Abstract
Research on modulation of iodine uptake by thyroid cells could help improve radioiodine treatment of dogs with thyroid tumors. The aim of this study was to characterize the immunohistochemical expression of thyroid transcription factor-1 (TTF-1), thyroglobulin, thyrotropin receptor (TSHR), sodium iodide symporter (NIS), pendrin, thyroid peroxidase (TPO), vimentin, and Ki-67 in follicular cell thyroid carcinomas (FTCs) and medullary thyroid carcinomas (MTCs), and to compare protein expression between FTC causing hyperthyroidism and FTC of euthyroid dogs. Immunohistochemistry was performed in 25 FTCs (9 follicular, 8 follicular-compact, and 8 compact) and 8 MTCs. FTCs and MTCs were positive for TTF-1, and expression was higher in FTCs of euthyroid dogs compared with FTCs of hyperthyroid dogs (P= .041). Immunolabeling for thyroglobulin was higher in follicular and follicular-compact FTCs compared with compact FTCs (P = .001), while vimentin expression was higher in follicular-compact FTCs compared with follicular FTCs (P = .011). The expression of TSHR, NIS, pendrin, and TPO was not significantly different among the different subtypes of FTCs or between FTCs causing hyperthyroidism and FTCs in euthyroid dogs. TSHR, NIS, pendrin, and TPO were also expressed in MTCs. Ki-67 labeling index was comparable between FTCs and MTCs, and between FTCs causing hyperthyroidism and FTCs in euthyroid dogs. Proteins of iodine transport were also expressed in canine MTCs, which could have implications for diagnosis and treatment. The different expression of thyroglobulin and vimentin between FTC histological subtypes could reflect variations in tumor differentiation.
Keywords
Thyroid carcinomas are common endocrine neoplasms in dogs. 2 According to recent studies, approximately 64% of canine thyroid tumors are follicular cell thyroid carcinomas (FTCs) and 36% are medullary thyroid carcinomas (MTCs).6,9 Canine FTC can be classified histologically as well differentiated (follicular, compact, follicular-compact, papillary), poorly differentiated, anaplastic, and carcinosarcoma.21,26 Histologically, canine MTCs have a solid growth pattern and may be difficult to distinguish from compact FTCs based on histology alone. Immunohistochemistry (IHC) for thyroglobulin, calcitonin, or markers of neuroendocrine tissue (eg, chromogranin A, synaptophysin) can be used to differentiate these 2 tumor types.26,27 Radioiodine-131 (131I) is an effective treatment modality for invasive and/or metastatic canine FTC and is associated with a median survival time of 27 to 30 months, while untreated dogs have a median survival time of only 3 months.23,46 In people with well-differentiated follicular thyroid cancer, 131I therapy is also highly effective, especially in patients with metastasis. 28 The loss of differentiation of thyroid features, such as iodide uptake and organification, can lead to radioactive iodine-refractory cancer cells. 1 Although not yet investigated in dogs, the use of 131I has also been reported in the treatment of MTCs in people.16,29 Research on the expression of proteins of iodine transport by thyroid cancer cells could help develop new strategies to improve 131I treatment in dogs with thyroid cancer.
Thyrotropin receptor (TSHR), sodium iodide symporter (NIS), pendrin, and thyroid peroxidase (TPO) are proteins essential for iodine transport into thyroid cells, iodine organification, and production of thyroid hormones.
Recently, IHC has been validated for TSHR, NIS, pendrin, and TPO proteins in canine thyroid tissue. 22 However, these markers have not yet been evaluated in the different types of canine FTCs and MTCs. Furthermore, differences in the expression of these proteins between FTCs causing hyperthyroidism and FTCs in euthyroid dogs have not been studied. If the expression of these proteins is associated with tumor histological subtypes and the occurrence of hyperthyroidism, then this could have implications for research on the modulation of iodine uptake and improvement of 131I therapy. Thyroid transcription factor-1 (TTF-1) is essential for thyroid organogenesis and governs thyroid functions by regulating the expression of thyroid-specific genes, including genes encoding thyroglobulin, TPO, TSHR, and NIS in thyroid follicular cells. 4 In human FTCs, high Ki-67 labeling index is associated with shorter time to metastasis and shorter disease-free survival. 43 In canine thyroid carcinomas, an increased Ki-67 labeling index has also been associated with shorter time to metastasis. 6 Vimentin is regarded as a canonical marker of epithelial–mesenchymal transition, a cellular reprogramming process in which epithelial cells acquire a mesenchymal phenotype that causes them to dramatically alter their shape and exhibit increased motility.33,45 Vimentin has been reported to be highly expressed in canine thyroid carcinosarcoma and rarely expressed in canine MTC.20,34,36
The aim of this study was to characterize the immunohistochemical expression of TTF-1, thyroglobulin, TSHR, NIS, pendrin, TPO, vimentin, calcitonin, and Ki-67 in a collection of canine FTCs and MTCs and to compare protein expression between FTCs causing hyperthyroidism and FTCs in euthyroid dogs.
Materials and Methods
Tumor Samples
Twenty-eight canine thyroid tumors collected between 2018 and 2020 from different clinics in Switzerland, Germany, and Italy were included in this study. All tumor samples were surgical specimens taken during thyroidectomy and were archived at the Institute of Animal Pathology of the University of Bern. All tumors were fixed in 10% neutral-buffered formalin for 24 to 48 hours before embedding in paraffin. In addition, 5 formalin-fixed paraffin-embedded samples of thyroid tumors causing hyperthyroidism archived at the University of Ghent were also included. Gross tumor size (maximum diameter of the tumor in cm) was recorded immediately after surgery using a ruler (n=20). If this information was not available, operative imaging measurements were taken from the medical records, with preference to those measurements taken by computed tomography (CT) (n=11) or cervical ultrasound (n=2). Histopathological evaluations were performed by a board-certified pathologist (M.D.), and tumors were classified according to World Health Organization guidelines as follicular cell tumors (follicular, compact, follicular-compact, papillary, poorly differentiated, undifferentiated, carcinosarcoma) or medullary tumors. 26 IHC for thyroglobulin and calcitonin was performed on all tumors to aid classification (Table 1). Distinction between adenoma and carcinoma was based on histologic evidence of invasion through/penetration of the capsule, vascular invasion, or metastasis. Mitotic count (MC) was determined by counting the number of mitotic figures in 10 fields of 400× magnification (total area of 10 HPFs = 2.37 mm2).
