Abstract
One of the primary objectives of the Oncology Pathology Working Group (OPWG), a joint initiative of the Veterinary Cancer Society and the American College of Veterinary Pathologists, is for oncologists and pathologists to collaboratively generate consensus documents to standardize aspects and provide guidelines for oncologic pathology. Consensus is established through review of relevant peer-reviewed literature relative to a subgroup’s particular focus. In this article, the authors provide a critical review of the current literature for grading of canine cutaneous mast cell tumors, suggest guidelines for reporting, and provide recommendations for its clinical interpretation. The article mainly focuses on histologic grading, but relevant information on mitotic count and cytological grading are also discussed. This document represents the opinions of the working group and the authors but does not constitute a formal endorsement by the American College of Veterinary Pathologists or the Veterinary Cancer Society.
Canine cutaneous mast cell tumors (MCTs) account for approximately 20% of all canine skin tumors. The wide range of biologic behavior exhibited by these tumors represents a clinical challenge for veterinary general practitioners and oncologists. 4 The diagnostic cornerstone for determining the prognosis of canine cutaneous MCTs has been for many years and still is the histological grade with a considerable number of publications available in the literature on this topic. 1,2,4 –10,12 –23 The Oncology Pathology Working Group (OPWG) is a joint initiative of the Veterinary Cancer Society and the American College of Veterinary Pathologists. This group aims to promote an integrated working relationship between veterinary oncologists and pathologists to facilitate and ensure the highest standard of pathology support and reporting for the advancement of veterinary clinical oncology and cancer research. Through this interdisciplinary effort the OPWG works to establish consensus documents designed to standardize and provide guidelines for oncologic pathology. The OPWG consensus documents represent a critical appraisal of the available literature on a specific topic and are established by a subgroup of individuals with a level of expertise in the area of focus. Thereafter, these documents are reviewed by and voted on (for adoption or rejection) by the OPWG membership at large.
In 2013, the OPWG published on its website (http://vetcancersociety.org/Consensus_CCMCT-Grading/Consensus_CCMCT-Grading.pdf) the first OPWG consensus document on the grading of canine cutaneous MCTs, which was updated in 2017. Since then, additional articles on grading of canine cutaneous MCTs have been published, continuing to expand and improve the body of knowledge on the subject. 1,5,8,9,16,19 This article represents the most recently updated OPWG consensus on grading of canine cutaneous MCTs, and it was approved by the OPWG membership at large in June 2020. The concluded recommendations represent the opinions of the authors, with the support of the OPWG membership, based on critical review of the literature as determined by assessing the robustness of the data presented in the publications reviewed. This consensus should be updated regularly as additional research and associated data come to light to ensure currency and progress in this area of veterinary medicine. It should be noted that this consensus does not address the literature related to canine subcutaneous MCTs, although this topic may be incorporated into future updates of this consensus or in a separate and independent OPWG consensus solely focused on canine subcutaneous MCTs.
Development of the Consensus Report
A review of the literature was performed by a subgroup of OPWG members who are all coauthors of this article. These OPWG members include 6 board-certified veterinary oncologists (DB, JBF, CAC, LG, JI, PJ), 3 board-certified veterinary anatomic pathologists (RR, DAK, AP), 1 board-certified veterinary clinical pathologist (RP), and 1 board-certified veterinary surgeon (JML). All subgroup members separately and critically reviewed the same publications, 1,2,5,6,8,9,16 –23 but to expedite the last update including 6 publications 1,5,8,9,16,19 the members were divided into 2 groups reviewing 3 papers for each group. Each member independently completed a template for each respective article providing a summary of the study objective, study design, materials and methods, and a critical assessment of the statistical soundness and conclusions drawn, while also offering their own overall conclusions and assessment relative to the strength and merit of the study. Taking into account all the independent reviews of the subgroup members, the subgroup chairs and authors (DB and RR) generated an initial draft of the consensus. This draft was then circulated, discussed, and edited, as deemed appropriate, among the subgroup as a whole in an open setting. The inclusion of background information originally incorporated in the 2013 OPWG Consensus documents and discussion points that arose during drafting of the 2017 and 2020 consensus documents led to the citation within the 2020 final consensus of additional nonreviewed publications. 4,7,10 –15 As a final step, the consensus document was made available for review by the OPWG membership at large, which then approved the document in June 2020 following minor edits.
