Abstract
Histopathologic differentiation between deep dermal or subcuticular equine sarcoids (ie, nodular sarcoids) and other spindle cell tumors in the dermis and subcutis such as peripheral nerve sheath tumors (PNSTs) can be challenging based on morphologic criteria alone. It has been proposed that polymerase chain reaction (PCR) for bovine papillomavirus (BPV) DNA and S100 immunohistochemistry be used as diagnostic tests to separate equine sarcoids from PNSTs. We reviewed 197 skin-associated spindle cell tumors (ie, soft tissue sarcomas), including PNSTs and sarcoids, received at the University of Florida between 1995 and 2013 and performed BPV PCR and S100 immunohistochemistry on archived paraffin-embedded tissues. We found that BPV DNA was demonstrable in 70% of the sarcoids, 59% of the PNSTs, 37% of the fibrosarcomas, and 22% of other tumors (myxosarcomas, fibromas, and other sarcomas) diagnosed on histomorphologic characteristics. Positive S100 staining was only seen in 12 tumors in the study (5 fibrosarcomas, 3 sarcoids, 2 PNSTs, and 2 other sarcomas). The results demonstrate that BPV is associated with many skin-associated spindle cell soft tissue tumors in horses in addition to sarcoids. S100 was rarely expressed in equine soft tissue sarcomas in the skin but was expressed in many tumor types, including PNSTs and sarcoids. Because 41% of the PNSTs classified by histomorphology in this study were BPV negative and 94% were S100 negative, it is reasonable to classify these as soft tissue sarcomas with nerve sheath tumor histomorphology rather than as either PNSTs or sarcoids.
Keywords
Equine sarcoids and soft tissue sarcomas are among the most commonly diagnosed cutaneous neoplasms in horses. 30 Jackson first described sarcoids in 1936 21 as cutaneous neoplasms with irregularly arranged fibroblastic components that were distinct from fibrosarcomas and fibromas and that included an epithelial component with acanthotic epidermis with elongate rete pegs. Clinical and laboratory classifications of equine sarcoids have evolved over the years, and histologic and other laboratory features of verrucous, fibroblastic, occult, nodular, and mixed sarcoids have been described. 24
Initial diagnostic classification systems based solely on histologic laboratory methods recognized that most sarcoids have both epidermal and dermal components. 19,29 The epidermal and epidermis-associated histologic patterns, including rete pegs, acanthosis, and picket fence fibroblastic pattern with epidermis, were considered important for reliable histopathologic diagnosis of sarcoid. This is because the interwoven or storiform pattern of fibroblastic spindle cells composing the majority of the nonepidermal component of sarcoids can be highly variable, especially in large tumors that are invasive, fast growing, and/or ulcerated. Cellular pleomorphism and other patterns of fibroblastic cells in deep sarcoids have overlapping morphology with those of other spindle cell/soft tissue sarcomas, including fibrosarcoma and peripheral nerve sheath tumors (eg, schwannoma, neurofibroma). 19,31 –33 More recent sarcoid classification systems recognize nodular sarcoids that may be largely or completely separated from epidermis and that are often more difficult to distinguish from peripheral nerve sheath tumors (PNSTs) and other soft tissue sarcomas on histomorphology alone. 4,24
Bovine papillomaviruses 1 and 2 (BPV-1/2) have been associated with most equine sarcoids. 2,3,26,36 A third delta bovine papillomavirus, BPV-13, has been associated with several sarcoids in Brazil. 23 Bovine papillomavirus DNA in sarcoids is in an epigenetic location, and the precise role of the virus in tumorigenesis is uncertain. 26 BPV DNA has been demonstrated in sarcoid tumors in associated epithelial cells of epidermis, follicles, and sebaceous glands as well as in epidermis of horses without sarcoid or those with inflammatory skin disease. 5,8,18,26,37
The difficulty in accurately diagnosing nodular sarcoids vs PNSTs was brought into focus more recently 4 in a study of 10 deep dermal and subcuticular spindle cell neoplasms initially diagnosed as schwannomas. All 10 neoplasms were positive for BPV-1 or BPV-2 DNA by polymerase chain reaction (PCR) and were negative for S100 protein immunohistochemical expression. On the basis of these findings, the authors concluded that these tumors were nodular sarcoids. They proposed that immunohistochemistry for S100 protein and molecular screening for BPV DNA should be pursued for all equine skin tumors exhibiting features consistent with schwannoma or PNST.
At the University of Florida, review of archived equine surgical pathology specimens between 1995 and 2013 resulted in identification of more than 50 tumors that had been diagnosed as PNSTs (nerve sheath tumors, neurofibroma, neurofibrosarcoma, schwannoma). Many of these were around the eye. Since many of these were diagnosed based on histopathology alone before the BPV PCR test and immunohistochemistry were commonly used on routine equine surgical pathology accessions, it was considered important to evaluate them further for BPV DNA and S100 expression. Other cutaneous soft tissue spindle cell tumors, including sarcoids diagnosed on the basis of histopathology alone, were similarly studied for comparison.