Details of antibodies and immunohistochemistry (IHC) methods.
Abbreviations: anti-TPO, anti-thyroid peroxidase; anti-SLC26A4, anti-pendrin; anti-TSHR, anti-thyroid-stimulating hormone receptor; anti-SCL5A5, anti-sodium iodide symporter; anti-TTF1, anti-thyroid transcription factor-1.
Immunohistochemistry
Commercially available antibodies previously validated for IHC in the canine thyroid gland were selected (Table 1). 22 IHC for TSHR, NIS, pendrin, and TPO was performed on 5-µm sections mounted on positively charged glass slides. After dewaxing and rehydration, endogenous peroxidase was blocked with 3% H2O2 for 20 minutes. Antigen retrieval was performed in citrate buffer (10 mM Na-citrate, 0.05% Tween 20, pH 6.0) (Table 1). Ten percent goat serum was used as blocking agent. Sections were incubated with primary antibodies horizontally in a humid chamber (Table 1). Healthy canine thyroid gland was used as a positive control. Negative controls were performed for all antibodies by replacing the primary antibody with phosphate-buffered saline. After washing with phosphate-buffered saline, incubation with biotinylated link secondary antibodies (Dako LSAB2 System Streptavidin-HRP, Dako North America, California, USA) was performed. 3-amino-9-ethylcarbazole (AEC) in substrate buffer solution (Dako AEC Substrate Chromogen, Dako North America, California, USA) served as chromogen and was allowed to react for 9 minutes. After washing, sections were counterstained with hematoxylin, rinsed in tap water, and mounted with coverslips.
All sections were examined by 2 investigators (J.J. and board-certified pathologist M.D.), who were blinded to the clinical information of the patients. The number of immunohistochemically labeled cells was subjectively scored as 0, no positive cells; 1, 1% to 33% positive cells; 2, 34% to 66% positive cells; or 3, 67% to 100% positive cells. Labeling intensity was subjectively scored as 0, no positive cells; 1, barely perceptible; 2, positive but weaker than the positive control; or 3, comparable to the positive control. An overall IHC score was calculated by multiplying the score of percent positive cells by labeling intensity as previously described. 18
IHC for TTF-1, thyroglobulin, calcitonin, vimentin, and Ki-67 was performed using a Bond III automated immunostainer (Leica Biosystems, Wetzlar, Germany) in an external laboratory (Pathologie Länggasse, Ittigen, Switzerland) with reagents and protocols recommended by the manufacturer (Table 1). Two percent dog serum (LabForce, Muttenz, Switzerland) was used in the negative controls. Healthy canine thyroid gland was used as a positive control for TTF-1, thyroglobulin, and calcitonin. A canine cutaneous mast cell tumor was used as a positive control for Ki-67. The percentage of positive cells for TTF-1, thyroglobulin, calcitonin, and vimentin was estimated subjectively by a board-certified veterinary pathologist (M.D.). To determine the Ki-67 labeling index, 2 photomicrographs were taken at 400× magnification (0.237 m2 field of view) in the region of highest positivity. At least 1000 neoplastic cells were evaluated in each photomicrograph using image analysis software (Fiji, ImageJ, National Institutes of Health, Bethesda, USA). The higher index of the 2 photographs was reported.
Thyroid Status
Thyroid status was determined at the time of diagnosis (before surgery) by measurement of serum T4 and TSH concentrations. This took place in different laboratories. Dogs were considered euthyroid if the T4 concentration was within reference range; hyperthyroid if the T4 concentration was above reference range; and hypothyroid if the T4 concentration was below reference range and the TSH concentration was above reference range.
Statistical Analysis
Statistical analysis was performed using MedCalc Statistical Software version 20.111 (MedCalc Software Ltd, Ostend, Belgium, 2022). To determine whether data were normally distributed, the D’Agostino-Pearson test was used. For comparisons between FTCs and MTCs and between FTCs causing hyperthyroidism and FTCs in euthyroid dogs, a T-test was used if data were normally distributed, while a Mann-Whitney (MW) test was used if data were not normally distributed. For comparisons between the different histological subtypes of FTC, the Kruskal-Wallis (KW) test was used. Significance level was set at 5% for all analyses.
Results
IHC was performed on 25 FTCs (9 follicular, 8 compact, and 8 follicular-compact) and 8 MTCs. There were 20 tumors from euthyroid dogs (14 FTCs and 6 MTCs), 8 tumors from hyperthyroid dogs (all FTCs), 2 tumors from hypothyroid dogs (1 FTC and 1 MTC), and thyroid function was not assessed in 3 dogs. Results for all tumors are summarized in Supplemental Table S1. For all parameters, median and interquartile ranges (IQRs) are provided below. For results of immunohistochemistry, median and IQR for percentage of positive tumor cells are given below, in Table 2 and in Supplemental Table S1.
Median and interquartile range for maximum diameter of the tumor, T4 concentration, mitotic count, Ki-67, vimentin, TTF-1, thyroglobulin, TSHR, NIS, pendrin, and TPO in 25 canine FTCs and 8 MTCs.