Review of the Literature
Histological Grading
Since 1984, the Patnaik grading system 14 has been the basis for determining the prognosis of canine cutaneous MCTs in the routine pathology and clinical setting. 14 This grading system divides cutaneous MCTs in 3 different grade categories (grade I, grade II, and grade III) based on extent of tissue involvement, cellularity, cellular and nuclear morphology, mitotic activity, stromal reaction, and edema/necrosis (Table 1). Numerous studies have proven its validity in the clinical setting. 4 Grade I MCTs have an excellent long-term prognosis, while grade III are associated with a guarded to poor prognosis because of a higher recurrence and metastatic rate. 12 More difficult to predict is the behavior of grade II MCTs; in fact, the majority of them follow a benign clinical progression, while about 20% are characterized by an aggressive clinical behavior. 4 Another limitation of this system is the subjectivity of its application by different pathologists with the resultant variability in the grade assignment. 13 This problem is more relevant for grade I and grade II MCTs, while it seems to be less of an issue for grade III MCTs. 10,23 A possible cause for this subjectivity could be the lack of guidelines to assign the grade in a MCT where histological features typical of different grades coexist.
Patnaik Histological Grading Criteria, 1984. 14
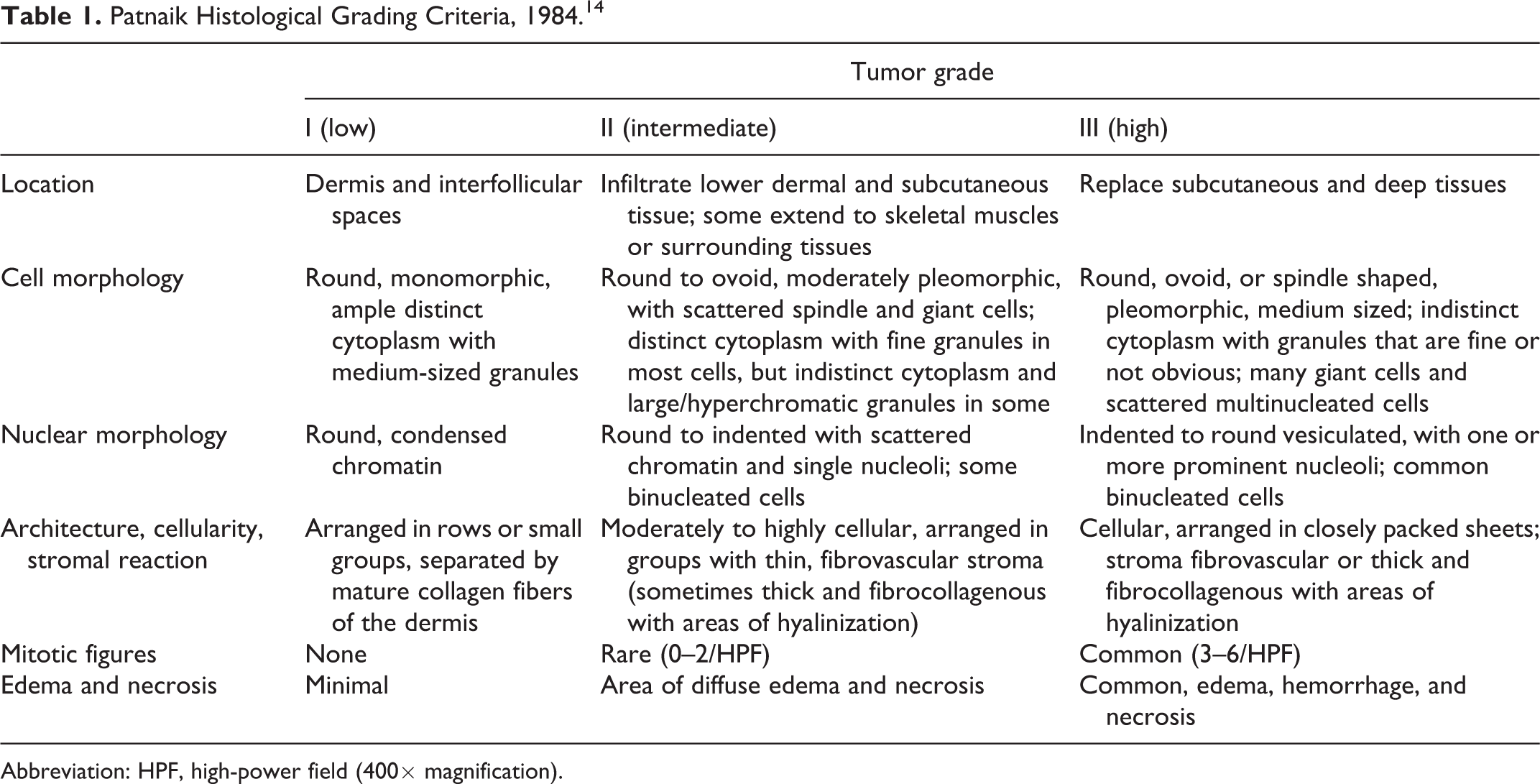
Abbreviation: HPF, high-power field (400× magnification).
