Abstract
Ten equine skin tumors that had been classified as schwannomas on routine histological examination were analyzed by polymerase chain reaction for bovine papillomavirus DNA. All 10 were positive for bovine papillomavirus 1 or 2, and all 10 were immunohistochemically negative for S-100 protein and strongly positive for vimentin. Nine tumors were moderately positive for laminin and 8, for smooth muscle actin. Five tumors were variably and weakly positive for type IV collagen. The lack of S-100 protein expression made Schwann cells an unlikely cell of origin, as opposed to peripheral nerve sheath tumors, which typically express S-100 protein, at least in some neoplastic cells. The immunohistochemical reactivity is consistent with myofibroblastic origin of the neoplastic cells, although smooth muscle cell or pericyte origin cannot be ruled out. These tumors represent an atypical form of equine sarcoid. Polymerase chain reaction for bovine papillomavirus and S-100 immunohistochemistry are strongly recommended for all equine skin tumors with histological characteristics typical of schwannoma or peripheral nerve sheath tumor.
Keywords
Equine sarcoids, attributed to nonproductive infection with bovine papillomavirus (BPV) type 1 or 2, 1,13 are the most common cutaneous neoplasm in the horse. Although they can occur anywhere on the body, the sites most frequently involved are the legs, ventral abdomen, and head. 7 Histologically, equine sarcoids are biphasic tumors with a pseudoepitheliomatous epidermal and fusiform dermal component. The latter is generally arranged in whorls, tangles, and/or herringbone patterns, but a neurofibroma-like proliferation has also been described. 7
Peripheral nerve sheath tumors (PNSTs) have been reported in cutaneous and noncutaneous locations in horses. 9,14,16 –18 Schwannoma, the most common equine PNST, typically has Schwann cell proliferation with Antoni type A and B patterns and Verocay body formation. 16,17 Other PNSTs can appear as well-demarcated cellular neurofibromas or as diffuse collagenous and infiltrative neurofibromas. 9,15 Although there are no specific immunohistochemical markers for PNST, expression of vimentin and S-100 protein is generally present. 14,16
Signalment and Clinical Features
Dermal tumors from 10 horses were collected at the University of California–Davis and the Veterinary Pathology Laboratory of Ghent University between 1987 and 2002. The average age of the horses was 7.5 years (range, 3 to 15 years). Five tumors were on the head (2 on the eyelids, 2 on the ears, 1 submandibular); 3 were in inguinal skin; 1 was in axillary skin; and in one case, the location was unknown.
Histologic Findings
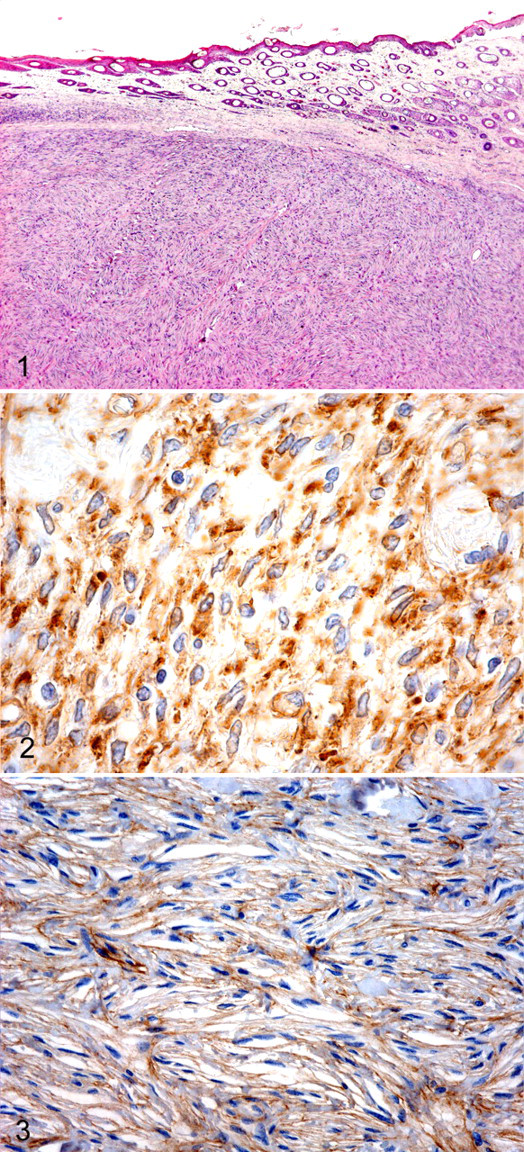
Tumors had been surgically removed, immersion fixed in 10% buffered formalin, embedded in paraffin wax, processed for routine histology, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). In one specimen, the epidermis was absent. All other specimens contained relatively well-circumscribed nodular dermal masses without epidermal changes. The masses were composed of interlacing bundles of neoplastic spindle cells arranged in a storiform pattern (Fig. 1 ). Most tumors contained palisading bundles in which the nuclei were prominently aligned, consistent with an Antoni type A pattern. The neoplastic cells had moderate amounts of fibrillar amphophilic cytoplasm, indistinct cell borders, and oval nuclei with finely stippled to vesiculated chromatin. There was moderate to marked anisokaryosis and an average of 1 mitotic figure per high-power field (40×). Based on the histological characteristics, all tumors were originally diagnosed as schwannomas.
