Abstract
Thyrotropin receptor (TSHR), sodium iodide symporter (NIS), pendrin, and thyroid peroxidase (TPO) are essential for the uptake of iodine by follicular thyroid cells. The aim of this study was to establish immunohistochemistry (IHC) protocols for TSHR, NIS, pendrin, and TPO in canine tissues and characterize their expression in organoids derived from canine follicular cell thyroid carcinoma (FTC) and in the respective primary tumors. This constitutes a fundamental step to establish organoids as a model to study the uptake of iodine in canine FTC. Commercially available antibodies directed against human proteins were selected. Antibody specificity was confirmed by western blot using lysates of the HTori-3 human thyroid cell line and healthy canine thyroid gland. IHC was validated using HTori-3 cells and a set of canine normal tissues including healthy thyroid gland. The expression of TSHR, NIS, pendrin, and TPO was evaluated in 3 organoid lines derived from FTC and respective primary tumors. All 4 antibodies produced specific bands by western blot and cytoplasmic labeling in follicular cells by IHC in both human HTori-3 cells and canine thyroid gland. NIS also showed basolateral membrane immunolabeling in follicular cells. All 4 proteins were highly expressed in organoids derived from FTC. The expression was similar or higher compared to the primary tumors. The results of this study characterize organoids derived from canine FTC as a suitable in vitro model to investigate iodine uptake, opening new research possibilities in the field of canine thyroid cancer therapy.
Keywords
Thyroid tumors represent 1% to 2% of all neoplasms in dogs. Median age at presentation is 9 to 10 years, and Boxers, Beagles, Golden Retrievers, and Siberian Huskies are overrepresented. 6,37 Ninety percent of canine thyroid tumors detected clinically are malignant and can be classified as either follicular cell thyroid carcinoma (FTC), which arises from thyroid follicular cells, or medullary thyroid carcinoma (MTC), which arises from the parafollicular cells (C-cells). 18 FTC accounts for 70% of canine thyroid carcinomas while MTC has a prevalence of 30%. 8 Radioiodine-131 (131I) is an effective treatment modality for invasive canine FTC and is associated with a median survival time of 27 to 30 months, while untreated dogs have a median survival time of 3 months. 30,36 Furthermore, 131I could constitute the only effective therapy against thyroid cancer metastases. However, treatment efficacy critically depends on the uptake of 131I by the primary tumor and its metastases. To improve treatment efficacy in dogs, research on modulation of 131I uptake by thyroid cancer cells is needed. One particularly important aspect therein is the development of an adequate in vitro model of canine FTC and the evaluation of the expression of proteins involved in iodine uptake.
Thyrotropin receptor (TSHR), sodium iodide symporter (NIS), pendrin, and thyroid peroxidase (TPO) are essential for the uptake and organification of iodine by thyroid follicular cells and play a crucial role in the production of thyroid hormones (Fig. 1). 16,25 TSHR is a G-protein-coupled receptor composed of 2 subunits (an extracellular thyrotropin-binding region and a transmembrane and cytosolic part responsible for intracellular signaling) and is located in the basolateral membrane of thyroid follicular cells. 39 Thyrotropin binds to the TSHR activating intracellular signaling via G-proteins and cyclic adenosine monophosphate (cAMP), which stimulates transcription of NIS, thyroglobulin, and TPO, as well as NIS-mediated iodide uptake (Fig. 1). 24,29 Functional TSHR is also present in extrathyroidal cells such as in cardiomyocytes, hematopoietic cells, human abdominal preadipocytes, orbital fibroblasts, and hair follicles. 4,5,11,19 NIS is a membrane glycoprotein mainly expressed in thyroid tissue, but also in salivary glands, gastric mucosa, and lactating mammary glands. 2 In thyroid follicular cells, NIS mediates the active transport of iodide ions across the basolateral membrane. 39 Pendrin is an anion transporter with 11 or 12 transmembrane domains and transports iodide across the apical membrane of thyroid follicular cells. 39 TPO is a crucial enzyme responsible for organification of iodine and biosynthesis of thyroid hormones. It catalyzes the iodination and coupling of tyrosine residues in thyroglobulin, leading to the synthesis of triiodothyronine and thyroxine. 20 Research on the expression of TSHR, NIS, pendrin, and TPO in thyroid follicular cells with the aim of modulating iodine uptake could lead to improvement of radioiodine treatment of thyroid diseases.
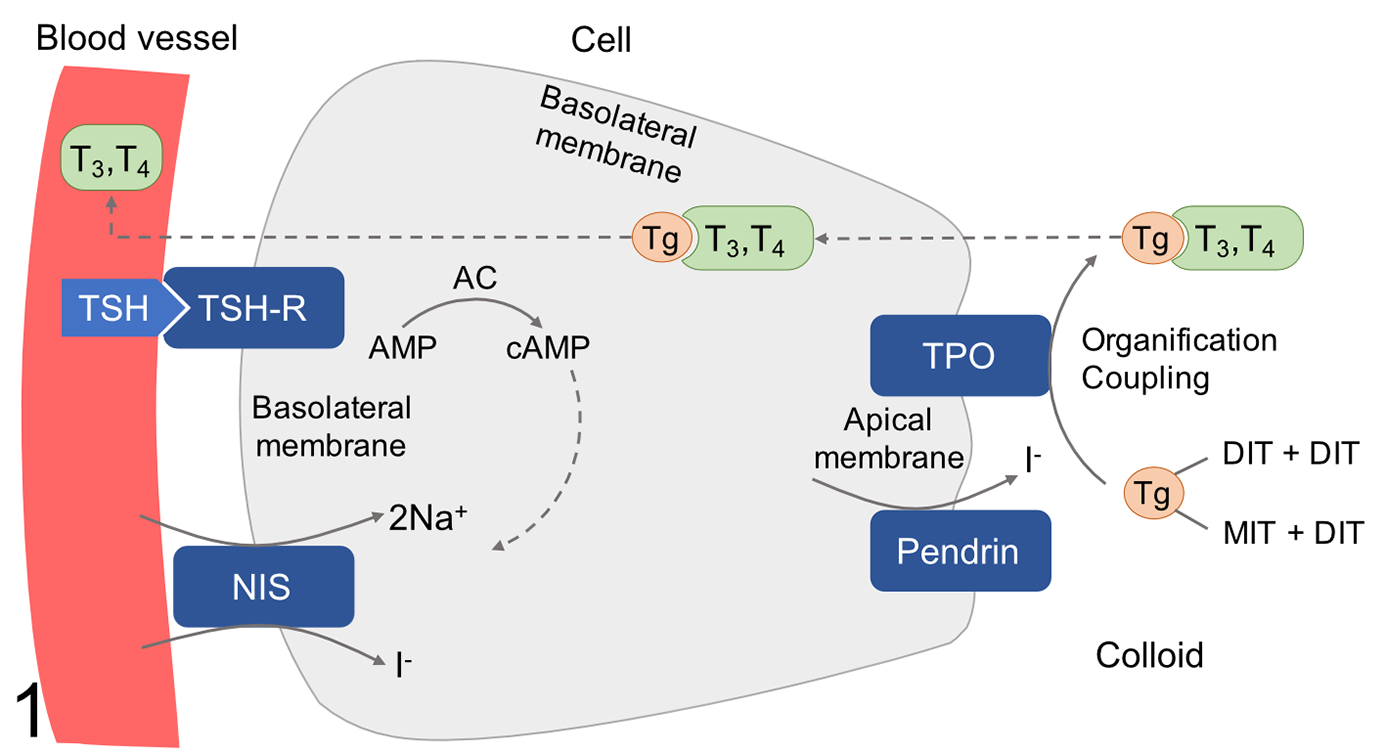
