Abstract
Equine sarcoid is the most common skin tumor of horses. Clinically, it occurs as a locally invasive, fibroblastic, wart-like lesion of equine skin, which has 6 clinical classes: occult, verrucose, nodular, fibroblastic, mixed, and malignant. Sarcoids may be single but multiple lesions are more frequent. The typical histological feature is increased density of dermal fibroblasts which form interlacing bundles and whorls within the dermis. Lesions are mostly persistent, resist therapy, and tend to recur following treatment. In general, sarcoids are not fatal but their location, size, and progression to the more aggressive form may lead to the withdrawal of a horse from use and serious infringement of their welfare leading to the loss of valuable animals. Bovine papillomavirus (BPV) type 1 and less commonly type 2 contribute to the development of equine sarcoid. The viral genome and proteins are detected in a high percentage of cases. Furthermore, viral oncoprotein activity leads to changes in the fibroblastic tissue similar to changes seen in other types of tumors. Equine sarcoids are characterized by a loss of tumor suppressor activity and changes allowing abnormal formation of the affected tissue, as well as y immune defense abnormalities that weaken the host’s immune response. This impaired immune response to BPV infection appears to be crucial for the development of lesions that do not spontaneously regress, as occurs in BPV-infected cows.
Equine sarcoid is the most common skin tumor in equids worldwide. 72 It is non-metastasizing, yet locally aggressive and has been reported in horses, donkeys, mules, and zebras of all ages, types, and colors. 27,51,60 Sarcoid tumors were first described and characterized as a separate clinical entity in 1936 by Jackson. 45 He defined the sarcoid as “a unique locally invasive, benign neoplastic like tumor of the skin with a variable epidermal component which has a high propensity for recurrence.” Currently, equine sarcoid represents a significant problem in equine dermatology. The reported incidence ranges from 12% to 67% of all neoplasms, 8,102 and a recent study reported a 24% incidence in the United Kingdom. 52 It is estimated that these tumors affect between 1% and 11.5% of all horses. 51,101 Equine sarcoids cause a significant horse welfare problem that requires intensified research since its cause and progression are not fully understood.
The Role of Bovine Papillomavirus in Induction of Equine Sarcoid
Bovine papillomavirus (BPV) is widely accepted as an essential extrinsic factor contributing to equine sarcoid development. In cattle, dermal fibropapillomas induced by BPV usually regress spontaneously due to a local immune response; however, horses do not appear to have an effective immune response to BPV infection. Phylogenetic analysis of the BPV-1 transcription promoter region (long control region [LCR]) demonstrates geographical segregation into an ancestral group (African, South American, and Australian samples), and a more recently derived, mainly European clade. 112 From the BPV family, BPV-1 viruses and less frequently BPV2 are associated with and believed to induce equine sarcoid. 105 BPVs are in the Papillomaviridae family. They form small, 50 to 60 nm diameter, nonenveloped icosahedral capsids containing a single circular double-stranded DNA genome of about 7.2 to 8.0 kb. 125 The BPV genome consists of 6 early (E) and 2 late (L) genes. 25,29,83 Early genes encode proteins that possess transforming capability—E5, E6, and E7—as well as proteins that regulate their replication and transcription—E1, E2, and E4. 27 The late region encodes structural proteins: L1, which forms pentamers termed capsomers, and L2, which appears in the capsid structure as a monomer. 76 Among others, E2, E5, E6, and E7 function as the major oncoproteins, stimulating sarcoid development. 83
Olson and Cook 80 were the first to suggest the relationship between BPV and equine sarcoid. BPV primarily causes warts in its natural host, the cow. Its virulence toward horses provides a rare, contemporary example of cross-species transmission of papillomavirus infection. 109,112 Since Jackson’s observation, 45 a number of studies have shown the presence of BPV types 1 and 2 in equine sarcoids. One of the first studies to detect DNA fragments of BPV in affected horses used DNA hybridization techniques. 4,6,56,58,111 In addition, the presence of BPV-1 and BPV-2, 12,68,69,78,81,104 as well as BPV-13, 59 have been demonstrated in several PCR analyses in tissues from sarcoid-positive horses. Lately, the results of in situ hybridization have shown the presence of viral nucleic acid in fibroblastic cells of all examined cases (18 sarcoids). 37 Various reports have demonstrated the presence of BPV in most cases of equine sarcoid, ranging from 73% 12 through 88% to 91% 69 to 95% to 100%. 81,104 In horses, the BPV seems not to be found only in sarcoids, but the presence of BPV DNA in normal skin or nonsarcoid lesions is not consistent. BPV DNA has not been detected in cutaneous squamous cell carcinoma, melanoma, intestinal squamous cell carcinoma, or papillomas of horses, 28,81 but its presence has been reported in some cases of equine dermatitis. 6,28,121 Studies of Gaynor et al 37 using in situ hybridization showed a lack of viral DNA in nonsarcoid tissues (mainly in hyperplastic and neoplastic lesions). On the other hand, Bogaert et al 16 reported the presence of the BPV DNA in the normal skin of 24 of 42 horses (57%). It was detected mainly in sarcoid-affected horses and horses living in contact with cattle (73%). The BPV DNA was also reported in 50% of the horses living in contact with sarcoid-affected horses and in 30% of healthy horses that did not have contact with affected animals. Another study by Bogaert et al 13 showed that all examined cases of schwannomas (n = 10) were positive for BPV DNA. In addition, Epperson and Castelman 34 demonstrated BPV DNA in 59% of nerve sheath tumors, 37% of fibrosarcomas, and 22% of other tumors (myxosarcomas, fibromas, and other sarcomas). Thus, with some exceptions, the presence of BPV viral DNA is highly associated with equine sarcoids. However, it cannot be used as the sole indicator for sarcoid due to its presence in other lesions and even in normal skin.