The objective of this study was to determine whether BPV DNA tests and S100 immunohistochemistry would facilitate differentiation of sarcoids from PNSTs and distinguish each from other skin-associated spindle cell neoplasms in the skin of horses.
Materials and Methods
Case Selection Criteria
Searches were conducted within the University of Florida College of Veterinary Medicine (UFCVM) surgical pathology files over a 19-year period from 1995 through 2013. Files maintained from 2007 to 2013 were in pdf format converted from Word documents and were searched with the Adobe Acrobat search engine. Files stored from 1995 to 2006 were WordPerfect documents that were searched via the WordPerfect file search engine. Search terms used for each year for equine accessions included nerve sheath tumor, sarcoid, sarcoid and eyelid, sarcoid and eye, schwannoma, neurofibrosarcoma, neurofibroma, fibroma, fibrosarcoma, sarcoma, spindle cell tumor, and myxosarcoma. Rhabdomyosarcoma and leiomyosarcoma were not included in the search because they were considered to have sufficiently unique histomorphology not to be confused with either sarcoid or PNST. Since content word searches were conducted, files were retrieved in which the search terms appeared in the clinical history, suspected clinical diagnosis, or as a differential diagnosis mentioned in the comments.
All cases with hits in the searches were reviewed. Equine accessions represented 18% (9494/51724) of all accessions. Search and review yielded 922 masses associated with skin, of which 705 (76%) were diagnosed as sarcoids. The most extensive study focused on 304 cases, which had as their original diagnosis the following: 40 sarcoids around the eye or eyelid, 98 sarcoids not associated with the eye, 53 nerve sheath origin tumors, and 113 sarcomas/fibromas. S100 immunohistochemistry and BPV-1/2 PCR were performed on 197 neoplasms after review and classification and based on the availability of archived paraffin blocks with adequate samples for recuts and analysis. Original diagnoses on these neoplasms were PNST (n = 44), sarcoid (n = 62), sarcoma other than neurofibrosarcoma (n = 79), and fibroma (n = 12). We also selected nonneoplastic cutaneous lesions (ie, habronemiasis, fibrosis), carcinomas and papillomas in the skin, and noncutaneous sarcomas (ie, intestinal leiomyosarcoma and oral hemangiopericytoma) as controls and comparisons with the tests on the skin-associated tumors.
Because 19 pathologists had been responsible for the diagnoses over the 19 years and because diagnostic practices had changed (eg, acceptance of the nodular sarcoid classification), it was important to review diagnoses on all cases using the same histomorphologic criteria.
Histomorphologic Classification
All neoplasms were characterized and diagnosed histologically while blinded to the original diagnosis using criteria that follow from the first author (E.D.E.). Features of malignancy such as vascular or lymphatic invasion, atypical mitotic figures, cellular atypia, anisocytosis and anisokaryosis, necrosis, and hemorrhage were noted if present. The mitotic count (ie, mitoses per ten 400× power fields) was recorded for each neoplasm. The field of view was 0.196 mm2. Cases with revised diagnoses as well as all cases diagnosed as PNST were reviewed by the senior pathologist (W.L.C.) with knowledge of the original and second diagnosis. After review, diagnoses of 7 of the 197 cases were changed to sarcoid from PNST (n = 6) and fibrosis/epithelial dysplasia (n = 1). All 7 cases had originally been diagnosed as sarcoids.
