Abstract
Nonocular melanocytic neoplasia is considered uncommon in cats yet is routinely encountered in diagnostic pathology and recognized to exhibit a wide variation in biological behavior. Accurate prediction of clinical outcomes is challenging with no widely recognized prognostic criteria. Signalment and tumor location were retrospectively evaluated in 324 cats diagnosed with nonocular melanocytic neoplasia. Histologic features were described in 141 neoplasms and outcome data were available in 79 cases. Immunohistochemistry using Melan-A, PNL-2, cyclooxygenase 2 (COX-2), and E-cadherin was performed in a subset (n = 24). Multivariate analysis identified tumor site, mitotic count, and the presence of intratumoral necrosis to be independent predictors of tumor-related death. On the basis of these findings, we propose a novel histologic grading scheme in which nonocular melanocytic neoplasms involving the lips, oral or nasal mucosa, or nasal planum are considered high grade if they fulfill 1 or both of the following criteria: at least 4 mitoses in 10 high-power fields (HPF) or presence of intratumoral necrosis; those arising elsewhere are considered high grade if they fulfill both of the above criteria. Of 79 tumors with outcome data, 43 (54%) were low grade and 36 (46%) were high grade. The grading system had an 80% sensitivity and 92% specificity for predicting tumor-related death in this population of cats. Median survival for cats with low-grade tumors was not reached, and the median survival was 90 days for those with a high-grade tumor. PNL-2 and Melan-A were sensitive markers for feline nonocular melanocytic neoplasia, and although not significantly associated with prognosis, a large proportion expressed COX-2, suggesting a potential therapeutic role for COX-2 inhibitors.
Nonocular melanocytic neoplasms are often considered rare neoplasms in cats, reported to account for less than 1% of all feline oral neoplasms and approximately 0.5% of feline cutaneous tumors. 14 Nevertheless, these neoplasms continue to be frequently reported in the literature. 2 –4 Unlike in dogs, 13 however, there are no defined prognostic features for feline nonocular melanocytic neoplasms, making provision of prognostic information, as well as guiding treatment options, challenging to both the pathologist and clinician. In dogs, oral melanocytic neoplasms generally have a worse prognosis compared to cutaneous melanocytic neoplasms, 14,15 but this was not found to be the case in a recent study evaluating the clinical outcome of nonocular melanomas in cats. 2 In addition to tumor location, histologic features, including mitotic index, nuclear atypia, and degree of pigmentation, are used to ascertain whether prognosis is favorable, uncertain, or poor in dogs with melanocytic neoplasia. 13 Given the known examples of where the biological behavior of neoplastic disease in dogs and cats differs, 12 extrapolation of these parameters may not be appropriate for use in cats. In addition, the information currently available for prognostication of melanocytic neoplasia in dogs may be inadequate in cases where features of both a favorable and poor prognosis coexist. Histologic grading has become a tool routinely used by veterinary pathologists for consistent and accurate assessment of the potential biological behavior for many neoplasms. Given the now-wide range of surgical, chemotherapeutic, and radiotherapeutic treatment options available to animals, as well as the implications on the animal and owner, accurate diagnosis and information regarding prognosis are critical. Further to the lack of prognostic information available regarding the biological behavior of feline nonocular melanocytic neoplasms, accurate diagnosis can also be problematic, particularly when tumors are amelanotic. Melan-A and PNL-2 antibodies are routinely used for diagnosis of amelanotic melanomas in dogs, 10 and the use of these markers in feline melanocytic neoplasia also has been described previously. 2,11,17 Cyclooxygenase 2 (COX-2), an enzyme involved in the synthesis of prostaglandins not normally expressed by melanocytes, has been shown to be expressed in canine melanocytic tumors 8,9 and related to tumor localization, proliferation, and overall survival. 5 Expression of COX-2 may suggest a role for nonsteroidal anti-inflammatory drugs (NSAIDs), particularly COX-2 inhibitors, in the treatment of melanocytic neoplasia. E-cadherin is a transmembrane protein responsible for forming junctions between melanocytes and neighboring keratinocytes. 14,17 E-cadherin expression recently has been shown to increase in feline diffuse iris melanoma associated with metastatic disease. 17 Conversely, in human malignant melanomas, downregulation of E-cadherin has been associated with metastasis. 6
We conducted a multi-institutional study to retrospectively evaluate available clinical, histopathologic, and outcome data in cats diagnosed with nonocular melanocytic neoplasia to identify potential prognostic factors. Using this information, the final aim of this study was to devise a novel grading scheme to accurately predict outcome in cats diagnosed with nonocular melanocytic neoplasia. In addition, a subset of feline nonocular melanocytic neoplasms was assessed for expression of Melan-A, PNL-2, COX-2, and E-cadherin in this study to determine sensitivity and potential associations with outcome.
Materials and Methods
Animals and Clinical Data
This retrospective study of client-owned cats used data obtained from 2 diagnostic veterinary histopathologic laboratories in the United Kingdom and was approved by the Clinical Research Ethical Review Board at The Royal Veterinary College (URN 2016 1578). Cases were selected from the searchable electronic databases available at each diagnostic laboratory between 2006 and 2017. Inclusion criteria included cats from primary care veterinary practices with a histologic diagnosis of a melanocytic neoplasm that included “melanocytic,” “melanocytoma,” or “melanoma.” All cats were grouped together as melanocytic neoplasms, regardless of whether the neoplasm was designated as benign or malignant in the original report. All cats in which lesions were intraocular were excluded. Where formalin-fixed, paraffin-embedded (FFPE) tissue or hematoxylin and eosin (HE)–stained tissue sections were available, the histopathologic diagnosis of melanocytic neoplasia was reviewed. Histological and immunohistochemical evaluation was performed blinded to outcome data.
The age, sex, neuter status, and breed of each cat, as well as the location of the neoplasm and any relevant clinical features, were directly obtained from that recorded on the original histopathology report, which was translated from the submission form completed by the primary veterinary practice. Follow-up data were obtained by telephone questionnaire with the primary practice. Cat coat color was recorded if known. The occurrence or absence of tumor-related death was recorded for outcome. Overall survival was recorded for all cats and defined as the time between histologic diagnosis and tumor-related death. Cats that were still alive at the last time of follow-up or died (or were euthanized) due to unrelated causes were censored in the survival analysis. Cats whose records were not obtainable or that were not seen by the practice since the time of diagnosis were not included in the survival data. Any evidence of local recurrence, clinical staging, or metastatic disease was also recorded. A total of 324 cats diagnosed with nonocular melanocytic neoplasia were identified electronically to provide information on signalment and tumor location. Histologic features were evaluated in 141 neoplasms in which FFPE tissue or HE-stained sections were available, and outcome data were able to be obtained in 79 cases. In 24 cases where FFPE tissue was available, immunohistochemistry was performed.
