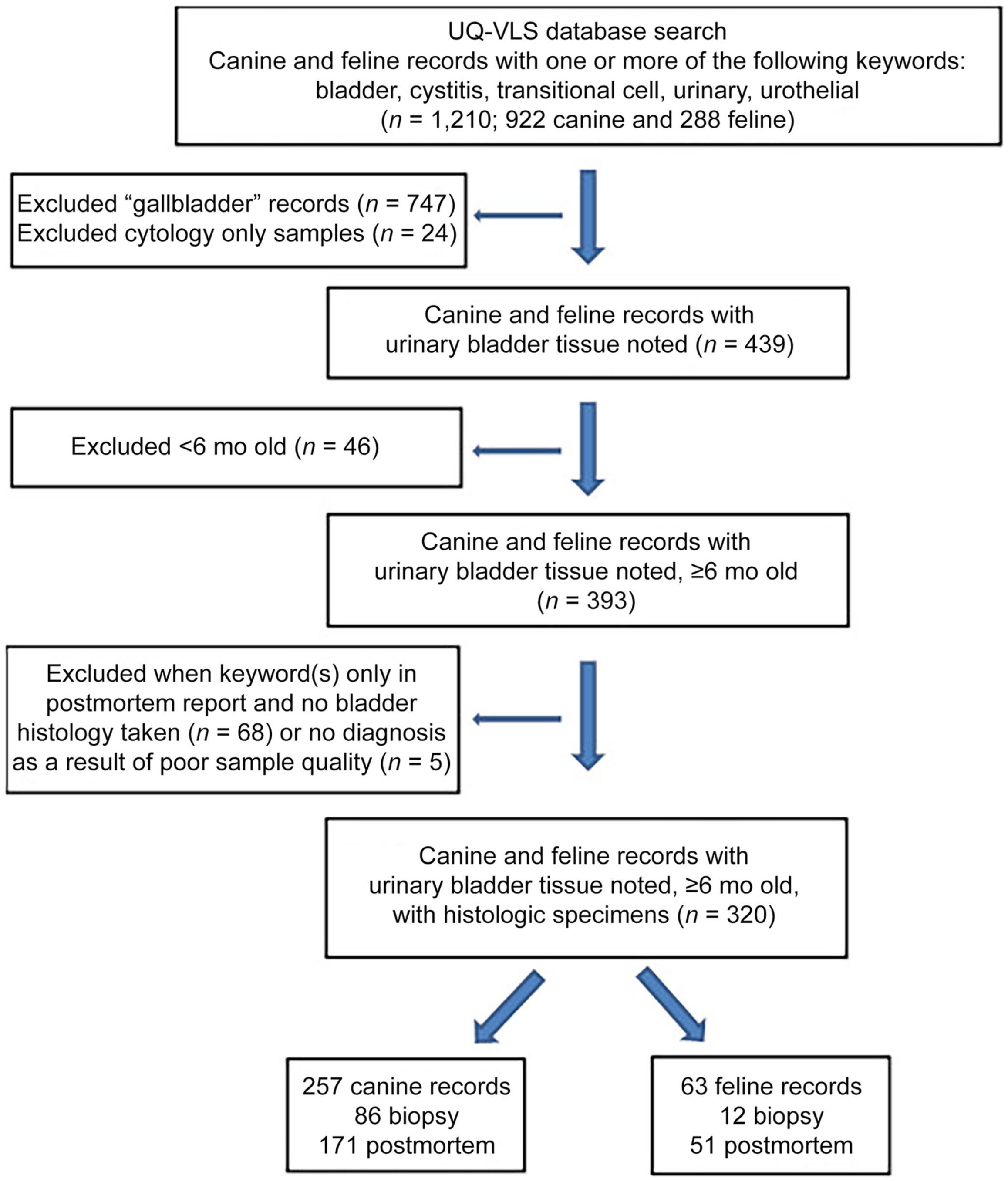
Flow chart for selection of pathology records of dogs and cats with urinary bladder tissue submitted to the University of Queensland Veterinary Laboratory Service (UQ-VLS), January 1994–March 2016.