Sarcoid
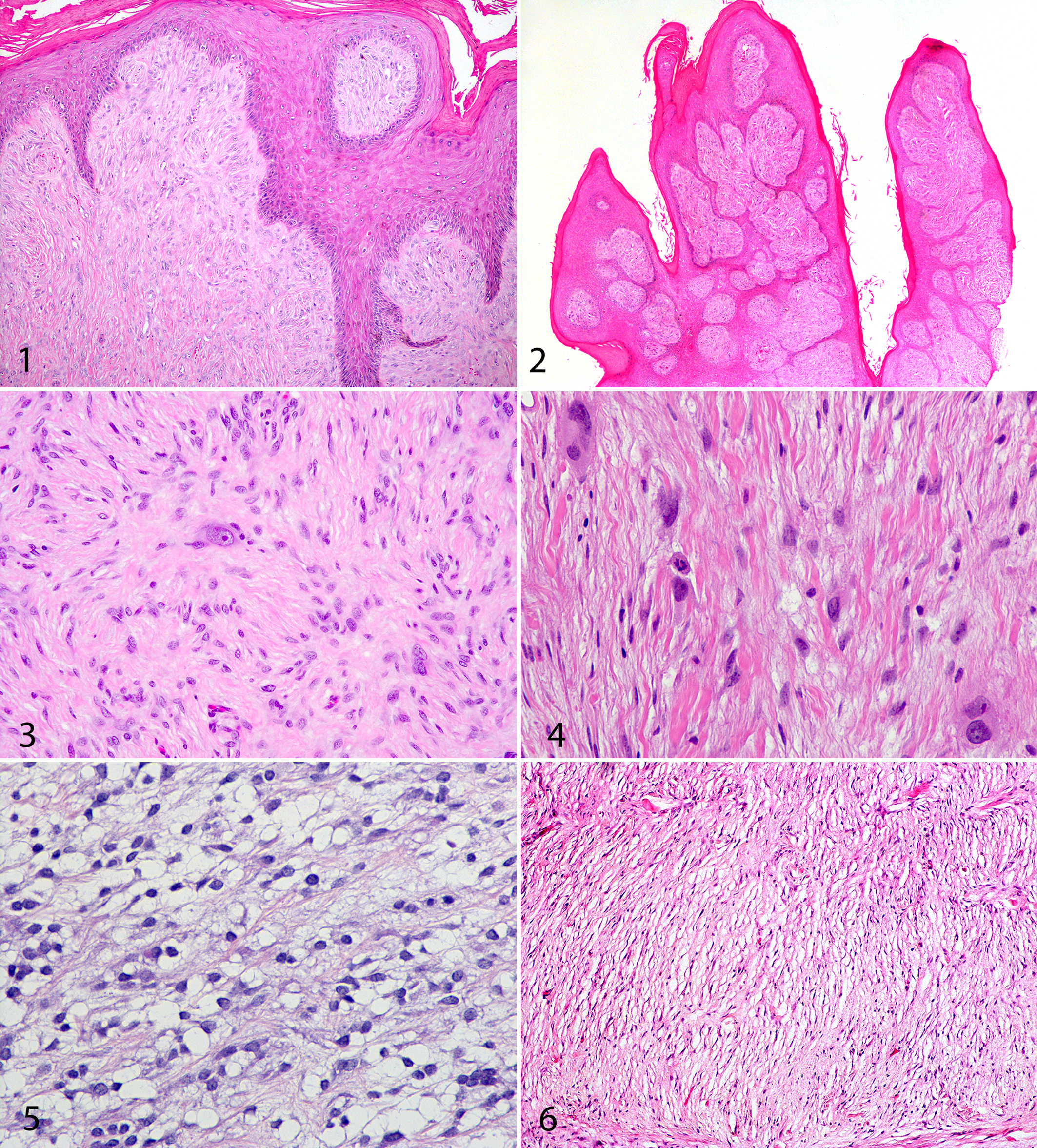
Tumors classified as classical sarcoids were poorly demarcated, infiltrative, unencapsulated dermal masses composed of spindle-shaped fibroblasts with indistinct cellular borders and a scant to small amount of eosinophilic cytoplasm. Spindle cells were arranged in a storiform, herringbone, and/or whorled pattern and were closely associated with the epidermis. Nuclear palisade patterns (ie, Antoni A patterns) were occasionally present in tumors classified as sarcoid for other characteristics (eg, close epidermal association with rete pegs and acanthosis). Epidermal changes included acanthosis and prominent epidermal rete pegs. 20,33 Presence of a “picket fence” perpendicular orientation of superficial dermal fibroblasts to the basal epidermis, decreased density of adnexa, and variable ulceration were also defining criteria for a diagnosis of sarcoid. 20,29,33 Mitotic count was variable but was generally low, averaging no more than 3 to 4 mitotic figures in ten 400× fields (field of view, 0.196 mm2). There were several sarcoids with the nodular subtype of sarcoid that were characterized by an infiltrative deep dermal or subcuticular, poorly to well-demarcated nodular mass of spindle-shaped fibroblasts with indistinct cellular borders arranged in a storiform pattern similar to that of classic sarcoid. Involvement of the overlying dermis and epidermis was minimal.
PNSTs
For diagnostic categorization, tumors of peripheral nerve sheath origin (ie, so-called malignant/benign PNST, schwannoma, neurofibroma, neurofibrosarcoma) were grouped together. These neoplasms minimally comprised spindle cells with indistinct cellular borders arranged in a whorled, herringbone, and/or storiform pattern that exhibited concentric lamination centered on nerves or loose bundles of connective tissue that resembled nerves. Some of these tumors were also variably characterized by densely cellular Antoni A areas that had scant collagenous stroma, with short fascicles of cells with palisading nuclei and Verocay bodies. Antoni A areas were occasionally admixed with hypocellular Antoni B areas, which had abundant myxoid stroma. 31,32 Less commonly observed features of nerve sheath origin neoplasms included hyalinization of vascular walls and stroma, tumor encapsulation, and irregular cytoplasmic vacuolation of neoplastic cells.