Histopathology
Tumors of all cases where FFPE tissue or HE-stained tissue was available were included for histopathologic examination. The cases were evaluated simultaneously by 2 board-certified pathologists (R.P. and S.L.P.). Histopathologic examination was performed on 4-μm-thick sections stained with HE using light microscopy and confirmed the diagnosis of melanocytic neoplasia. Tumors were evaluated for the predominant histological pattern (cell type), percentage of neoplastic cells that contained melanin pigment, pigment intensity, nuclear atypia, cellular pleomorphism, mitotic count, the presence of intratumoral necrosis, surface ulceration, junctional activity, and the presence of intraepithelial nests of neoplastic cells (Figs. 1–6). Sections were also evaluated for the presence or absence of tumor-free histologic tissue margins (whether or not neoplastic cells extended to any of the histologic margins available) and evidence of vascular or lymphatic invasion. If required, bleached sections were examined to assess nuclear detail. If neoplastic cells were predominantly (>60%) polygonal and arranged in nests or cords, these were classified as epithelioid. If >60% of neoplastic cells were spindloid or elongate and formed streams or bundles, these were classified as spindloid, and if both patterns were present in approximately equal proportions, these were classified as mixed. If neoplastic cells were polygonal with a large volume of pale-staining cytoplasm, these were categorized as the balloon cell variant. Degree of pigmentation was recorded as the percentage (<50% = 1, ≥50% = 2) of neoplastic cells containing melanin pigment and pigment intensity (0 [no pigment] to 2 [high pigment]). These scores were then multiplied to give an overall pigmentation score. Nuclear atypia was subjectively graded as mild, moderate, or marked (mild = 0%–33%, moderate = 34%–66%, and marked = 67%–100% of neoplastic cells contained large, irregular, and often multiple nucleoli) and given a score of 1, 2, or 3, respectively. Cellular pleomorphism according to variation in cell size and shape was also subjectively assessed as mild, moderate, or marked and allocated a score of 1, 2, or 3, respectively. A mitotic count consisted of the number of mitoses in 10 randomly selected, consecutive, nonoverlapping (400×) high-power fields (HPF) (2.37 mm2) starting at an area of high mitotic activity and avoiding areas of ulceration or necrosis. Neoplasms were further grouped according to whether the mitotic count was <4 or ≥4. Intratumoral necrosis was defined as confluent aggregates of necrotic neoplastic cells and recorded as present or absent and excluded areas associated with ulceration. The presence of surface ulceration, junctional activity, and intraepithelial nests of neoplastic cells was assessed where possible, with junctional activity considered as neoplastic cells abutting the dermoepidermal junction and intraepithelial nests being neoplastic cells forming nests within the overlying epithelium.
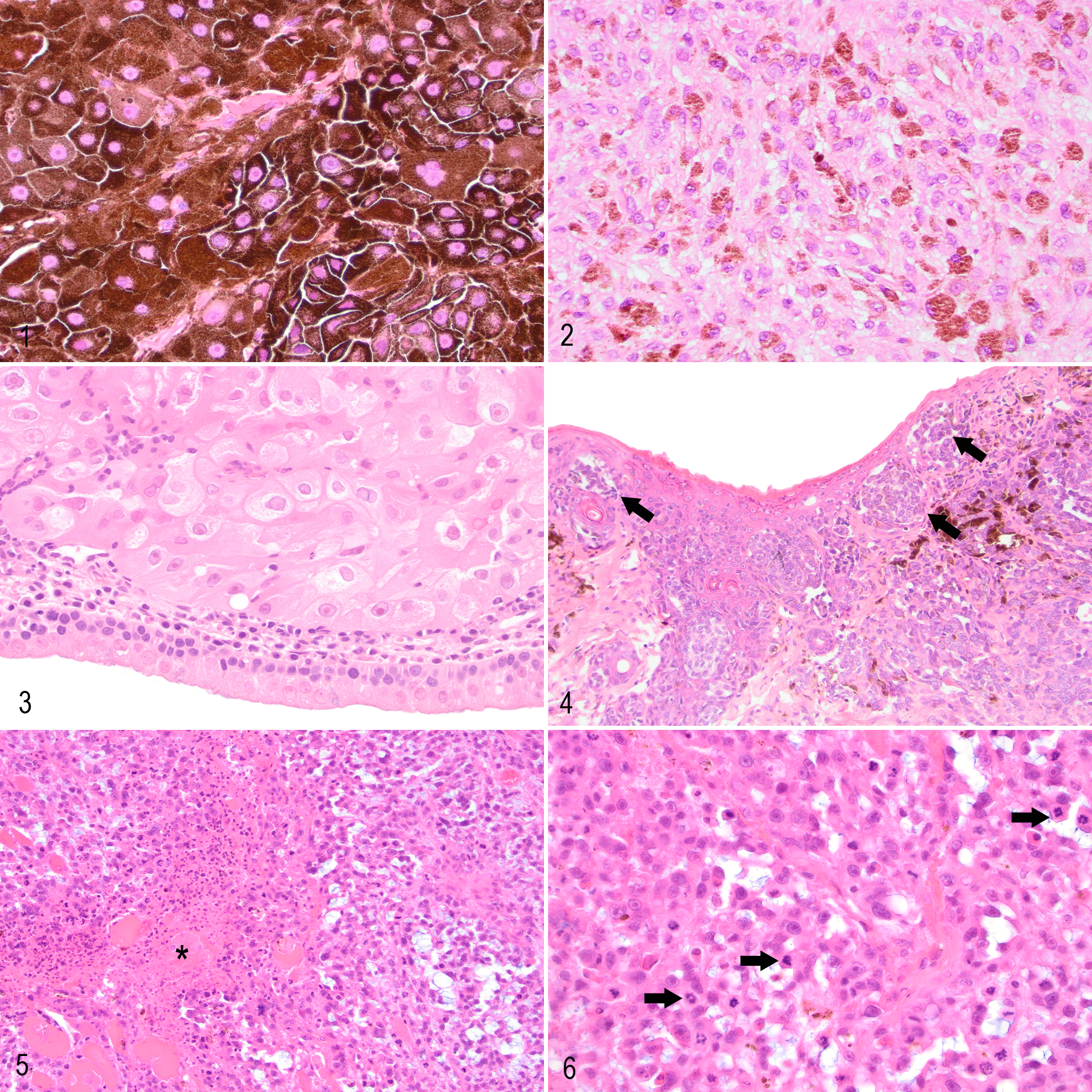
Melanocytic neoplasms, cat. Hematoxylin and eosin.