The following information was exported from the UQ-VLS database: case reference code; species (canine or feline); pathology report, including clinical history (if submitted); date of pathology report; animal date of birth; animal breed; and animal sex and neuter status (e.g., spayed female = SF, intact male = M). Dogs were grouped into gundog, hound, pastoral, terrier, toy, utility, and working dog breed categories based on The Kennel Club (UK) breed categories (https://www.thekennelclub.org.uk/). The gundog category included sporting breeds such as Labrador Retrievers, Springer Spaniels, Hungarian Vizslas, German Shorthaired Pointers, and Weimaraners; the hound category included Wolfhounds, Dachshunds, Beagles, Whippets, Greyhounds, and Rhodesian Ridgebacks; pastoral breeds included herding dogs such as Australian Cattle Dogs, Australian Kelpies, Collies, Shetland Sheepdogs, and Australian Shepherds; terrier dogs included Bull Terriers, Fox Terriers, and Staffordshire Terriers; toy breeds included Bichon Frise, Cavalier King Charles Spaniels, Maltese, Pomeranians, Papillons, and Pugs; the utility category included non-sporting breeds such as Bulldogs, Poodles, Schnauzers, and Shih Tzus; the working dog category included Boxers, Bull Mastiffs, Dobermans, Rottweilers, and German Shepherds. Cats were grouped as Burmese, domestic (short-, medium-, and long-hair varieties), foreign cat (Devon Rex, Korat, and Bengal breeds), Persian cats, or semi-longhair (2 cats; 1 Himalayan and 1 Somali cat), based on the Governing Council of the Cat Fancy (https://www.gccfcats.org/Cat-Breeds). The patient’s breed was recorded as unknown if not specified on the pathology report.
Diagnosis was classified as the primary diagnosis assigned to bladder tissue by the original pathologist ranked in the order neoplasia, urolithiasis, cystitis (infectious or non-infectious), normal bladder wall, or other diagnoses. Infectious bladder diseases were combined with cystitis without uroliths (most commonly FIC cases) into the “cystitis” category. Records were assigned to the urolithiasis category if uroliths were mentioned in the history regarding the current clinical presentation or cause of death or reason for euthanasia. When uroliths were present in the history or at autopsy, the original histologic diagnosis was often “cystitis,” given that bladder wall inflammation can occur in these cases. However, we separated these records into their own “urolithiasis” group because inflammation was likely attributable to the physical trauma of the stone in addition to potential bacterial infection. Given the retrospective and histologic nature of our study, and variations in case management, many cases lacked urine culture results, making it impossible to categorize cases as having or not having bacterial cystitis.
If neoplasia of any type had been identified in the bladder wall, records were placed into the “neoplasia” category (Suppl. Table 2), even if there was concurrent inflammation or urolithiasis because we considered neoplasia to be the predominant pathologic process. Records were categorized as “normal” if the bladder was reported as normal or was recorded as being sampled for histology but not mentioned in the diagnosis section. We assumed that the absence of comments regarding the urinary bladder, in the presence of comments on other diseased tissues, meant that no abnormalities were found in the bladder tissue. All other diagnoses were grouped together in the “other” category, including peritonitis, traumatic bladder rupture, and healed scar tissue (Suppl. Table 3).
For statistical analysis, infectious and non-infectious cystitis were grouped together given the similar pathologic processes and the logistic limitation of being unable to confirm bacterial cystitis in these retrospective cases. Retrieved pathology records were interrogated by E. Jones and R. Allavena. Data were managed in Excel (Microsoft).