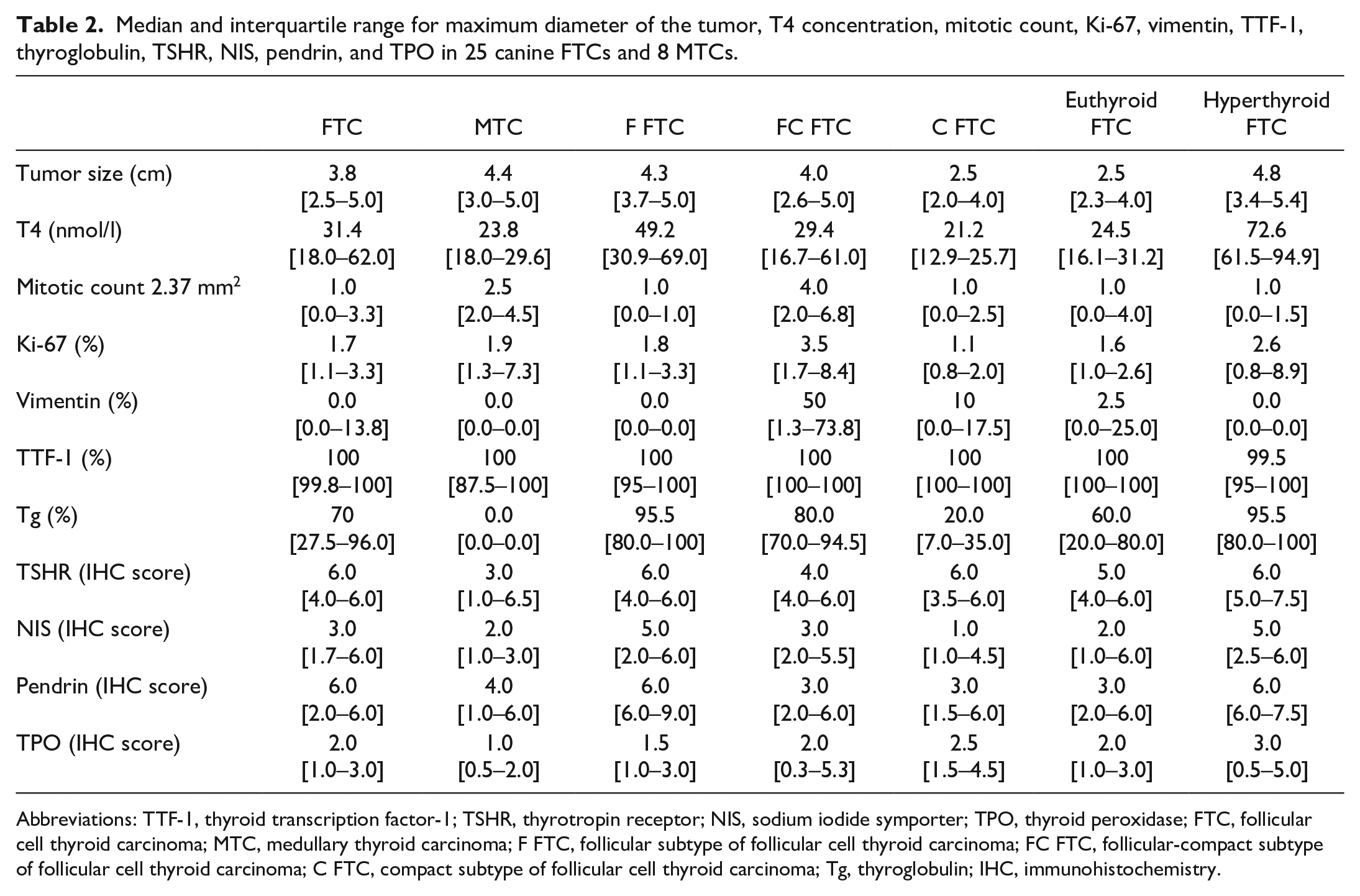
Abbreviations: TTF-1, thyroid transcription factor-1; TSHR, thyrotropin receptor; NIS, sodium iodide symporter; TPO, thyroid peroxidase; FTC, follicular cell thyroid carcinoma; MTC, medullary thyroid carcinoma; F FTC, follicular subtype of follicular cell thyroid carcinoma; FC FTC, follicular-compact subtype of follicular cell thyroid carcinoma; C FTC, compact subtype of follicular cell thyroid carcinoma; Tg, thyroglobulin; IHC, immunohistochemistry.
Comparison of FTCs and MTCs
Maximal tumor dimensions did not differ significantly between FTCs (3.8 [2.5–5.0] cm) and MTCs (4.4 [3.0–5.0] cm) (Table 2). T4 values were lower in dogs with MTCs (23.8 [18.0–29.6] nmol/l) compared with those with FTCs (31.4 [18.0–62.0] nmol/l), but the difference was not statistically significant (Table 2). Expression of TTF-1 was comparable between FTCs (100% [99.8%–100%]) and MTCs (100% [87.5%–100%]) (Table 2, Suppl. Fig. S1a). All FTCs were negative for calcitonin, and all MTCs were negative for thyroglobulin. MCs were not significantly different between FTCs (1.0 [0.0–3.3]) and MTCs (2.5 [2.0–4.5]) (Table 2, Suppl. Fig. S1b). Ki-67 labeling indices were similar between FTCs (1.7% [1.1%–3.3%]) and MTCs (1.9% [1.3%–7.3%]) (Table 2, Suppl. Fig. S1c). Eleven of 25 (44%) FTCs and 1 of 8 (12.5%) MTCs were positive for vimentin with a wide range of percent positive cells (5%–95%) (Suppl. Fig. S1d). TSHR, NIS, pendrin, and TPO were expressed in both FTCs and MTCs. In agreement with several human and canine studies, we observed basolateral membranous and cytoplasmic immunolabeling for NIS, and diffuse cytoplasmic labeling for TPO, pendrin, and TSHR.17,22,35 The IHC scores were lower in MTCs than in FTCs (Figs. 1, 2), but the differences were not statistically significant (Table 2, Suppl. Fig. S2).
