Abstract
Sarcoids are the most common cutaneous tumor of equids and are caused by bovine papillomavirus (BPV). Different clinical subtypes of sarcoids are well characterized clinically but not histologically, and it is not known whether viral activity influences the clinical or histological appearance of the tumors. The aim of this study was to verify whether the development of different clinical types of sarcoids or the presence of certain histological features were associated with BPV distribution within the tumor. The presence of BPV was assessed by polymerase chain reaction (PCR) and visualized in histological sections by chromogenic in situ hybridization (CISH) in 74 equine sarcoids. Furthermore, to better characterize the molecular features of neoplastic cells, immunohistochemistry for S100, smooth muscle actin-α (αSMA), and fibroblast-associated protein-α (FAPα) was performed. The presence of BPV was confirmed in all tissues examined by either or both PCR and CISH (72/74, 97% each). Of 70/74 CISH-positive cases, signal distribution appeared as either diffuse (61/70, 87%) or subepithelial (9/70, 13%); the latter was more frequently observed in the verrucous subtype. However, no statistically significant association was found between clinical subtypes and specific histological features or hybridization pattern. Moreover, CISH signal for BPV was not detected in the epidermis overlying sarcoids nor in the tissue surrounding the neoplasms. By immunohistochemistry, αSMA confirmed the myofibroblastic differentiation of neoplastic cells in 28/74 (38%) sarcoids. Using tissue microarrays, FAPα labelling was observed in neoplastic fibroblasts of all sarcoids, suggesting this marker as a potential candidate for the immunohistochemical diagnosis of sarcoids.
Keywords
Sarcoids are the most common cutaneous spindle cell tumors affecting equids, representing 90% of all skin neoplasms. 2 Equine sarcoids are classified as non-metastatic and locally aggressive tumors, demonstrating a high tendency to recur after surgical treatment. 11 The association of sarcoids with BPV-1 (bovine papillomavirus), BPV-2, and, less commonly, BPV-13 has been widely reported, but the pathogenetic mechanisms are still to be fully elucidated. 13,23 Interestingly, BPV DNA has been demonstrated in the skin of healthy horses, suggesting that additional factors such as skin trauma and genetic predisposition may be required for sarcoid development. 4
Sarcoids are classified into 6 distinct subtypes based on their clinical features. These include the occult, verrucous, nodular, fibroblastic, mixed, and rarely the malignant (malevolent) type. 16 The histological features of sarcoids are well-known, characterized by a proliferation of interlacing bundles and whorls of fibroblasts, epidermal hyperplasia with rete-peg formation, and “picket-fence” arrangement of neoplastic cells at the dermo-epidermal junction. 20 However, previous histological studies failed to demonstrate an association between clinical types and histological appearance as these features are rarely always present together and similar histological patterns can be present in other mesenchymal neoplasms. 14 The presence of epidermal hyperplasia and “picket fence” arrangement of fibroblasts are typically considered pathognomonic for the diagnosis of sarcoid; however, they can be absent or lost due to epidermal ulceration. Thus, a definitive diagnosis may require polymerase chain reaction (PCR) and/or immunohistochemistry (IHC). Currently, there are no specific immunohistochemical markers for sarcoids; however, the combined presence of BPV DNA and S100 negativity are considered the main diagnostic features. 9 FAPα is a cell surface serine protease that is upregulated during tissue remodeling events and expressed in stromal fibroblasts of many carcinomas as well as under some inflammatory conditions. 17,21 Its expression has been verified in a subset of human sarcomas, 8 but the role of FAPα in the diagnosis of spindle cell tumors in domestic animals has never been assessed.
In the study presented here, BPV nucleic acid was localized with chromogenic in situ hybridization (CISH) in sarcoid tissues, in order to verify whether the development of different clinical types or presence of certain histological features were associated with viral distribution. Moreover, immunohistochemical markers S100, α-smooth muscle actin (SMA), and FAPα were applied to better define the molecular features of sarcoids.
Materials and Methods
Case Selection and Histology
Tissue samples from surgical excision or incisional biopsies with a histological diagnosis of sarcoid were retrieved from the archives of the Veterinary Pathology Centre at the University of Surrey (UK) and from the Pathology Service of the Department of Veterinary Medical Science of the University of Bologna (Italy), from 2010 to 2018. Clinical information, including signalment, tumor location, and clinical subtypes were retrieved from the submission forms. Samples were available as formalin-fixed, paraffin-embedded (FFPE) and hematoxylin and eosin (HE)-stained slides. Two veterinary pathologists (BB, GT) reviewed each case and the initial diagnosis of sarcoid was confirmed based on histopathologic features.