In order to overcome these 2 limitations a 2-tier grading system was developed by Kiupel and colleagues in 2011 (Table 2). 10 This system divides cutaneous MCTs into 2 categories (low and high grade) based only on cellular morphologic criteria including mitotic figures (mitotic count), multinucleation, bizarre nuclei, and karyomegaly. The Kiupel grading system has been validated in multiple studies 6,17,20,21,23 as an independent prognostic factor in canine cutaneous MCTs able to predict local recurrence, 6 metastatic propensity, 20 and overall survival. 17,21,23 An advantage of the Kiupel grading system is that the criteria for assigning the grade are more objectively defined, increasing the concordance between pathologists. 21,23 A limitation of this grading system is that it does not take into account the growth pattern or extension of the MCT (infiltrative or not). Currently, there is no information as to whether the lack of assessment of the growth pattern could lead to underestimation of the local aggressiveness of the tumor in some cases.
Kiupel (2-Tier) Histologic Grading Criteria, 2011. 10

Abbreviation: HPF, high-power fields (400× magnification).
Few studies have applied both grading systems (Patnaik and Kiupel) to the same population of dogs. 6,9,17,20,23 In those studies, all grade I tumors were low-grade Kiupel and all grade III tumors were high-grade Kiupel, while grade II split with the majority being low-grade and a smaller proportion (ranging from 7.5% to 21%) being high grade. 6,9,17,20,23 Both systems were able to predict outcome in dogs with cutaneous MCTs, but interestingly the Patnaik system was more sensitive, while the Kiupel system more specific, in detecting dogs with aggressive disease. 23 The evidence is still weak, but it appears that the 2 systems could be complementary in that the Kiupel system might help refine the prognosis of grade II Patnaik MCTs, 10,23 while the Patnaik system might clarify the behavior of high-grade MCTs. 17,20
Using the Patnaik and Kiupel grading system together, cutaneous MCTs can be divided into 4 categories with different prognoses:
Grade I/low-grade. The prognosis is excellent with virtually no tumor-related deaths. 9,17 One study reports a low risk of lymph node metastasis (6%) and distant metastases (2%) at initial staging, stressing the importance of staging even in this group of patients. 20
Grade II/low-grade. Three articles describe this category of patients. 9,17,20 The prognosis is supposedly good with 3% to 17% of dogs dying of causes related to MCTs. 9,17 In one study, 17 the median survival time for this group of patients was not reached after 92 months, and 94% were alive at 1 year. At initial staging, metastatic rate to the lymph nodes was 16%, while distant metastases were seen in 2% of the cases. 20
Grade II/high-grade. Three articles describe this category of patients. 9,17,20 The prognosis is fair to guarded with 14% to 56% of dogs dying of causes related to MCTs. 9,17 The median survival time was between 7.5 and 23.3 months, and only 46% of dogs were alive at 1 year. 9,17 At presentation, the metastatic rate to the lymph nodes was 15%, while distant metastases were seen in 2% of the cases. 20
Grade III/high-grade. The prognosis is guarded to poor, with 67% to 75% tumor-related deaths in studies with adequate numbers of cases. 9,12,17 The median survival time in 2 studies was 3.6 and 6.8 months. 9,17 The metastatic rate to the lymph nodes was 46%, with distant metastases in 21%. 20
According to data available, it appears that the difference in survival seen between Grade II/low-grade and Grade II/high-grade might be due to local recurrence rather than metastatic disease, because metastatic rates between these 2 groups appear similar. This hypothesis requires further investigation, although the study by Donnelly and colleagues 6 on grade and margins as predictors of local recurrence provided corroborative findings. In this study, the removal of MCTs with histologic clean margins, even if ≤3 mm, appeared adequate to prevent local recurrence in 96% of low-grade MCTs, while 36% of high-grade MCTs recurred regardless of the width of the histologic tumor-free margins. 6
One study described the value of biopsies prior to curative-intent surgery and found an overall concordance rate of 96% based on the Patnaik grading system and 92% based on the Kiupel grading system. 19 Discordance in grade was more likely to underestimate tumor grade. While the study did not directly evaluate concordance of incisional and curative-intent excisional biopsies using combined grading schemes (as proposed above), extrapolating the study’s data in this manner suggested pretreatment biopsies underestimated up to 44% of Grade II/high-grade MCTs with less than 6% discordance for all other combined grades. Moreover, the majority of biopsies in the study were large (wedge or punch) raising the concern of increased expense and morbidity.