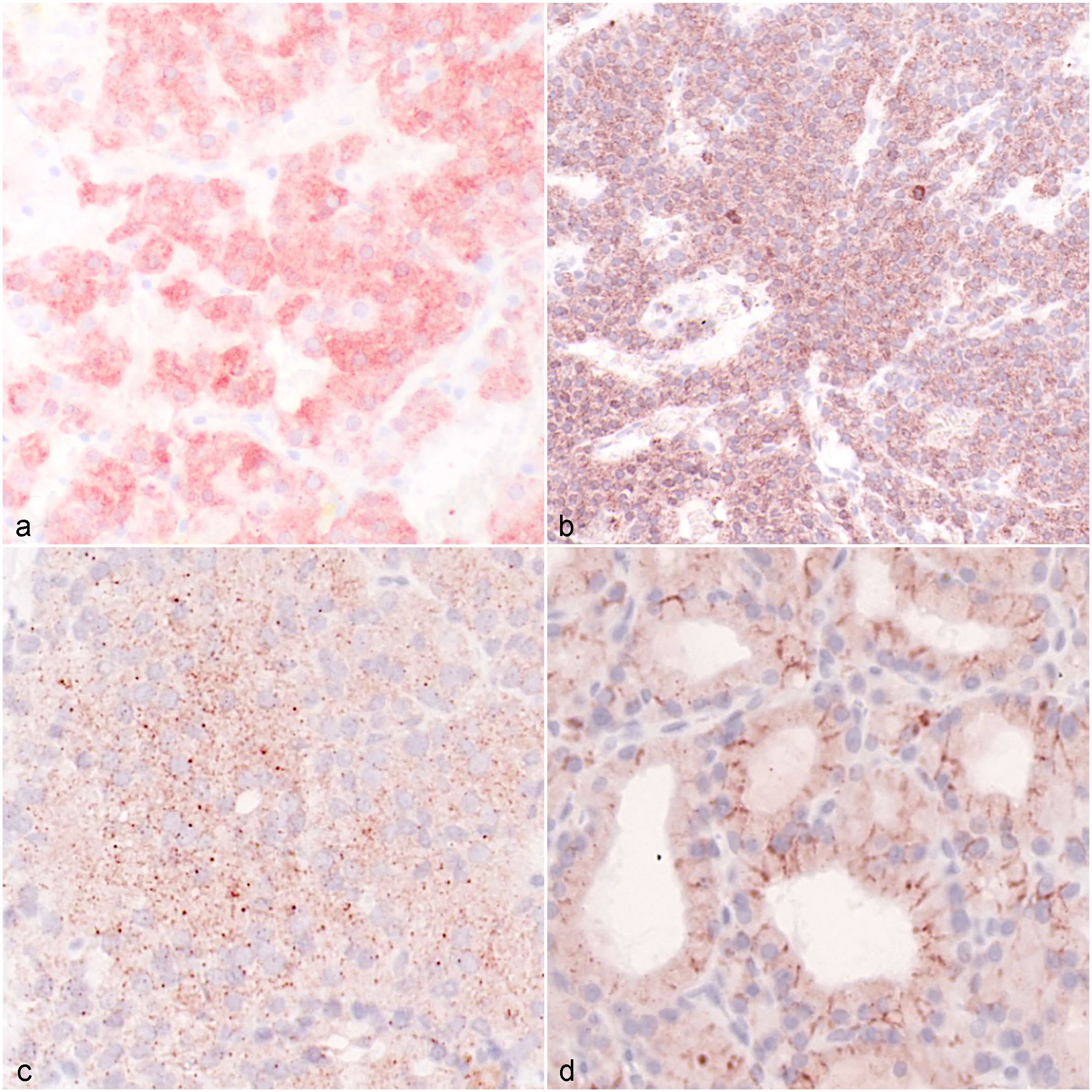
Immunohistochemistry (IHC) in medullary thyroid carcinoma (MTC) and follicular thyroid carcinoma (FTC). (a) MTC, mild to moderate (IHC score 4) cytoplasmic labeling for thyrotropin receptor (TSHR). Case 1. (b) FTC with strong (IHC score 6) cytoplasmic labeling for TSHR. Case 2. (c) MTC with moderate (IHC score 3) granular cytoplasmic immunolabeling for sodium iodide symporter (NIS). Case 3. (d) Follicular FTC with strong (IHC score 6) membranous and weak cytoplasmic labeling for NIS. Case 4.