To compare histopathological features with clinical subtypes, each tumor was evaluated for 6 histological features: hyperkeratosis, hyperplasia of the epidermis, rete pegs, picket fence arrangement of fibroblasts (rows of fibroblasts with perpendicular orientation to the epidermal basement membrane), 20 ulceration/crusting, and inflammation. Each feature was recorded as present or absent. Moreover, 2 growth patterns of fibroblasts in the dermis were identified and categorized into either interstitial (neoplastic fibroblasts arranged between and around collagen fibers) or fascicular (neoplastic cells arranged in interlacing streams and bundles), based on the prevalent pattern.
Protein Extraction and Western Blot Analysis
For protein extraction, fresh tissue from equine dermis, intestine, and sarcoid was collected. From each, 25 µg of tissue was homogenized with 500 ml RIPA buffer (Thermofisher Scientific) and 100 µl of protease inhibitor (Halt Protease Inhibitor Cocktail EDTA-free, Thermofisher Scientific). Tubes were centrifuged at 12 000 rpm for 30 minutes at 4 °C and the supernatant was collected and stored at −80 °C. Protein concentration was calculated using the DC Protein Assay Kit (Bio-Rad). To evaluate FAPα antibody specificity, a Western blot analysis was performed. Briefly, 40 µg of proteins were denatured at 100 °C for 10 minutes. Proteins were separated using an 8% gel and electrophoretically transferred to a nitrocellulose membrane. Nonspecific binding sites were blocked with milk 5% in TBS-T for 1 hour at room temperature. The blot was incubated at 4 °C overnight with an antibody against FAPα (1:500, Abcam). Then, the membrane was incubated with anti-rabbit peroxidase conjugated secondary antibody (1:200, Thermofisher) for 1 hour at room temperature. Reactive bands were visualized with a chemiluminescent detection kit (Westar nC 2.0 Cyanagen, XLS075.0020) using the Chemidoc instrument (Bio-Rad). β-Actin was used as the loading control and was detected using a β-actin-specific antibody (1:500 Santa Cruz Biotechnology).
Immunohistochemistry
Immunohistochemistry for S100 and αSMA was performed to differentiate sarcoids from schwannoma and other nerve sheath tumors (NSTs) as previously described. 5,9 Additionally, immunohistochemistry for FAPα was performed using either full sections or tissue microarray blocks, according to tumor size.
Using an online sequence alignment tool (https://blast.ncbi.nlm.nih.gov/Blast.cgi) the FAPα sequences for equine were compared to sequences for human, and a 100% sequence homology indicated that the antibody for FAPα would be predicted to label equine tissues.
Three-micrometer-thick sections were dewaxed and rehydrated. For antigen retrieval, sections were immersed in 200 ml citrate buffer (pH 6.0) or in tris-EDTA (pH 9.0), and heated in a microwave oven at 750 W for two 5-minute cycles for αSMA and FAPα immunostaining, respectively. Enzymatic antigen retrieval was performed in 37 °C oven for 15 minutes with 0.05% trypsin for S-100. Slides were incubated with the primary antibody overnight at 4 °C (αSMA: mouse, monoclonal, clone 1A4, M0851, dilution 1:500 Dako; S-100: rabbit, polyclonal, Z0311, dilution 1:1000, Dako; FAPα: rabbit, polyclonal, ab53066, dilution 1:300, Abcam). The reaction was developed using a commercial streptavidine-biotine-peroxidase technique (ABC Kit Elite, Vector) and visualized with 3-amino-9-ethylcarbazole (Dako). Slides were counterstained with Harris hematoxylin. Positive controls consisted of normal equine intestine for αSMA, equine melanoma for S100, and both a WB-positive equine sarcoid and an equine squamous cell carcinoma for FAPα. As a negative control, the primary antibody was replaced with an isotype-matched non-relevant antibody.
For FAPα, cells were considered positive when cytoplasmic labelling was observed, while any nuclear staining was considered unspecific. 1
Tissue Microarray Technique
Ten sarcoid cases were selected for tissue microarray validation as previously described. 15,18 From each case, both a full section and a punch section were used to perform IHC for FAPα. IHC positivity was assessed blindly and the results of IHC on full section and tissue microarray were compared to assess consistency.