Immunohistochemistry
Immunohistochemistry (IHC) was performed on cases where FFPE tissue was available. The 4-μm-thick sections from representative blocks were mounted onto positively charged slides (SuperFrost Plus; Menzel Gläser, Braunscheig, Germany). Antigen retrieval, labeling, and counterstaining were performed on a Bond-Max Autostainer (Leica Biosystems, Newcastle-upon-Tyne, UK) using the Bond polymer refine detection system (Leica Biosystems). Primary antisera were specific for Melan-A (A103 clone, monoclonal [Leica Microscopy, Milton Keynes, UK]; 1 in 100 dilution; antigen retrieval in buffer pH 9.0 buffer [ER2, Leica Biosystems] for 20 minutes), PNL-2 (sc-59306, monoclonal [Santa Cruz, Heidelberg, Germany]; 1 in 300 dilution; antigen retrieval in pH 6.0 buffer [ER1, Leica Biosystems] for 20 minutes), COX-2 (SP21 clone, monoclonal [Fisher Scientific, Loughborough, UK]; 1 in 150 dilution; antigen retrieval in pH 6.0 buffer [ER1, Leica Biosystems] for 10 minutes), and E-cadherin (NCH-38 clone, monoclonal [Agilent, Stockport, UK]; 1 in 100 dilution; antigen retrieval in pH 6.0 buffer [ER1, Leica Biosystems] for 10 minutes). Sections from a canine oral melanocytic neoplasm, known to express Melan-A and PNL-2, were used as a positive control. Canine kidney was used as a positive control for COX-2 and normal feline skin as a positive control for E-cadherin. Negative controls were used where primary antibodies were replaced with normal serum.
Immunolabeling characteristics for Melan-A and PNL-2 were characterized according to both the percentage of immunopositive cells and the intensity of labeling. The percentage of positively labeled neoplastic cells was recorded as 0 = ≤5%, 1 = 5% to 10%, 2 = 11% to 50%, and 3 = >50%. Intensity was subjectively scored as 0 = none, 1 = mild, 2 = moderate, and 3 = intense. Scores were then multiplied to give an overall score. 10 COX-2 labeling was characterized according to percentage of positive neoplastic cells and intensity of labeling with 0 = negative, 1 = 1% to 19%, 2 = 20% to 49%, and 3 = >50% and 0 = negative, 1 = weak, 2 = moderate, and 3 = strong. These were then multiplied to give a total immunoreactivity score. 9 E-cadherin immunoreactivity was recorded according to the intensity of labeling (0 = less than control, 1 = control) and whether labeling was membranous and/or cytoplasmic. Areas adjacent to ulceration and/or necrosis were avoided for interpretation.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics (v. 23; SPSS, Inc, an IBM Company, Chicago, IL). Differences were considered significant when P < .05. For survival analysis, outcome was defined as time from histologic diagnosis to death due to melanocytic disease; cats that were still alive at the last time of follow-up or died/were euthanized due to unrelated causes were censored in the survival analysis and the time between diagnosis and follow-up or death recorded. Median survival time was estimated by the Kaplan-Meier method, and univariable and multivariable Cox proportional hazard model was used to identify significant prognostic factors. Multivariable Cox proportional hazard regression analysis was performed to include all of the significant variables identified in the univariable analysis. Those with the highest P values were sequentially excluded until the remaining variables all had P values of <.05. For analysis, ages of cats were grouped into quartiles. Mitotic count was grouped into those cases with a mitotic count of <4 and those with a mitotic count of ≥4. Tumor location was grouped into “lip/oral/nasal” or “other nonocular.” A single case where the neoplasm was present within a lymph node was excluded for survival analysis. The data analyzed in this study for all cats where outcome data were obtained are available in Supplemental Table S1.
Results
Animals
A total of 324 cats diagnosed with nonocular melanocytic neoplasia were identified electronically to provide information on signalment and tumor location. Histologic features were evaluated in 141 neoplasms where FFPE tissue or HE-stained sections were available. Outcome data were able to be obtained in 79 cases. In 24 cases where FFPE tissue was available, immunohistochemistry was performed.
Signalment and Tumor Location
This study included 324 diagnoses of nonocular melanocytic neoplasia in cats. The age of the cats at the time of diagnosis ranged between 10 months and 20 years, with the age of 11 cats not recorded (median, 11 years; interquartile range, 6 years). Of the 324 cats, 167 (51.5%) were female, 146 (45.1%) were male, and most cats (261/324, 80.6%) were neutered, with 11 cats being of unknown sex or neuter status. The breeds affected were domestic short hair (264/324, 81.4%), domestic long hair (29/324, 9.0%), or other breeds: Maine Coon (5/324, 1.5%), British Shorthair (5/324, 1.5%), Siamese (4/324, 1.2%) Burmese (3/324, 0.9%), “Domestic” (2/324, 0.6%), and Abyssinian, Bengal, Birman, British Blue, Turkish Angora, Ragdoll, Persian, Persian X, and Burmese X (each 1/324); 3 of 324 were of unknown breed. The most frequently affected anatomic locations included the pinna/ear (80/324, 24.7%), elsewhere on the skin of the head (66/324, 20.4%), the oral cavity (56/324, 17.3%), and the truncal skin (32/324, 9.9%), with the remainder affecting other locations, including the lip (17/324, 5.2%), the limb skin (14/324, 4.3%), the nose (13/324, 4.0%), digital skin (7/324, 2.2%), the conjunctiva or third eyelid (4/324, 1.2%), the tail (3/324, 0.9%), and prepuce (1/324, 0.3%). Six of 324 (1.9%) were primarily diagnosed within lymph nodes, and 25 of 324 (7.7%) were described as being on the skin without further being specified. Of cats in which follow-up information was obtained, 6 of 80 (7.5%) were of unknown color, with 33 of 74 (44.6%) black or black and white, 23 of 74 (31.1%) “tabby” or “tabby and white,” 7 of 74 (9.5%) tortoiseshell, 5 of 74 (6.8%) ginger or ginger and white, 2 of 74 (2.7%) gray, 2 of 74 (2.7%) white, 1 of 74 (1.4%) sealpoint, and 1 of 74 (1.4%) brown.