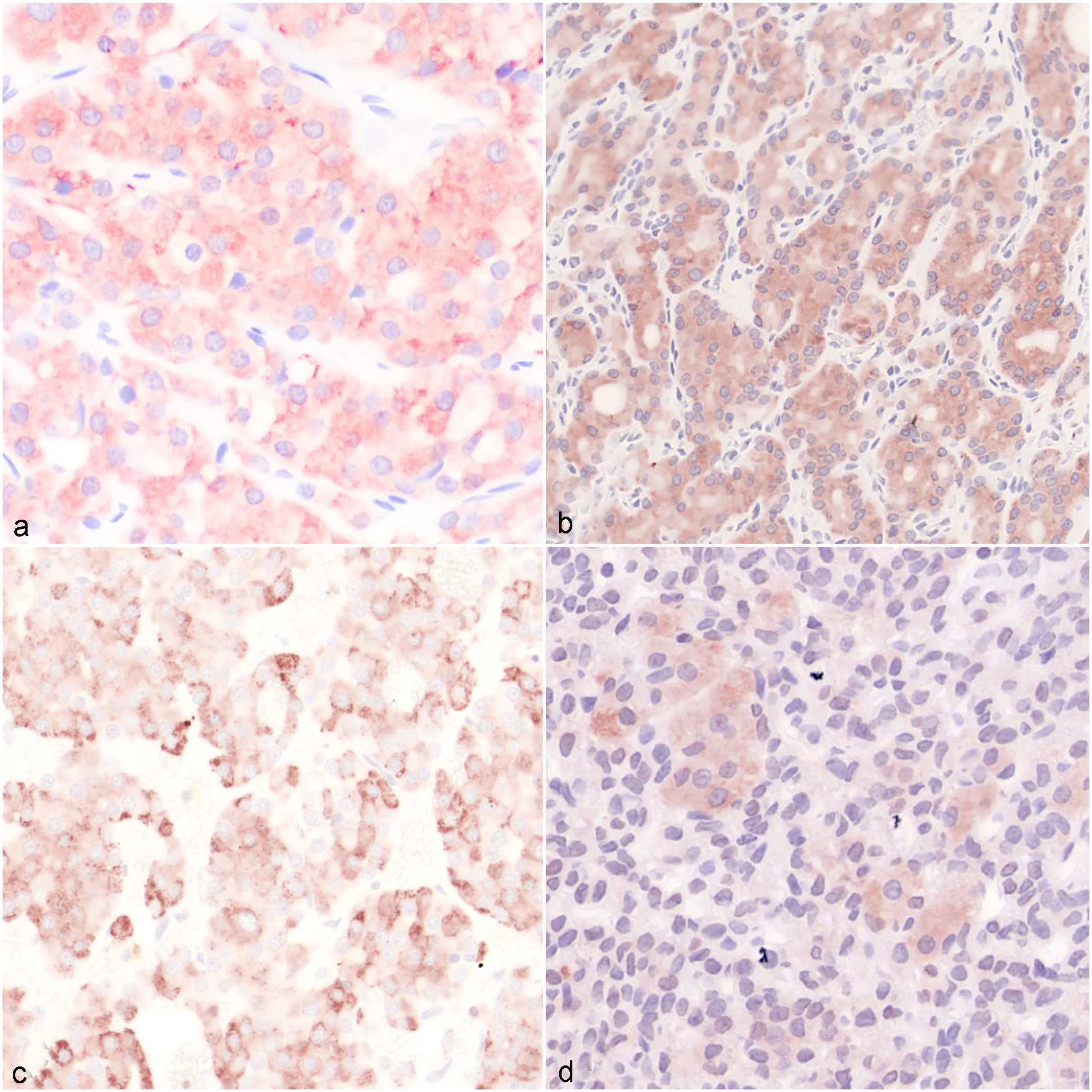
Immunohistochemistry (IHC) in medullary thyroid carcinoma (MTC) and follicular thyroid carcinoma (FTC). (a) MTC with mild to moderate (IHC score 6) cytoplasmic labeling for pendrin. Case 5. (b) FTC with strong (IHC score 8) cytoplasmic labeling for pendrin. Case 6. (c) MTC with showing moderate (IHC score 6) cytoplasmic granular immunolabeling for thyroid peroxidase (TPO). Case 7. (d) Follicular-compact FTC with mild to moderate (IHC score 4) cytoplasmic immunolabeling for TPO. Case 8.
Comparison Between the Different Histological Subtypes of FTCs
Maximal tumor dimensions did not differ significantly between subtypes of FTCs (follicular 4.3 [3.7–5.0] cm, follicular-compact 4.0 [2.6–5.0] cm, and compact 2.5 [2.0–4.0] cm) (Table 2). Although all dogs with FTCs causing hyperthyroidism had follicular FTCs, T4 concentrations were not significantly different between the different FTC subtypes (follicular 49.2 [30.9–69.0] nmol/l, follicular-compact 29.4 [16.7–61] nmol/l, compact 21.2 [12.9–25.7] nmol/l) (Table 2). All FTCs were highly positive for TTF-1 (follicular 100% [95%–100%], follicular-compact 100% [100%–100%], compact 100% [100%–100%]) (Table 2). MCs (follicular 1.0 [0.0–1.0], follicular-compact 4.0 [2.0–6.8], compact 1.0 [0.0–2.5]) (Table 2) and Ki-67 labeling indices (follicular 1.8% [1.1%–3.3%], follicular-compact 3.5% [1.7%–8.4%], compact 1.1% [0.8%–2.0%]) were higher in follicular-compact FTCs, but there were no significant differences (Table 2, Fig. 3a, b). Immunolabeling for vimentin was significantly higher in follicular-compact FTCs (50% [1.3%–73.8%]) compared with follicular FTCs (0.0% [0.0%–0.0%]) (P = .011, Fig. 3c). Expression of thyroglobulin was significantly higher in follicular (95.5% [80.0%–100%]) and follicular-compact FTCs (80.0% [70.0%–94.5%]) compared with compact FTCs (20.0% [7.0%–35.0%]) (P = .001, Fig. 3d). IHC scores for TSHR, NIS, pendrin, and TPO were not significantly different between the 3 subtypes of FTCs (Table 2, Suppl. Fig. S3).