Mitotic Count
In dogs with cutaneous MCTs, the literature reviewed provides evidence that the mitotic count is an independent prognostic indicator. 1,2,7,15,22,23 This was previously referred to as mitotic index, but the appropriateness of the term mitotic index (defined as the number of cells in mitosis divided by the number of cells not in mitosis) has recently come into question and the adoption of the term mitotic count (number of mitotic figures within a given area) has been indicated as more appropriate. 11 It has also been recently proposed that the mitotic count (MC) be determined and reported in a standardized area of 2.37 mm2. With most microscopes, which have an ocular field number equal to 22 (objective diameter = 0.55 mm), the standardized area of 2.37 mm2 corresponds to 10 high-power fields (40× objective). Guidelines for the adjustment of the mitotic count are available for microscopes with ocular field numbers larger or smaller than 22 as well as for measuring the 2.37 mm2 area using digitized whole slide images, the usage of which is increasing. 11
In the literature reviewed by the OPWG subgroup, the mitotic count (MC) was consistently obtained by counting the number of mitoses in 10 high-power fields (40× objective) in the region with highest mitotic activity, as determined initially on a low power scan (10× objective) of the specimen. 15 However, only in 2 of the most recent studies did the authors state that they examined the standard 2.37 mm2 area. 1,9 Most studies not providing that information were published before it was recognized that the MC should be reported using a standardized area. 2,6,8,16 –23 For future studies, standardization of the parameters for MC assessment in cutaneous MCTs is considered to be of paramount importance. Defining the best method for selecting the region of highest mitotic activity within each tumor should be emphasized, as interpathologist variability in identifying these “hot spots” has been recently documented. 3
In 4 studies on canine cutaneous MCTs evaluating the MC as a prognostic factor with a threshold of MC = 5, 1,2,15,23 the sensitivity of MC to predict tumor-related death ranged from 39% to 55% and the specificity was 86% to 99%. The median survival time for cutaneous MCTs with a MC ≤ 5 was >70 months, while the survival for MCTs with a MC > 5 varied between 2 and 5 months. 1,2,15,23 Mitotic count is significantly associated with metastatic rate, but not with recurrence rate. 15 Some authors 7,9,22 suggest that the stratification of the MC into 3 categories might be superior, but these methods are less easily applicable in the clinical setting.
Cytological Grading
The application of a cytological grading system based on the Kiupel histological grading system has been investigated in 3 studies. 5,8,18 The Kiupel grading system is appealing for clinical pathologists because it relies on cellular morphological features rather than on tissue architecture, as opposed to the Patnaik system. Morphologic features used in cytological grading include presence of mitotic figures, nuclear pleomorphism, binucleation/multinucleation, and anisokaryosis. In the Camus grading system, 5 another important criterion for assigning the cytological grade was mast cell granulation, which is not assessed in the Kiupel grading system but is used in the Patnaik grading system.
According to these studies, 5,8,18 based on histopathology as the gold standard, cytological grading has a sensitivity of 85% to 88%, a specificity of 95% to 97%, and an overall accuracy of 94%, which is comparable with the performance of an incisional biopsy prior to curative-intent surgery. 19 The cytological grade was significantly associated with median survival times and tumor-related deaths in one study. 5 In this study, the criteria applied to assign a high cytological grade were poor granulation, or at least 2 of the following features: presence of mitotic figures, binucleation/multinucleation, nuclear pleomorphism, or anisokaryosis (Table 3). Despite these promising findings, cytological grading presents some limitations and challenges, which are difficult to resolve and require further investigation.
Camus Cytologic Grading Criteria, 2016. 5

The first limitation is the difficulty for cytology to differentiate between cutaneous and subcutaneous MCTs, potentially leading to subcutaneous MCTs also being graded. For this aspect of cytological evaluation, the clinical pathologist must rely on the historical information provided by the submitting clinician; however, this information may not be included or may not be reflective of the true location of the tumor.