Fibrosarcoma
Well-differentiated, infiltrative, unencapsulated, well-demarcated fibroblastic tumors in the dermis or subcutis that comprised spindle-shaped cells with indistinct cellular borders forming interlaced herringbone or storiform patterns supported by a prominent collagenous stroma were consistent with the diagnosis with fibrosarcoma. 20 In addition, these neoplasms exhibited moderate to marked anisocytosis, anisokaryosis, and cellular and nuclear pleomorphism. Mitotic count was moderate to high, averaging 5 to 6 mitotic figures in ten 400× fields, with up to 38 mitotic figures per ten 400× fields in one neoplasm. In addition, necrosis and hemorrhage were variably evident in some tumors. These tumors did not exhibit features of epidermal association such as pronounced epidermal rete pegs or a perpendicular “picket fence” arrangement of superficial dermal fibroblasts.
Myxosarcoma
Well-demarcated, unencapsulated, infiltrative masses that comprised spindle- to stellate-shaped fibroblasts with indistinct cellular borders and hyperchromatic nuclei, loosely suspended in an abundant pale basophilic (myxomatous) matrix, were classified as myxosarcoma. 20 The cell population was predominantly homogeneous, with minimal to mild pleomorphism and mitoses averaging 5 in ten 400× fields.
Fibroma
Fibromas were characterized as well-circumscribed, expansile, unencapsulated dermal masses composed of a low-density, monomorphic population of mature fibroblasts with indistinct cellular borders and scant basophilic cytoplasm amid an abundant amount of repetitive, dense collagen fibers. 20 Mitotic figures were rare, averaging less than 1 in ten 400× fields.
Liposarcoma
Liposarcoma was defined as an infiltrative, unencapsulated, poorly to well-demarcated dermal or subcuticular neoplasm composed of sheets of polygonal to round or spindle cells supported by a small amount of collagen-rich stroma, multifocally interspersed with areas of myxomatous and edematous stroma. 20 Cytoplasm was moderately to markedly expanded by variably sized empty, nonstaining round spaces (extracted lipid droplets). There were few multinucleated cells. Mitotic count was low, averaging 2 in ten 400× fields.
Histiocytic Sarcoma
Histiocytic sarcomas were infiltrative, well-demarcated, unencapsulated dermal and subcuticular neoplasms composed of dense sheets of round to polygonal cells to sometimes spindle-shaped cells with distinct cellular borders and a moderate to abundant amount of eosinophilic cytoplasm that rarely contained small vacuoles. 9 There were small to moderate numbers of multinucleated giant cells and apoptotic neoplastic cells. Anisocytosis, anisokaryosis, and cellular and nuclear pleomorphism were marked, with a high mitotic count, ranging from 11 to 62 in ten 400× fields. There were small to moderate numbers of bizarre mitotic figures, and necrosis and hemorrhage were frequent features of these tumors.
Malignant Fibrous Histiocytoma (Malignant Giant Cell Tumor of Soft Parts)
Cutaneous neoplasms composed of infiltrative, well-demarcated, unencapsulated storiform patterns of stellate- to spindle-shaped cells with variably distinct cellular borders and round to polygonal cells with distinct cellular borders and a moderate amount of eosinophilic cytoplasm, admixed with moderate to large numbers of multinucleated giant cells, were categorized as malignant fibrous histiocytoma. 20,33 Necrosis, hemorrhage, and edema were frequent features, along with prominent nuclear and cellular pleomorphism, anisocytosis, anisokaryosis, and occasional atypical mitotic figures. Mitotic count was moderate to high, at 8 to 9 mitoses in ten 400× fields.
Bovine Papillomavirus DNA PCR
PCR was used to detect a common sequence of the E5L2 open reading frame of both BPV1 and BPV2 as originally described by Teifke and Weiss 34 and used by others. 3,4,24 To avoid cross-contamination, each paraffin block was cut with a new disposable microtome blade, and forceps that were newly cleaned with detergent and alcohol for each sample were used to transfer paraffin sections from the microtome blade to microcentrifuge tubes. DNA was extracted with a Qiagen FFPE QIAmp DNA extraction kit (Qiagen GmbH, Hilden, Germany) according to kit directions. The following primer set 34 used was: forward, 5′-CAAAGGCAAGACTTTCTGAAACAT-3′; reverse, 5′-AGACCTGTACAGGAGCACTCAA-3′. Taq Master mix (2×; Genescript, Piscataway, NJ) was diluted with nuclease-free water, and final concentrations of primers were 0.26 μM. DNA template concentration varied between 2 and 20 ng/μl. Initial denaturation in the thermocycler was at 95°C for 3 minutes. This was followed by 40 cycles: 95°C for 30 seconds, 63°C for 30 seconds, and 72°C for 1 minute. The final extension was at 72°C for 7 minutes. Products (244 bp) were detected in 2% agarose gels. Each assay run included negative controls and positive controls. The positive control for each assay was a sarcoid that had previously reacted positively. The negative control for each assay was complete mix with primers with nuclease-free water replacing the DNA template. Other negative controls were included periodically (ie, squamous cell carcinoma or cutaneous habronemiasis samples). Except for positive and negative controls, samples were analyzed without knowledge of sample diagnosis. Selected PCR products amplified from a sarcoid and a fibrosarcoma were purified and sequenced at the University of Florida Interdisciplinary Center for Biotechnology Research (ICBR) Sanger Sequencing core.