Further analysis has shown the presence of BPV mRNA and proteins in equine sarcoids. RNA containing both BPV early (E2, E5, E6, E7) and late (L1) phase transcripts has been identified in equine sarcoids. 17,78,114 However, Wilson et al 114 noticed that the late gene mRNAs was less than 0.3% of the transcribed BPV RNA. On this basis, they postulated that BPV infection in equids is abortive, and it does not undergo productive infection in the epidermis overlying equine sarcoids at levels comparable to those found in its natural cattle host. This is in line with the very first studies based on in situ hybridization which failed to detect BPV DNA in the epidermis of sarcoids. 4,54,55 Recently, Gaynor et al 37 using the same technique showed lack of BPV nucleic acid within the epidermal cells even when it was present in the fibroblast layers. Western blot analysis, immunohistochemistry, and immunofluorescence showed the presence of E5, E7, and L1 proteins in the affected tissues. 20,21,26 In contrast to Wilson et al, 114 Brandt et al 20,21 and Bogaert et al 15 provided evidence that the L1 late phase protein is present not only in biopsies of the affected dermis but also in the epidermis, where virion formation and release may occur, suggesting that BPV infection in equids is productive. Moreover, the hypothesized presence of productive BPV virus infection is supported by the discovery of a deletion of 4 residues from within the L2 protein in BPV DNA isolated from horses, that may prove the existence of a distinctive equine subtype of BPV1. 1,114,115 However, electron microscopy and immunohistological studies, which detect virus particles as well as late proteins in bovine fibropapillomas, failed to demonstrate the presence of BPV virus or late proteins within sarcoids. 96,103,106,114,115 The productivity of BPV infection in horses remains questionable, and it remains unexplained why the immune response in horses fails to eliminate the virus as it does in cows.
The E2, E5, L1, and L2 DNA levels, 41 E2, E5, E6, and E7 mRNA abundance, 17 as well as E5 protein content 26 have been positively correlated with severity of the disease. The highest expression for these molecules was observed in the clinically aggressive fibroblastic variant of sarcoid, in comparison to their expression in the cells of quiescent tumors. 41 Additionally, nodular sarcoids show the highest expression levels of each of the above-mentioned BPV mRNAs, followed by verrucous, fibroblastic, and occult tumors.
In horses, BPV inoculation leads to a sarcoma-like lesion. This confirms the involvement of BPV in the development of equine sarcoid. In 2011, about 50 years after the first experiments on the induction of sarcoid by deliberate infection, 80,85,113 Hartl et al 43 confirmed the results of their predecessors. They inoculated 4 foals with infectious BPV-1 virions. This caused a viremic phase, which led to the formation of pseudosarcoids (sarcoid-like granulomas) only at sites inoculated with virions. 43 In an in vitro experiment, equine fetal palate fibroblasts were transfected with BVP-1 DNA. The obtained cells had the characteristics of horse sarcoid cells—morphological transformation, faster proliferation, extended viability, and growth independent of the substrate compared to uninfected cells. 123
The crucial role of BPV in sarcoid progression may provide a protective effect of vaccination with chimeric virus-like particles. Immunization of horses with BPV-1 L1 papillomavirus-like particles leads to development of a BPV-1-neutralising antibody and, in consequence, induces long-lasting protection against experimental BPV-1 virion-induced disease. 1,39,40,42 Similar effects were observed in donkeys vaccinated when they already had a sarcoid. Animals immunized with papillomavirus-like particles containing BPV-1 L1 and E7 proteins showed a tendency toward enhanced tumor regression and reduced progression. 7 These promising results provide the possibility of immunotherapy directed against BPV virus in equids with sarcoids.