First, representative areas containing neoplastic tissue without necrosis, inflammation, hemorrhage, or non-neoplastic tissue were selected from the HE-stained sections. Selected areas were marked on the slide and compared with the donor block to identify the sampling area, then were punched with a 3 mm diameter biopsy punch. Tissue microarrays were constructed allocating the obtained tissue cores into new empty receiving paraffin blocks. One tissue core per tumor was used for the construction. A sector map was then created to recognize the position of each tumor core. Each tissue microarray contained 8 tumor cores and 1 canine liver core as a landmark. From each tissue microarray, a HE-stained section was evaluated to confirm the presence of the selected tissue. After validation of the tissue microarray technique, IHC for FAPα was performed in each case; full sections were used for small tumor samples (<5 mm) while large tumor samples were performed on tissue microarray.
Polymerase Chain Reaction
Tumor tissue was obtained from FFPE blocks. Briefly, multiple 4-µm sections were cut and purification was carried out using NucleoSpin DNA FFPE XS kit (Macherey Nagel) following manufacturer’s instructions. To test for successful DNA extraction and presence of BPV-1/-2-/13, all DNA isolates were subjected to qualitative β-actin 19 and E2/E5 PCR respectively. The primers BPV1213F 5′-CCACTACCTCCTGGAATGAAC-3′ and BPV1213R 5′-GGCAGACCTGTACAGGAGCA-3′ were designed to amplify a 219 base pair region of the E2/E5 open reading frames (ORF). Thermal profile for amplification consisted in initial denaturation step at 94 °C for 5 minutes followed by 35 cycles with 3 steps: 94 °C for 30 seconds, 58 °C for 30 seconds, and 72 °C for 30 seconds with a final extension time of 72 °C for 7 minutes. Six amplicons of positive samples were sequenced (Bio-Fab Sequencing Service). The sequences were aligned to the E5 ORF sequence of BPV-1, BPV-2, and BPV-13 (GenBank Accession No. NC_001522.1, M20219, and MF741676, respectively), and Pairwise distances were calculated using BioEdit (https://bioedit.software.informer.com/7.2/).
Chromogenic In Situ Hybridization
Chromogenic in situ hybridization was performed using the RNA scope kit (Advanced Cell Diagnostics). The target genes and probe regions were designed to hybridize to messenger RNA (mRNA) and viral genomic DNA of both BPV-1 and BPV-2 as previously reported. 10 Three-micrometer-thick sections were deparaffinized in xylene, followed by dehydration in ethanol. Following endogenous peroxidase blocking, sections were incubated in 200 ml 1X Target Retrieval solution (Advanced Cell Diagnostic) maintained at 100 °C to 103 °C using a water bath for 15 minutes, rinsed in deionized water, and immediately treated with 4 drops of protease plus (Advanced Cell Diagnostic) at 40 °C for 30 minutes in a HybEZ hybridization oven (Advanced Cell Diagnostics). Target probe (4 drops) was added to the slides and hybridized at 40 °C for 2 hours in the HybEZ oven. Amplification steps were performed at first in HybEZ hybridization oven at 40 °C for 30 minutes (from AMP1 to AMP4) than at ambient temperature for 30 and 15 minutes for AMP5 and AMP6, respectively. Chromogenic detection was obtained by adding Red-working solution (60 µl) for 10 minutes at room temperature. Slides were counterstained with Gill’s hematoxylin and mounted with VectaMount (Vector Laboratories). Ec-PPIB probe (Equus caballus peptidylprolyl isomerase B [cyclophilin B] [PPIB] mRNA) was used as positive control to assess gene housekeeping while Bacillus subtilis strain SMY methylglyoxal synthase dihydrodipicolinate reductase (dapB) gene was used as negative control. Negative control consisted in equine cutaneous lymphoma. To analyze the probe signal a semiquantitative score system was developed, utilizing the estimated number of punctate dots present in 10 high power fields (HPFs) visible at 400× magnification (2.37 mm2). A score of 0 indicated absence of positive cells, a score of 1 indicated low numbers (20% to 50%) of positive cells, a score of 2 identified an intermediate number (50% to 80%), and a score of 3 indicated that a high number (80% to 100%) of positive cells. 10
Statistical Analysis
For statistical analysis, χ2 test (xy contingency table) was applied with the level of significance set at P < .05. All tests were performed using GraphPad Prism 8.02.
Results
Signalment and Clinical Data
The study included 74 sarcoids from 62 equids (49 horses, 4 donkeys, and 9 ponies). Equids ranged from 2 to 17 years old with a median age of 8 ± 0.5 years; 21 were female (28%), 36 (49%) were male, and 17 were of unknown sex. Among horses, Warmbloods (11%), Thoroughbreds (8%), and Cobs (8%) were the most represented breeds.