Schematic representation of proteins involved in iodine uptake and thyroid hormone production in thyroid follicular cells. Thyrotropin (TSH) stimulation of TSH receptor (TSHR) activates adenylate cyclase (AC) generating cyclic AMP (cAMP). This stimulates transcription of sodium-iodide symporter (NIS), thyroglobulin (Tg), and thyroid peroxidase (TPO), as well as NIS-mediated iodide uptake. Iodide is transferred across the apical membrane by pendrin. TPO mediates the oxidation of iodide (I−) to iodine (I0) and the iodination of tyrosine residues within the thyroglobulin backbone, a process known as organification. The coupling of monoiodotyrosine (MIT) and diiodotyrosine (DIT) leads to the production of triiodothyronine (T3) and tetraiodothyronine (T4). These ultimately undergo endocytosis and are released to the blood stream.
Tissue-derived adult stem cells and cancer cells can be grown in 3D matrix with high efficiency into self-organizing organotypic structures termed organoids. 10 Cancer organoids recapitulate features of the original tumor and can model clinically relevant drug responses, providing a unique opportunity for personalized cancer therapy and could constitute an ideal in vitro model to investigate modulation of iodine uptake by thyroid cancer cells. 10 In humans, long-term organoid cultures have been developed from colon, esophagus, pancreas, stomach, liver, retina, and endometrium, as well as from metastatic colon, prostate, and breast cancer. 7,10 Our group has recently developed stable organoid cultures derived from canine FTC, to our knowledge the first organoid culture from spontaneous thyroid cancer in any species. In order to establish patient-derived thyroid organoids as a model for iodine uptake and therapeutic studies in canine FTC, we aimed to validate the expression and subcellular localization of TSHR, NIS, pendrin, and TPO, matching it to the primary tumors. To date, immunohistochemistry (IHC) for canine proteins involved in iodine uptake has only been validated for TSHR. 22 The aim of this study was to establish IHC protocols for TSHR, NIS, pendrin, and TPO in canine tissue and to evaluate their expression in matched canine primary FTC and FTC-derived organoids.
Materials and Methods
Tissue Samples
Samples of healthy canine thyroid gland (n = 3) and FTC (n = 3) were available through the biobank of the Vetsuisse Faculty, University of Bern. This tissue collection comprises samples of thyroid tumors collected during surgery for diagnostic and curative purposes, and of healthy thyroid glands collected after euthanasia from dogs that had to be euthanized for other reasons. Parts of the thyroid samples were either stored in DMSO-containing freezing medium (45% FCS, LubioScience; 10% DMSO Sigma-Aldrich; 45% DMEM ThermoFisher) at −150 °C (for culture), were snap frozen in liquid nitrogen and stored at −80 °C (for western blot), or were fixed in 10% neutral buffered formalin for 24 to 48 hours. The latter underwent paraffin embedding and subsequent histopathological evaluation by a board-certified pathologist (MD). Thyroid tumors were classified according to World Health Organization guidelines as follicular cell tumors (follicular, compact, follicular-compact, papillary, poorly differentiated, undifferentiated, carcinosarcoma) or medullary tumors. 18 IHC for thyroglobulin (rabbit polyclonal, Agilent Technologies) and calcitonin (rabbit polyclonal, Agilent Technologies) was performed in all tumors to aid classification. The distinction between adenoma and carcinoma was based on histologic evidence of capsular invasion, vascular invasion, or metastases.
Two-Dimensional Cell Culture (HTori-3)
The human thyroid epithelial cell line HTori-3 cell line was cultured as described and maintained in complete medium (RPMI-1640, 10% FBS, 1% L-glutamine,1% penicillin-streptomycin-amphotericin B; ThermoFisher,) in standard T-75 flasks in a humidified cell incubator (21% O2, 5% CO2, 37 °C). 35 Cells were split and plated into 6-well plates 3 days before embedding, allowing an initial 72-hour baseline of uninterrupted growth acquiring 70% to 80% confluency before cell collection. For immunohistochemical analyses, HTori-3 cells at 3 passages post-thawing were processed into micro-cell-blocks (MCB), as previously described. 3 In brief, 1.0 × 106 trypsinized cells were collected in a 1.5 mL Eppendorf tube. Cells were washed with warm DPBS and pelleted by centrifugation at 450g. After aspiration of the supernatant, cells were resuspended in human plasma derived from whole blood (Interregional Blood Transfusion SRC) and Thrombin (Diagnotec) (ratio 5:1) followed by 3-minute incubation at room temperature (RT). The clot was fixed with 4% PFA for 45 minutes protected from light. After a DPBS wash, supernatant was aspirated and cells were incubated in hematoxylin and DPBS solution (ratio 1:8) on a rocker shaker for 15 minutes at RT. The counterstained clot was transferred to a plastic micro-cassette for paraffin embedding for IHC.
For western blot analysis, HTori-3 cells at 3 passages post-thawing were harvested, 1.0 × 106 cells for each replicate (n = 8 technical replicates). Cells were washed with warm PBS before plates were put on ice at 4 °C. Using a cell scraper cells were detached and instantly collected in a 1.5 mL Eppendorf tube followed by a short spin 450g at 4 °C. Subsequently, the cells were snap frozen in liquid nitrogen and stored at −80 °C until processing.