When we found that approximately 30% of the sarcoids were PCR BPV negative, we tested for evidence of inhibitory substances for PCR by spiking DNA samples of 5 negative reacting samples with maximally diluted samples of DNA from positive sarcoids but found no evidence of inhibitors of PCR. We tested 35 sarcoid DNA samples that were negative for BPV-1/2 for BPV-13 using degenerate papillomavirus primers as described by Lunardi et al. 23 However, no definite positive PCR reactions indicative of BPV-13 DNA were obtained from cases that were negative for BPV-1/2 DNA. We analyzed frequency of negative BPV-1/2 PCR results in sarcoids and other spindle cell tumors in the skin for each accession year. We also expanded the total number of sarcoids examined from the archives and conducted BPV-1/2 PCR tests. There was no pattern in the frequency of negative results that suggested that prolonged storage of paraffin blocks was decreasing the efficiency of DNA extraction and BPV-1/2 detection (Suppl. Table S1).
S100 Protein Immunohistochemistry
Immunohistochemistry for S100, a calcium binding protein expressed by Schwann cells in equine schwannomas and neurofibromas, 31,32 was conducted as previously described. 11 Paraffin sections mounted on charged slides were deparaffinized and dehydrated prior to quenching endogenous peroxidase activity in 0.3% H2O2 in methanol for 30 minutes. Heat-induced epitope retrieval was accomplished by treatment for 20 minutes with Tris EDTA buffer (pH 9) at 95°C. Nonspecific staining was suppressed with Background Punisher (Biocare Medical, Concord, CA) according to the manufacturer protocol. Sections were treated with primary S100 monoclonal antibody cocktail (CM089; Biocare Medical) at 1:150, which recognizes proteins of 21 to 24 kDa, identified as A and B subunits of S100 protein. A Mach 4 polymer detection system (Biocare Medical) was used as described by the manufacturer. Irrelevant antibody (mouse anti-rat CD8) at 10 ng/ml was used as a control. Peroxidase activity was detected with Metal Enhanced DAB (3,3′-diaminobenzidine) Substrate Working Solution (Thermo Scientific, Rockford, IL) as described in the kit. Slides were lightly stained with Harris hematoxylin. For many of the suspected PNSTs, we repeated S100 staining using Dako rabbit anti-S100 diluted 1:200 (Z0311; Dako, Glostrup, Denmark) after antigen retrieval treatment (S1700; Dako), which gave essentially identical results as the monoclonal cocktail. Samples were performed and interpreted without knowledge of sample diagnosis. Cells staining positively for S100 generally had strongly staining cytoplasm and nuclei compared with background. A smaller number of cases were recorded as positive for S100 that had cells with strong cytoplasmic staining and only moderate to weak nuclear staining.