Molecular Features
The invasiveness of sarcoids is associated with dysregulation in the functioning of matrix metalloproteinases (MMPs). 75,119,120,124 MMPs, a family of zinc-dependent endopepdidases, exhibit higher expression during cancer invasion. MMPs have been demonstrated to play a crucial role in initiating degradation of the basement membrane and extracellular matrix, enabling the invasion of tumor cells. Human papillomavirus type 8 (HPV-8) E7 protein has been shown to stimulate overexpression of MMP-1, MMP-8, and membrane type 1 MMP (MT1-MMP), leading to invasion of human keratinocytes. 1 Similarly, a large-scale analysis revealed an elevated level of MMP-1 transcript in equine fibroblasts transformed by BPV-1. 124 Fibroblasts in sarcoids are invasive and destroy the matrix. Their invasiveness is associated with increased expression of MMP-1, MMP-2, and MMP-3. 75,119,120 Expression of MMP-1 can be increased by BPV-1 oncoproteins E6 and E7 through the activator protein 1 (AP-1) and by E5 via an indirect mechanism. 119 In line with these studies are results by Martano et al 65 who reported abnormalities in turnover of the extracellular matrix deposition and degradation, which were the result of altered expression of MMPs (MMP- 2, MMP-9, and MMP-14) and tissue inhibitor of metalloproteinase 2 (TIMP-2) in equine sarcoid tissues. Additionally, MMP production is also stimulated by vascular endothelial growth factor (VEGF), which is strongly expressed in equine sarcoid. 66 MMPs in equine sarcoid cells likely contribute to the invasion of tumor cells into the surrounding tissues allowing localized spread of viral DNA. The invasion and migration of affected cells to sites distant from the main tumor may be one of the reasons for the high propensity to relapse, which leads to poor prognosis for complete recovery. 120
Over the past decade, studies on equine sarcoid have provided further insights on its pathogenesis. For instance, the transformation activity of BPV is associated with binding of E5 protein to platelet-derived growth factor β receptor (PDGFβ), causing its phosphorylation and activation. 2 Indeed, PDGFβ activation, which is associated with pathological cell proliferation, morphological changes, and motility, plays a great role in the progression of horse sarcoids. 19 PDGFβ affects the activation of the PI3K-AKT signaling pathway. This pathway exhibits elevated activity in various human tumors, because it controls apoptosis, anoikis, and cell cycle progression. Binding E5 oncoprotein to the PDGFβ possibly leads to activation of transforming signal by c-Jun N-terminal kinases (JNKs), which are involved in augmentation of AP-1 transcription factor, which in turn mediates metalloproteases transcription. Activation of PDGFβ has also been observed in equine sarcoid derived cell lines in which PDGFβ was phosphorylated and bound downstream molecules related to Ras-mitogen-activated protein kinase-ERK pathway, thus resulting in Ras activation. 2
PDGFβ may also trigger the activation of the p38 mitogen-activated protein kinase (MAPK). Indeed, E5 and E6 oncoproteins have been shown to activate p38 MAPK by enhancing its phosphorylation in equine sarcoids. This process is essential for transformation of equine fibroblasts by BPV-1 and further proliferation of sarcoid cells. 118
There are conflicting publications regarding the role of p53 in equine sarcoid pathogenesis. p53 is a transcription factor and is considered a “guardian of the genome” because it is activated in response to DNA damage and is involved in the regulation of DNA repair, cell cycle, apoptosis, cellular metabolism, and initiation of cell death if DNA damage is unrepairable. 61 In the human papillomavirus, E6 protein has been shown to complex with p53 and to enhance its degradation. 91 Similarly, the transformation of human mammary cells with BPV E6 protein results in reduced levels of p53. 9 Some publications state that equine sarcoid progression is associated with the loss of tumor suppressor p53 function caused by BPV and leads to an imbalance between cell growth and cell death. 17,79 Immunohistochemical studies showed that in some cases of equine sarcoid, p53 was overexpressed, while in others it remains unchanged. 47,67 Immunostaining and western blot analysis confirmed this phenomenon in an equine fibroblast sarcoid cell line; however, further analysis revealed that its function was compromised. 79 It has been established that this is due to the presence of nuclear and cytoplasmic p53 pools. While p53 is a nuclear protein, it is believed that its cytoplasmic localization completely deactivates its function. On the other hand, Bogaert et al 17 using immunohistochemical staining showed that there was no significant difference in p53 expression between the different clinical types of sarcoids and suggested that p53 expression might not be associated with the aggressive behavior of an equine sarcoid. Moreover, Finlay 36 showed using western blot analysis that p53 level was increased in 2 sarcoid-derived cell lines. However, immunohistochemical staining demonstrated that in one of these lines, nuclear p53 expression was detectable, while in the second, a distinct punctate cytoplasmic pattern with no evidence of nuclear staining was shown. Additionally, Bucher et al 24 and Nasir et al 77 demonstrated that p53 gene mutations are not associated with sarcoid development in horses and donkeys. Certainly, further studies based not only on immunohistochemistry are needed to determine the impact of BPV on p53 activity along with its nuclear and cytosolic localization in equine sarcoid cells.
Fragile histidine triad is another tumor suppressor that shows altered expression in BPV-associated equine sarcoids. Alterations of fragile histidine triad are observed in several animal cancers; however, its role is not fully understood. It possibly regulates proliferative and apoptotic cellular processes. 100 Another tumor suppressor with decreased activity in equine sarcoids is S100 calcium-binding protein A14 (S100A14), which inhibits proliferation and cancer cell metastasis. 93 Horse sarcoid samples had a significantly higher rate of DNA methylation in the S100A14A region than the corresponding normal skin tissue. Increased methylation of S100A14A may cause previously reported loss of expression of this gene in the progression of sarcoids. 94 Changes in the methylation of genes associated with carcinogenesis process play a significant role in equine sarcoid progression. The regions of both stratifin tumor-suppressor gene, a negative regulator of cell cycle that plays a key role in regulating signal transduction pathways and cell proliferation, as well as SPARC (secreted protein acidic and cysteine-rich) gene, encoding for a protein involved in extracellular matrix synthesis and promotion of cell shape changes, have different methylation patterns in equine sarcoids compared to healthy skin. 95 On the other hand, the development of equine sarcoid is associated with an elevated level of the transcription factor hypoxia-inducible factor-1 alpha (HIF-1α). 64 This transcription factor is activated in response to hypoxia and leads to angiogenesis, the development of new vessels necessary for neoplastic invasion, growth, and metastasis.