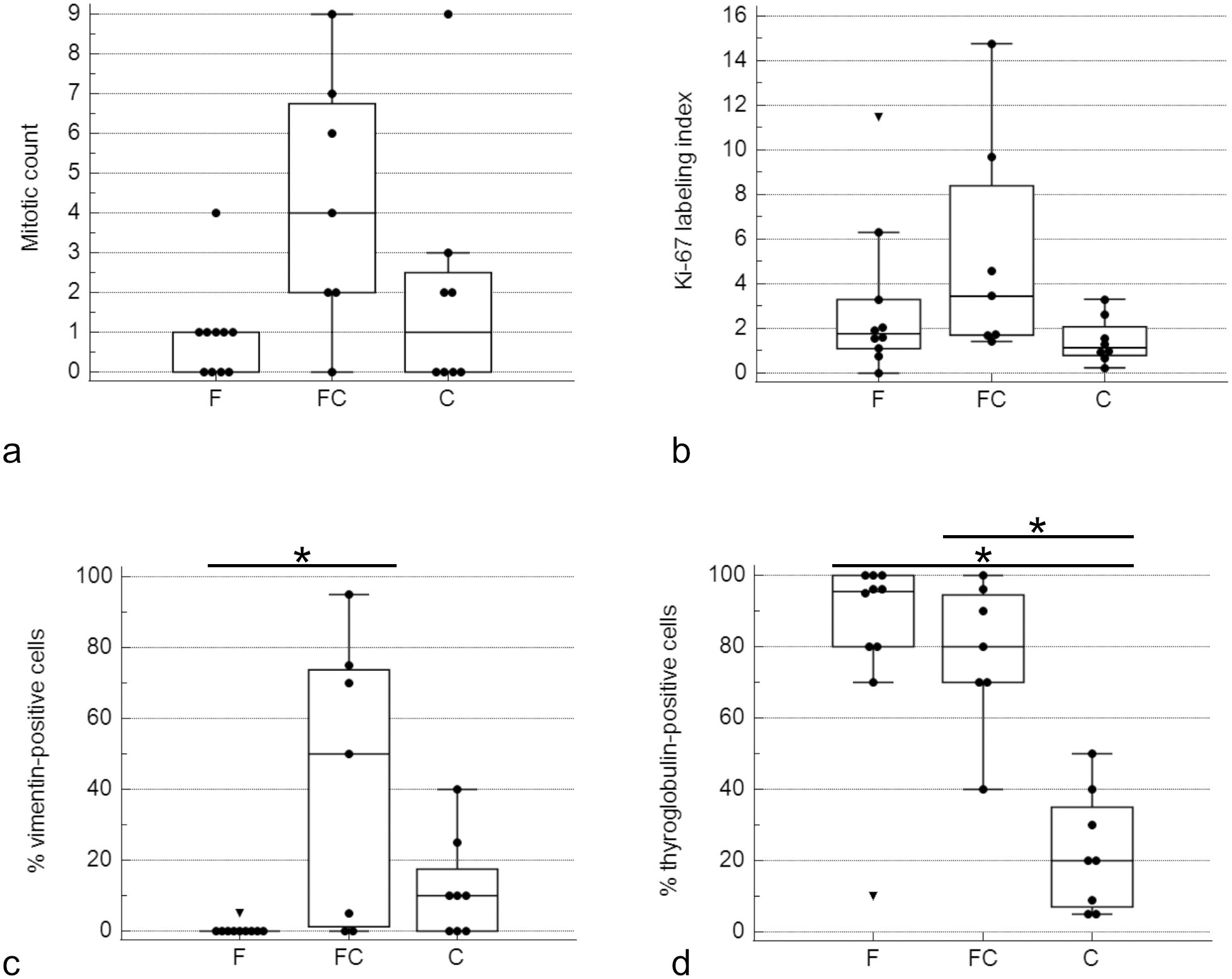
Boxplots of (a) mitotic count, (b) Ki-67, (c) vimentin, and (d) thyroglobulin expression in 9 F (follicular), 8 FC (follicular-compact), and 8 C (compact) follicular cell thyroid carcinomas (FTCs). The dots are “outside values,” values smaller than first quartile minus 1.5× interquartile range. The triangles are “far-out values,” values smaller than first quartile minus 3× interquartile range. (c) Immunolabeling for vimentin was significantly higher in FC FTCs compared to F FTCs (*P = .011). (d) The percentage of thyroglobulin-immunolabeled cells was significantly higher in F and FC FTCs compared to C FTCs (*P = .001).
Comparison of FTCs Causing Hyperthyroidism and FTCs in Euthyroid Dogs
FTCs causing hyperthyroidism were significantly larger (4.8 [3.4–5.4] cm) than those from euthyroid dogs (2.5 [2.3–4.0] cm) (P = .007, Table 2). Expression of TTF-1 was slightly higher in FTCs from euthyroid dogs (100% [100%–100%]) compared with FTCs causing hyperthyroidism (99.5 [95–100]) (P = .041, Table 2). MCs and Ki-67 proliferation indices did not differ significantly between FTCs causing hyperthyroidism (MC: 1.0 [0.0–1.5] and Ki-67: 2.6% [0.8%–8.9%]) and FTCs from euthyroid dogs (MC: 1.0 [0.0–4.0] and Ki-67: 1.6% [1.0%–2.6%]) (Table 2, Fig. 4a, b). Vimentin was mainly expressed in FTCs from euthyroid dogs; only one FTC causing hyperthyroidism was mildly positive for vimentin (P = .057, Fig. 4c). The percentage of cells immunolabeled for thyroglobulin was above 80% in all but 1 FTC causing hyperthyroidism (95.5% [80.0%–100.0%]), while it was highly variable in FTCs in euthyroid dogs (60.0% [20.0%–80.0%]) (P = .0258, Fig. 4d). IHC scores for TSHR, NIS, pendrin, and TPO were slightly higher in FTCs causing hyperthyroidism compared with FTCs from euthyroid dogs, but the differences were not statistically significant (Suppl. Fig. S4).