Descriptive statistics and proportionate morbidities
Descriptive statistics (median, range, and frequency, as appropriate) were determined using R (https://www.r-project.org/). First, associations between species and each of age, diagnosis, sex, neuter status, breed, and sampling method were assessed using a chi-squared or Fisher exact test (more accurate test when sample size is small) for categorical variables, or Wilcoxon rank-sum test to evaluate the equality of the medians for continuous variables.
We used proportionate “bladder disease” morbidity as the measure of risk for our study, and Poisson distribution was assumed. Proportionate morbidity (PM, expressed as a percentage) was deemed an appropriate measure of risk because the denominator in this dataset is a group of pathology records that had bladder tissue processed through the UQ-VLS, which do not necessarily represent the general population of pet dogs and cats in which bladder diseases could potentially occur. 9 PM for each bladder disease outcome in dogs or cats (i.e., cystitis, neoplasia, and urolithiasis) was calculated by dividing the total number of cases for that outcome by the total number of canine or feline cases in the dataset (i.e., cystitis, urolithiasis, neoplasia, and normal combined). “Other” diagnoses were not included in the PM calculation. The 95% confidence interval (CI) for PM was estimated using exact binomial distribution. Animal age was separated into 4 categories based on the mean overall age, which was similar for dogs and cats (Table 1). Animals were grouped by age into < 4 y, 4 to ≤ 8 y, 8 to ≤ 11 y, and > 11 y. PM ratios (PMR) were used to describe the proportion of each disease, and to evaluate associations between disease outcomes and animal factors based on proportionate morbidities. PMRs and their 95% CIs were calculated based on the Poisson distribution.
Summary data for canine and feline records with urinary bladder tissue submitted to UQ-VLS, January 1994–March 2016. Counts are for each variable; totals are 257 canine and 63 feline records. The p-value reports the overall difference between canine and feline cases for each variable.

NA = not applicable; UQ-VLS = University of Queensland Veterinary Laboratory Service. Breeds could not be compared between cats and dogs given that each species has its own breeds. Numbers in parentheses are percentages, unless otherwise noted.
Multivariable simple logistic regression modeling
A multivariable logistic regression model with binomial link functionality was built in R to explore the association between potential explanatory variables and each of the disease outcomes (cystitis, neoplasia, or urolithiasis). “Other” diagnoses were not included in the logistic regression models. The dataset used for the logistic regression models comprised the following potential explanatory variables: a) species (categorical variable with 2 categories: canine, feline); b) age (separated into 4 categories: < 4 y, 4 ≤ 8 y, 8 ≤ 11 y, > 11 y); c) sex (categorical variable with 2 categories: female, male); d) neuter status (categorical variable with 2 categories: spayed, or castrated, and intact); e) breed (categorical variable with 5 categories for cats: domestic, foreign, Burmese, Persian, semi-longhair; and 7 categories for dogs: pastoral, toy dog, utility, terrier, hound, gundog, working dog); and f) biopsy (categorical variable with 2 categories: “yes” for biopsy sampling, “no” for postmortem sampling).
First, the association between each potential explanatory variable and each outcome was examined using univariable logistic regression models. The “best fit” of the explanatory variable (continuous or categorical) was determined by graphical assessment of the relationship between the log odds of the outcome by categories of each explanatory variable. 11 A likelihood ratio test p ≤ 0.25 from the univariable analysis was used as a criterion to screen the explanatory variable for entry into the multivariable logistic regression model, with variables selected by a forward stepwise elimination procedure. All variables eliminated in the multivariable model were reintroduced in the final model, one at a time, in a search for statistical significance at alpha level ≤ 0.05. Variables were retained in the multivariable model if the likelihood ratio test p was at an alpha ≤ 0.05 when establishing the model involving main effects, 15 or when an excluded variable (likelihood ratio test p > 0.05) included in the final multivariable model changed covariate estimates by 20% and was therefore declared a confounder. Species and biopsy variables were forced into the final model once it was stable. Overdispersion (or variance inflation factor; the ratio of the Pearson chi-squared goodness-of-fit statistic to its degrees of freedom) was estimated. A chi-squared test was then used to test for overdispersion in the data at p ≤ 0.05. Overdispersion was declared, and adjustment was required, if the chi-squared p was ≤ 0.05. The overall fit of the final model was evaluated by the Hosmer–Lemeshow goodness-of-fit statistic. 15 Further testing, including the calculation of leverage and delta-betas, was used to identify any outliers or highly influential observations using the influence.ME package 25 in R.