BPV-1 has been shown to influence apoptosis in cells affected by equine sarcoid. Apoptosis may be activated by infected host cells in order to avoid completing virus replication. Viruses evolved the ability to avoid triggering apoptosis through activation of signaling pathways inhibiting cell death. One such pathway involves the BAG family molecular chaperone regulator 3 (BAG3). Studies on equine sarcoid samples and on a sarcoid-derived cell line carrying the BPV-1 genome exhibited selective expression of the BAG3 protein, which has been shown to protect human cancer cells from programmed death. 31,89
Similarly to human cancers, abnormal microRNA expression has been observed in equine sarcoids. MicroRNAs are small, noncoding molecules involved in RNA silencing and posttranscriptional regulation of gene expression, which are promising biomarkers and potential therapeutic agents. Studies on equine fibroblasts transformed with the BPV-1 genome revealed 219 microRNAs with different expressions. Interestingly, 68% of these microRNAs were not expressed in affected cells in relation to controls, which suggests that in normal cells certain processes are silenced by microRNA, while BPV infection leads to pathological induction of these processes leading to development of the disease. 110 Further studies on BPV-infected horses revealed 110 differentially expressed microRNAs in sarcoid tumors compared to normal skin. 18
In general, equine sarcoid fibroblasts exhibit cancer cell traits. Cells affected by BPV show increased ploidy and genomic instability, which may be promoted by altered global DNA methylation and oxidative stress. 83 Moreover, infection of equine fibroblasts by BPV-1 leads to oncogenic stress manifested by an abnormal, uncontrolled proliferation supported by aerobic glycolysis or induction of a defense response, namely, oncogene-induced premature senescence. 82
The presence of sarcoids in horses is associated with a number of abnormalities in immune mechanisms. In transcriptome-wide studies, Semik et al 94 identified 901 significantly differentially expressed genes between lesional and healthy skin samples of horses. A large subset of differentially expressed genes, with decreased expression, was associated with a suppression of malignant transformation, while some overexpressed genes were involved in the processes associated with growth and progression of the tumor or immune system activity. Further studies have shown that BPV-1 upregulates transcription of inflammatory cytokines, inhibitors of apoptosis, and promoters of cell motility and invasion. Equine sarcoid progression subverts both the adaptive and the innate immune response. 63,124 Among genes whose expression is affected by BPV, great attention is paid to the upregulation of CXC and CC chemokines (CXCL5, GRO2/CXCL2, CXCL6, and CCL7) involved in the activation of immune response and cancer development, as well as downregulation of Toll-like receptor 4 (TLR4), major histocompatibility complex class I (MHC I), and interleukin-12B, which play central roles in cell-mediated response. 124 In subsequent studies, the reduction in TLR4 expression was confirmed and evaluated as a consequence of exogenous expression of BPV-1 E2 and E7 oncoproteins. It is suggested that a decrease in TLR4 expression in horse sarcoids disrupts cytokine production and contributes to virus persistence and tumor recurrence. 122
E5, a major oncoprotein in equine sarcoids, has been proven to inhibit MHC I. 63 MHC I presents viral peptides to cytotoxic T lymphocytes, effector cells capable of recognizing and destroying transformed or infected cells. MHC class I is therefore a central player of the adaptive immune response. 62 BPV has developed the ability to downregulate MHC I, which leads to poor immune clearance of tumor cells, lack of spontaneous regression, and their persistence and recurrence. Detrimental effect of E5 oncoprotein on MHC I occurs in 2 stages: (1) inhibition of MHC I heavy chain formation on a transcriptional level and (2) retention of MHC I complex in the Golgi apparatus rather than allowing it to reach the cell surface where it could be active. Further analysis of immune mechanisms has revealed increased densities of cytotoxic and T helper cells in lesional and perilesional tissues of equine sarcoids. 38 Lesions of affected horses were shown to have increased numbers of E5 BPV-1 mRNA and increased levels of anti-inflammatory mRNAs for FOXP3 (fork 3 protein) and interleukin 10 (IL10), as well as pro-inflammatory factors, such as interferon gamma (IFNG). 115 Independent studies confirmed the increased transcription of FOXP3 mRNA in horse sarcoids compared to healthy skin. 115 FOX3P is a marker of immunosuppresor, regulatory T cells (Treg), which inhibits the action of antigen-reactive lymphocytes and regulates the intensity and duration of immune response by limiting degradation of affected cells. 88 Upregulation of Tregs in equine sarcoid lesions compared to intact skin, distant from the tumor suggests local, Treg-induced immune suppression.