Results
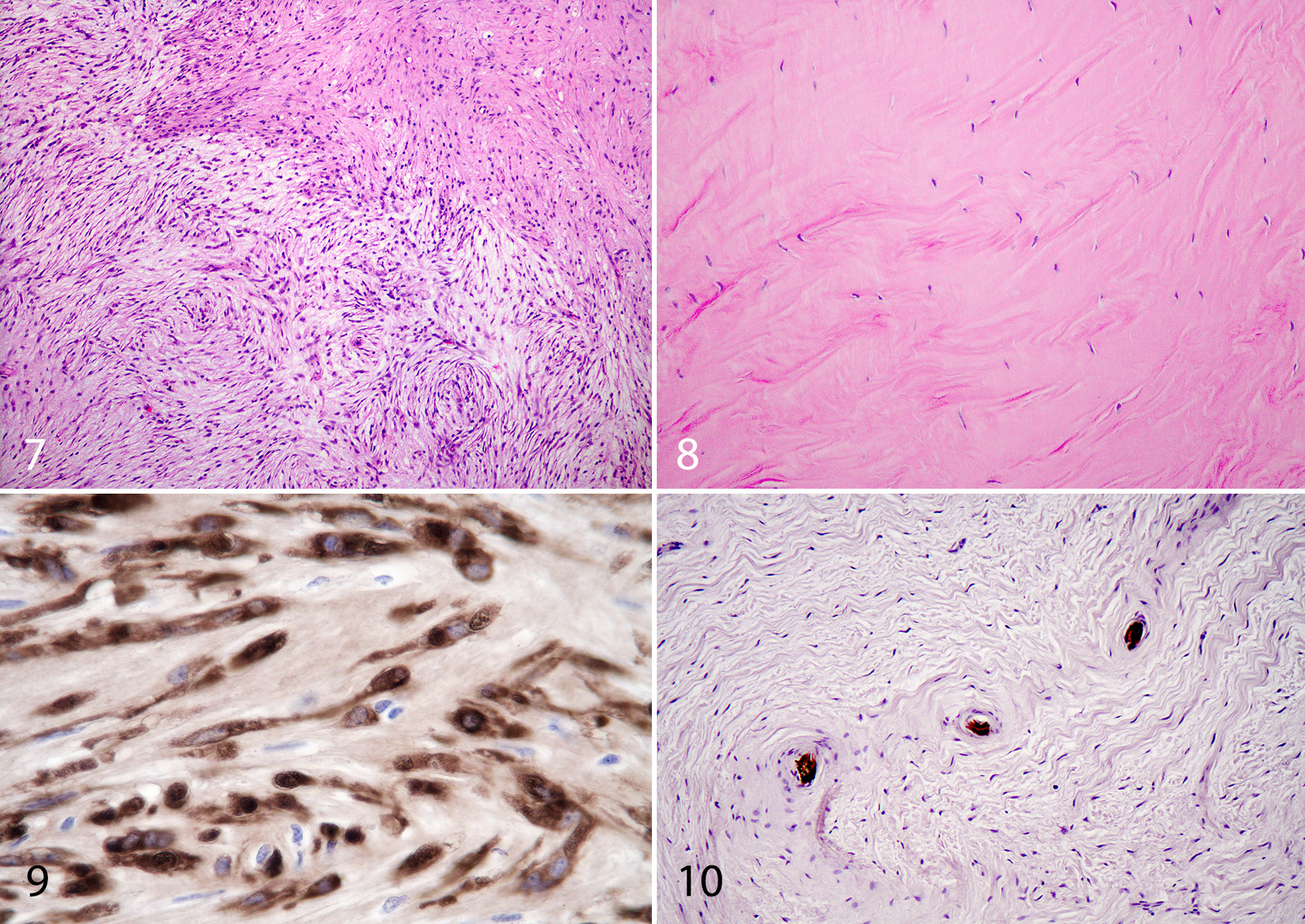
Results for major tumor categories and the corresponding profiles of BPV DNA content and S100 immunoreactivity for the 197 fully studied tumor accessions are presented in Table 1. There was considerable heterogeneity of BPV DNA positivity and S100 staining among the diagnostic groups. Thirty percent of the tumors diagnosed as sarcoids were negative for BPV-1/2 DNA (Table 1; Figs. 1, 2). Many spindle cell tumors other than sarcoids were positive for BPV-1/2 DNA. These included 20 of 54 (37%) fibrosarcomas (Fig. 3); 6 of 23 (26%) other sarcomas, including malignant fibrous histiocytoma (Fig. 4) and liposarcoma (Fig. 5); 1 of 8 (12.5%) myxosarcomas (Fig. 6); and 19 of 32 (59%) PNSTs (Fig. 7). In addition, 2 of 10 (20%) fibromas were BPV-1/2 positive (Fig. 8).
Presence of Bovine Papillomaviruses 1/2 DNA by PCR and S100 Immunoreactivity in Sarcoids and Other Equine Soft Tissue Tumors.

BPV, bovine papillomavirus; PNST, peripheral nerve sheath tumor.
aHistomorphology indicative of nerve sheath origin.
bAnaplastic histiocytic sarcoma, poorly differentiated sarcoma, liposarcoma, and malignant fibrous histiocytoma.
BPV PCR products from a fibrosarcoma and a sarcoid were sequenced and both found to be 99% homologous with the E2 and E5 genes of both BPV-1 and BPV-2 (Basic Local Alignment Search Tool, National Center for Biotechnology Information). Since BPV-1/2 can be detected in normal epidermis, blocks of sarcomas and fibromas that tested positive for BPV-1/2 were evaluated for the presence of epithelium that might have contributed to a false-positive PCR reaction. Of the 48 nonsarcoid BPV-positive neoplasms, 21 of the samples analyzed by for PCR did not contain epidermis. For 4 of the fibrosarcomas that tested positive for BPV-1/2 and had skin in the biopsy block, additional blocks from the cases were available that did not include skin, and these were tested. Three of 4 of these cases remained positive by PCR for BPV-1/2 DNA when no skin was in the embedded sample.
Nonneoplastic lesions (cutaneous habronemiasis and dermal fibrosis), cutaneous squamous cell carcinomas, intestinal leiomyosarcoma, and gingival hemangiopericytoma all tested PCR negative for BPV-1/2 DNA.