Immunohistochemistry
All tumors were subjected to immunohistochemistry as described. 2 Four-micrometer sections were deparaffinized and treated with 3% hydrogen peroxide–methanol for 5 minutes to quench endogenous peroxidase activity. Next, the sections were incubated with the following antibodies: monoclonal mouse anti-bovine vimentin (M0720, clone 3B4, Dako, Glostrup, Denmark; 1:400), polyclonal rabbit anti-bovine S-100 protein (Z0311, Dako; 1:1600), polyclonal rabbit anti-laminin (Z0097, Dako; 1:320), monoclonal mouse anti-human smooth muscle actin (clone 1A4, Dako; 1:200), and monoclonal mouse anti-human collagen type IV (clone CIV94, Zymed Laboratories, San Francisco, CA, 1:10). Binding sites were visualized in a three-step biotin–streptavidin–horseradish peroxidase complex method.
Strong positive cytoplasmic reactivity for vimentin was demonstrated in all 10 tumors, yet all tumors were negative for S-100 protein. Mainly membranous and occasionally cytoplasmic laminin reactivity (Fig. 2 ) was present in 9 tumors (Nos. 1–9); smooth muscle actin was expressed in the cytoplasm of 8 tumors (Nos. 1, 3, 4, 6–10). Five tumors (Nos. 1, 2, 5, 7, 9) expressed type IV collagen with marked variation in staining intensity and distribution (Fig. 3 ). A control PNST stained positive for S-100, vimentin, and laminin but negative for collagen IV and smooth muscle actin.
Polymerase Chain Reaction
BPV detection was performed by polymerase chain reaction (PCR), both on complete paraffin sections and on trimmed dermal nodules without the surrounding tissue. A 5-μm section of paraffin-embedded tissue was deparaffinized with three xylene and two ethanol washes. DNA isolation was then performed with a commercial protocol (Puregene DNA Isolation Kit, Gentra Systems, Minneapolis, MN) resulting in a pellet of DNA, which was dissolved in a final volume of 100-μl DNA hydration solution. The PCR was used to detect BPV DNA as described. 11 Briefly, an oligonucleotide primer set complementary to a common sequence of the E5L2 open reading frame of BPV-1 and BPV-2 was used (upper primer: 5′-CAAAGGCAAGACTTTCTGAAACAT-3′; lower primer: 5′-AGACCTGTACAGGAGCACTCAA-3′). The initial denaturation was performed at 94°C for 10 minutes. Thirty-five amplification cycles were performed. Each cycle involved a denaturation step of 30 seconds at 94°C, followed by 30 seconds of primer annealing at 58°C and 1 minute of primer extension at 72°C. After the last cycle, the PCR mix was heated for 10 minutes at 72°C to complete the final extension. Samples were considered positive for BPV DNA when a specific fluorescent band at 250 base pairs was present. The PCR products were cloned (pCR 2.1 vector, Invitrogen, Merelbeke, Belgium) and sequenced for verification on an Applied Biosystems 3730xl DNA analyzer (96 capillaries) with BigDye Terminator 3.1 Cycle Sequencing Kit (Applied Biosystems, Lennik, Belgium). As confirmed by histology and positive S-100 immunohistochemistry, samples of normal equine skin and equine PNST were processed, with the test samples as negative controls. Cloned BPV-1 and BPV-2 were used as positive controls. 4 In all cases, BPV DNA was amplified both in tumor samples with surrounding tissue and in trimmed dermal masses (Fig. 4 ). Six samples had a sequence consistent with BPV-1 and 4 samples, with BPV-2 (data not shown).
