Abstract
Oral malignant melanoma (OMM) is considered the third most common oral malignant neoplasm in cats, but its variable morphology and frequent lack of melanin pigment make it a diagnostic challenge. Twenty-two cases of cats with malignant oral neoplasms that were diagnosed as OMM or listed OMM as a suspected differential diagnosis on the biopsy report were examined using an immunohistochemistry (IHC) panel for S100, melan-A, PNL2, laminin, CD34, and pan-cytokeratin. Although OMM was suspected (n = 14) or previously diagnosed (n = 8), only 2 cases were immunohistochemically confirmed as OMM. Seven cases were classified as soft tissue sarcoma based on positive expression of CD34 or laminin, and one was classified as carcinoma based on positive expression of pan-cytokeratin. The majority of cases (n = 12) were categorized as unclassified malignant neoplasms because they did not express melan-A, PNL2, laminin, CD34, or pan-cytokeratin; however, a proportion of these did express S100 (n = 7). Long-term prognosis of all 22 cats was poor, with a median survival time of 87 days (range = 2–249 days). Cases with longer survival times (>100 days) were treated with surgery, radiation therapy, or a combination. For feline oral malignant neoplasms thought to be OMM, routine use of IHC is required for an accurate diagnosis.
Oral malignancies in the cat account for approximately 6% of all feline neoplasms. 11 The vast majority of oral neoplasms in the cat are malignant, with squamous cell carcinoma being the most common. 33 Several other neoplasms have been recognized in the oral cavity of the cat, including fibrosarcoma, malignant melanoma, osteosarcoma, lymphoma, mast cell tumor, and undifferentiated carcinoma. 33 Histologically, malignant melanoma, osteosarcoma, and fibrosarcoma may have a spindle cell morphology without further distinguishing microscopic features. These are often referred to as malignant spindle cell tumors in initial biopsy reports.
Cats with oral tumors often present with ptyalism, oral pain, bleeding, dysphagia, weight loss, reduced grooming, lethargy, and oral malodor. Clinical signs can be caused by the invasive nature of the tumor and its potential for metastasis. Regional lymph nodes tend to be initial targets, followed by distant metastasis most commonly to the lungs. 32 Soft tissue sarcoma (STS), specifically fibrosarcoma, have been documented to be locally aggressive with common recurrence after surgical removal. 20
Melanocytic neoplasms in the cat have been most commonly reported in the eye, followed by cutaneous and oral forms. 23 Location of feline melanomas correlates with survival; those of the oral cavity, lip, or nose have a much shorter median survival time compared to those of the skin or digit. 24 OMM is thought to be the most common oral malignancy in the dog, but it is rare in the cat, accounting for less than 1% of all feline oral malignant neoplasms. 5,32,33 While variation in incidence exists between dogs and cats, what seems to be consistent among the species is a shared propensity for local tumor recurrence and high risk for metastasis to regional lymph nodes and the lungs. 6,14,23,25,26,32,34
Melanocytes are derived from neuroectodermal melanoblasts and retain the ability to differentiate into multiple morphologic phenotypes which can make the diagnosis of melanoma challenging. 32 Neoplastic cells in OMM can have a spindle cell or polygonal/epithelioid appearance, or a mix of both. They may lack melanin pigment or junctional activity that if present would indicate melanoma. 31 Other morphologic variations of neoplastic melanocytes include clear-cell, signet-ring, pseudolipoblastic, rhabdoid, plasmacytoid, or balloon cell, and rarely tumors exhibit chondroid or osseous differentiation. 2,15,22,30,32 Many other neoplasms can mimic melanoma microscopically including, but not limited to, STS, carcinoma, chondrosarcoma, osteosarcoma, and lymphoma. Confirmation of the diagnosis is based on characteristic histologic features or via positive immunohistochemical labeling with antibodies that have been shown to have a high specificity for melanocytes in the absence of such features. 31 Melanomas often express S100, melan-A, and PNL2. While S100 is a commonly used immunohistochemical marker for both human and animal melanocytic neoplasms, it lacks specificity and can be expressed in STS, another common malignant neoplasm in dogs and cats. 27 Other markers such as laminin and CD34 have been used to identify STS in dogs. 17,18,30 Despite advances in immunohistochemistry (IHC), detailed studies about the sensitivity and specificity of various antibodies for differentiating feline OMM from oral STS are lacking. The purpose of the present study was to evaluate the histologic and immunohistochemical phenotype of oral malignant neoplasms in cats diagnosed with OMM or with OMM listed as a suspected differential diagnosis on the biopsy report, and to describe their clinical presentation and progression.