Of the 62 equids in the study, 54 had one tissue sample included in the study and 8 had multiple tissue samples ranging from 2 to 4 per horse. Sarcoid clinical subtype was known in all cases and was categorized as occult (4/74, 5%), verrucous (16/74, 22%), fibroblastic (9/74, 12%), nodular (35/74, 47%), and mixed (10/74, 13%). Sarcoids were located on the head/neck region (23/74, 31%), genital area (23/74, 31%), trunk (15/74, 20%), limbs (12/74, 16%), and not known (1/74, 1%; Supplemental Table S1).
Histological Features
Histological features were evaluated in all cases and in relation to the clinical subtypes. Distribution of the histological features according to clinical subtype is summarized in Table 1. The predominant feature in occult sarcoids was the presence of epidermal hyperplasia, picket fence arrangement, and interstitial fibroblast pattern which were present in 4/4 cases. Verrucous sarcoids were predominantly characterized by interstitial fibroblast pattern in 15/16 cases (94%), hyperkeratosis in 12/16 cases (75%), and epidermal hyperplasia with rete pegs in 16/16 cases (100%). The most prominent feature of nodular sarcoids was the presence of clear separation between the dermis and the epidermis, often with epidermal thinning observed in 22/35 cases (51%), while all the other features were detected in variable proportions. The most prominent feature in fibroblastic sarcoids was the presence of epidermal ulceration observed in 9/9 cases, while the other epidermal and dermal features were present in variable proportions.
Histological features of 74 sarcoids according to the clinical typea.
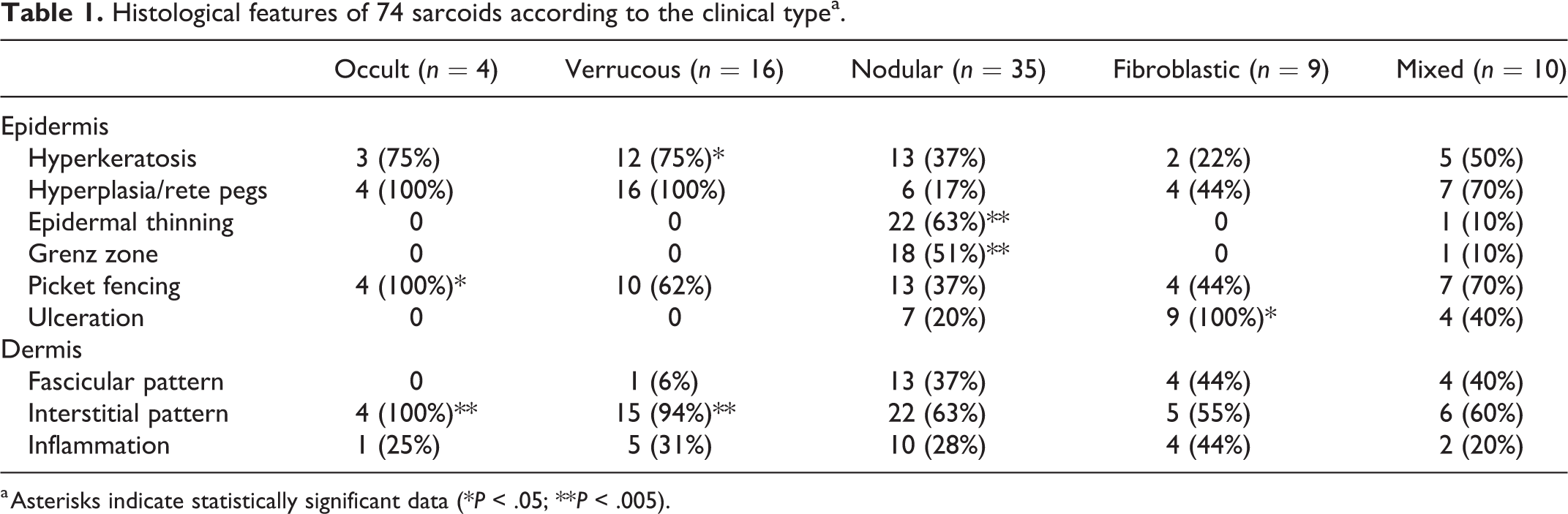
a Asterisks indicate statistically significant data (*P < .05; **P < .005).