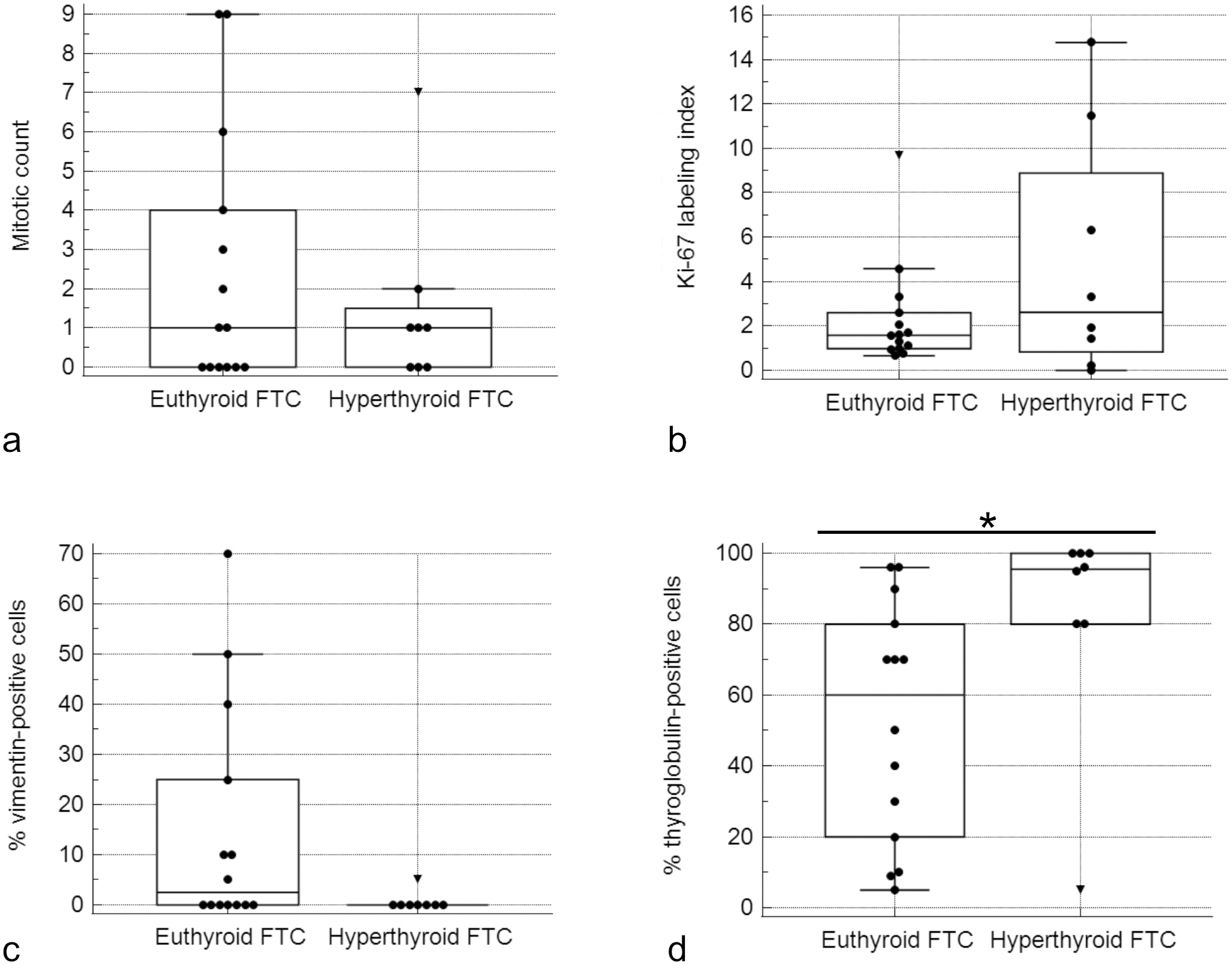
Boxplots of (a) mitotic count, (b) Ki-67, (c) vimentin, and (d) thyroglobulin expression in 20 tumors from euthyroid dogs and 8 tumors from hyperthyroid dogs. The dots are “outside values,” values smaller than first quartile minus 1.5× interquartile range. The triangles are “far-out values,” values smaller than first quartile minus 3× interquartile range. (a) Mitotic counts and (b) Ki-67 labeling indices were comparable between follicular thyroid carcinomas (FTCs) causing hyperthyroidism and FTCs of euthyroid dogs. (c) Vimentin was mainly expressed in FTCs of euthyroid dogs. (d) The percentage of cells immunolabeled for thyroglobulin was higher in FTCs causing hyperthyroidism compared to FTCs from euthyroid dogs. (*P = .0258).
Discussion
To develop adequate in vitro models aimed at improving 131I treatment in dogs with FTC, it is important to evaluate the expression of proteins involved in iodine transport. This study evaluated the immunohistochemical expression of TTF-1, thyroglobulin, TSHR, NIS, pendrin, TPO, vimentin, calcitonin, and Ki-67 in different histological types of canine thyroid carcinoma. Our results show that TSHR, NIS, pendrin, and TPO were also expressed in MTCs. Furthermore, we found no significant differences in immunohistochemical expression of TSHR, NIS, pendrin, and TPO between the different subtypes of canine FTCs or between FTCs causing hyperthyroidism and FTCs from euthyroid dogs.