Materials and Methods
The University of Pennsylvania School of Veterinary Medicine (Penn Vet) Diagnostic Laboratory electronic medical records were searched from 2007 to 2016 for cats diagnosed with any of the following terms: OMM, suspected/probable OMM, oral malignant neoplasm, or oral malignant spindle cell neoplasm. Additional inclusion criteria were availability of medical records and paraffin-embedded tissue samples for immunohistochemical testing. Cases were eligible for inclusion regardless of their treatment modality. For all cases, the following clinical data was collected: breed, sex, age at diagnosis, tumor location, results of staging procedures when available, treatments, confirmed metastasis, and outcomes. Histopathologic data included mitotic count, presence of junctional activity (defined as neoplastic cells nesting within the overlying epithelium), presence of pigment (present, scant, or not present), and cellular morphology (polygonal, spindle-shaped, or mixed). Clinical data was obtained via phone interviews with the referring veterinarians when needed. Survival time was defined as the time from histopathologic diagnosis to death.
Differentiation between rostral and caudal location was made from the referring veterinarians’ medical notes or photographs of tumors. When available, tumors were classified as rostral if they affected the area of the incisor or canine teeth. Tumors affecting the area of the premolar or molar teeth were considered caudal. If photographs were not available or rostral versus caudal location was not noted in the referring veterinarians’ notes, the location was described as maxilla, mandible, or unknown.
All tissue specimens were fixed in buffered 10% formalin, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. Paraffin blocks were submitted to the Michigan State University Veterinary Diagnostic Laboratory where a single pathologist (MK) reevaluated the tissue samples, and 5-µm serial sections were immunohistochemically labeled for S100, melan-A, PNL2, laminin, and CD34 and assessed by light microscopy (Suppl. Table S1). 27,30,35 Immunohistochemical labeling for pan-cytokeratin was performed at the Penn Vet Diagnostic Laboratory and evaluated by a single pathologist (AD). IHC was performed on a Bond autostainer automated system (Leica Microsystems), using a biotin-free, polymeric alkaline phosphatase-linked antibody detection system with fast red chromogen and hematoxylin counterstain (Bond Polymer AP Red Detection Kit, Leica Microsystems) for melan-A, PNL-2, and CD34; and a biotin-free, polymeric horseradish peroxidase conjugated detection system with 3,3′-diaminobenzidine and hematoxylin counterstain (Bond Polymer HRP Detection System, Leica Microsystems) for S100 and laminin. 30 Immunoreactivity for all markers was assessed by subjectively determining the percentage of positive neoplastic cells. Neoplasms were considered positive for CD34 if at least 30% of neoplastic cells had positive labeling. 30 Immunoreactivity for melan-A, PNL2, S100, laminin, and pan-cytokeratin was assessed via the percentage of positive cells using a scoring system previously reported: a score of 0 was given if less than 5% positive cells were found, a score of 1 if 5% to 10% positive cells, a score of 2 if 11% to 50% positive cells, a score of 3 if 51% to 80% positive cells, and a score of 4 if 80% to 100% positive cells. 30 Samples scored as either 2, 3, or 4 were considered positive for that antibody. 30 Neoplasms with a score of 1 were considered positive if intraepithelial nests consisting of at least 5 clustered neoplastic cells labeled positively or if there were aggregates of at least 20 cells that labeled positively anywhere within the neoplasm. If only individual scattered positive cells were observed in neoplasms with a score of 1, the neoplasm was considered negative for that antibody. 30 Neoplasms were also considered positive for melan-A and PNL2 if intraepithelial nests consisting of at least 5 clustered neoplastic cells labeled positively or if there were aggregates of at least 20 cells that labeled positively anywhere within the neoplasm. If only individually scattered positive cells were observed, it was considered negative for that antibody. Positive controls of tissues known to express the markers (Suppl. Table S1) and negative test tissue controls (via omission of primary antibody) were performed and all were labeled as expected.
IHC labeling consistent with a diagnosis of OMM was defined as positive labeling for melan-A and/or PNL2 with the absence of CD34, laminin, and pan-cytokeratin expression. A neoplasm was considered a STS if labeling was positive for laminin or CD34 and lacked labeling for melan-A, PNL-2, and pan-cytokeratin. A neoplasm was considered a carcinoma if labeling was positive for pan-cytokeratin and negative for labeling for melan A and PNL2. The mitotic count was determined by a single pathologist (MK) as the number of mitotic figures in neoplastic cells in 10 high power fields using an ocular with a field of view measuring 0.237 mm2.