To verify the presence of distinctive features in each clinical subtype, the frequency of each histological feature was evaluated according to the clinical aspect. Hyperkeratosis was significantly associated with verrucous type (P = .0003), while picket fence arrangement was significantly associated with the occult type (P = .0297). The presence of separation of the tumor from overlying dermis and epidermis was significantly associated with the nodular type (P = 0.0001). Regarding the fibroblast arrangement, the interstitial pattern was significantly associated with both the verrucous and occult subtypes (P = 0.0003 and P = 0.0014, respectively). No other histological features were significantly associated with clinical subtypes.
Immunohistochemistry
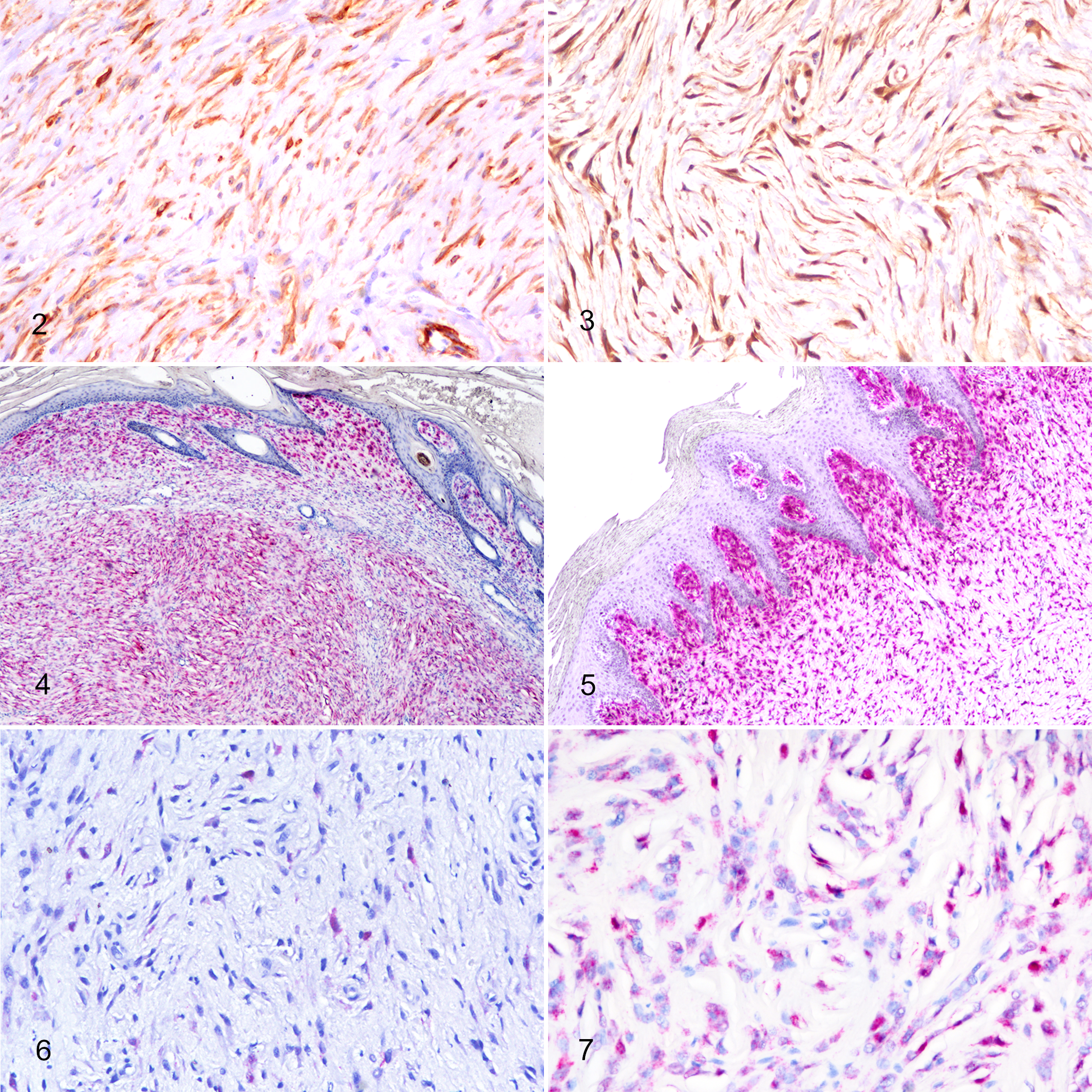
All 74 equine sarcoids were negative for S100 and 28/74 (38%) tumors were positive for αSMA displaying variably intense cytoplasmic positivity (Fig. 2). Three different positivity patterns were observed, diffuse intratumoral, peritumoral, and subepithelial. According to the histologic subtypes, αSMA positivity was recorded as follows: 1/4 occult (25%), 5/16 verrucous (31%), 12/35 nodular (34%), 4/9 fibroblastic (44%), and 5/11 mixed (45%). No significant association was observed between either positivity or distribution of αSMA labelling and clinical subtype.