Clinical Characteristics
Of the 141 cases where FFPE or HE-stained tissue was available, submission data were reviewed. Of these, 59 of 141 (41.8%) were described or suggested as being macroscopically pigmented and 25 of 141 (17.7%) were described as being eroded or ulcerated. Of the 80 cases where follow-up information was obtained, 8 cases (10%) recorded some form of clinical staging, which consisted of local lymph node assessment in 7 cases and chest radiographs in 1 case. All 7 cases where lymph node assessment was performed reported evidence of metastatic disease. Confirmed or suspected metastatic disease was reported in 16 of 80 (20%) cases.
Histopathology
HE-stained tissue was evaluated in 141 of the 324 cases where FFPE or HE-stained tissue was available. Of the141 cases, 14 (9.9%) were amelanotic, with 7 of these affecting the oral cavity, 2 arising on the pinna, and 1 each affecting the conjunctiva, lip, nose, skin of a limb, and trunk. Mitotic count ranged from 0 to 133 per 10 HPF (2.37 mm2). Epithelioid was the most common histologic pattern (81/141, 57.4%), with 48 of 141 (34.0%) being spindloid, 10 of 141 (7.1%) being mixed, and 2 of 141 (1.4%) being the balloon cell variant. Histologic tissue margins were considered free of neoplastic cells in 82 (58.2%) cases, with incomplete margins in 59 (41.8%) cases. In 6 cases (4.3%), lymphatic or vascular invasion was observed, 3 of which were in the oral cavity. Ulceration was present in 94 of 141 cases (66.7%), and 110 of 141 (78.0%) were found to have junctional activity, with 14 of 141 (9.9%) having intraepithelial nests. Intratumoral necrosis was present in 29 of 141 (20.6%) cases.
Clinical Outcome and Survival Analysis
Follow-up data were available for 80 of the 141 cases (57%) in which histopathologic evaluation was performed. Cats that were lost to follow-up included cases where primary practice information was not available, the cat had not been seen at the practice since time of diagnosis, practice recording systems had changed and historical records were not retrievable, or cats could not be found on the records system. Survival of all cats ranged from 4 days to >2022 days, with a median survival time of 427 days. Of the 80 cats with follow-up data, 42 (52.5%) were euthanized or died of causes perceived to be related to their melanocytic neoplasia, with a median survival of 244 days (Fig. 7).
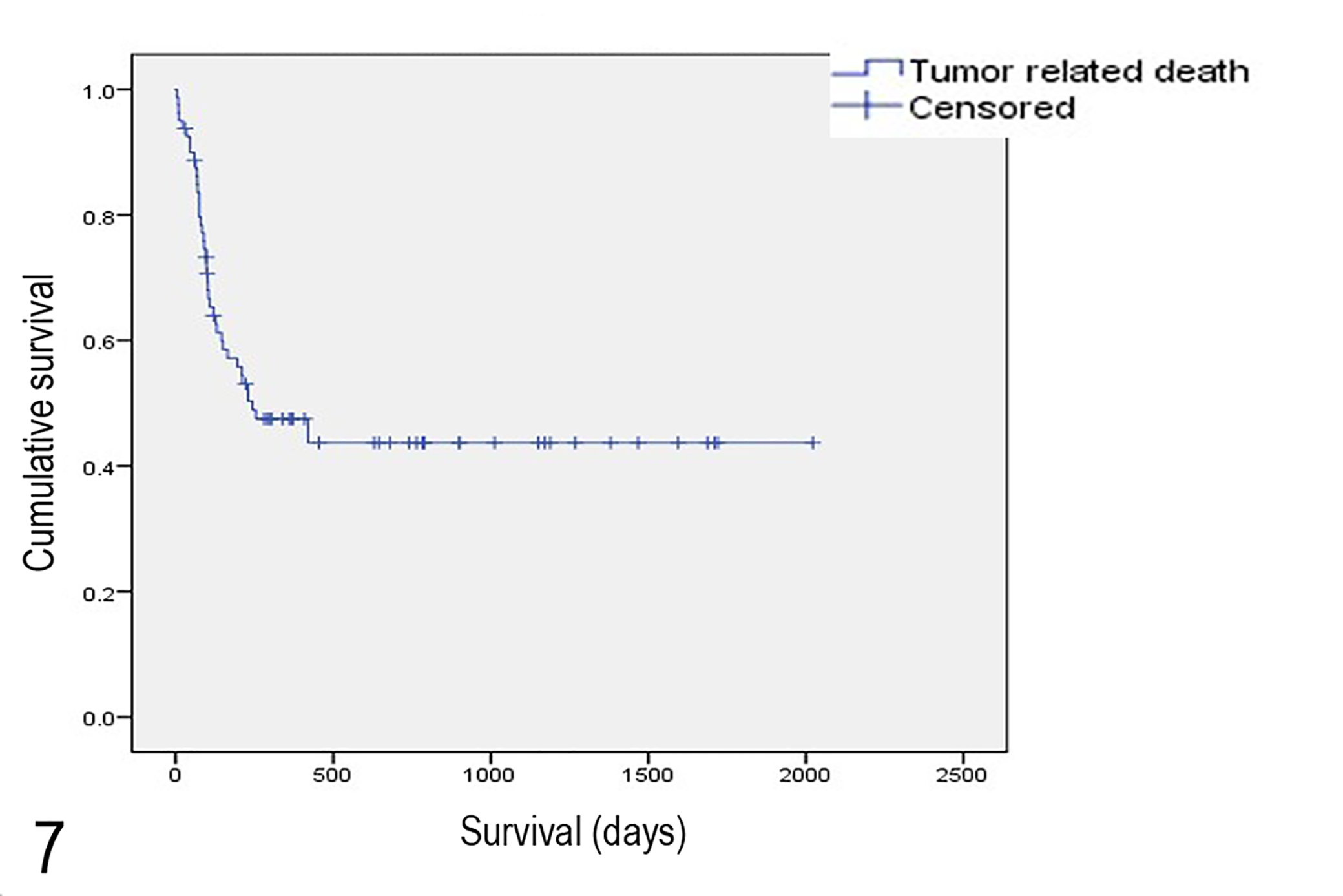
Kaplan-Meier survival curve for cats with nonocular melanocytic neoplasia.