Results
The clinical results are summarized in Table 1. The median age at the time of diagnosis for the 22 cats was 11 years (range, 3 to 18 years). Seven were castrated male, 14 were spayed female, and 1 was an intact female. The predominant breed was domestic shorthair (n = 19), followed by 2 Maine coons and 1 Siamese.
Treatment, survival time, staging data, and immunohistological results for 22 cats with suspected or previously diagnosed oral malignant melanoma.
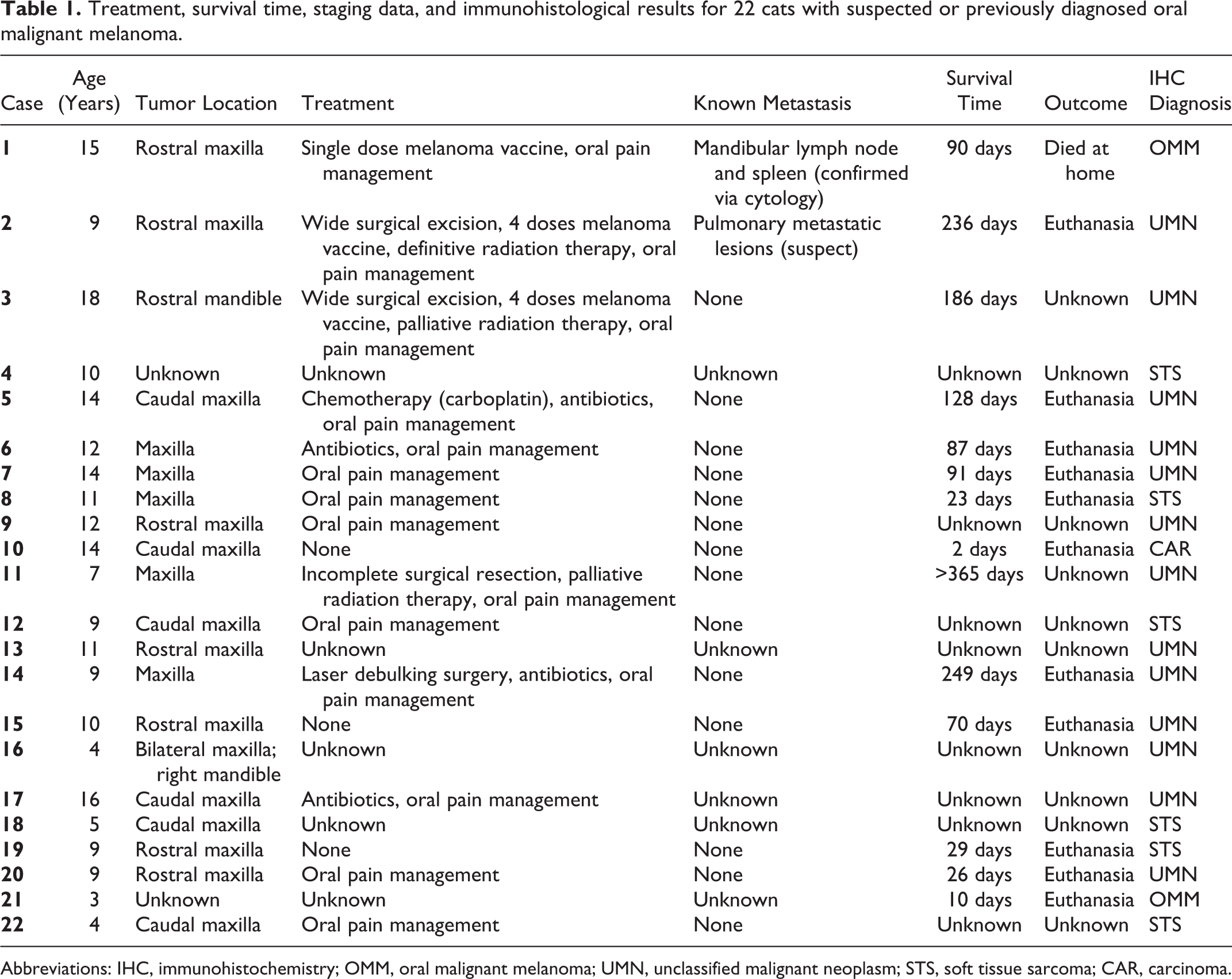
Abbreviations: IHC, immunohistochemistry; OMM, oral malignant melanoma; UMN, unclassified malignant neoplasm; STS, soft tissue sarcoma; CAR, carcinoma.
Intraoral tumor location was available for all but 2 cases (cases 4 and 21). The majority of neoplasms (18/20, 90%) were maxillary masses. The remaining 2 cases had a mandibular neoplasm and multifocal oral cavity involvement, with neoplasms in the right and left maxilla and right mandible. Neoplasms involved the gingiva, alveolar mucosa, buccal mucosa, palatal mucosa, and mucocutaneous junction.