Results
Data summary
Our UQ-VLS database search yielded 320 records for dogs and cats that had urinary bladder wall histology fitting the inclusion criteria (Fig. 1, Table 1). There were 257 canine submissions, with bladder diagnoses comprised of 85 (33%) cystitis cases, 66 (25%) neoplasia cases, 51 (20%) normal bladders, 20 (8%) urolithiasis cases, and 35 (14%) other diagnoses. There were 63 feline submissions with urinary bladder diagnoses comprised of 24 (38%) cystitis, 17 (27%) normal bladders, 7 (11%) neoplasia, 4 (6%) urolithiasis, and 11 (18%) other diagnoses. There was a significant difference in the proportions of neutered and intact animals between dogs and cats (p = 0.05).
There were 98 biopsy (86 dogs and 12 cats) and 222 postmortem records (171 dogs and 51 cats). Dogs accounted for 257 of 320 records (80% of the entire dataset; 88% of the biopsy records, and 77% of the postmortem records).
Signalment descriptive data
For 50 (16%) records, the animal age could not be ascertained: 37 dogs (14% of canine cases) and 13 cats (21% of feline cases). The means for age were not significantly different between dogs and cats. Both dogs and cats had a mean age of < 8 y (7.8 y and 7.2 y, respectively). The median age was 8 y for dogs and 5 y for cats (Table 1). Breed was recorded for 203 of 257 (79%) dogs and 52 of 63 (83%) cats. The most common breed groups were pastoral breed dogs (19%; such as Australian Cattle Dogs and German Shepherd) and domestic cats (60%).
Of the 257 dogs in the dataset, 115 (45%) were male, 124 (48%) were female, and 18 (7%) did not have sex recorded. For cats, there were 36 (57%) male, 25 (40%) female, and 2 (3%) that did not have sex recorded. The proportions of each sex were not statistically different between dogs and cats (p = 0.17).
Seventy-one percent (n = 228) of all records had some form of clinical history submitted with the tissue or body: 185 (72%) dogs and 43 (68%) cats. Complete signalment information comprising animal age, sex, neuter status, and breed was recorded in only 130 (41%) animals: 99 (39%) dogs and 31 (49%) cats. All animals with complete signalment information had clinical historical information recorded as well.
Proportionate morbidity and proportionate morbidity ratios
For neoplasia, cats had a PMR of 0.45 (95% CI = 0.21–0.99, p = 0.05) compared to dogs (Suppl. Table 4). For dogs and cats combined, there was a marked association between age and neoplasia, with increasing PMRs for neoplasia in each progressive age category (Suppl. Table 4). The reference group was < 4 y old. The 4 ≤ 8-y-old group had a PMR of 8.59 (95% CI = 1.97–37.35, p < 0.01), the 8 ≤ 11-y-old group had a PMR of 9.25 (95% CI = 2.15–39.88, p < 0.01), and the > 11-y-old group had a PMR of 17.62 (95% CI = 4.20–73.97, p < 0.01).
Cases of bladder neoplasia were ~3 times more likely (PMR = 2.7, 95% CI = 1.70–4.27, p < 0.01) to have had the tissue sample obtained via biopsy than via postmortem examination. Sex was documented in 61 (92%) neoplasia records and, in the PMR calculations, was not statistically significantly associated with neoplasia. Of dogs with neoplasia outcome and sex recorded (n = 61), the proportion of males compared to females was the same for all neoplasms (57% male, 43% female) as well as when evaluating urothelial carcinoma (UC) alone (56% male, 44% female). Cats with neoplasia and sex recorded (n = 7) had a proportion of 28% male and 72% female across all neoplasms, and 25% male, 75% female for UC alone.