Factors Predisposing to Equine Sarcoid Development
Several environmental and genetic factors appear to correlate with disease frequency. Geldings and young horses were thought to be predisposed to equine sarcoids; however, recent studies demonstrate lack of sex and age predispositions. 51 Little is known about genetic predisposition toward equine sarcoid. Encouraging the study of genetic factors related to the increased risk of disease onset is that some researchers observed increased prevalence of equine sarcoid in particular families, 86,98 as well as differences in the occurrence in various breeds, relatively high in Quarter horses and Franches-Montagnes horses, medium in Thoroughbreds, and low in Standarbreds. 5,30,74
Strong evidence of a link between genetic variations and appearance of equine sarcoid has been demonstrated for some MHC haplotypes. Studies conducted on Thoroughbred horses in the United States revealed that animals with high frequency of equine leukocytes antigen (ELA) haplotypes W3 and B1 appeared to be more vulnerable to sarcoid development. 73 Lazary et al 57 also found ELA W3, B1 haplotype to be more frequent in sarcoid-positive Irish, Swiss, and French horses. Additionally, W11 haplotype is more common in horses of Irish background, and W5 haplotype in Swiss and French horses with sarcoids. However, the results obtained for affected Swedish Halfbreds are different. In these horses, A3 and W13 haplotypes appeared more frequently; additionally, animals expressing W13 haplotype showed increased percentage of recurrence after surgery, while presence of A5 haplotype was associated with an early onset. 22,23 A genome-wide association study performed on 82 sarcoid-positive horses and 270 controls revealed 2 marker loci with high levels of significance for the emergence of the disease. One of the loci occurred in an intron of the MHC Class II receptor, DQ alpha 1 gene in the ELA region. 97 This gene plays a central role in the immune system by presenting peptides derived from extracellular proteins such as in B lymphocytes, dendritic cells, or macrophages. Associations between genetic leukocyte antigen variants and papillomavirus-induced tumors have been also reported for human papillomavirus, suggesting a common pathogenic mechanism. 33 The effect of these ELA haplotypes on the development of equine sarcoid is probably due to impaired elimination of the virus-induced tumor cells and impaired cell-mediated immunity at the level of epitope recognition in animals representing particular ELA haplotypes. 57,73
Scanning the entire genome of 222 horses belonging to 2 half-sibling sire families, 26 of which suffered from equine sarcoid, revealed 3 new candidate regions possibly associated with sarcoid on chromosomes 20, 23, and 25, including genes that regulate virus replication and host immune response. 46 Recent studies investigated the heritability and mode of inheritance for explanation of susceptibility to equine sarcoid. They confirmed that the susceptibility to equine sarcoid is polygenic, and a strong influence of one major gene is rather unlikely. These findings are promising for introduction of breeding values for horse sarcoid in the selection process. 30
Sarcoid Transmission
The mechanism by which horses are infected with BPV leading to equine sarcoid is not fully understood. The postulated mode of infection was direct contact with cattle or horses carrying BPV or through bites of a virus-carrying fly. However, the results of BPV transmission studies leading to development of sarcoid are not consistent. Recently, Haspeslagh et al 44 performed interesting studies on BPV spread. They exposed 420 stable flies (Stomoxys calcitrans) to BPV-positive equine sarcoids or bovine papilloma tissue. There was a significant increase in BPV viral load (copies/cell) both in sarcoid-exposed and papilloma-exposed flies; however, the viral load was higher and lasted longer in the group of flies exposed to papilloma tissue compared to sarcoid tissue. The results indicate that BPV transmission by stable flies is possible and may be more likely after contact with bovine papillomas than with horse sarcoids. It is also worth noting that virus transmission is more likely soon after tissue exposure. BPV-1 DNA can be detected both in biting and nonbiting flies trapped in the proximity of sarcoid-affected animals. These flies harbor BPV-1 variants that are often detected in equine sarcoids, indicating flies as a potential transmitter. 35
Studies on donkeys revealed higher risk of disease development in animals in close contact with individuals suffering from sarcoid. 87 Nasir et al 77 questioned the theory of cattle-to-horse transmission of BPV, based on the lack in equine sarcoids of BPV-1 variants, which are characteristic for infection in cattle. On the contrary, other studies showed a higher percentage of carriers of BPV DNA among horses kept together with horses that had sarcoids or with cattle that had papillomas. 14,16,17 No significant differences were found between the DNA sequences of bovine- and equine-origin BPVs, suggesting that horses and cows are common host species for BPV1 and viral transfer between these species is common. 53 It seems that a considerable number of horses are infected with BPV. However, additional, individual triggers contribute to the disease. Possibly in immunocompetent horses, an effective immune response might inhibit the development of disease, while in animals with genetic susceptibilities or reduced resistance to BPV, it causes the development of the disease. 16
Regland et al 84 observed a relationship between equine sarcoid and skin trauma, which appeared shortly after injury or in response to prolonged injury. Skin trauma may allow direct access of BPV to subepidermal fibroblasts, which may lead to abnormal proliferation. This phenomenon is rare; however, such complications of wound healing are still reported. Nevertheless, most sarcoids appear without any signs of skin injury, although micro-injuries such as insect bites cannot be excluded. 16
Interestingly, recent studies performed on blood and placental tissue of mares that did not have sarcoids and of their respective foals revealed the presence of BPV DNA in the placenta. 90 This might suggest vertical BPV DNA transmission from mares to foals although this remains to be confirmed by other analysis.