Cats that were lost to follow-up (n = 10) were censored from median survival time determination. Median survival time was 87 days (range = 2–249 days). One cat was alive more than 365 days after the initial diagnosis but was diagnosed with local recurrence and subsequently lost to follow-up. Follow-up was available for 12 cats, of which 1 died at home and the remaining 11 were euthanized. Reasons for euthanasia included progression of the primary tumor (n = 8), poor prognosis (1), anorexia (1), and nasal discharge and dyspnea (1).
Diagnostic evaluation performed in the 22 cases included complete blood count and serum chemistry in 10 cats, intraoral dental radiography in 10 cats, thoracic radiography in 5 cats, regional lymph node cytology in 5 cats, computed tomography of the head in 3 cats, computed tomography of the thorax in 1 cat, and abdominal ultrasound in 3 cats. No cats had known metastatic disease at the time of initial diagnosis of the oral neoplasm; however, thoracic radiography or computed tomography and relevant lymph node cytology were not routinely performed in all cats. Subsequent diagnostic testing confirmed lymph node and splenic metastasis via cytology in 1 cat. Another cat had pulmonary nodules on thoracic radiography; however, cytology was not performed.
Treatments were variable. Palliative analgesia was provided in 14 cats, with medical management alone used in 9 cats. Surgery was performed on 4 cats: 2 had curative-intent excision but both failed to obtain tumor-free margins, and 2 cases underwent palliative debulking. One of the surgically treated cats was lost to follow-up, and the other 3 all had local recurrences. Radiotherapy was performed in 3 cats. The xenogeneic human DNA vaccine (Oncept, Merial) was administered to 3 cats, with 1 to 4 vaccinations given per case. One cat received chemotherapy of 3 doses of intravenous carboplatin. Treatment was unknown or not performed due to poor prognosis in 8 cats.