Another controversial element is the stain used to prepare the sample. May-Grünwald-Giemsa or Wright’s stains routinely used in clinical pathology allow rapid and accurate diagnosis of MCTs because of the intense coloration of the granules. However, the abundance of granules in well-differentiated mast cells might obscure some nuclear criteria, especially pleomorphism, making it difficult to assign a cytological grade based only on Kiupel criteria. With the evidence available at this time, the procedures of de-staining and re-staining cytological samples with hematoxylin and eosin to allow better visualization of the nucleus do not appear to be cost-effective and could lead to staining artifacts. 8 Moreover, some nuclear features have been associated with inaccurate grading; for example, karyomegaly is the main reason for assigning a false positive cytological high grade to a histologically low-grade tumor, 8,18 while nuclear pleomorphism (bizarre nuclei) is consistently poorly predictive when correlated to the histologic grade across all 3 studies. 5,8,18 The presence of binucleated cells may also contribute to discrepancies between cytological and histological grading; binucleated cells are considered in cytological grading whereas the Kiupel histological grading only considers cells with 3 or more nuclei as a criterion for high grade. 5,10 Aqueous-based rapid cytological stains should also be critically evaluated in the future, as these are widely used in practice and may give more variable staining of cytoplasmic granules. 16 For now, there is no available evidence for their use in grading MCTs. As with histopathology, the increasing use of whole-slide imaging or selected scans for digital cytology requires separate validation and assessment, especially when involving computational algorithms and in-house staining with rapid stains.
The last challenge is the difficulty of assessing the mitotic activity in cytological preparation, which is likely in part due to the heterogeneous distribution of mitoses within the tumor and variation in the quality of aspirated samples, but also due to the poor visualization of the nucleus in well-granulated mast cells.
Recommendations
Based on the critical review of the currently available literature on grading of canine cutaneous MCTs, the OPWG recommends the following: Histopathologic reporting of canine cutaneous MCTs should include both histologic grading systems (Patnaik and Kiupel). Additionally, as recommended since the initial (2013) consensus, all diagnostic pathologists reporting on canine cutaneous MCTs should have the criteria for both systems (Tables 1 and 2) available for reference at the time of grading. All histologic reports of canine cutaneous MCTs should also include the MC, which should be expressed as the absolute number of mitoses counted in the region of the tumor with the highest mitotic activity, covering a standardized area of 2.37 mm2. This usually means performing the MC in 10 high-power fields (40× objective), but adjustments should be made as needed based on the microscope’s optics or for digital images to guarantee the 2.37 mm2 area. It is important to highlight that a subset of tumors with a histologically benign appearance (grade I/low-grade or grade II/low-grade with MC ≤5) can still manifest an aggressive clinical behavior. Regardless of the grading system used, grade must be considered as only one prognostic factor and used in conjunction with the overall clinical picture: age, clinical progression, size, site, stage, completeness and quality of surgical margins, and other prognostic markers such as proliferation markers, kit expression, and c-kit mutation. Cytological grading is promising. The Camus grading system (Table 3) should be further validated, but may provide valuable preoperative information. Based on current information, a cytological diagnosis of low-grade MCT correlates well with histologic grading and clinical outcome. However, a diagnosis of high-grade MCT should be received with caution if only based on 2 morphological criteria, mainly if anisokaryosis and nuclear pleomorphism is one of them, because of the risk of false positives when compared to histological grading. Until more fully evaluated, other factors including clinical presentation and clinical signs should be included to support the suspicion of a more aggressive MCT when guiding therapy and staging. Incisional biopsies prior to curative-intent surgery are moderately accurate to grade cutaneous MCTs prior to definitive treatment, but they may underestimate the grade, particularly in grade II/high-grade tumors. Subjectively, members of the OPWG rely on cytology as an initial screening test, but rarely recommend an incisional biopsy to guide staging or treatment planning because of its limited clinical benefit and associated morbidity.
Footnotes
Acknowledgements
The authors would like to acknowledge all other members of the OPWG Canine Cutaneous Mast Cell Tumor Subgroup on Grading who contributed to the earlier versions (2013, 2017) and initial foundation of this current consensus: Jeffrey Bryan (2013), Charles Halsey (2013), Elizabeth Hershey (2013, 2017), Luca Malfassi (2013), Karen Oslund (2013), John Peauroi (2013, 2017), Kerry Rissetto (2013), Mona Rosenberg (2013), Ted Valli (2013), and Laurel Williams (2013). The authors would also like to acknowledge the entire OPWG membership for their review and helpful comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
