Abstract
HER2 is overexpressed, amplified, and mutated in a subset of human lung cancer. The aim of this study was to investigate HER2 protein overexpression and gene amplification in feline pulmonary carcinomas. Thirteen pulmonary carcinomas were selected and TTF-1 and HER2 expression was evaluated by immunohistochemistry. Fluorescence in situ hybridization (FISH) was performed with a HER2 probe and a BAC probe for the feline chromosome E1p1.12-p1.11 region. Twelve adenocarcinomas and 1 squamous cell carcinoma were diagnosed. TTF-1 was positive in 7 carcinomas (58%). HER2 was overexpressed in 2 (15%), equivocal in 5 (38%), and negative in 6 cases (46%). FISH analysis of HER2 was indeterminate in 2 cases. Three pulmonary carcinomas (27%) had HER2 amplification and 8 cases were not amplified (73%). The significant correlation between HER2 protein overexpression and gene amplification are promising preliminary data, but study of additional cases is needed to confirm HER2 as a target for possible innovative treatments.
Keywords
Lung cancer is the most commonly diagnosed cancer in humans and the leading cause of cancer death. 2 In cats, pulmonary carcinomas are rare and aggressive neoplasms that have a high rate of metastasis. 5,19 Surgery is the therapeutic strategy for pulmonary carcinoma in dogs and cats, but most feline tumors are classified as inoperable at diagnosis because of the extensive disease. 19 Several molecular alterations have been documented in humans, but information regarding the molecular carcinogenesis in feline pulmonary carcinomas is limited. 5,9
Human lung adenocarcinoma is characterized by genomic rearrangements, and targeted therapy is available for patients harboring activated oncogenes such as mutant EGFR, BRAF, and HER2, or translocated ALK, fused RET or rearranged ROS1. 1,4 HER2 encodes for a tyrosine kinase receptor that undergoes ligand-independent dimerization, phosphorylation, and linking with downstream proliferation pathways. 15 In humans, overexpression of HER2 protein is present in 0.87% to 36% of patients with non–small cell lung cancer, caused by gene amplification or mutations. 11,14,16,17 Therapeutic strategies are available for the targeted treatment of human HER2+ lung cancers using monoclonal antibodies. 11,14 Furthermore, HER2 overexpression in lung cancer is an unfavorable prognostic factor. 17 HER2 is reported to be overexpressed and amplified in a subset of feline mammary carcinomas, 3,7,13 but studies of HER2 in feline pulmonary carcinoma are not yet available. The aim of this study was to investigate HER2 protein overexpression and gene amplification status in feline pulmonary carcinoma.
Thirteen primary feline pulmonary carcinomas were retrospectively collected. The experiments were in compliance with the national legal treatment of animal tissue samples. The samples were surgical biopsies or necropsy samples and were available as formalin-fixed paraffin-embedded material, routinely stained with hematoxylin and eosin. The diagnoses were revised based on the current classification system. 19
For antibody validation by western blot, proteins from p248, Fc2Lu cell lines, murine lung tissue, and human MCF10 cell line were extracted in lysis buffer (1% Triton X-100, 10% glycerol, 50 mM Tris, 150 mM sodium chloride, 2 mM EDTA, pH 8.0, and 2 mM magnesium chloride) containing protease inhibitor cocktail (P8340 Sigma). Total proteins (20 μg) were separated by SDS-PAGE (10% or 15%) and transferred onto a PVDF membrane. After washing, membranes were incubated in TBS with 10% bovine serum albumin at room temperature for 1 hour and then overnight at 4 °C with antibodies against-HER2 (CB11 clone Thermofisher Scientific) and TTF-1 (clone 8G7G3/1, Dako Glostrup).
For immunohistochemistry (IHC), serial 3-μm-thick sections were processed using an antibody against TTF-1 or 2 antibodies against-HER2 (polyclonal, Dako Glostrup, known to be reactive in cats; 7 and clone CB11, Thermo Fisher Scientific). Immunohistochemistry methods are summarized in Table 1. Positive controls used were feline mammary carcinoma (score 3+) for HER2 and normal feline lung for TTF-1. For the negative controls, the primary antibody was omitted. TTF-1 labeling was considered positive when present in >10% of neoplastic cells. 5 HER2 protein was scored, based on the ASCO CAP guidelines, and grouped in tumors with positive (3+), equivocal (2+), and negative expression (1+, 0). 20
Immunohistochemical Specifications and Antibody Details.