Histopathologic and immunohistochemical data are summarized in Table 2. Neoplasms were predominantly composed of spindle cells in 9 of 22 cats, mixed cell types (a combination of spindle and polygonal) in 9 cats, and polygonal cells in 4 cats. None of the 22 samples contained melanin pigment.
Histopathologic and immunohistochemical data for 22 cats with suspected or previously diagnosed oral malignant melanoma.
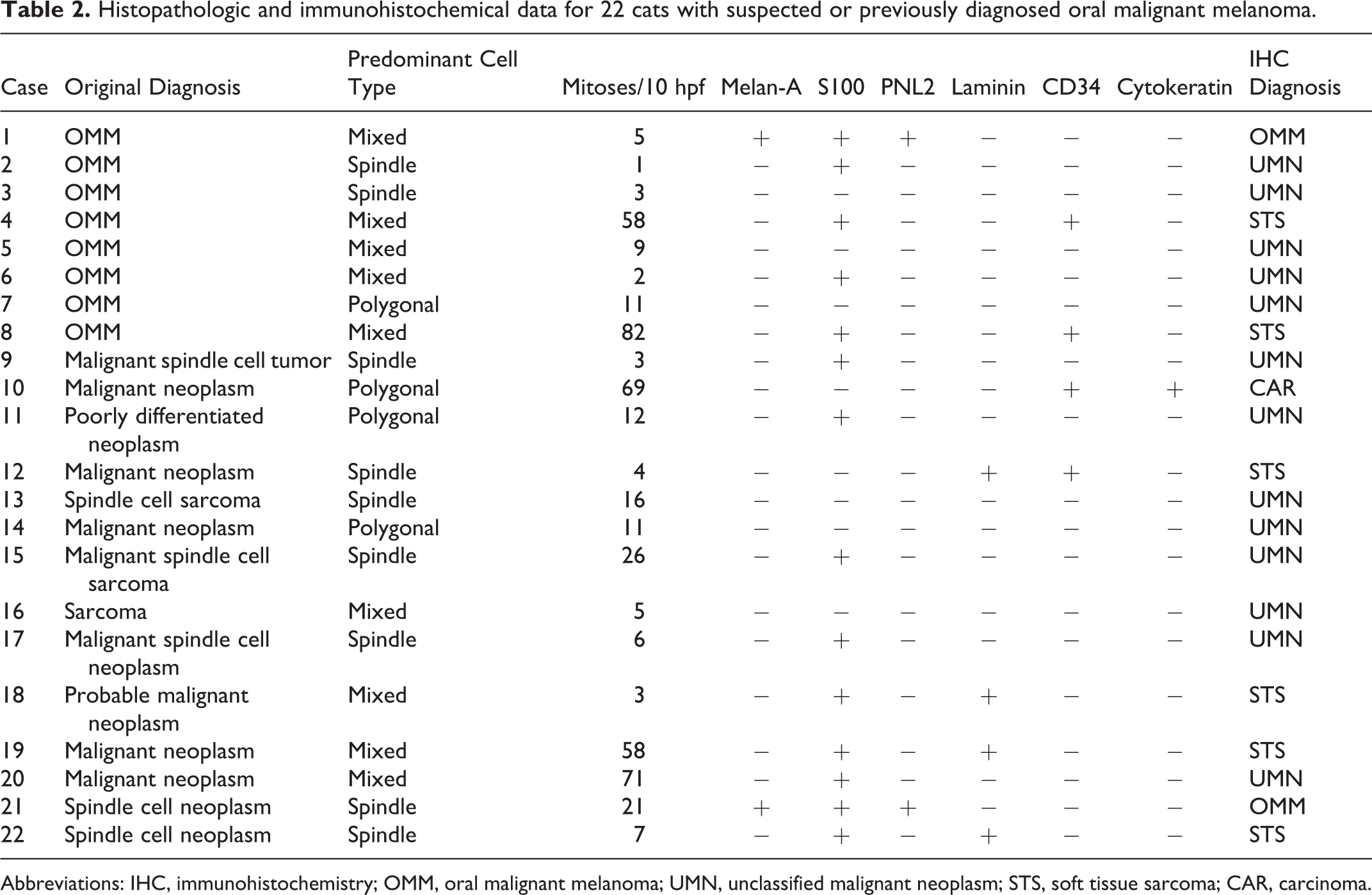
Abbreviations: IHC, immunohistochemistry; OMM, oral malignant melanoma; UMN, unclassified malignant neoplasm; STS, soft tissue sarcoma; CAR, carcinoma.
IHC labeling was consistent with a diagnosis of OMM in only 2 cats. One was originally diagnosed as OMM based on the presence of junctional activity (Fig. 1a), and it had positive immunolabeling for PNL2, melan-A, and S100 but was negative for CD34, laminin, and pan-cytokeratin. For the other OMM, an initial diagnosis of STS had been favored based on microscopic examination due to the absence of melanin pigment and junctional activity, but a diagnosis of OMM was ultimately confirmed based on immunohistochemical labeling for PNL2 and melan-A. It was also positive for S100 but negative for CD34, laminin, and pan-cytokeratin. Thus, only 1 of the 8 neoplasms originally diagnosed as OMM based on routine hematoxylin and eosin staining was confirmed to be OMM by IHC (Fig. 1b, c). Mitotic counts for the 2 cats with OMM were 5 and 21, and their overall survival times were 90 and 10 days, respectively. Metastatic disease was documented in the ipsilateral mandibular lymph node and spleen based on cytology in 1 case approximately 6 weeks after the diagnosis of oral OMM. Both were euthanized as a result of their disease, but necropsies were not performed.
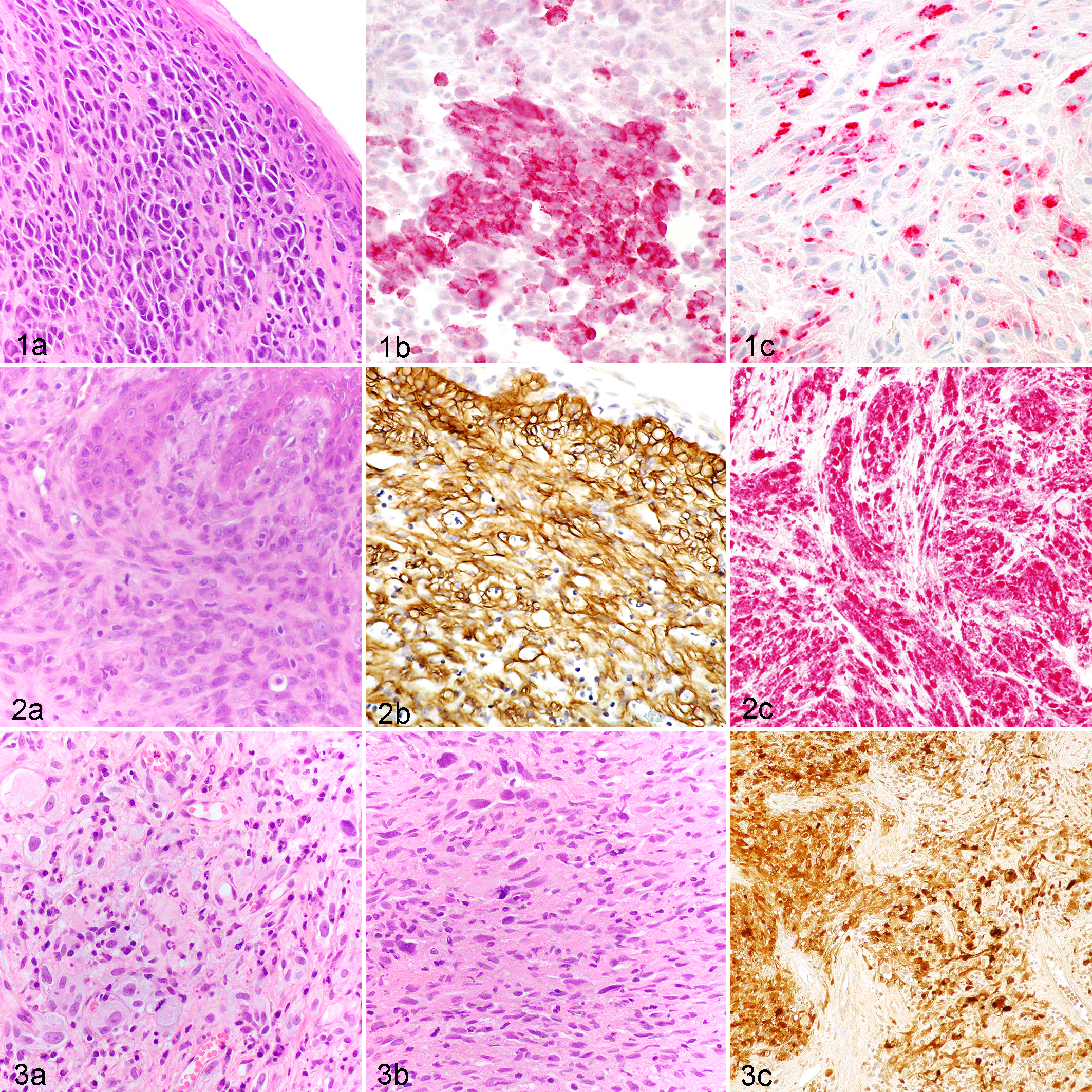
Oral malignant melanoma (OMM), oral cavity, cat. (a) Neoplastic cells are round to polygonal, arranged in sheets and nests, and exhibit epitheliotropism. Hematoxylin and eosin (HE). (b) Neoplastic cells exhibit strong cytoplasmic immunolabeling for melan-A. (c) Variably strong cytoplasmic immunolabeling for PNL2 is present.