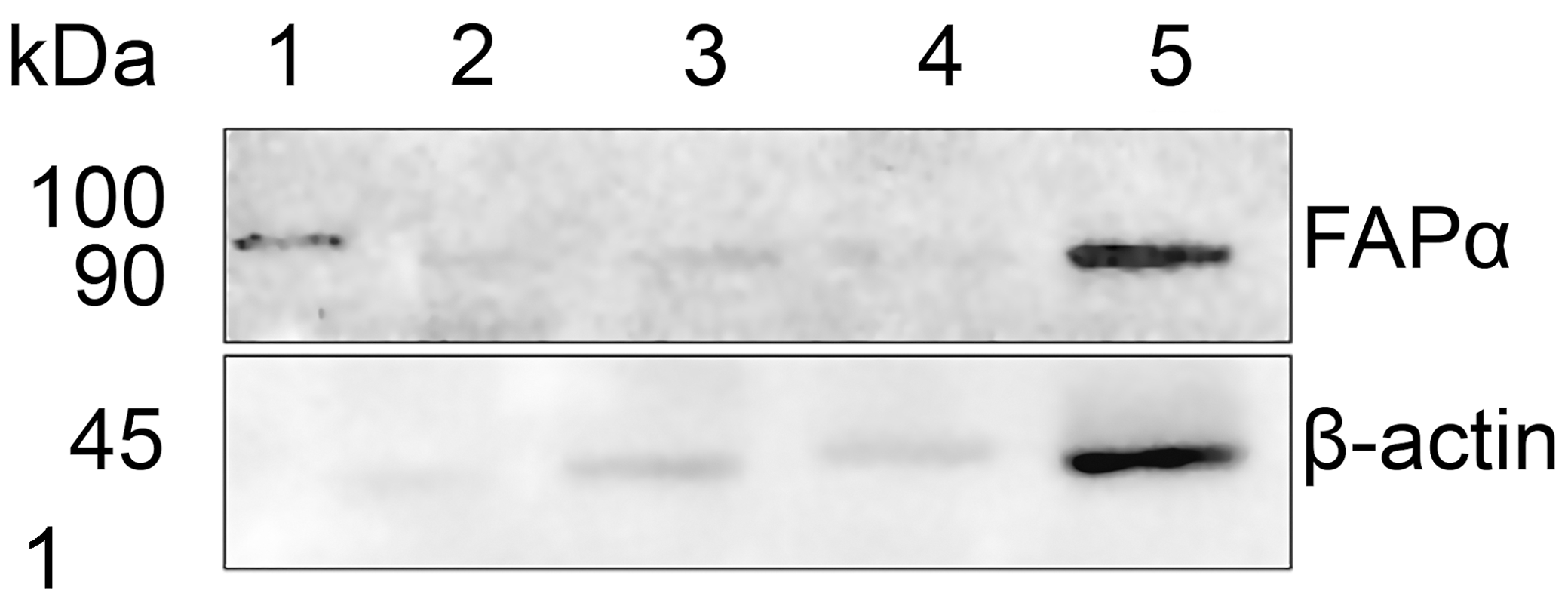
Western blot analysis of protein lysates from equine dermis (lanes 2 and 3), intestine (lane 4) and sarcoid (lane 5). Marker proteins are shown in lane 1. The polyclonal antibody to fibroblast-associated protein-α (FAPα) revealed a strong band at 90 kDa protein in equine sarcoid and weaker band in other tissues. An antibody to β-actin was used as a loading control.
Sarcoid, skin, horse.
Western blot analysis using the polyclonal antibody to FAPα revealed a 90 kD protein that corresponded to the molecular weight of FAPα. A strongly positive band was detected in the sarcoid sample, while in the dermis and intestine a weaker signal was present (Fig. 1). FAPα specificity for equine fibroblasts was confirmed by positive cytoplasmic and nuclear labelling of sarcoid fibroblasts as well as stromal fibroblasts in equine squamous cell carcinoma; no labelling was observed in normal equine dermal fibroblasts. The cytoplasm of neoplastic cells were labeled for FAPα in all 74 sarcoid cases (Fig. 3).