Organoid Cultures
The protocol for establishing organoid cultures derived from canine FTC is described extensively elsewhere (Tièche et al, unpublished data). Briefly, tumor samples from 3 euthyroid dogs with naturally occurring FTC of compact type (case 1), follicular-compact type (case 2), follicular type (case 3) were frozen in DMSO-containing freezing medium (45% FCS, LubioScience; 10% DMSO, Sigma-Aldrich; 45% DMEM, ThermoFisher) within 24 hours after thyroidectomy and stored at −150 °C until processing. After thawing, the tissue was digested with collagenase IV and dissociated mechanically. Cells were subsequently seeded in Cultrex Basement Membrane Extract (Amsbio) in 30 µl droplets on 24-well culture plates (Huberlab). Five hundred microliters of prewarmed culture medium was added after 30 to 45 minutes. The basic culture medium composed of Advanced DMEM/F12 (ThermoFisher), supplemented with L-glutamine (ThermoFisher), HEPES buffer (Sigma-Aldrich), penicillin-streptomycin (ThermoFisher), N-acetylcysteine (Sigma-Aldrich), B-27 supplement (ThermoFisher), epithelial growth factor (Prepotech), Noggin, Rspondin-1 (both self-produced using stably transduced 293 HEK cells, provided by Dr Calvin Kuo, Stanford University), and ROCK-inhibitor Y-27632 (AbMole Bioscience). Organoids were cultured in a 5% CO2 atmosphere and medium was changed twice a week. Organoids were passaged after 9 to 14 days using TrypLE Express (ThermoFisher) for 10 minutes and mechanical dissociation. After 15 to 24 days (passage 1), organoids were formalin-fixed, pelleted in 2.5% agarose, paraffin-embedded, and processed for hematoxylin-eosin staining and IHC. Thyroid follicular cell origin of the organoids was confirmed with IHC for thyroid transcription factor-1 (TTF-1) and thyroglobulin.
Antibodies
Commercially available antibodies directed against human proteins were selected according to the homology of the epitopes (accessed in NCBI) to the canine protein. Rabbit polyclonal anti-TPO antibody (ab203057, Abcam), rabbit polyclonal anti-SLC26A4 (pendrin) antibody (ab98091, Abcam), rabbit polyclonal anti-TSHR (C-terminal) antibody (ab188902, Abcam), and mouse monoclonal anti-SLC5A5/NIS antibody (LS-C16858, LifeSpan BioSciences, LabForce) were used.
Western Blot
To prepare the protein lysate, HTori-3 cells and samples from snap frozen healthy canine thyroid gland were homogenized in RIPA buffer with phosphatase (Thermo Scientific) and protease inhibitor (Thermo Scientific), sonicated (amplitude 30 for 1 minute), and centrifuged at 12 000 rpm for 20 minutes. The supernatants were then recovered and protein concentration was determined by BCA protein assay kit (Thermo Scientific). Samples were mixed in Protein Loading Buffer with 2-mercaptoethanol (made in Institute of Animal Pathology, Vetsuisse Faculty, Bern, Switzerland). After electrophoresis (SDS-PAGE) on a 7.5% polyacrylamide gel, samples were transferred to PVDF membranes for 1 hour at 100 V in transfer buffer (25 mM TRIS, 192 mM glycine, 20% methanol, pH 8.3). Membranes were blocked in 5% nonfat dry milk in TBS-T (TRIS 100 mM, pH 7.5, NaCl 0.9%, Tween 0.05%) for 1 hour at RT. Antibodies for NIS, pendrin, and TPO were diluted according to the manufacturer’s instructions. TSHR was diluted 1 in 750 after a trial with 1 in 1000 dilution produced a blot of insufficient quality. Incubation with primary antibodies for TSHR (1 in 750 dilution), NIS (1 in 1000 dilution), pendrin (1 in 500 dilution), and TPO (1 in 1000 dilution) was performed in TBS-T overnight at 4 °C. Membranes were washed 3 times for 10 minutes in TBS-T and incubated with anti-rabbit (TSHR, pendrin, TPO 1 in 2500 dilution, Cell Signaling Technology) and anti-mouse (NIS, 1 in 2500 dilution, Cell Signaling Technology) secondary antibodies for 1 hour at RT. After washing 6 times for 5 minutes in TBS-T, blots were revealed using a commercial ECL mixture (GE Healthcare Life Sciences). Images were taken using a Vilber Fusion FX (Collégien, France) machine.
Immunohistochemistry
For antibody validation, the following formalin-fixed and paraffin-embedded (FFPE) canine tissues were used as a positive control: healthy thyroid gland (all antibodies), salivary gland (NIS), stomach (NIS), and skin (TSHR). In addition, FFPE pellets of human HTori-3 cells were also used as positive control for all antibodies. As a negative control, the following FFPE canine tissue were used: small intestine (TSHR), liver (NIS, pendrin, TPO), lymph node (NIS, pendrin, TPO), skin (NIS, pendrin, TPO), and brain (NIS, TPO). This tissue selection was based on the expression of the respective antigens in human tissues that were reported to highly express or to specifically lack expression of respective proteins. 2,5,9,15,25
These tissues were obtained from dogs euthanized for different reasons, few hours after euthanasia, and were processed as described above. After successful antibody validation, protein expression was evaluated on FFPE sections of the 3 FTCs and the organoid lines derived from these tumors.
From all tissues, pelleted cells, and organoids, 5-µm sections were mounted on positively charged glass slides. After dewaxing and rehydration, endogenous peroxidase was blocked with 3% H2O2 for 20 minutes. Antigen retrieval was performed in citrate buffer (10 mM sodium citrate, 0.05% Tween 20, pH 6.0) by microwaving at 90 °C for 5 minutes for TSHR, and by pressure cooking for NIS (115 °C, 0.7 bar for 20 minutes), pendrin (107 °C, 0.3 bar for 20 minutes), and TPO (107 °C, 0.3 bar for 10 minutes). Ten percent goat serum was used as blocking agent. Sections were incubated with primary antibodies horizontally in a humid chamber for 1 hour at RT for pendrin (1 in 100 dilution) and overnight at 4 °C for TSHR (1 in 50 dilution), NIS (1 in 100 dilution), and TPO (1 in 100 dilution). Evaluation of the optimal concentration of each primary antibody was performed with serial antibody dilutions using healthy canine thyroid gland as positive control. As a negative control for all antibodies, the primary antibody was replaced with PBS. After washing with PBS, incubation with biotinylated link secondary antibodies (Dako LSAB2 System Streptavidin-HRP, Dako North America) was performed. 3-Amino-9-ethylcarbazole (AEC) in substrate buffer solution (Dako AEC Substrate Chromogen, Dako North America) served as chromogen and was allowed to react for 9 minutes. After washing, sections were counterstained with hematoxylin, rinsed in tap water, and mounted with cover slips.