Median survival of cats with melanocytic tumors arising within the oral cavity, lips, or nose (n = 25) was 83 days. Median survival time of cats with melanocytic neoplasia arising in the skin or digit was not reached, with 15 of 51 (29.4%) undergoing tumor-related death. Mean estimated survival was 1406 days. Median survival of conjunctival/third eyelid melanocytic tumors (n = 3) was also not reached, with a mean estimated survival of 555 days. Clear histologic tissue margins were present in 42 of 80 (52.5%) of cases, of which local recurrence was recorded in 10 cases (23.8%).
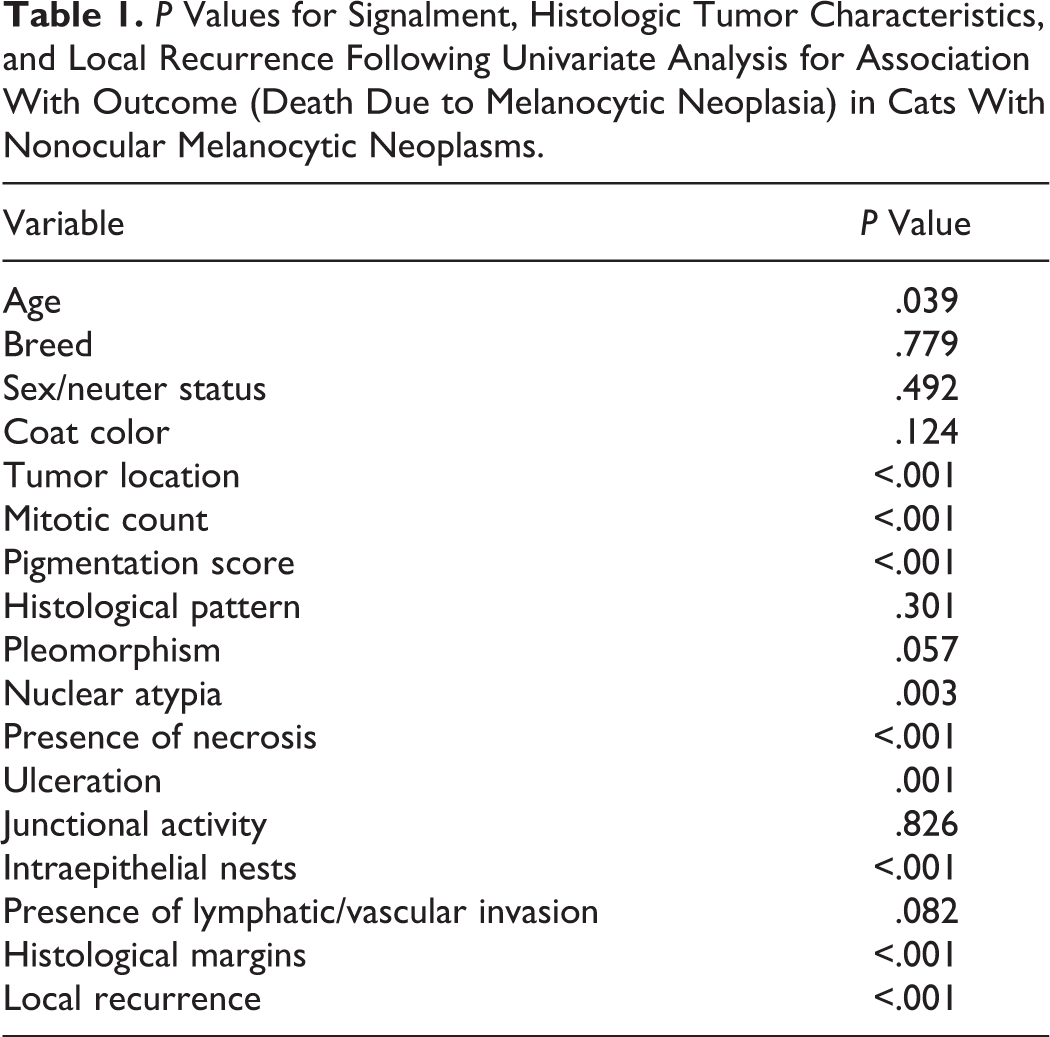
The single case where the diagnosed melanocytic neoplasm was within a lymph node was excluded from additional survival analysis. Univariate Cox proportional hazard regression analysis was performed on signalment and histologic criteria recorded (n = 79). Age at diagnosis, tumor location, mitotic count, pigmentation score, nuclear atypia, intratumoral necrosis, ulceration, intraepithelial nests, and tumor-free histologic tissue margins were all significantly (P < .05) associated with outcome (Table 1). Local recurrence was also significantly associated with outcome. Breed, sex and neuter status, coat color, histological pattern, cellular pleomorphism, junctional activity, and the presence of lymphatic or vascular invasion were not significantly associated with outcome.
P Values for Signalment, Histologic Tumor Characteristics, and Local Recurrence Following Univariate Analysis for Association With Outcome (Death Due to Melanocytic Neoplasia) in Cats With Nonocular Melanocytic Neoplasms.
Tumor location (lip/oral/nasal or other nonocular), mitotic count (<4 or ≥4 per 10 HPF), and intratumoral necrosis (presence or absence) were identified to be independent predictors of survival in the multivariable analysis (Figs. 8–10). Based on these, a grading system was devised (Table 2), and each nonocular melanocytic neoplasm with follow-up data available was then graded according to these criteria. Of these neoplasms, 43 were low grade and 36 were high grade. Median survival for low-grade neoplasms was not reached, with 19% (8/43) of cats dying or euthanized due to their melanocytic neoplasia, with an estimated mean survival of 1642 days. Median survival for high-grade neoplasms was 90 days, with an estimated mean survival of 131 days and 92% (33/36) of cats dying or euthanized due to their melanocytic neoplasia (Fig. 11). Sensitivity of the grading scheme to identify those cats that were likely to die due to their melanocytic neoplasia was 80% with a specificity of 92%. The positive and negative predictive values of the grading scheme were 92% and 81% respectively.