Polymerase Chain Reaction
Seventy-four sarcoids from 62 equids were tested by PCR for BPV-1/-2/-13. In equids with multiple sarcoids, only one tumor was tested by PCR except in 2 cases. The presence of BPV-DNA was confirmed in 72/74 samples. The 2 samples that tested negative were also negative for β-actin, indicating unsuccessful DNA extraction. Furthermore, sequencing of 6 cases suggested the presence of viral DNA referable to BPV-1.
Chromogenic In Situ Hybridization
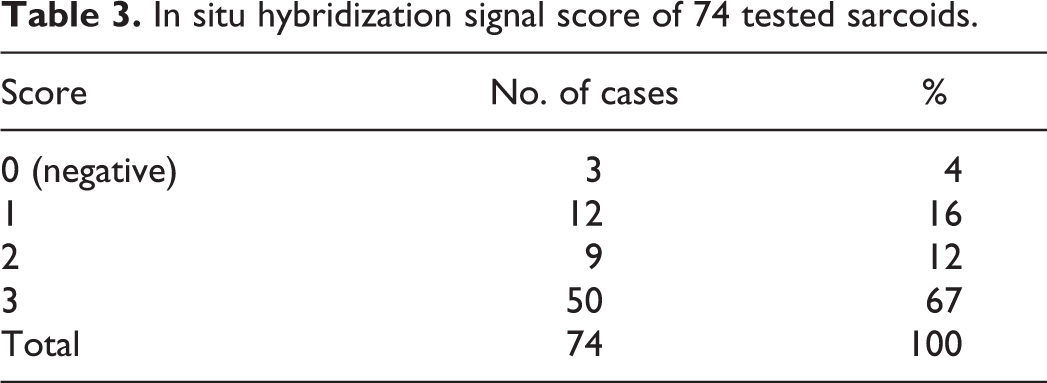
Using CISH for BPV-1 and BPV-2, a positive hybridization signal was observed in 70 of 74 (94%) cases tested. Positivity appeared as strong nuclear labeling and occasionally finely granular punctate dots in the cytoplasm of neoplastic fibroblasts. The patterns of CISH positivity were evaluated within the tumor: 61/70 (87%) cases had a diffuse, uniform BPV viral distribution throughout the tissue (Fig. 4) while the remaining 9/70 (13%) cases showed a predominantly subepithelial CISH positivity (Fig. 5; Table 2). No association was observed between BPV viral distribution and clinical subtype, although the 9 cases that had a predominantly subepithelial distribution all displayed picket fence arrangement. Of these, 5 cases were verrucous, 1 was occult, and 3 were mixed. However, no significant association was observed between the histological variables and nucleic acid distribution. In addition, no signal was observed in the epidermis overlying the neoplasm nor in the adnexa and/or dermis surrounding the neoplasm. Labeling was categorized into 4 grades as previously reported: 10 3/74 cases (4%) were negative, 12/74 cases (16%) were scored 1 (Fig. 6), 9/74 cases (12%) were scored 2, and 50/74 were scored 3 (67%) (Fig. 7; Table 3).
In situ hybridization signal distribution and pattern in fibroblasts of 70 CISH-positive sarcoids of different clinical subtypes.

Abbreviation: CISH, chromogenic in situ hybridization.
In situ hybridization signal score of 74 tested sarcoids.
All cases tested by CISH were also tested with PCR. Seventy out of 74 had concordant results, with 68/74 cases being positive for both CISH and PCR, 2 cases negative for both, 2 cases CISH positive/PCR negative, and 2 cases CISH negative/PCR positive.
Discussion
Equine sarcoids are typically described as biphasic tumors with both dermal and epidermal components. 5 In fact, epidermal hyperplasia with rete peg formation and “picket fence” arrangement of fibroblasts beneath the epidermis are considered pathognomonic features for the histological diagnosis of sarcoids. 5 As previously reported, 14 we found that the histopathologic features of sarcoids can be variable. In fact, although features such as hyperkeratosis, picket fence arrangement, and interstitial fibroblast pattern were more prevalent in the occult and verrucous clinical subtypes, there were no histologic features that allowed distinction of the clinical subtypes. Hence, the main aim of the study was to evaluate if the distribution of BPV might be associated with clinical or histologic characteristics of the neoplasm. BPV distribution, assessed by in situ hybridization, was diffuse and uniform throughout the tumors in the majority (87%) of cases, irrespective of the clinical subtype, displaying strong signal in neoplastic fibroblasts. Only in a minority (13%) of cases was BPV DNA localized in the subepithelial area, and this occurred exclusively in cases where epidermal hyperplasia and/or picket fence arrangement were present (the occult, fibroblastic, and mixed subtypes). Previous in situ hybridization studies have also revealed this subepithelial pattern, 12 but we demonstrated that there was no significant association with either clinical subtype of sarcoid or anatomic site. Therefore, we speculate that cases with stronger signal in the subepithelial areas might be related to either the phase of infection or stage of tumor development.