All sections were examined by 2 investigators (JJ, MD), who were blinded to the clinical information of the patients. The distribution of immunohistochemical labeling in neoplastic cells was subjectively scored as follows: 0 (no positive cells), 1 (1% to 33% positive cells), 2 (33% to 66% positive cells), and 3 (67% to 100% positive cells). Labelling intensity was subjectively scored as follows: 0 (negative), 1 (weak), 2 (moderate), and 3 (strong). 13 Photomicrographs from immunolabeled healthy thyroid gland and tumors were taken from scanned slides using a Pannoramic p250 scanner (3D HISTECH), and from organoids with an Olympus BX51 microscope (Olympus) using the CellSens Standard software (Olympus).
Results
Western Blot
TSHR, NIS, pendrin, and TPO produced specific protein bands in the western blot of canine thyroid tissue and human thyroid cell line HTori-3. TSHR showed bands at 50 and 60 kDa for healthy canine thyroid gland and at 60 kDa for HTori-3 cells. NIS revealed bands at 50 kDa for canine thyroid gland and at 58 kDa for HTori-3 cells. Pendrin showed a band at 85 kDa for both healthy canine thyroid gland and HTori-3 cells. TPO showed bands at 50 kDa and 120 kDa for both canine thyroid gland and HTori-3 cells (Fig. 2).
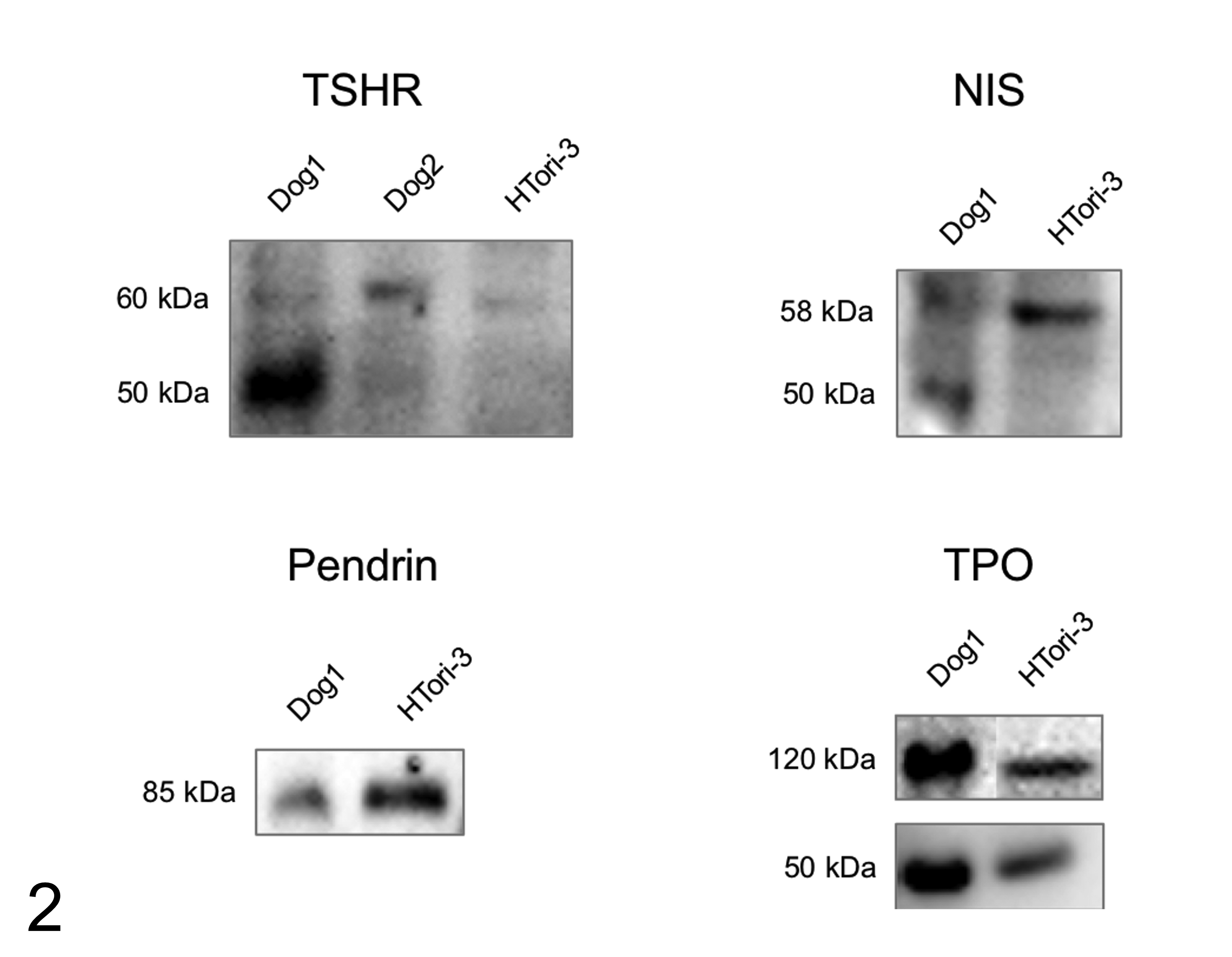
Western blot results for thyrotropin receptor (TSHR), sodium iodide symporter (NIS), pendrin, and thyroid peroxidase (TPO) protein lysate from healthy canine thyroid gland (dog 1 and dog 2 samples) and HTori-3 healthy human thyroid cell line. TSHR showed bands at 50 and 60 kDa for healthy canine thyroid gland and at 60 kDa for HTori-3 cells. NIS revealed bands at 50 kDa for canine thyroid gland and at 58 kDa for HTori-3 cells. Pendrin showed a band at 85 kDa for both healthy canine thyroid gland and HTori-3 cells. TPO showed bands at 50 kDa and 120 kDa for both canine thyroid gland and HTori-3 cells.