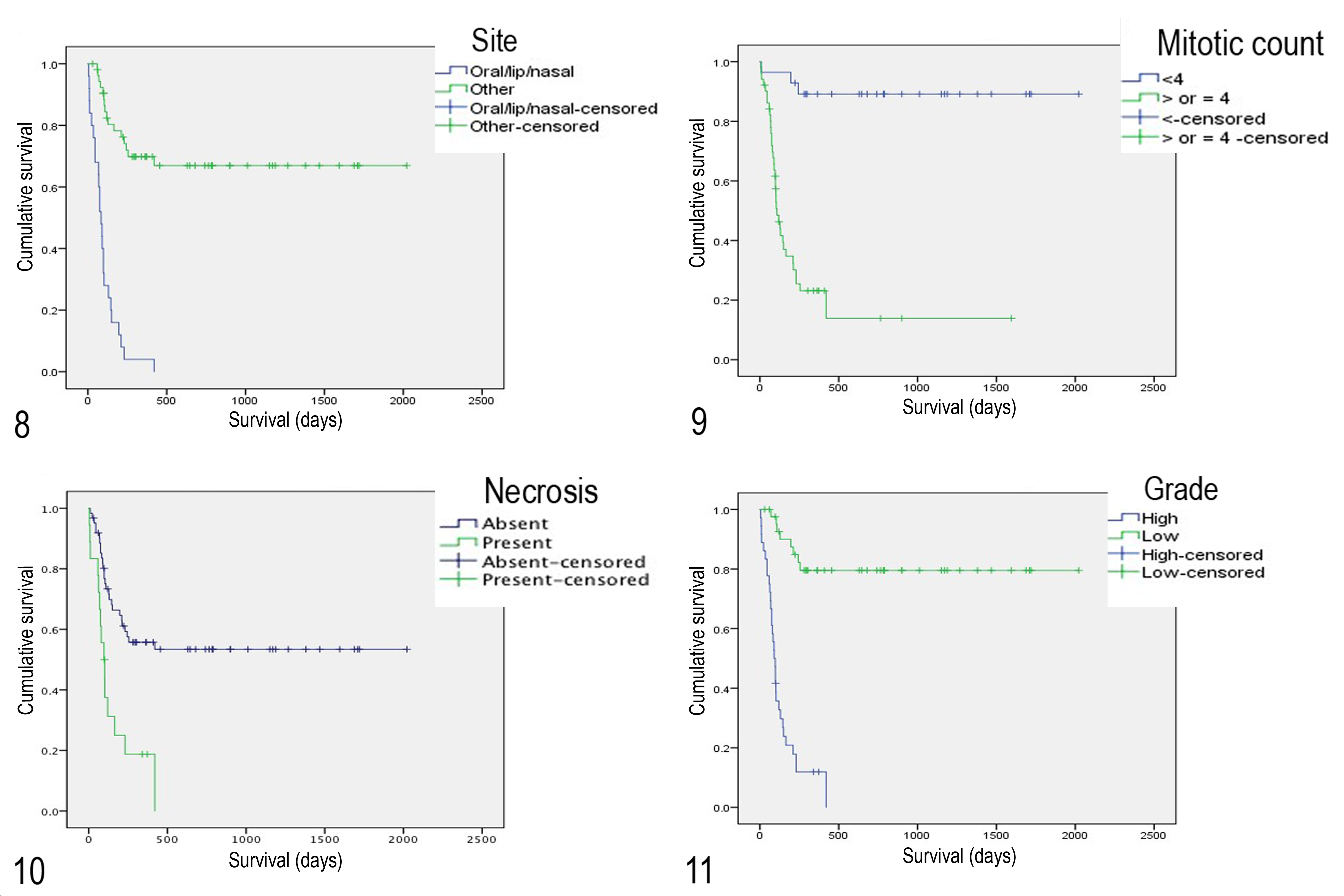
Kaplan-Meier survival curves for cats with nonocular melanocytic neoplasia according to tumor site, mitotic count, presence or absence of intratumoral necrosis, and histologic grade. Points indicate censored events. Overall survival according to location (lip/oral/nasal or other nonocular; Figure 8), mitotic count (<4 or ≥4 per 10 high-power fields; Figure 9), presence or absence of intratumoral necrosis (Figure 10), and histological grade using the proposed grading scheme (Figure 11).
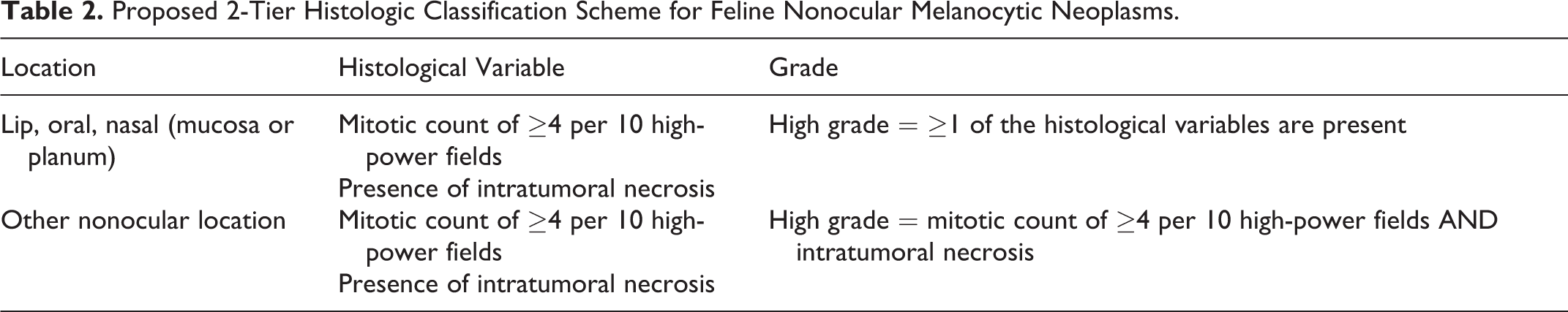
Proposed 2-Tier Histologic Classification Scheme for Feline Nonocular Melanocytic Neoplasms.