A diagnosis of STS was based on the absence of junctional activity and melanin pigment combined with the lack of immunohistochemical labeling for melan-A, PNL2, and pan-cytokeratin, but positive labeling for CD34 or laminin (Fig. 2a–c). STS was identified in 6 cats. OMM had been originally diagnosed in 2 of these cats. Survival time was known for only 2 cats, which was 23 and 29 days. Median mitotic count was 32 (range = 3–82). Five of these neoplasms were also positive for S100, most consistent with a diagnosis of peripheral nerve sheath tumor. The 2 cats with a known outcome were euthanized; both due to progression or complications of the primary tumor. Metastatic disease was not reported, and necropsies were not performed for either of these cats.
One cat was diagnosed with a carcinoma due to positive immunolabeling with pan-cytokeratin. The mitotic count was 69. Euthanasia was performed 2 days after the biopsy results were available due to poor perceived prognosis.
The majority of cats (n = 13) were negative for melan-A, PNL2, CD34, laminin, and pan-cytokeratin. Seven of these neoplasms were S100-positive (Fig. 3a–c). These tumors were considered to be unclassified malignant neoplasms. Within this category, 5 tumors had originally been diagnosed as OMM. This group of 13 cats had the most variable treatments and survival time. Four had surgical therapy, 1 had chemotherapy, 3 had radiation therapy, and 2 had a complete course (4 doses, 2 weeks apart) of the canine melanoma vaccine. Median mitotic count was 9 (range = 1–71). Survival time was documented in 8 of these 13 cats, with a mean of 134 days (range = 26–249 days). Outcome was known in 6 cats, and included euthanasia due to progression of the primary tumor (4), anorexia (1), and nasal discharge and dyspnea (1). Necropsies were not performed in any of the cats. One cat was diagnosed with metastatic pulmonary lesions based on thoracic radiography; however, cytologic or histopathologic evaluation of these lesions was not performed.