The predicted molecular weight of NIS in dogs is 68 kDa (Uniprot E2RMU7) and in humans 68.6 kDa (Uniprot Q92911) to 87 kDa have been reported. 17,31,39 The predicted molecular weight of TSHR in dogs is 86.4 kDa (Uniprot P14763) and in humans 84 to 86.8 kDa (Uniprot P16473). 32,39 The predicted molecular weight of pendrin in humans and dogs is 85.8 kDa (Uniprot O43511, E2RRQ1) and the predicted molecular weight of TPO in dogs is 101.4 kDa (Uniprot Q8HYB7) and in humans 100 to 110 kDa (Uniprot P07202). 15,26,33,34
Validation of Immunohistochemistry
Immunohistochemistry for all antibodies showed specific positive labeling in human HTori-3 cells as well as in healthy canine thyroid gland. Specific positive labeling was also observed in the additional canine positive controls: salivary gland (NIS), stomach (NIS), and skin (TSHR). Describing the IHC labeling in detail, HTori-3 cells showed moderate membranous and weak cytoplasmic labeling for TSHR, weak membranous and cytoplasmic labeling for NIS, strong membranous and moderate cytoplasmic labeling for pendrin, and very weak cytoplasmic labeling for TPO (Suppl Figs. S1–S4). In healthy canine thyroid gland, thyroid follicular cells showed weak to moderate cytoplasmic labeling for TSHR, weak cytoplasmic labeling for TPO, moderate cytoplasmic labeling for NIS, and strong cytoplasmic labeling for pendrin (Figs. 3 –6). For NIS, additional immunolabeling of the basolateral membrane was observed in thyroid follicular cells, stronger than the cytoplasmic labeling (Fig. 4). Parathyroid gland and C-cells were negative for all 4 antibodies. In healthy canine salivary gland and stomach (additional positive controls for NIS), NIS showed labeling of salivary gland ductal epithelial cells and of gastric mucous neck cells (Suppl Figs. S5, S6). In healthy canine skin (additional positive controls for TSHR), TSHR showed labeling in epidermal and hair follicle keratinocytes and hair follicle mesenchyme (Suppl Fig. S7). No immunolabeling was observed in any of the respective negative controls: healthy canine small intestine (TSHR), skin, brain, liver, and lymph nodes (NIS and TPO), and skin, liver, and lymph nodes (pendrin). Furthermore, the sections incubated with PBS without primary antibody remained unlabeled for all antibodies (Suppl Figs. S8–S10).
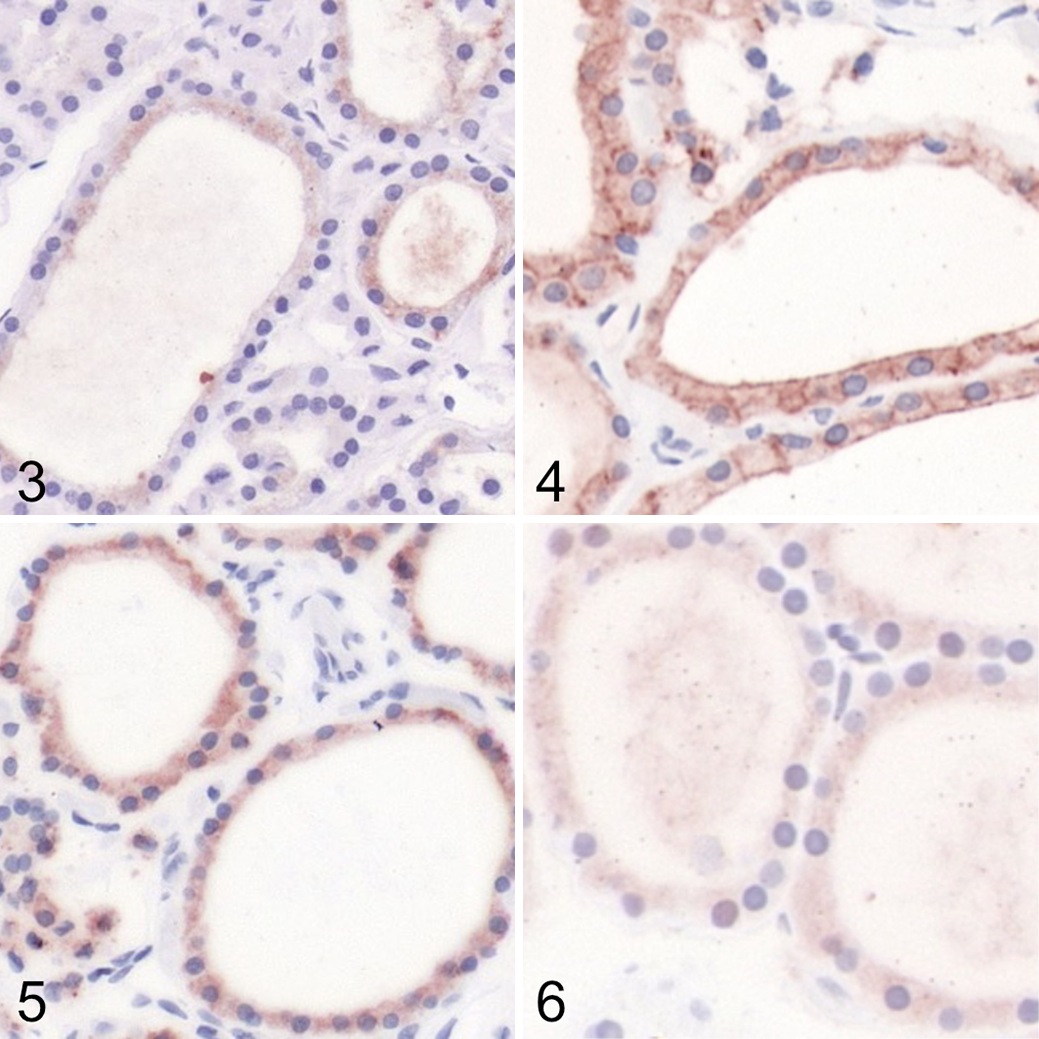
Healthy thyroid gland, dog. Immunoperoxidase; AEC chromogen with hematoxylin counterstain.
IHC of Organoids and Primary Tumors
The results of the IHC of the 3 primary follicular cell carcinomas (follicular, compact, and follicular-compact) and 3 organoid lines derived from these tumors are summarized in Table 1 and illustrated in Figures 7 to 16. All tumors and organoid lines showed at least weak expression of all markers tested. Distinct labelling patterns were mostly consistent between primary tumors and the corresponding organoids.