Human thyroid carcinomas can be classified as follicular thyroid carcinoma, invasive encapsulated follicular variant of papillary thyroid carcinoma, papillary thyroid carcinoma, oncocytic carcinoma of the thyroid, high-grade follicular-derived carcinomas (poorly differentiated and differentiated high-grade thyroid carcinoma), anaplastic thyroid carcinoma, MTC, and mixed MTC and FTC. 24 Ki-67 can be used as a prognostic indicator in several tumors, and in human papillary thyroid carcinoma, high expression of Ki-67 is associated with disease recurrence.31,44 In our study, there were no significant differences in the Ki-67 labeling indices or MCs between canine FTC and MTC, in agreement with previous reports.6,41 Immunolabeling for vimentin was observed in 11 of 25 FTCs and 1 of 8 MTCs. In people with differentiated thyroid carcinoma, 53% of papillary thyroid carcinomas and 75% of follicular thyroid carcinomas are positive for vimentin.3,5 Overexpression of vimentin in human cancers correlates with increased tumor invasiveness and metastasis. 38 In the healthy canine thyroid gland, vimentin is expressed by the immature follicular cells and is not present in the typical thyroid follicles, suggesting it may be associated with decreased cellular differentiation. 25 Vimentin expression had also been previously reported in canine thyroid carcinomas and in one case report of canine MTC.11,20,34,36
Surprisingly, immunolabeling for TSHR, NIS, pendrin, and TPO was also observed in canine MTCs. The cause for the expression of proteins of iodine transport and thyroid hormone production in canine MTC is uncertain but may offer an explanation for the scarce reports of iodine uptake by these tumors.7,47 In a recent study, neither non-neoplastic thyroid C cells nor non-neoplastic parathyroid chief cells expressed these markers. 22 Immunohistochemical expression of NIS and TPO, as well as gene expression of TSHR, has been reported in human MTC.14,15,39 A possible explanation could be that, due to neoplastic transformation, MTC cells start to express these markers. Mixed medullary and follicular cell-derived carcinomas are reported in people, but this seems unlikely in our study given that all MTCs were immunohistochemically negative for thyroglobulin. 24 The expression of proteins of iodine transport and organification by canine MTCs is of clinical importance because it implies that 131I could potentially be of use for diagnosis and treatment purposes. The use of 131I as adjunctive therapy to surgery has been reported in people with MTC, with the rationale that 131I trapping (and delivery of radiation) by the neighboring healthy thyroid cells would provide a clinical benefit. 16 Recent controlled studies, however, did not find any benefit in disease-free or disease-specific survival and do not support its use in the treatment of MTC. 29
In our study, we found no differences in the expression of NIS and TSHR between the different subtypes of FTCs. In people, TSHR is similarly expressed in FTC and papillary thyroid carcinoma. 19 NIS has a greater expression in human FTC compared with papillary and insular thyroid carcinoma, and it is also expressed in MTCs.14,19 The expression of TSHR, TPO, and especially NIS in cancerous tissue could serve as predictive biomarkers for tumors that could be targeted with 131I.8,32,48
In our study, TPO was similarly expressed in the different subtypes of FTCs. In people with follicular cell–derived tumors, the absence of TPO expression in cytological and histological specimens is considered a marker of malignancy.12,13 In our study, pendrin was highly expressed in all subtypes of FTCs. This finding is in agreement with studies in human thyroid carcinoma, in which pendrin is highly expressed in follicular as well as papillary thyroid carcinoma. 40
All our tumors were highly positive for TTF-1. In human studies, it has been shown that TTF-1 and thyroglobulin are highly expressed only in well-differentiated follicular and papillary thyroid carcinomas.37,39 Thyroglobulin was expressed less frequently in compact FTC compared with follicular and follicular-compact FTCs, as previously reported. 35 The greater expression of thyroglobulin in follicular and follicular-compact FTCs could be related to a higher degree of differentiation of these FTC subtypes. Follicular FTCs histologically resemble non-neoplastic thyroid tissue, which could also support the notion that they are more differentiated than compact FTCs. 26 Furthermore, the greater expression of vimentin in follicular-compact FTCs compared with follicular FTCs also supports differences in differentiation, given the association of vimentin with epithelial-mesenchymal transition. 30 In one study, canine patients with compact FTCs had a shorter survival time, and the authors hypothesized that this more aggressive behavior could be due to a lesser degree of differentiation. 10 Still, the expression of other markers (TSHR, NIS, pendrin, and TPO) was not significantly decreased in compact FTCs.
Tumors causing hyperthyroidism were larger than tumors of euthyroid dogs. However, this has not been investigated in previous studies.2,6 Immunolabeling for thyroglobulin was greater in FTCs causing hyperthyroidism compared with FTCs from euthyroid dogs. Similar findings have been reported in humans. 17 The apparent lower expression of vimentin in FTCs causing hyperthyroidism may also be related to greater tumor differentiation. Furthermore, in our study, all tumors causing hyperthyroidism were follicular FTCs, as previously reported. 10 Together, these findings suggest that tumors causing hyperthyroidism may be associated with greater tumor differentiation. Immunolabeling for TSHR, NIS, pendrin, and TPO was greater in FTCs causing hyperthyroidism, but the differences were not statistically significant. In people, the expression of proteins of iodine transport and production of thyroid hormones are associated with the functional status of thyroid tumors. 42
Limitations of our study include the limited sample size and the fact that T4 values were determined in different laboratories with different reference ranges.
In conclusion, proteins of transport and organification of iodine are commonly expressed in both canine FTCs and MTCs. This was an unexpected finding for MTCs and could have relevant implications for diagnosis and treatment. The expression of these proteins was similar across histological subtypes of FTC and between functional and non-functional tumors. The different subtypes of canine FTC could be associated with different degrees of tumor differentiation.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231217245 – Supplemental material for Canine follicular cell and medullary thyroid carcinomas: Immunohistochemical characterization
Supplemental material, sj-pdf-1-vet-10.1177_03009858231217245 for Canine follicular cell and medullary thyroid carcinomas: Immunohistochemical characterization by Jana Jankovic, Eve Tièche, Martina Dettwiler, Kerstin Hahn, Stephanie Scheemaeker, Martin Kessler, Sylvie Daminet, Sven Rottenberg and Miguel Campos in Veterinary Pathology
Footnotes
Acknowledgements
We thank Sohvi Blatter and Olivia Richard for trimming the tumor samples, and Erika Bürgi, Manuela Bozzo, and Bettina de Breuyn for preparing the slides for IHC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Albert Heim Stiftung, the Tierärztliche Verrechnungsstelle (TVS), the European Society of Veterinary Endocrinology, and the Small Animal Clinic of the Vetsuisse Faculty—University of Bern.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