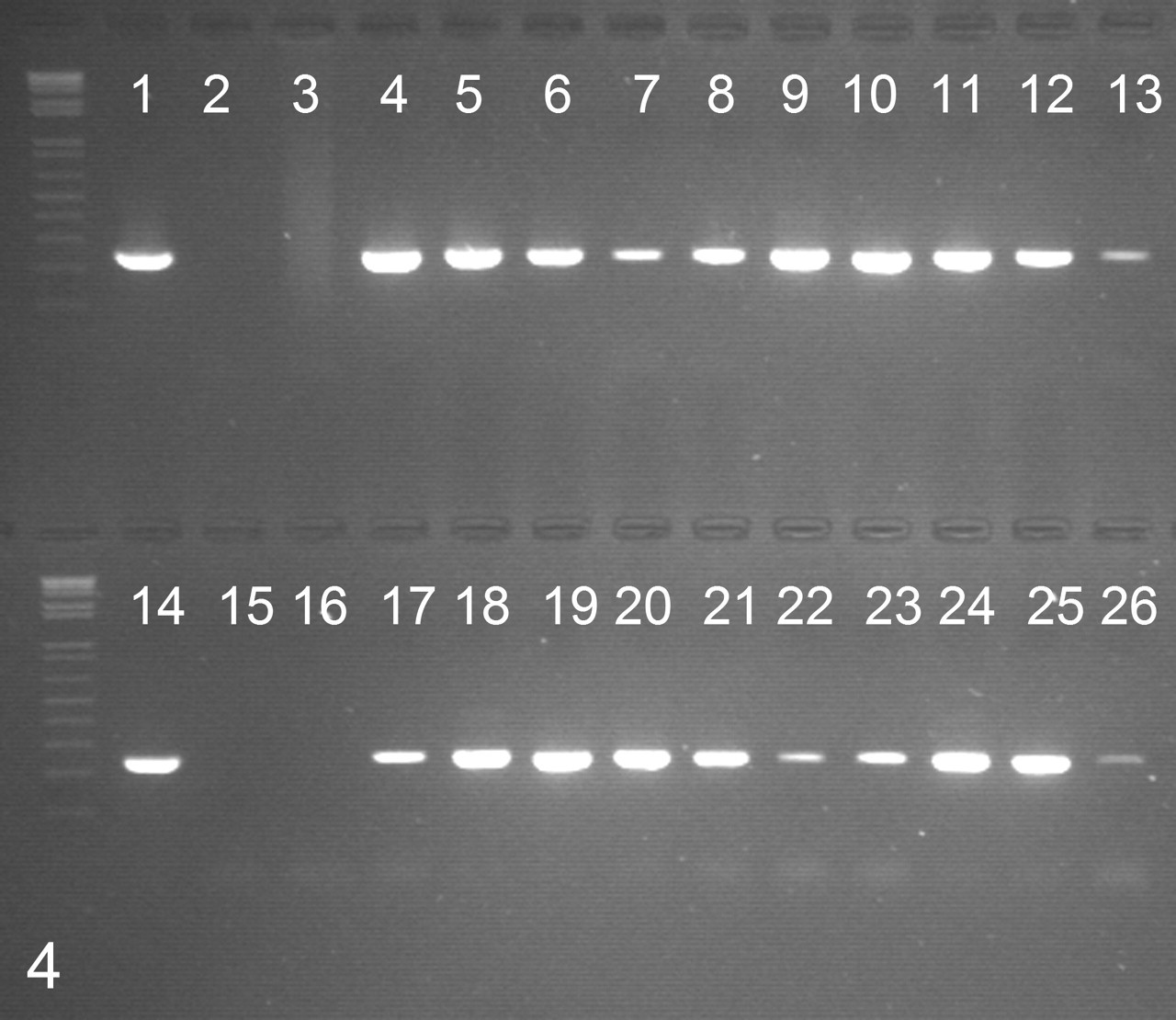
Detection of bovine papillomavirus DNA by polymerase chain reaction with E5L2 consensus primers. Positive samples show a fluorescent band at 250 base pairs. 1, cloned bovine papillomavirus type 1 DNA; 2, water; 3, peripheral nerve sheath tumor; 4–13, case Nos. 1–10, complete paraffin sections; 14, cloned bovine papillomavirus type 2 DNA; 15, water; 16, normal equine skin; 17–26, case Nos. 1–10, trimmed nodules without the surrounding tissue.