There were no significant associations between diagnosis and species, age, sex, or whether the sample was obtained via autopsy or biopsy for the cystitis outcome (Suppl. Table 5) or the urolithiasis outcome (Suppl. Table 6).
Multivariable models
Animal species (dog or cat) was not a statistically significant explanatory variable; therefore, a parsimonious modeling approach was followed in which dogs and cats were combined. The small sample size for cats would have made it difficult to obtain meaningful results, whereas a larger combined dataset would be more likely to provide this. Age, sex, and undergoing bladder wall biopsy influenced the odds of being diagnosed with neoplasia. After controlling for the effect of other variables in the model (Table 2), animals ≥4 y old were at increased risk for neoplasia (p < 0.01). Dogs and cats combined had a 6.8 times higher risk of neoplasia at 4–8 y old (odds ratio [OR] = 6.84, 95% CI = 1.76–45.38, p = 0.01), 13 times the risk at 8–11 y old (OR = 13.04, 95% CI = 3.38–86.51, p < 0.01), and 20 times the risk of neoplasia when > 11 y old (OR = 20.63, 95% CI = 5.58–134.36, p < 0.01). Male animals were almost twice as likely to be diagnosed with neoplasia compared to females (OR = 1.84, 95% CI = 1.00–3.46, p = 0.05). In addition, dogs or cats having a biopsy procedure were 3 times more likely to be diagnosed with neoplasia than those undergoing postmortem examination (OR = 3.29, 95% CI = 1.76–6.28, p < 0.01).
Odds ratios of final multivariable logistic regression models fitted separately on records diagnosed with cystitis (n = 109), neoplasia (n = 73), and urolithiasis (n = 24) for canine and feline pathology records containing bladder histology submitted to UQ-VLS, January 1994–March 2016.

UQ-VLS = University of Queensland Veterinary Laboratory Service. Numbers in parentheses are 95% confidence intervals.
Statistical significance interpreted at p ≤ 0.05. The p-values are derived from the likelihood ratio test comparing the reduced model with saturated model for each variable. The p-values show the statistical significance of the explanatory variable to overall model fit.
Neuter status and age both influenced the odds of cystitis diagnosis. After controlling for the effect of other variables in the model (Table 2), neutered dogs and cats were 78% less likely (OR = 0.22, 95% CI = 0.07–1.09, p = 0.01) to be diagnosed with cystitis compared with intact animals. Animals 4–8 y old had a reduced risk of cystitis (OR = 0.25, 95% CI = 0.08–0.71, p < 0.01) compared to all other age categories combined. No significant associations were found for cases with a diagnosis of urolithiasis in the multivariable models.
Discussion
Multivariable modeling revealed some associations that were not evident when evaluating the PMs and PMRs, highlighting the utility of logistic regression modeling in adjusting for confounding variables. Disease diagnosis, age, and sex were not significantly different between dogs and cats in the initial analysis; therefore, we combined dogs and cats in the modeling process. The mean age was similar for both dogs and cats; therefore, the same age groupings were used for both species. The median age was lower for cats, indicating a larger number of young cats than young dogs, with the mean likely increased by a small number of very old cats.
Dogs had more than double the odds of the overall dataset population for bladder neoplasia, consistent with reports of bladder neoplasia occurring much more commonly in dogs than in cats. 18 There were increased odds of neoplasia with increasing age, with up to 20 times higher odds in dogs and cats > 11 y old, consistent with the knowledge that older animals are at increased risk for bladder neoplasia. 33 Our result of increased odds for neoplasia with increasing age is unlikely to have been affected by the combination of canine and feline records, given the very low number of cat neoplasia cases in our dataset.