Clinical Classification and Biologic Behavior
Equine sarcoid has been categorized as a benign tumor of fibrous tissue (Fig. 1). Lesions can occur on any part of the body; however, the head, ventral abdomen, and limbs are most commonly affected. 72 Six distinct clinical forms can be recognized including occult, verrucose, nodular, fibroblastic, mixed, and malignant (malevolent) types. 10,51 Equine sarcoid is a disorder of varied biologic behavior. Horses may have a single sarcoid, but most commonly, affected animals develop multiple sarcoids. 68 The progress of the disease is dynamic, and the less severe forms can rapidly develop into the more aggressive types. 51 It is characterized by a high frequency of recurrence after treatment. Recurrent tumors are often aggressive and regrow more rapidly than the initial tumor. 109 Regland et al 84 reported that 49% of equine sarcoids recurred within 3 years of surgical removal, while another group found a 64% recurrence after conventional surgery. 32 Injection of 5-fluorouracil resolved 62% of sarcoid cases (no recurrence during 3 years follow-up). 99 Studies performed by Martens et al 70 revealed that the recurrence rates for particular methods of treatment were as follow: 21% for cryosurgery, 33% for BCG vaccination, 29% for carbon dioxide laser, and 18% for conventional excision. However, it must be mentioned that the follow-up in these studies were relatively short (mean 20 months). No clinical form of equine sarcoid, including the malignant form, has been reported to metastasize to local lymph nodes or beyond. 51,117 Sarcoids can also undergo spontaneous regression. Studies on Swedish horses revealed that one third of the affected horses became free of sarcoids without treatment. 22 Even higher rates of spontaneous regression were observed in Franches-Montagnes horses. Occult equine sarcoids underwent complete spontaneous regression in 65%, while verrucous lesions regressed spontaneously in 32% of animals. 11
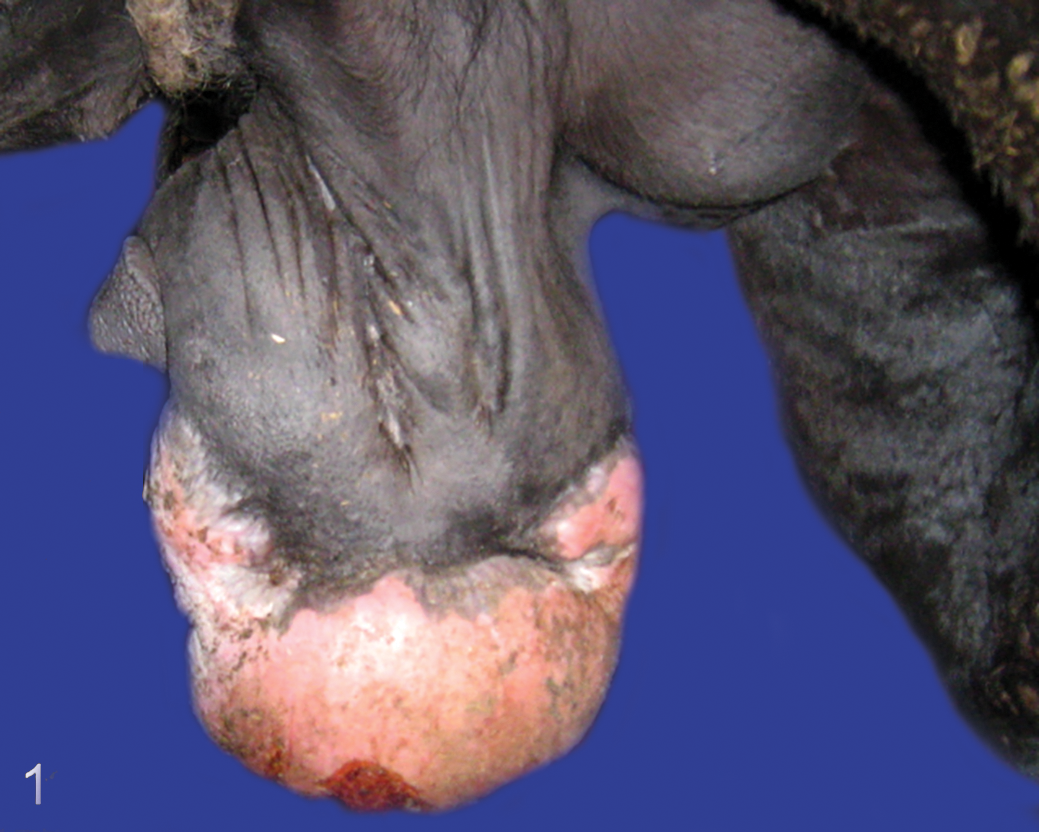
Fibroblastic sarcoid type 2, prepuce, horse. The mass is sessile without a pedicle and the margin between neoplastic and nonneoplastic tissue is not well-defined.
Occult Sarcoid
The occult sarcoid possibly represents the mildest, most stable, and superficial form of the sarcoid types. 16 The predilection sites include the skin around the mouth and eyes, the neck, and other relatively hairless areas of the body, including the inside of the forelimb, axilla, and thigh. They usually contain one or more small cutaneous nodules (2–5 mm diameter) or roughened areas with a mild hyperkeratotic appearance. The lesions are characteristically slow growing and may remain unchanged for years. However, extensive development of verrucous sarcoid or conversion into fibroblastic-type sarcoid can occur. 10,49 –51
Verrucous (Verrucose) Sarcoid
This type has a rough hyperkeratotic appearance with variable degrees of flaking and scaling over limited or wider areas of the body. Most often this type is seen on the face, trunk, and groin/sheath areas. Verrucous sarcoids are predominantly slow growing and are rarely aggressive until injured. Nevertheless, small nodules may appear at any stage or over any area of the affected skin, which may further develop into a fibroblastic type. 49 –51
Nodular Sarcoid
The nodular form of sarcoid is easily recognizable as firm, well-defined subcutaneous, spherical nodules. Based on the level of skin involvement, 2 types of nodular sarcoid are recognized. Nodular type A are entirely subcutaneous, while nodular type B resemble type A but have varying involvement of overlying skin. Individual nodules can vary from 0.5 to 20 cm or more in diameter. The number of nodules varies widely from single to hundreds. This type can be found most often in the groin, sheath, or eyelid areas. Similarly to verrucous type, nodular sarcoids display moderate growth. However, accidental or intentional interference can agitate these sarcoids leading to rapid growth and possible transformation into the more ominous forms of sarcoid, particularly the fibroblastic type. 16,49 –51
Fibroblastic Sarcoid