Few of the tumors studied were positive for S100 by immunohistochemistry (Table 1). Five fibrosarcomas, 3 sarcoids, 2 PNSTs, and 2 other sarcomas (myxosarcoma and poorly differentiated sarcoma) stained positively for S100 protein. There were no histomorphologic criteria that allowed prediction of which fibrosarcoma or other sarcoma would be S100 positive. All 3 of the sarcoids that were S100 positive were biphasic with both epithelial changes (epithelial thinning with reduction of follicular density) and fibroblastic spindle cell populations with storiform pattern. The second S100-positive sarcoid had epithelial pegs and ulceration. There was multifocal nuclear palisading and concentric lamination of spindle cells around muscle fibers and large collagen bundles but not nerves. The third S100-positive sarcoid had thinned epidermis and hair follicle reduction and storiform spindle cell pattern with areas of nuclear palisading. It was originally diagnosed upon accession as a sarcoid with PNST as the differential diagnosis. S100 immunohistochemistry was done at that time and was positive, so the diagnosis was revised to PNST. When reclassified on histomorphology alone in this review, it was categorized as a sarcoid and tested positive for BPV-1/2 and was again S100 positive.
Only 2 of 32 (6%) tumors that were histologically consistent with PNST had positive S100 protein immunoreactivity (Fig. 9) and were negative for BPV-1/2 DNA PCR. There were no unique histomorphologic features distinguishing classified PNSTs that were S100 positive from those that were S100 negative. Spindle cell tumors with frequent palisading nuclei that wrapped around nerves in a concentrically laminated pattern and followed nerves as they branched were generally S100 negative and had S100-positive nerves in the middle of the laminated spindle cells (Fig. 10).
Tumors originally classified as PNST in the selected 197 cases from the archives had the most dramatic reduction in number after histomorphologic review and classification. The number of PNSTs diagnosed histologically in this study went from 44 before review to 32 after review. Eight were reclassified as sarcoids, and 14 were reclassified as sarcomas (usually fibrosarcomas). Four tumors originally classified as sarcoids were reclassified as PNSTs. Six tumors originally classified as sarcoma were reclassified as PNSTs. If the histomorphologic criteria for equine schwannoma and neurofibroma are used 31,32 and coupled with the additional diagnostic criteria suggesting 4 that all schwannomas and other PNST should be BPV negative and S100 positive, the number of PNSTs in the study would further decrease from 32 to 2. The first of these two was from the girth area of an 8-year-old female Rocky Mountain horse and was well demarcated, with whorled spindle cells around nerve-like structures and Antoni A areas. No mitotic figures were observed in ten 400× fields. The S100 staining pattern was mixed with approximately 60% of the cells having nuclear and cytoplasmic staining. There was partially ulcerated epidermis and granulation tissue over the mass. The second S100-positive and BPV-1/2–negative PNST was from the skin under the tail of a 5-year-old Arabian gelding. The mass was originally diagnosed as a fibrosarcoma or soft tissue sarcoma with PNST as a differential diagnosis. It was widely separated from the epidermis and extended deeply into subcutis. It had repeating whorled patterns throughout associated with nerve-like structures. Many of the whorled areas had centrally mineralized areas. There were widespread multifocal Antoni A areas. One mitotic figure was seen in 10 fields. The S100 staining pattern was mixed with approximately 50% of the cells having nuclear and cytoplasmic S100 staining patterns.
Discussion
This study was undertaken with the initial goal to further characterize archived skin-associated tumors originally diagnosed as equine PNSTs (schwannoma, neurofibroma, neurofibrosarcoma, nerve sheath tumor) at the University of Florida and to distinguish them from other spindle cell tumors associated with the skin that might be confused with PNST because of overlapping histomorphology. Bogaert et al 4 suggested that differentiation between PNSTs and sarcoids could be made on the basis of BPV-1/2 PCR tests and S100 immunohistochemistry. The diagnostic guideline predicted that sarcoids would be BPV-1/2 positive and S100 negative, whereas PNSTs such as schwannomas would be BPV-1/2 negative and S100 positive. Other spindle cell tumors in the skin such as fibrosarcoma, myxosarcoma, and fibroma were predicted by logical extension of the suggested diagnostic guideline to be BPV-1/2 negative.
The results from the study differed significantly from the predicted results. Most significantly, many spindle cell tumors other than sarcoids were often positive by PCR for BPV-1/2 DNA. These included fibrosarcomas, PNSTs, myxosarcomas, malignant fibrous histiocytomas, and fibromas.