Immunohistochemistry Results for Thyrotropin receptor (TSHR), Sodium Iodide Symporter (NIS), Pendrin, and Thyroid Peroxidase (TPO) in 3 Primary Canine Follicular Cell Thyroid Carcinomas (FTC) and in 3 Organoid Lines Derived From These Tumors. Case 1, Compact FTC; Case 2, Follicular-Compact FTC; Case 3, Follicular FTC.
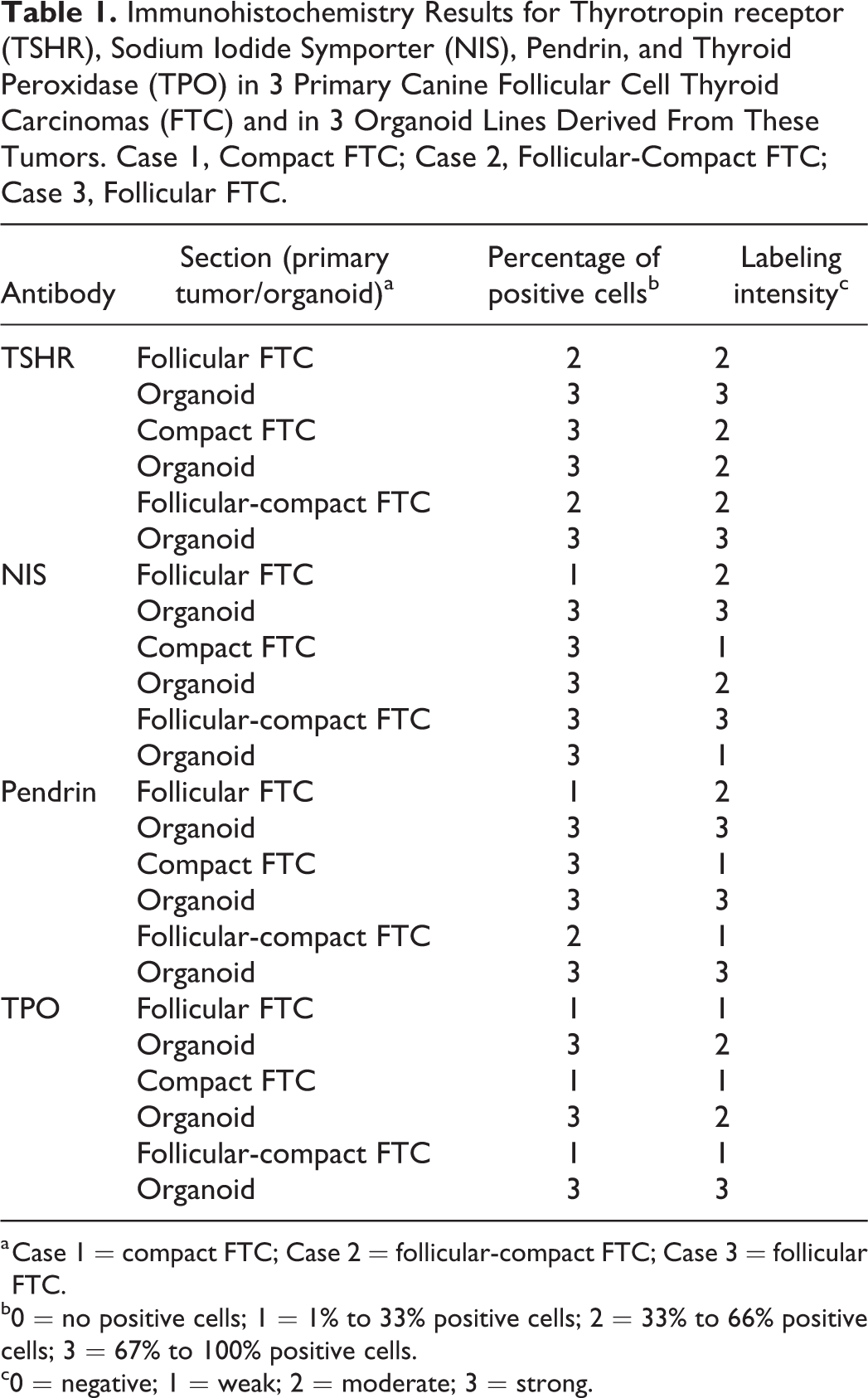
a Case 1 = compact FTC; Case 2 = follicular-compact FTC; Case 3 = follicular FTC.
b0 = no positive cells; 1 = 1% to 33% positive cells; 2 = 33% to 66% positive cells; 3 = 67% to 100% positive cells.
c0 = negative; 1 = weak; 2 = moderate; 3 = strong.
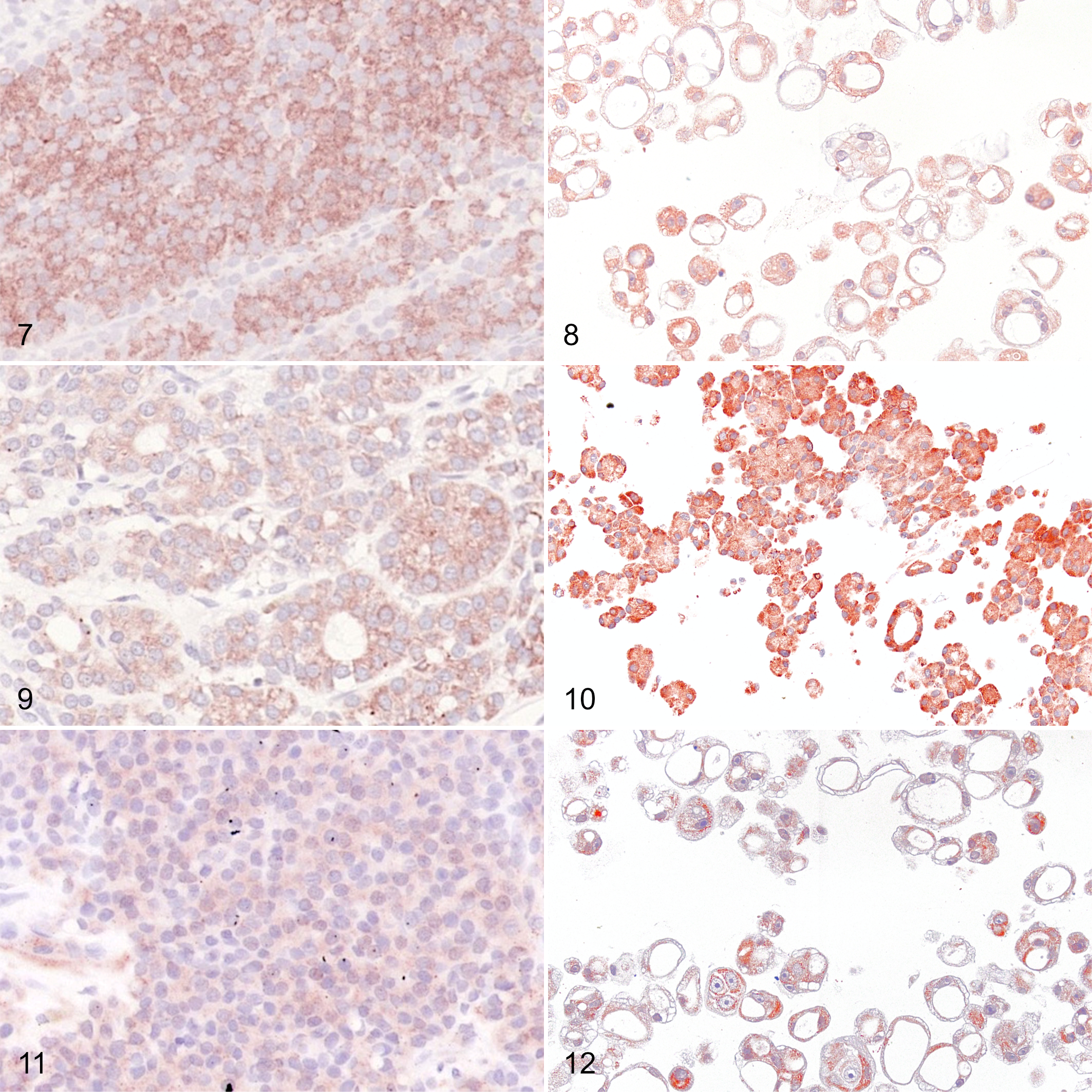