These tumors have a characteristic fleshy, ulcerated, aggressive appearance and can be divided into 2 types. Type 1 fibroblastic sarcoids are pedunculated with a limited or faint base palpable within the skin, whereas type 2 fibroblastic sarcoids are broader-based (sessile) without a recognizable pedicle and often have a diffuse, ill-defined margin. Predilection sites include the groin, eyelid, lower limbs, and coronary band, as well as sites of skin wounds at any location, and sites of any other types of sarcoid subjected to trauma or insult. Despite their outwardly aggressive appearance, fibroblastic sarcoids do not metastasize but can spread by local invasion into the dermis, and may carry a very poor prognosis. 49 –51
Mixed Sarcoid
In this category, an individual lesion has complex mixtures of any or all of the above types. This type of sarcoid probably represents a progressive or transient state between verrucous, occult, nodular, and fibroblastic types. 49 –51
Malignant (Malevolent) Sarcoid
This form is most severe, with infiltration of lymphatic vessels that results in nodules and cords of palpable tumor. The characteristic of this form is that cords of tumors are visible or palpable. It is highly invasive and destructive with an apparent predilection for the skin of the jaw and face, elbow, and medial thigh areas. It can rapidly spread over a wide body area and grows quickly. This type of sarcoid is rare, often develops following trauma or failed treatment, and can be ulcerated and bleed. 16,41,50,51
The frequency of occurrence of particular types of equine sarcoids varies between studies. Among 61 affected Franches-Montagnes horses, 17 possessed occult, 28 verrucous, and 2 nodular sarcoids. In 14 horses, there was no clear distinction between occult and verrucous lesion. 11 In another study of 194 sarcoids in 48 equids (34 horses of various breeds, 2 ponies, 11 donkeys, and 1 mule), there were 15 occult, 80 verrucous, 30 nodular, 35 fibroblastic, and 34 mixed forms. 107 Similarly, within 467 sarcoids in horses of various breeds from western Canada, the following clinical types were diagnosed: fibroblastic (145), nodular (104), occult (72), verrucous (72), and mixed (74). Malignant sarcoids are described rarely. 51,116 Furthermore, it must be mentioned that many equine sarcoids of mild manifestation, especially occult, verrucose, and sometimes nodular, are not presented to veterinarians. Therefore, the above-mentioned frequencies of particular forms may not be representative of the general population.
Histopathological Characteristics
The clinical diagnosis of equine sarcoids can be insufficient. Koch et al 53 studied 40 equine skin lesions (26 sarcoid and 14 other skin lesion) classified by 14 equine sarcoid experts, 39 board-certified equine specialists, 103 equine practitioners, and 25 novices, and visual gross examination of the lesion results corresponded to the histologic diagnosis in 82%. Sarcoids can be confused with fibromas, fibrosarcomas, and neurofibromas. Therefore, histopathological confirmation can help in equine sarcoid diagnosis and implementation of treatment.
The common histologic feature of all equine sarcoids is increased density of dermal fibroblasts compared to normal skin. 67 The typical histological changes include a proliferation of interlacing bundles and whorls of spindle-shaped fibroblasts within the dermis. The epidermis is hyperplastic and hyperkeratotic with long rete peg-like extensions into the dermal mass in the majority of cases. Frequently, at the dermoepidermal junction, there is a characteristic perpendicular orientation of fibroblasts toward the basement membrane, reminiscent of a picket fence 71,116 (Fig. 2). Histopathologically, equine sarcoid may be confused with fibroma, fibrosarcoma, or neurofibroma. 71,109
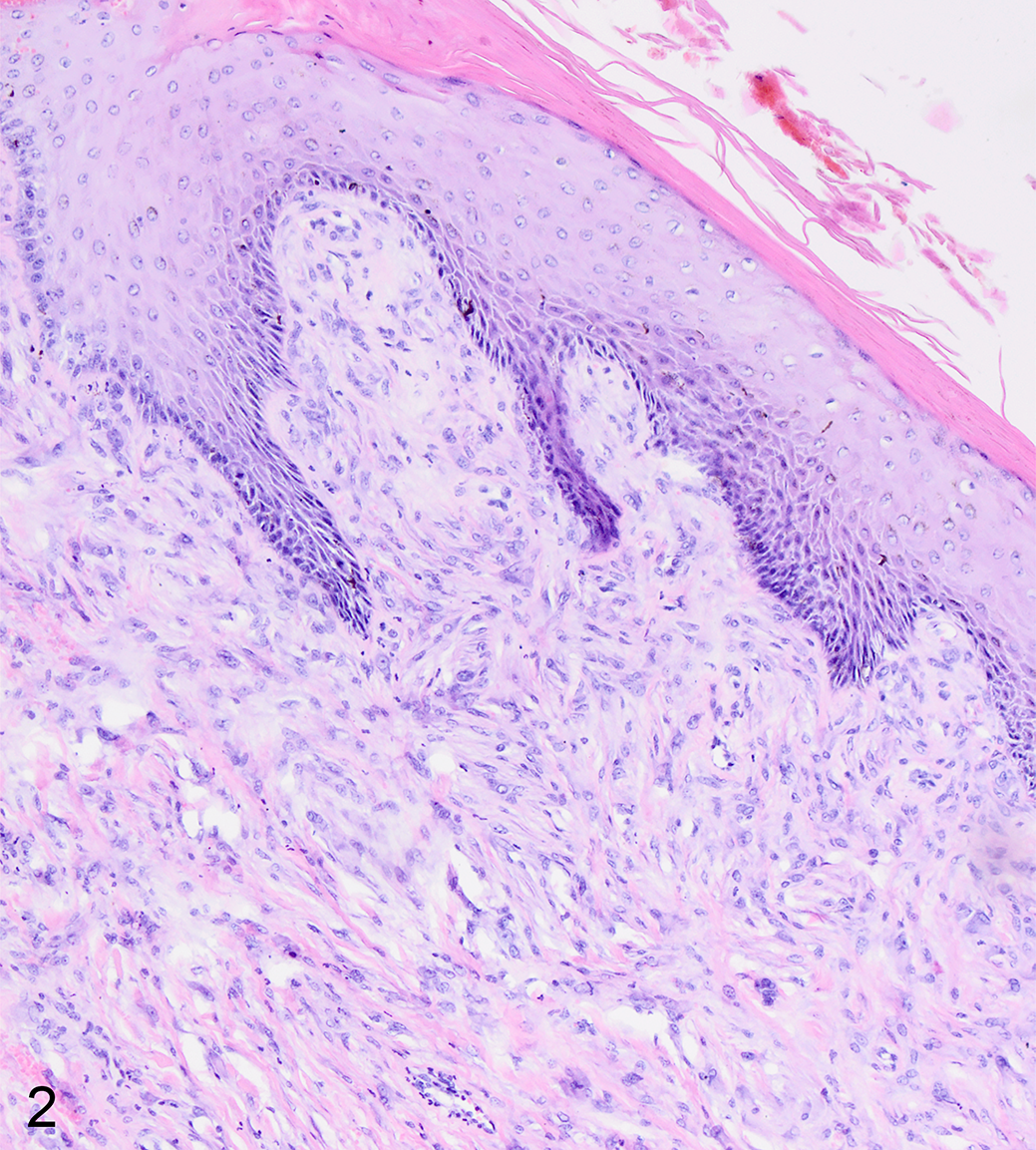
Fibroblastic sarcoid type 2, skin, horse. Spindle-shaped neoplastic cells form a mass in the dermis with the typical “picket fence” arrangement of neoplastic cells along the dermoepidermal junction. The epidermis is hyperplastic with a rete peg-like structure extending into the dermal mass, and there is compact hyperkeratosis. Hematoxylin and eosin.