Discussion
Feline amelanotic OMM are rare based on the findings of the current study. An accurate diagnosis requires both histology and IHC, as the latter is crucial to differentiate OMM from other oral malignances in the cat. Most neoplasms for which OMM was the primary favored differential diagnosis were reclassified based on IHC results; however, it is important to note that a proportion of the unclassified neoplasms may be poorly differentiated melanocytic neoplasms. Interestingly, all samples that had been initially misdiagnosed as OMM (n = 7) were originally evaluated from 2007 to 2010 (with 4 in 2007). Advances in pathology and the reliance on IHC to confirm a diagnosis of a challenging tumor may account for fewer such cases in recent years. In fact, IHC is advocated for the diagnosis of amelanotic melanoma in dogs. 30 In the absence of melanin pigment and/or junctional activity, a definitive diagnosis of OMM is not possible without the use of IHC.
The characteristic microscopic features of OMM include pigmentation, junctional activity (intraepithelial nests), lentiginous spread (radial extension of intraepithelial neoplastic cells), and extension to the overlying epithelial basement membrane (compound component). However, routine microscopic examination alone has several limitations. Oral neoplasms often have ulcerated surfaces thereby precluding evaluation of the overlying epithelium for junctional activity and lentiginous spread. Spindle cell variants of OMM closely mimic sarcomas, whereas polygonal/epithelioid variants mimic large-cell lymphomas and some poorly differentiated sarcomas or carcinomas. Based on our study, spindle cell STS occurs more commonly in the oral cavity of cats than OMM, and one neoplasm with polygonal morphology was reclassified as a carcinoma based on positive pan-cytokeratin immunolabeling. Furthermore, many feline OMM are not pigmented. All confirmed OMM cases in the present study were amelanotic. This could have some prognostic significance as shorter survival time in cats with melanoma has been significantly correlated with a lower degree of pigmentation, 7,24 and there are reported longer survival times in dogs with highly pigmented oral and cutaneous melanocytic neoplasms compared to dogs with amelanotic melanomas. 3,9,31 Mitoses and necrosis are also used as prognostic features and a histologic grading scheme proposed for feline melanocytic tumors of the oral cavity, lips, or nose categorized melanomas as high-grade if they had at least 4 mitoses in 10 high-power fields and/or the presence of intratumoral necrosis. 24
There are only a limited number of studies of feline OMM, 7,10 –12,16,17,27,28 and the diagnosis of OMM was confirmed with IHC only in a single study. 7 Given the findings of the current study, cases diagnosed as OMM without published histopathologic criteria and the use of IHC are of questionable validity. Therefore, reported survival times and response to treatment from these previous studies may not be representative of feline OMM, as these were most likely based on heterogeneous tumor populations. For example, in one study, feline amelanotic OMMs comprised 10 of 13 cases, but the diagnosis for 4 of these cases was based entirely on positive S100 immunoreactivity despite an absence of pigmentation, junctional activity, or labeling for melan-A. 7 In our study, histologically similar S100-positive cases were reclassified as STS based on an expanded IHC panel. This underscores the importance of an IHC panel when amelanotic melanoma is suspected in cats.
There is no single antibody that is both highly specific and highly sensitive for melanocytic neoplasms and thus antibodies are typically used in panels. While melan-A, PNL2, tyrosinase reactive protein 1 (TRP-1), and TRP-2 have been shown to have a specificity of 100% for differentiating canine oral melanocytic neoplasms from other spindle cell neoplasms, their sensitivity was reported as 82%, 90%, 55%, and 80%, respectively. 30 In contrast, other commonly used antibodies to detect canine melanocytic neoplasms, such as S100 or MITF, only had a 20% specificity while their sensitivity was above 80%. 30 Based on these results, an antibody cocktail composed of melan-A, PNL2, and TRP-1 and TRP-2 has been shown to provide the highest sensitivity and specificity for accurately diagnosing canine melanocytic neoplasms, but such a cocktail has not been tested in cats. 30 TRP-1 and TRP-2 were not used in the current study due to their relatively lower sensitivity for canine oral amelanotic melanomas compared with that of PNL2 and melan-A. 30
In cats, only melan-A, PNL2, and S100 have been investigated as melanocytic markers. 7,19,24,27 The published sensitivity of melan-A for feline amelanotic melanocytic neoplasms varies greatly, and it is uncertain how the diagnosis of amelanotic melanoma was originally established in these studies. 7,19,27 In one study in cats, 67% of melanocytic neoplasms were melan-A positive, indicating a moderately low sensitivity, but good specificity. 27 S100 was found to be sensitive in detecting amelanotic melanomas, but studies did not clearly distinguish these from other spindle cell neoplasms (eg, peripheral nerve sheath tumor) that are expected to label with S100, and thus basing a diagnosis of OMM only on reactivity with S100 is inappropriate. 4,15,27,31 PNL2, a monoclonal antibody that recognizes both normal and neoplastic melanocytic cells, was used in a recent study evaluating nonocular melanocytic tumors in cats and shown to be significantly associated with outcome; cats with higher PNL2 reactivity were more likely to die from their tumor. 24 That study showed both melan-A and PNL2 are sensitive markers for feline nonocular melanocytic (and amelanotic) tumors, thus both were evaluated in the present study to increase diagnostic accuracy. PNL2 labeled the same 2 OMMs that were diagnosed with melan-A, thereby suggesting a similar sensitivity and specificity for these 2 antibodies. More importantly, neither antibody labeled any of the other spindle cell neoplasms, further supporting the conclusion that amelanotic OMM is rare in cats.