Discussion
In this study, 10 equine sarcoids so resembled schwannomas that they were originally misdiagnosed on routine histopathology. Without additional diagnostic techniques or awareness by pathologists of this mimicry, the association with BPV infection would have been missed. Because the prognosis and treatment for schwannomas differ from those for BPV-associated sarcoids, their distinction has significant implications. Sarcoids frequently recur after inadequate surgical excision and thus require ancillary treatment, such as chemotherapy or immunotherapy. 12
In this study, a PCR amplification of a consensus BPV sequence allowed the identification of the tumors as equine sarcoids. Detection of BPV DNA in skin samples may also be the result of superficial contamination or latent BPV infection. 3 However, surface contamination unlikely caused the positive results in the present study, because PCR demonstrated BPV DNA in the trimmed sections of the tumor nodules without surrounding tissue. Although latent BPV infection is common, equine sarcoids have a much higher BPV load than that of normal skin (up to 10,000 times). 3 Although no quantitative analysis was performed, it seems unlikely that all 10 BPV-positive tumors were the result of latent BPV infection.
The tumors in this series were originally diagnosed as schwannomas based on routine histology; however, the true cell of origin remains unknown. There are no specific immunohistochemical markers for schwannomas. Positive immunohistochemistry for glial fibrillary acidic protein, type IV collagen, or laminin is inconsistent and not reliable for diagnosis. 10 Vimentin is more consistently expressed in Schwann cells but identifies all mesenchymal cells. Most PNSTs express S-100 protein; therefore, its absence tends to eliminate PNST from the differential diagnosis. Probably the most definitive evidence of Schwann cell origin is the ultrastructural demonstration of a basal lamina around neoplastic Schwann cells and their processes. However, not all PNSTs contain an exclusive population of such cells, and intermingled fibroblasts and other non-basal-lamina-forming cells can lead to diagnostic uncertainty. 10 The basal lamina around Schwann cells consists predominantly of laminin and, to a far lesser degree, type IV collagen. 8
Equine dermal schwannomas have been identified on the basis of the ultrastructural presence of a distinct basal lamina surrounding the neoplastic spindle cells and immunohistochemical S-100 expression (though variable in intensity and distribution). 6,16 The lack of S-100 protein immunoreactivity in our cases tends to rule out Schwann cell origin. Nevertheless, most of the tumors were positive for laminin and/or type IV collagen, which is consistent with the presence of a basal lamina. The positivity for laminin and smooth muscle actin and the negative immunolabeling for S-100 protein could reflect smooth muscle cell or pericyte origin. Alternatively, this immunolabeling pattern could indicate a myofibroblast origin. Indeed, the intercellular spaces between myofibroblasts variably and weakly express laminin and type IV collagen. 5 Earlier studies described a discontinuous basal lamina surrounding myofibroblasts; however, more recent work reported the presence of fibronexus junctions rather than a basal lamina. The fibronexus is the point of convergence on the cell surface of intracellular myofilaments and the extracellular fibronectin fibril and is an important marker for myofibroblastic differentiation. 5
In conclusion, equine dermal neoplasms that mimicked schwannomas were confirmed as equine sarcoids by PCR and sequencing of BPV. This is a previously unrecognized presentation for equine sarcoid, lacking interaction with the epidermis. Other than the lack of S-100 protein expression, a uniform immunohistochemical reactivity pattern was not identified for this entity. Important characteristics of these tumors—including clinical progression, prognosis, and cell origin—require further investigation. Immunohistochemistry for S-100 protein and molecular detection of BPV are strongly recommended as ancillary diagnostic tools in all equine skin tumors with histologic features of schwannoma.
Footnotes
Acknowledgements
We thank Isabel De Bleeker for immunohistochemical assistance, Christian Puttevils for the photographs, and Cindy De Baere and Sarah Loomans for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