Compact follicular cell thyroid carcinoma (FTC), dog, case 1, and organoids derived from this tumor. Both primary tumor cells (Fig. 7) and organoid cells (Fig. 8) show moderate to strong cytoplasmic labeling for thyrotropin receptor. Immunoperoxidase; AEC chromogen with hematoxylin counterstain.
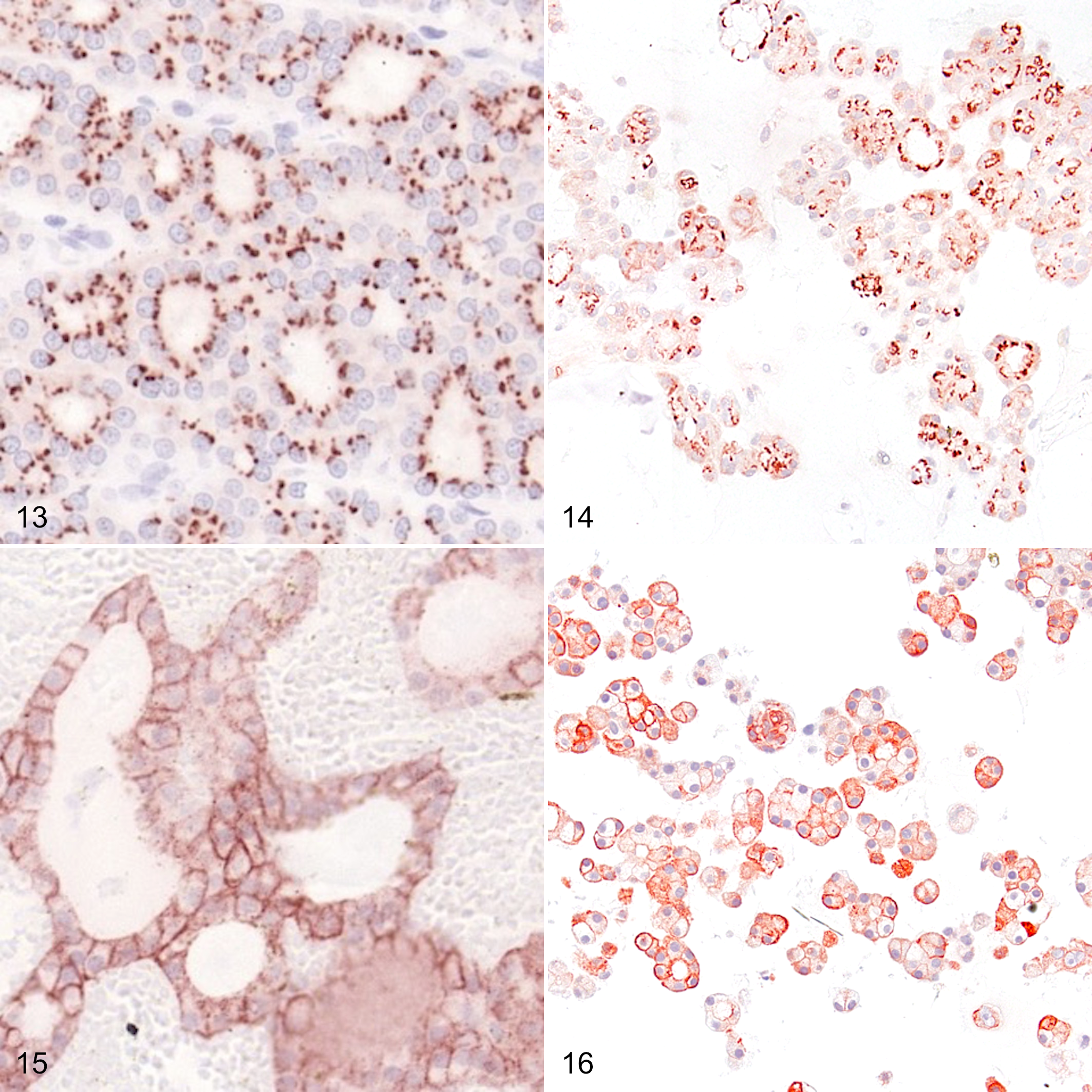
Follicular-compact follicular cell thyroid carcinoma (FTC), dog, case 2, and organoids derived from this tumor. Both primary tumor cells (Fig. 13) and organoid cells (Fig. 14) display focal granular immunolabeling for sodium iodide symporter (NIS) in the apical cytoplasm. Immunoperoxidase; AEC chromogen with hematoxylin counterstain.