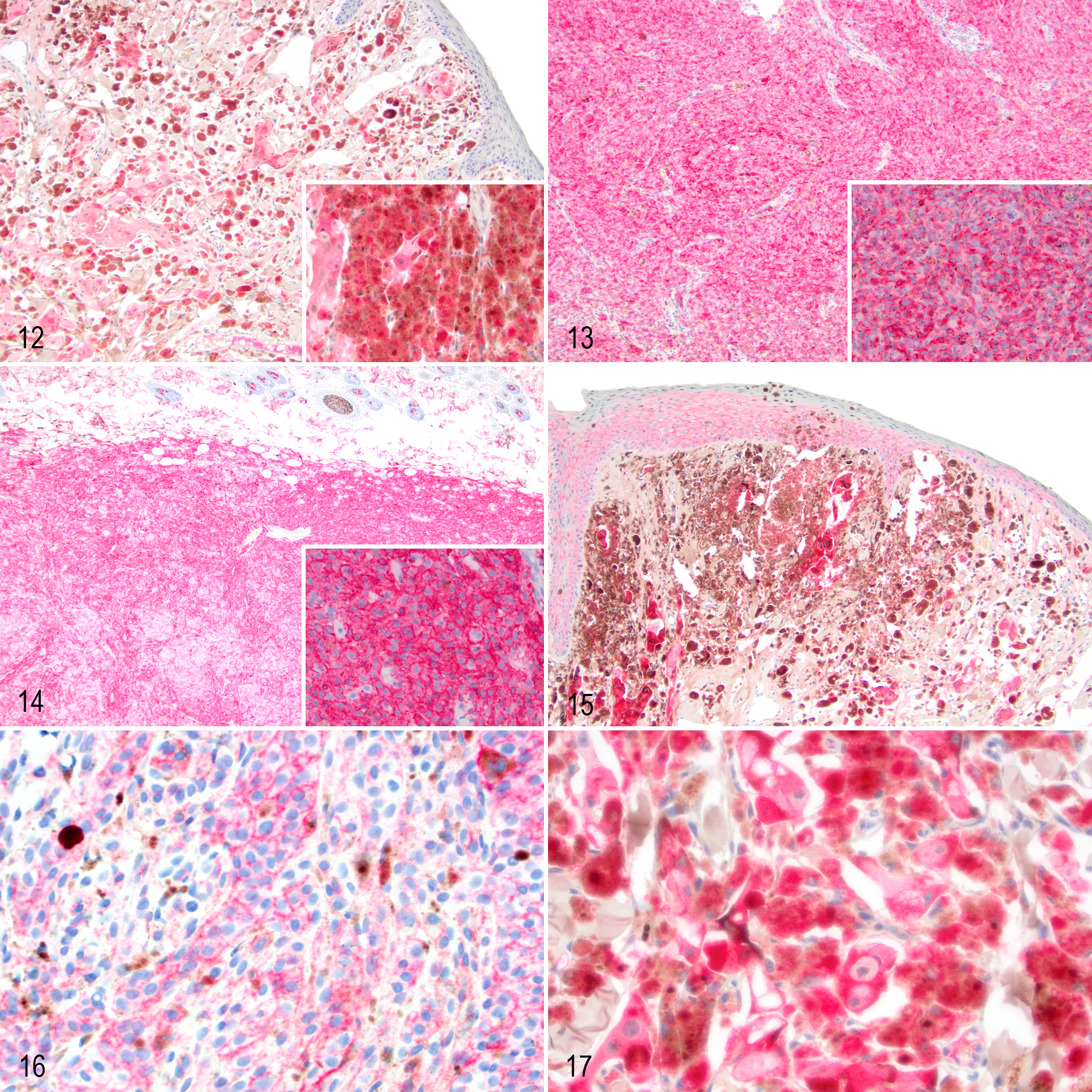
Immunohistochemistry
Immunohistochemical labeling was performed on the 24 feline nonocular melanocytic neoplasms where FFPE tissue was available (Figs. 12–17). Twenty-one of 24 (87.5%) immunohistochemically labeled for Melan-A, and 22 of 24 (91.7%) labeled for PNL-2. One neoplasm was negative for both (neoplastic cells, however, exhibited a low degree of pigmentation on HE staining, which identified the diagnosis). Total score for Melan-A expression was not significantly associated with outcome, whereas PNL-2 score was significantly associated with outcome (P = .035), with those with a higher PNL-2 more likely to die due to their tumor.
Melanocytic neoplasms, cat.
Of the 24 neoplasms, 15 (62.5%) exhibited labeling for COX-2 with no significant association between score and outcome. Labeling for E-cadherin was observed in 19 of 24 (79.2%) cases, with 13 of 24 (54.2%) exhibiting a reduced labeling intensity compared to controls with no significant association with outcome. Of these, 3 of 24 showed membranous labeling only, and 16 of 24 showed membranous and/or cytoplasmic labeling. E-cadherin intensity or distribution was not associated with outcome.
Discussion
The aim of this retrospective study was to identify clinical and histologic features of feline nonocular melanocytic neoplasia associated with outcome. Any original classification as benign or malignant at the time of initial diagnosis was disregarded in this study due to a lack of defined criteria of malignancy for feline nonocular melanocytic neoplasia and variability in terminology used, and therefore all tumors are referred to in this study as melanocytic neoplasms. Prospectively, for feline nonocular melanocytic neoplasms, the authors would recommend the restricted use of the term melanoma, or preferably malignant melanoma, to state a malignant designation where appropriate, such as where there is presence of intravascular invasion. A lesion considered benign should be designated melanocytoma. In frequent situations where benign or malignant biological behavior is not clearly apparent, the authors prefer the use of the umbrella term melanocytic neoplasm.
Assessment of signalment data from a large number of cats diagnosed with nonocular melanocytic neoplasia identified a wide age range to be affected, with a predominance of middle-aged to older cats. There was no apparent sex predilection, and most cats were neutered, with the predominant breed affected being the domestic short-haired cat, likely reflecting the general population in the United Kingdom. Common anatomic sites included the pinna and skin of the head as previously reported. 4,14 The oral cavity was also a common site affected. Although there was no significant association between coat color and tumor-related death, nearly half of all cats where coat color was recorded were either black or black and white, suggesting that pigmentation of the coat may predispose to development of melanocytic neoplasia. This is in contrast to white cats being predisposed to squamous cell carcinoma, 7 with only 2 of the cats in this study being described as white. A limitation in assessing this parameter is that it was only possible to obtain general coat color, and thus it is not known what color the hair coat was specifically at the site of tumor development in those cats that were not of uniform color. Nevertheless, a differential diagnosis of melanocytic neoplasia should be considered for masses arising on the skin of the head or within the oral cavity of cats, particularly in those cats that are not white in color. In addition, many of the neoplasms in this study (41.8%) were reported by the primary veterinarians to be grossly pigmented. The retrospective nature of this study, however, may mean that the true proportion that exhibits macroscopic pigmentation is likely to be higher. As the population of cats used in this study was from primary veterinary practices rather than from a referral population, the majority did not undergo any form of clinical staging, with only 10% reporting either local lymph node assessment or imaging. In the majority of cases where staging was performed, metastatic disease was reported, but selection bias based on the clinical presentation of cats may overestimate the likelihood of metastasis in this study, with clinical evidence of metastatic spread initiating staging in these cases. A total of 20% of cases reported suspected or confirmed metastatic disease, but given the inconsistent clinical staging and lack of histological confirmation of metastasis, this number may be either an underestimation or an overestimation of the true metastatic rate.