Population-level literature on bladder neoplasia typically includes only UC given that this is the most common neoplasm of the urinary bladder in these species. 18 Our neoplasia category includes all types of bladder neoplasia; however, the majority were UC (Suppl. Table 2), thus our findings can be compared to other reports.19,33
In dogs and cats combined, bladder neoplasia was significantly more common in males than females. This finding was unique to the logistic regression model, highlighting the utility of this method in considering any interacting or confounding variables. This finding was the same when evaluating only canine records. Increased odds of bladder neoplasia in males contrasts the literature on canine UC, in which females are at 1.7–1.95 times higher risk than males 17 ; however, the female-to-male ratio in our study did level out in the previously reported higher risk breeds (Shetland Sheepdogs, West Highland White Terriers, and Beagles). 18 The canine male-to-female ratio was comparable when evaluating all neoplasms together compared to evaluating UC alone. This finding may be attributable to a different sex distribution of urinary bladder neoplasms in Australian dogs (given that current literature is primarily North American or European), or perhaps differences in the disease process or socioeconomic factors leading to male dog bladders being sampled more frequently than females and skewing our dataset. For cats, there were more females than males in the neoplasia group. Bladder cancer is rare in cats; thus, literature is sparse, and there have been conflicting reports of sex association for this disease in cats. 6 Our small sample size for cats impedes evaluation of sex association for feline bladder neoplasia. The small sample size for cats in our study is thought to be a sampling bias as a result of a combination of factors: fewer cats undergoing postmortem examination at our institution, the difficulty of obtaining bladder biopsies from cats, particularly males, 7 as well as a lower proportion of owned cats compared to dogs in Australia; 27% of households have a pet cat, 40% of households have a pet dog. 1 The literature reports neutered dogs being at higher risk of bladder neoplasia 18 ; however, our PMRs and multivariable modeling showed no association between neoplasia and neuter status. We saw a trend toward increased risk for neoplasia in pastoral and terrier breeds; however, these changes were not statistically significant.
For the cystitis outcome, it is important to note that our definition of cystitis (i.e., histologic evidence of inflammation in the bladder wall diagnosed by the reporting pathologist) is different from the commonly used clinical definition of cystitis, which is lower urinary tract clinical signs, often with positive urine bacterial culture. The cystitis disease category in our study could not distinguish between bacterial infection causing cystitis, and FIC, given the absence of bacterial culture results for most cases. In addition, positive bacterial culture does not necessarily indicate colonization or infection of the urinary tract, which further complicates classification.27,31 When comparing our cystitis cases to the literature, we are forced to look to published work on canine bacterial cystitis for dogs and both bacterial cystitis and FIC for cats because both of these diseases are likely to be represented in our feline “cystitis” disease category.
Bacterial cystitis may lead to urolithiasis, as occurs with struvite stone formation secondary to infection by urease-producing bacteria.26,28 Conversely, uroliths can impair bladder defenses, facilitating the development of bacterial cystitis. 26 Up to 50% of canine uroliths are thought to be infection related, 29 a phenomenon that is much more common in dogs than in cats. 16 The small number of cases in our study with concurrent bacterial cystitis and urolithiasis (n = 4, 20% of urolithiasis records) were categorized with the urolithiasis rather than cystitis cases for a number of reasons. First, the urolithiasis category in our dataset is likely to already contain some cases with both urolithiasis and undocumented bacterial UTI. Second, the cystitis disease group is already a heterogeneous combination of cases, with bacterial or fungal infection confirmed by urine culture, cases with suspected bacterial infection without bacterial culture, and cases with suspected FIC (no infectious organism involved). Thus, the decision was made to not complicate the category further by adding urolithiasis and/or bacterial cystitis cases to this group. Finally, the mechanical damage caused by uroliths typically leads to a different inflammatory profile than bacterial cystitis alone; therefore, these cases were deemed more similar histologically to the urolithiasis cases.