Discussion
In this study, IHC protocols were established for TSHR, NIS, pendrin, and TPO in canine tissue. To the best of our knowledge, this is the first study to validate IHC for such a complete group of proteins involved in the uptake of iodine and thyroid hormone production in dogs. Furthermore, this study showed that organoids derived from canine FTC conserve the expression of these proteins and provide a new in vitro model to investigate modulation of iodine uptake. Such a model may help to improve and optimize radioiodine therapy for canine thyroid follicular carcinoma.
In order to confirm that our antibodies raised against human proteins could detect the target proteins, we performed western blot and IHC on the HTori-3 human thyroid cell line, which expresses features of thyroid epithelial function, including iodide trapping and thyroglobulin production. 1 Our results show that the antibodies used could detect not only human but also canine TSHR, NIS, pendrin, and TPO. Western blot for NIS showed bands at 50 kDa in canine healthy thyroid gland and around 58 kDa in the human cell line. Although NIS has a molecular mass of approximately 87 kDa in its fully glycosylated form, several studies have shown that it is possible to detect a partially glycosylated form with western blot at 50 to 56 kDa. 14,17,39 Western blot for TSHR showed bands at 50 and 60 kDa in the canine healthy thyroid gland and 60 kDa in the HTori-3 cells. Although the molecular mass of TSHR is 84 kDa, a glycosylated form at 63 kDa and a non-glycosylated form at 50 kDa have been detected by western blot in human thyroid gland tissue. 21,39 We believe our western blot detected the glycosylated form of TSHR in HTori-3 cells and both glycosylated and non-glycosylated forms in canine healthy thyroid gland, as already described in previous studies. 21,39 The predicted molecular weight for pendrin of 85 kDa corresponded to the observed bands in both healthy canine thyroid gland and HTori-3 thyroid cell line. Western blot of TPO revealed bands at 50 and 120 kDa in both canine healthy thyroid gland and the HTori-3 human cell line. The predicted molecular weight of TPO is approximately 100 to 110 kDa. 15,26 The detection of TPO bands at slightly higher molecular weight than 116 kDa has been attributed to varying reducing conditions of the technique. 23 Simultaneously, the detection of bands of TPO at lower molecular weights (around 54 kDa) has also been described to correspond to TPO cleavage during sample solubilization and trypsination. 23,38 Further developments in the project could include the use of synthetic proteins, both full-sized and isoforms in various states of glycosylation, as a positive control for western blot. In addition, mass spectrometry could help in the identification of the different bands.
Our results correlated with the reported human expression patterns for all markers. For example, we observed specific NIS immunolabeling in mucous neck cells of canine stomach and in ductal epithelial cells of canine salivary gland, identical to human tissues. This finding is in agreement with the fact that these organs typically show iodine uptake in canine scintigraphy studies. 2,28 Furthermore, TSHR immunolabeling was present in keratinocytes of the epidermis and hair follicles, and in the mesenchyme of hair follicles of canine skin. Previous human studies have shown that intracutaneous TSHR is fully functional in situ, and that hair follicle of female individuals are direct targets for nonclassical, extrathyroidal TSH bioregulation. 5,9 These findings additionally corroborate our western blot results; thus, the specificity of these antibodies for the canine protein orthologues can be presumed.
The second aim of this study was to investigate the expression of TSHR, NIS, pendrin, and TPO in canine follicular thyroid carcinomas and organoids derived from these tumors. Although TSHR is a membranous protein, we mainly observed cytoplasmic labeling in follicular cells in canine FTCs, but also in healthy thyroid gland. This is in agreement with several studies in both humans and dogs. 12,22
NIS also performs its function in the basolateral membrane of the thyrocyte. In our study, we observed basolateral membranous immunolabeling as well as cytoplasmic immunolabeling in both healthy canine thyroid gland and in FTC cells, although basolateral labeling was stronger. In humans, immunolabeling for NIS is also described at the basolateral membrane in healthy thyroid gland and in follicular carcinoma. 2,12
Although pendrin and TPO are functional at the apical membrane of thyroid follicular cells, we observed diffuse cytoplasmic labeling of these cells with both markers in healthy canine thyroid gland and in FTC cells as reported in humans. 12,25,27
Organoids derived from canine FTC showed immunolabeling for TSHR, NIS, pendrin, and TPO comparable to the primary tumors. This proves that these proteins are expressed in the cultured organoids and could constitute an interesting in vitro model for research on modulation of iodine uptake and improvement of radioiodine therapy. Interestingly, the labeling intensity for all markers was commonly stronger in the organoids than in the primary tumors. This could be explained by the fact that iodine was not included in the culture medium, and these proteins were overexpressed. Other explanations could be that organoids originated from a part of the tumor with higher protein expression than the part taken for the paraffin block, or that the disparities in expression are caused by different microenvironmental conditions during fixation in formaldehyde due to volume differences.
In conclusion, antibodies directed against human TSHR, NIS, pendrin, and TPO are suitable to detect the canine orthologues. IHC protocols using the validated antibodies were established for canine healthy thyroid gland tissue, FTCs, and organoids derived from FTCs. FTC organoids conserve expression of these proteins and hence potentially constitute an in vitro model to perform research on modulation of iodine uptake and improvement of radioiodine therapy for thyroid cancer.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211018813 - Validation of Immunohistochemistry for Canine Proteins Involved in Thyroid Iodine Uptake and Their Expression in Canine Follicular Cell Thyroid Carcinomas (FTCs) and FTC-Derived Organoids
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211018813 for Validation of Immunohistochemistry for Canine Proteins Involved in Thyroid Iodine Uptake and Their Expression in Canine Follicular Cell Thyroid Carcinomas (FTCs) and FTC-Derived Organoids by Jana Jankovic, Martina Dettwiler, Martin González Fernández, Eve Tièche, Kerstin Hahn, Simon April-Monn, Matthias S. Dettmer, Martin Kessler, Sven Rottenberg and Miguel Campos in Veterinary Pathology
Footnotes
Acknowledgements
We thank Sohvi Blatter and Olivia Richard, who trimmed the tumor samples, and Erika Bürgi, Manuela Bozzo, and Bettina de Breuyn for preparing the slides for IHC. We thank Paola Francica and Nadja Gerber for their support in western blot and IHC, and Renaud Sylvain Maire for assisting with 2D cell cultures (HTori-3). We thank Marine Inglebert and Ismar Klebic for their support in the culture of organoids.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Albert Heim Stiftung, the Tierärztliche Verrechnungsstelle (TVS), ECVIM-CA Clinical Studies Fund, the European Society of Veterinary Endocrinology, and the Small Animal Clinic of the Vetsuisse Faculty, University of Bern.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