This study confirms a wide variation in the histologic appearance of nonocular melanocytic neoplasia in cats. Although the epithelioid pattern was most common, there was no significant association between pattern and outcome. Age of cats was significantly associated with outcome, with older cats more likely to suffer from tumor-related death. In this study, as shown in previous studies, 4 a higher proportion of nonocular melanocytic neoplasms affecting the pinna was observed in younger cats. It may be that neoplasms in this location, more likely to be in younger cats, are recognized earlier or more likely to be surgically resectable. Tumor location was also associated with outcome in our study population, with those affecting the lips, nose, or oral cavity having a poorer prognosis. This may be due to innate biological features of the neoplastic cells arising in these locations, with all of the melanocytic neoplasms in the oral cavity in this study having a mitotic count of ≥4, or may be due to the relative difficulty in achieving early diagnosis and/or surgical resection in these locations, with oral neoplasms largely being biopsied for diagnostic purposes rather than being completely excised. Mitotic count and presence of intratumoral necrosis were also associated with occurrence of tumor-related death, with a higher mitotic rate and presence of intratumoral necrosis potentially suggesting more rapid growth. All of the cats with amelanotic tumors in our study population died of their melanocytic disease, with degree of pigmentation significantly associated with outcome. This supports the suggestion that poor pigmentation correlates with poor differentiation and a more aggressive biological behavior. Similarly, nuclear atypia was also associated with outcome, but this was felt to be a more subjective parameter to assess and was susceptible to increased interobserver variability. Interestingly, the presence of intraepithelial nests was associated with outcome, but junctional activity was not. Tumor-free histologic margins and absence of recurrence were both associated with reduced likelihood of tumor-related death. In this study, it was not possible to standardize the sectioning technique due to the retrospective nature involving multiple institutions, and this should be considered in any future prospective studies, in addition to further recording the size of tumor-free histologic tissue margins where complete excision is achieved. Surprisingly, the presence of lymphatic or vascular invasion was not significantly associated with outcome. However, of 5 cats with this feature and with follow-up information available, 4 died of their melanocytic neoplasm and 1 died after a short period of time for an unrelated reason. Therefore, the low number of cases in which this feature was observed may not have allowed identification of an association.
Based on multivariable analysis, the tumor location, mitotic count, and presence of any intratumoral necrosis were features used to devise a novel classification scheme. A 2-tiered grading scheme was favored to avoid the inclusion of an intermediate group where prognosis would remain uncertain. The grading scheme proposed here was able to identify 81% of cats that would die of their melanocytic disease, with 8% of those diagnosed as high grade not dying as a result of their tumor. Based on these results, this grading scheme should identify the majority of nonocular melanocytic neoplasms that are likely to exhibit aggressive biological behavior and thus warrant further staging and discussion regarding adjunctive therapy. In addition, the contrast in survival times between cats with tumors of each grade provides additional prognostic information not currently available. The parameters used in this grading scheme have been grouped in such a way as to minimize interpathologist variation and subjectivity.
All of the cats in our study had surgical biopsy or excision only with no adjunctive chemotherapy or radiotherapy. A limitation of this study is the presumption that all cats alive at the time of follow-up or deceased due to other causes would not have died due to their melanocytic neoplasia, and ongoing prospective validation of this classification scheme is required. Survival data from our population of cats suggest an adequate follow-up period of approximately 500 days to identify those cats that will suffer tumor-related death.
PNL-2 and Melan-A have been shown to be sensitive markers for feline nonocular melanocytic neoplasms in our study. The 1 neoplasm that did not label with either marker also had no labeling of internal positive controls (adjacent nonneoplastic melanocytes) within the section, suggesting that the tissue may have been unsuitable for immunohistochemistry such as due to overfixation. A large proportion of tumors labeled for COX-2 has been shown in canine melanocytic neoplasms, 8,9 but expression was not found to be associated with prognosis. E-cadherin expression was reduced or lost in 75% of cases, suggesting downregulation of intercellular junctions and potentially enabling increased tumor growth, but this was also not found to be a prognostic indicator. The lack of significant associations with outcome may be due to the small sample size used for immunohistochemical analysis.
For amelanotic tumors, where diagnosis may be challenging, both Melan-A and PNL-2 are sensitive markers to confirm melanocytic origin in cats. The significance of COX-2 and E-cadherin expression requires further investigation, but a large proportion of melanocytic neoplasms in these cats expressed COX-2. Given the recognition of COX-2 inhibitors in the treatment of other tumors in both dogs and cats, 1 this suggests a potential new therapeutic option for the treatment of melanocytic disease in cats. Although aberrant cytoplasmic E-cadherin expression has been associated with a more aggressive behavior in skin neoplasms, we cannot give further interpretation of these findings, and future works should address the role of E-cadherin in nonocular melanocytic neoplasms. 16
Based on this retrospective study, we propose a novel 2-tier histologic grading scheme to more accurately predict those feline nonocular melanocytic neoplasms that will exhibit aggressive biological behavior and are likely to result in tumor-related death, potentially warranting further clinical staging and adjuvant therapy. Prospective use of this grading scheme on additional cases is required for further validation.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819854435 - Nonocular Melanocytic Neoplasia in Cats: Characterization and Proposal of a Histologic Classification Scheme to More Accurately Predict Clinical Outcome
Supplemental Material, DS1_VET_10.1177_0300985819854435 for Nonocular Melanocytic Neoplasia in Cats: Characterization and Proposal of a Histologic Classification Scheme to More Accurately Predict Clinical Outcome by Rachel Pittaway, Melanie J. Dobromylskyj, Kerstin Erles, Charles E. Pittaway, Alejandro Suárez-Bonnet, Yu-Mei Chang and Simon L. Priestnall in Veterinary Pathology
Footnotes
Acknowledgements
We thank the laboratory staff involved in providing tissue sections required for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Pathobiology and Population Sciences at the Royal Veterinary College.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