Differential diagnoses for the STS diagnosed in the present study include fibrosarcoma, peripheral nerve sheath tumor, and perivascular wall tumor. Laminin is commonly expressed in peripheral nerve sheath tumors, 18 and CD34 is widely expressed by most perivascular wall tumors. 1,17 Thus, laminin and CD34 were used in the present study to exclude melanoma and to diagnose neoplasms as STS. STS was confirmed in 6 of 22 cases, 2 of which had been suspected or previously diagnosed as OMM. Although CD34 also labels vascular and hematopoietic neoplasms, it is useful for distinguishing non-melanocytic spindle cell tumors from melanomas. None of the neoplasms grouped as STS in our study had features of hematopoietic differentiation or evidence of vasoformative changes. Importantly, in canine oral amelanotic melanomas, CD34 labeled 80% of spindle cell sarcomas and none of the melanocytic tumors. 30 Sporadic case reports of aberrant expression of CD34 in melanomas in humans 6 and cats 13 have been reported, but the majority of the human and veterinary literature does not support CD34 as a marker for melanocytic tumors. 2,7,8,21,26,29,30,32
Despite advances in IHC, poorly differentiated neoplasms still present a diagnostic challenge, and we may be unable to definitively diagnose such cases with currently available tools. The plasticity of mesenchymal spindle cell neoplasms is extremely high as many most likely arise from mesenchymal cells through reactivation of genes that encode different lines of mesenchymal differentiation. Therefore, cross-reactivity of antibodies among the different entities is well recognized. 29 In human medicine, even the identification of recurrent chromosomal and genomic alterations in a significant subset of soft tissue neoplasms has yet to lead to major advances in the histological classification of mesenchymal neoplasms. 29 Such genetic alterations have not been characterized in mesenchymal spindle cell neoplasms in cats and thus the primary means to diagnose soft tissue tumors in cats is still microscopic evaluation of the morphologic features with some support of IHC. At this point, the clinical significance of further classifying such malignant oral neoplasms in cats is debatable, because the prognosis for all categories of tumors in the present study was quite poor. Future studies may be better directed at the potential of any of these markers to indicate a higher or lower risk for metastasis or to predict specific therapeutic responses.
Limitations of the present study were its retrospective nature, which produced a large variability of treatments making prognostic evaluation impossible. In addition, the relatively small sample size, specifically of cats immunohistochemically confirmed with OMM, may have prevented identification of histologic factors that influence prognosis. The largest group of tumors in our study were unclassified malignant neoplasms. A proportion of these unclassified neoplasms could be poorly differentiated melanomas that do not express melan-A or PNL2, because the consistency of PNL2 and melan-A immunoreactivity in feline OMM has yet to be validated in the literature. The majority of our samples were incisional biopsies with less than 1 cm of tissue available for testing. This may have negatively impacted diagnostic accuracy, but the size of the samples was typical for oral biopsies and thus our data reflect the accuracy of routine diagnostic histopathology.
In conclusion, feline OMM requires IHC for accurate diagnosis in cases that lack melanin pigment and junctional activity. Based on our small population, oral spindle cell malignancies in cats are more likely to be soft tissue sarcomas than melanomas but most remain unclassified. Regardless of tumor type, oral malignant neoplasms in cats likely carry a poor long-term prognosis.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211025793 - Immunohistochemical evaluation of suspected oral malignant melanoma in cats
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211025793 for Immunohistochemical evaluation of suspected oral malignant melanoma in cats by Kelly M. Saverino, Amy C. Durham, Matti Kiupel and Alexander M. Reiter in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff at both the Michigan State University and the University of Pennsylvania’s veterinary diagnostic laboratories for assistance with handling and preparing histologic samples for review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