In this study, none of the cases displayed BPV hybridization in the epidermis overlying the tumor, in contrast with previous studies which found BPV DNA in the epidermis. 3,10 Laser microdissection and real-time PCR were previously applied to investigate the presence of BPV in the epidermis and dermis separately, and they found that keratinocytes of 40% of early-stage sarcoids harbored BPV-DNA, but in lower amounts than fibroblasts. 3 In another study, immunohistochemistry for E5 and E7 oncoprotein revealed positive labeling of both neoplastic fibroblasts and epidermal cells. 6 More recently, the presence of BPV DNA in equine sarcoids was further investigated using CISH and in a small proportion of the analyzed cases, viral nucleic acid was also detected in epidermis, sebaceous glands, and hair follicles. 10 In our study, hybridization signal for BPV was not observed in the epidermis; based on our results we hypothesize that the hyperplastic epidermal component of sarcoids may merely depend on the presence of neoplastic cells in close contact with the epidermis rather than being a direct consequence of viral infection of keratinocytes. In this respect, we argue that sarcoids should be considered purely mesenchymal rather than biphasic tumors as commonly reported. 5,9 Moreover, the non-neoplastic dermis adjacent to the tumor showed no evidence of viral DNA by CISH, demonstrating that the presence of the virus is restricted to the neoplastic cells and does not extend to the surrounding tissue.
The histologic diagnosis of sarcoids is often not challenging, although a proportion of sarcoids can be confused with other spindle cell tumors such as fibromas, fibrosarcomas, and nerve sheath tumors of Schwann cell origin. 5,9 Earlier studies found that the absence of features such as Verocay bodies or Antoni patterns distinguishes sarcoids from schwannoma. 22 Other studies proposed that a combined S100 negativity and PCR positivity for BPV DNA could be used to distinguish the 2 entities. 5 Other authors suggested that S100 can be expressed in sarcoids, having found concurrent S100 positivity and BPV DNA in 2/70 sarcoids in the study. 9 In our case series, we found no S100-positive cases, confirming that sarcoids are indeed negative to this marker. To further confirm the fibroblastic origin of sarcoid neoplastic cells, we applied a novel fibroblastic marker that labels activated fibroblasts. 8 FAPα is an integral membrane serine proteinase encoded by FAP gene that is selectively expressed in reactive fibroblasts of epithelial tumors and malignant cells of bone and soft tissue sarcomas with no expression in normal fibroblasts or in other normal stromal tissue. 17,21 In all the analyzed cases, intense and diffuse FAPα expression in neoplastic cells supported the fibroblastic/myofibroblastic origin of sarcoids. Concurrent expression of αSMA and FAPα also confirms that a subset of sarcoids has a myofibroblastic differentiation. αSMA positivity was previously observed in the majority of the cases included in the study. 5 FAP-targeted therapies have been developed to specifically target cancer-associated fibroblasts in human carcinomas. Its consistent expression in sarcoids may be a basis for developing new immunotherapeutic strategies that target this molecule. 7
In conclusion, after validating the antibody for equine tissue, we propose FAPα as a candidate immunohistochemical marker to confirm of the diagnosis of sarcoids, although its specificity for sarcoid remains unknown because other sarcomas were not evaluated in this study. We also assessed the value of CISH in establishing a diagnosis of sarcoids, confirming that this technique can be used as an alternative to PCR. However, BPV distribution does not seem to be associated with specific histologic or clinical features of sarcoids, and thus the distinction of the different subtypes of sarcoids is based on the clinical findings.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211022696 - Fibroblast-associated protein-α expression and BPV nucleic acid distribution in equine sarcoids
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211022696 for Fibroblast-associated protein-α expression and BPV nucleic acid distribution in equine sarcoids by Giorgia Tura, Federica Savini, Laura Gallina, Roberto M. La Ragione, Andy E. Durham, Martina Mazzeschi, Mattia Lauriola, Giancarlo Avallone, Giuseppe Sarli, Barbara Brunetti, Luisa V. Muscatello, Cinzia Girone and Barbara Bacci in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