Our analysis found no difference between cystitis and species or sex, whereas the literature reports bitches to be at increased risk for bacteriuria.14,23 Our results may be attributable to the unavoidable combining of infectious cystitis and feline non-infectious cystitis; however, our multivariable model showed that even when species was accounted for, there was no difference in the risk of cystitis between males and females. Spayed/neutered animals were found to have a significantly decreased risk for cystitis, which is consistent with some published work on canine bacteriuria. 14 There is sparse published evidence on neuter status as a risk factor for canine bacterial cystitis; castrated males seemed to be at increased risk of bacterial cystitis as a result of exogenous steroid administration in one study 8 ; another study found castrated male dogs to have a higher risk of positive urine culture compared to intact dogs. 14
There were decreased odds of cystitis in animals 4–8 y old, which is consistent with published material. Increased odds for younger and older age groups makes sense biologically given that FIC and anatomical abnormalities such as ectopic ureters are more common in younger cats.4,12,32 UTI risk is higher in dogs and cats > 7 y old.20,22 The combined age groupings for dogs and cats may have resulted in a large < 4-y-old group for cats given the lower median age for cats. However, the majority of cystitis cases came from animals < 2 y old for both dogs and cats; therefore, a different age grouping system for cats is unlikely to have changed this result. There was a trend toward a higher risk for cystitis in pastoral, hound, and toy breeds; however, this was not statistically significant.
We found no significant associations with the urolithiasis outcome in the PMs, PMRs, or the multivariable model, potentially because of the limited case numbers. No significant associations with any bladder disease were detected for breed. Our sample size was limited for cats, preventing adequate analysis of breed associations.
Most veterinary disease studies evaluate the proportions of diseases in large or university veterinary hospital submissions.10,30 In contrast, our dataset was of animals that were presented to veterinarians at the UQ teaching hospital and general practices in southeast Queensland, Australia, that then underwent bladder biopsy, or had bladder tissue sampled during autopsy that was routed through a single diagnostic service. For this reason, PM was chosen as a measure of prevalence in our study population, given that our population may not necessarily be representative of the general Queensland or Australian dog and cat population. The uniqueness of our dataset means that we cannot make direct comparisons to other population-level studies; however, our information may still be useful for client and veterinarian awareness. The acquisition of case records was performed via a database search and then manual data extraction; however, there is increasing literature on the utility of machine learning for extracting information from veterinary pathology reports. 2 Therefore, this method could be utilized in future studies to more easily and more accurately extract animal signalment and disease information.
The retrospective nature of our study does have some inherent elements of recall bias and information bias that could affect external validity of our study. A major finding from our study was the lack of complete patient signalment data in 59% of pathology records, including absence of breed information in 20% of records. The absence of this information likely impaired the adjustment of records by breed, and therefore evaluation of breed as a risk factor for cystitis, neoplasia, or urolithiasis. Missing data in our dataset were likely to have had a bidirectional impact on the proportionate morbidities—some may be overestimated whereas others were underestimated. A further limitation is the lack of standardization of pathology reporting and the potential for missed records because of misspelling, or the disease diagnosis containing slightly different terms to the ones used in our search, although we endeavored to capture these variations.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211004008 – Supplemental material for Association between case signalment and disease diagnosis in urinary bladder disease in Australian cats and dogs
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211004008 for Association between case signalment and disease diagnosis in urinary bladder disease in Australian cats and dogs by Emily Jones, John Alawneh, Mary Thompson and Rachel Allavena in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We acknowledge the support of Drs. Chiara Palmieri and Karen Jackson during our study. We appreciate the University of Queensland Gatton Library staff for their support with Endnote referencing software and the literature review. We also thank the UQ-VLS team for their help in acquiring the veterinary pathology records.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We acknowledge the John and Mary Kibble bequest for companion animal research, companion animal research funds for the University of Queensland School of Veterinary Science, and the Dr. David Galbraith Postgraduate Research Scholarship in Companion Animal Health.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