Of the 70 sarcoids studied in detail, 70% were positive for BPV-1/2 DNA. Other studies using screening PCR tests for bovine papillomaviruses 1 and 2 on formalin-fixed, paraffin-embedded (FFPE) tissues have reported variability in the presence of BPV-1/2 DNA in sarcoids from 70% to 77%. 3,36 The 30% of sarcoids in this study that were negative for BPV-1/2 DNA by PCR can be explained at least in part by fixation and paraffin processing and storage artifact since PCR studies on unfixed sarcoid samples yield higher positive recoveries than those from FFPE tissues. 10,28 Furthermore, it has been proposed that the screening PCR assay used in this and many other studies is low in sensitivity and that large amounts of connective tissue in tumors may further reduce sensitivity of the assay. 12 Alternatively, induction of equine sarcoids is a multifactorial process, and it is reasonable to suggest that some sarcoids may develop without BPV infection. 1,2,25
This study is the first to report that bovine papillomavirus DNA is present in equine spindle cell tumors in the skin other than sarcoids. Although previous studies reported that BPV was not associated with neoplasms in horses other than sarcoids, 10,13,28 those studies examined carcinomas, melanomas, cystadenocarcinomas, papillomas, hemangiomas, and lymphomas (ie, not other cutaneous spindle cell tumors). In the present study, PCR demonstrated that BPV-1/2 DNA was present in 48 of 127 (37.8%) nonsarcoid spindle cell tumors associated with the skin. Positive tumors included fibrosarcoma, PNST, myxosarcoma, fibroma, and other types of sarcomas (poorly differentiated sarcoma, malignant fibrous histiocytoma, and liposarcoma).
It was considered possible that positive BPV-1/2 PCR reactions in nonsarcoid tumors may have been due to BPV present in epidermis over the neoplasm, rather than within neoplastic cells. However, half of the PCR-positive nonsarcoid tumors processed did not have epidermis covering the sample. So BPV-1/2 DNA detected in these assays most likely originated from the tumor cells. PCR products amplified from one epidermis-free fibrosarcoma were sequenced and found to be 99% homologous with the E2 and E5 genes of BPV-1 and BPV-2.
The pathogenic role of bovine papillomavirus in equine sarcoid development is only partially characterized. 1,2,7,12,16,25,26,35 Although BPV DNA is found in various locations within sarcoids, it also can be found in normal skin areas of horses with sarcoids as well as within skin of horses with dermatitis. 2,6,17,18,37 Sarcoid induction and other tumorigenic effects that might be relevant to the more widespread association of BPV-1/2 DNA with equine spindle cell tumors indicated by this study have been linked most closely with cellular expression of E5 protein. BPV-1 is capable of inducing proliferation and transformation in cultured dermal fibroblasts from horses, mice, and humans. 26 E5 protein from BPV-1/2 binds to and activates the cellular platelet-derived growth factor β (PDGF-β) receptor as one of its most important activities to stimulate cell proliferation. 16 It induces downregulation of major histocompatibility complex I (MHC I) expression on cells, which may allow them to evade immune destruction by cytotoxic T cells. E5 protein from BPV-1 has also been shown to have other biologic effects relevant to tumor cell development and survival, including changes in cell-to-cell communication via alterations in connexin 26 and reduction in apoptosis through several mechanisms, including increased degradation of Bax protein. 16 Other BPV-1 proteins may be important in sarcoid development, including E6 and E7. 26,27 Experimental infection of BPV in mice and hamsters will induce fibroblastic neoplasms. 12
S100 was expressed in only 12 of the 197 tumors in this study and was found in both PNSTs and sarcoids in addition to fibrosarcomas and other sarcomas. Although S100 is a marker of neural crest–derived cells and a diagnostic marker for equine PNST (schwannomas and neurofibromas), 22,31,32 it is not a specific marker and is also found in many sarcomas as well as melanomas. 22 In humans, malignant PNSTs are often S100 negative. 22 The recommendation by Bogaert et al 4 based on the study of 10 tumors that BPV PCR and S100 immunohistochemistry be used to distinguish sarcoids from PNSTs appears to be of limited practical value. They studied 10 tumors diagnosed as schwannomas. Our application of their recommendation to 197 tumors in the skin showed that classic sarcoids with biphasic morphology could be either S100 negative or positive as well as positive or negative for BPV DNA based on PCR. Similar variable test results were shown for PNSTs and fibrosarcomas.
PNSTs appear to have been overdiagnosed in our study based on histomorphology alone. As currently defined by the veterinary literature, PNSTs should be S100 positive. 31,32 Only 2 of 32 PNSTs classified morphologically were S100 positive. Forty-one percent of the tumors classified as PNSTs in our study were BPV-1/2 negative. In view of the S100 and BPV PCR results, it appears more reasonable to classify these tumors as soft tissue sarcomas with PNST histomorphology, rather than PNSTs or sarcoids. 14,15
In conclusion, bovine papillomavirus DNA is present in many spindle cell neoplasms associated with the skin in horses in addition to sarcoids. The findings suggest the possibility that BPV may play a role in tumorigenesis of many skin-associated equine tumors, including fibrosarcoma, fibroma, and other soft tissue sarcomas. S100 is expressed at low frequency in many skin-associated spindle cell tumors, including sarcoids and tumors with PNST morphology, and does not appear to be a strong marker for distinguishing whether a tumor is a sarcoid or other soft tissue sarcoma. Most tumors in our study that had been diagnosed as PNSTs were S100 negative, and 41% were BPV-1/2 negative. These may be better classified as soft tissue sarcomas with nerve sheath tumor morphology, rather than as nerve sheath tumors or sarcoids.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