Histopathological studies of Terwid et al 108 performed on 100 equine sarcoids showed a high frequency of epidermal hyperplasia, rete peg, and “picket fence” formation, which were present in 67%, 88%, and 91% of the cases, respectively. In studies of 40 equine sarcoids published 15 years later by Martens et al, 67 the above-mentioned pathological changes were recorded less frequently: 54% had epidermal hyperplasia, 46% had rete pegs, and 48% had “picket fence” arrangement. These features were absent more frequently in occult and nodular sarcoids compared to the other clinical types, and their absence may result in occult sarcoids being overlooked histologically, or may result in nodular sarcoids being misdiagnosed as fibromas, neurofibromas, or fibrosarcomas. 48,92 An important point is that the microscopic alterations in occult, verrucous, fibroblastic, nodular, and mixed equine sarcoids are not consistent; therefore, they cannot be used in the differentiation of the clinical types. To the best of our knowledge, the histologic features of malignant sarcoids have not been reported. It also must be mentioned that many veterinarians avoid biopsies to reduce risk of tumor progression, and histopathologic confirmation is not available for such cases.
Equine sarcoid exhibits several immunohistochemical features. The expression of oncosuppressors—O6-methylguanine-DNA methyltrasferase (MGMT) 3 and fragile histidine triad (FHIT) 100 —is reduced or absent in sarcoid tissues. Equine sarcoids are mostly S100-negative 13,34 and are positive for hypoxia-inducible factor-1 alpha (HIF-1α). 64 However, these immunohistochemical characteristics are insufficient for unambiguous distinction of equine sarcoids from other mesenchymal neoplasms. The discovery of a unique immunohistochemical marker would be a long-awaited milestone for equine sarcoid diagnosis.
Conclusions
Equine sarcoid is the most common skin cancer in horses with incompletely understood etiology and pathogenesis. It is difficult to treat and characterized by a high recurrence rate. Recent studies on equine sarcoid have revealed MMPs dysregulation, induction of growth factor signaling pathways, abnormal DNA methylation, as well as loss of activity by tumor suppressors, p53 and FHIT. Equine sarcoid is also strongly associated with impaired immune responses. BPV oncoproteins inhibit TLR4, MHC I, and MHC II, leading to disturbed cytokine production and poor immune clearance of tumor cells. BPV infection also causes induction of Treg immunosuppressor responses, which impairs the action of antigen-reactive lymphocytes.
An overwhelming number of studies indicate BPV to be the primary causative agent of equine sarcoid. The challenge remains as to why some horses develop lesions on BPV infection, while others remain healthy. Besides the essential BPV infection, it seems that additional contributing factors are required to develop equine sarcoid lesions. Various studies have reported that contact with sarcoid-affected horses or cattle with papillomas may increase the occurrence of sarcoids in horses. The fact that in cows, unlike horses, lesions caused by BPV infection regress spontaneously indicates that differences in the immune response are of crucial importance for development of tumors. Indeed, the presence of sarcoid has been associated with specific equine leukocyte antigens, as well as genetic variants such as those in genes regulating viral replication and the host immune response. The presence of BPV DNA is not sufficient for diagnosis of sarcoid, and the histopathological evaluation of lesions does not necessarily correlate with the clinical classification of different forms of equine sarcoid. During the last decade, high-throughput genomic, epigenomic, transcriptomic, and proteomic technologies have been applied to further our understanding of the molecular pathogenesis of this heterogeneous disease and to develop strategies that aim to improve the management of sarcoids. However, there is lack of clear diagnostic and prognostic markers that would allow for accurate repeatable classification. High morbidity and resistance to treatment highlights the need of further in-depth studies on the mechanisms underlying BPV-induced equine sarcoid progression, which will introduce new therapeutic and preventive strategies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