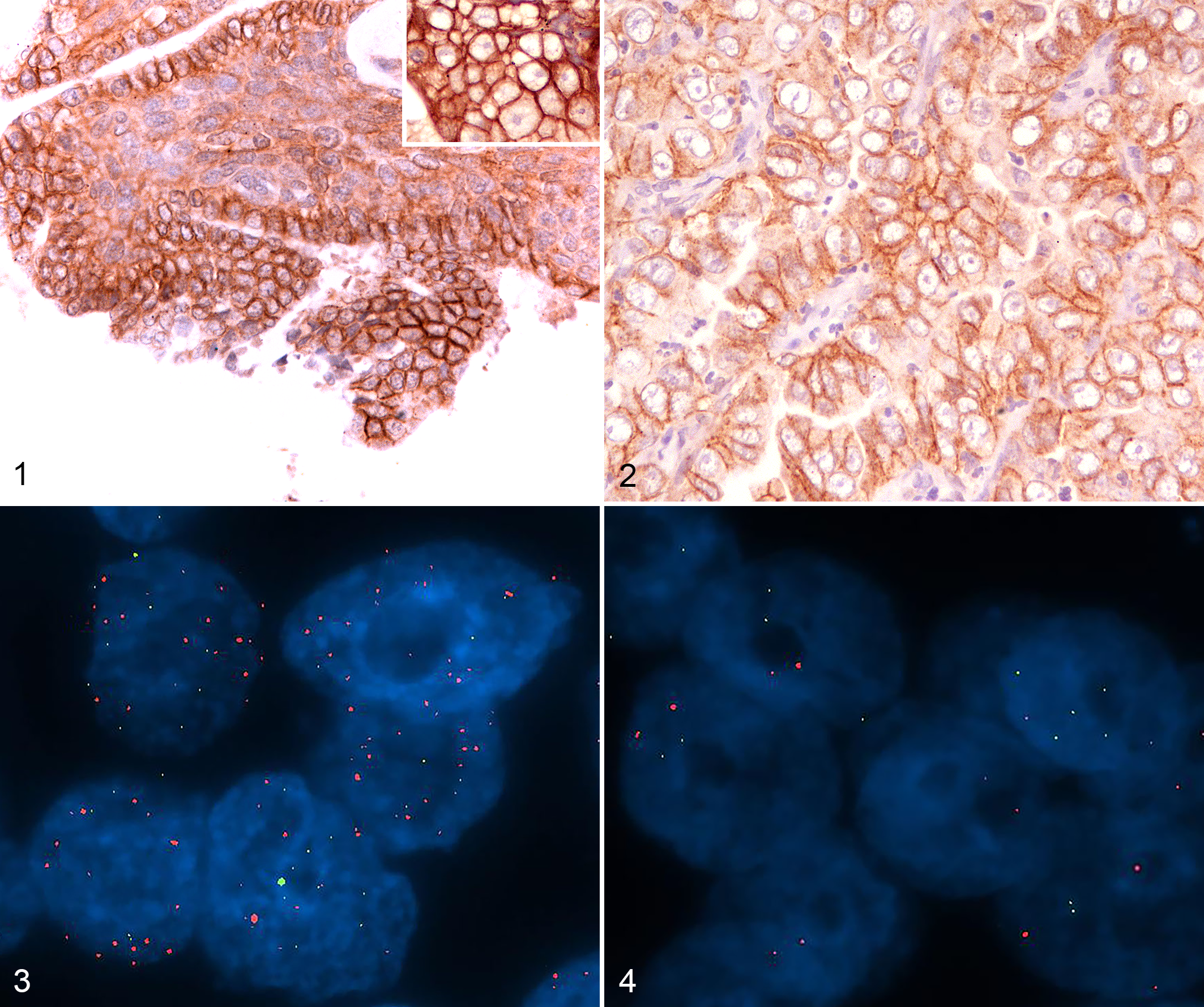
Tumors were subjected to fluorescence in situ hybridization (FISH) in a dual-core tissue microarray containing 6 cases (the layout is described in a previous study) 13 and 7 whole sections. Easy FISH pretreatment KIT (OaCP IE Ltd) was used. The sections were dewaxed, dehydrated, air-dried, and then incubated with permeation solution in water bath at 90 °C for 5 minutes and in protease solution at 37 °C for 23 minutes. The buffer (Smart-ISH Solve, OaCP IE) and a probe for HER2 was applied (HER2 spectrum Orange FISH probe, TITAN FISH probe, OaCP IE). The previously tested BAC probe 662A1H2 6,13 that hybridized the p1.12-p1.11 region (green spectrum) proximal to the nucleolus organized region of chromosome E1 was used as a control to verify the polysomy. The slides were incubated at 75 °C for 10 minutes for DNA denaturation and at 37 °C overnight for hybridization. The slides were washed in NP40 0.5%/2× SSC (pH 7.0–7.5) at 75 °C for 2 minutes, in 2° SSC for 2 minutes at room temperature, dehydrated, and counterstained using DAPI. Human breast cancer with amplified HER2 was used as positive control. The evaluation system was based on the ASCO/CAP guidelines. 20 FISH was performed twice for 4 of 13 cases in order to verify the reproducibility of the method.
Statistical evaluation was performed using GraphPad Prism 8.3. Data were not normally distributed according to D’Agostino and Pearson omnibus test. Mann-Whitney test was used to investigate the associations between HER2 protein and gene status, HER2 protein and TTF-1 expression. Spearman test was used to investigate the correlation between the HER2 antibodies tested. P < .05 was considered as significant.
The affected individuals were 6 males and 7 females with a mean age of 12.4 years. Breeds represented were the Domestic Short Hair (9/13), Persian (2/13), and Siamese (2/13). Twelve adenocarcinomas and 1 squamous cell carcinoma were diagnosed. Adenocarcinoma was the predominant subtype and showed multiple histological patterns: solid (7/12), lepidic (3/12), acinic (1/12), and micropapillary (1/12).
Western blot of HER2 (CB11) revealed a specific band migrating at 185 kDa in feline cell lines and human MCF10, demonstrating cross-reactivity of the antibody (Fig. S1). A slight positivity was also shown in murine tissue. On the contrary, no band corresponding to the expected size of TTF-1 (41 kD) was distinguishable in any of the analyzed samples, and several nonspecific bands were present (Fig. S2).
In order to confirm the pulmonary origin of the carcinomas, TTF-1 IHC was performed. Intense to moderate nuclear expression of TTF-1 was present in normal pneumocytes in 12 of 13 cases (Fig. S3). In a single case, no expression of TTF-1 was present in the internal control, but a pulmonary origin of the neoplasm was confirmed by the combined information of morphology and absence of other carcinoma in the clinical history.
There was moderate to intense nuclear expression of TTF-1 (Fig. S4) in >10% of neoplastic cells, in 7 out of 12 carcinomas (58%). There was no TTF-1 immunolabeling in the other 5 carcinomas.
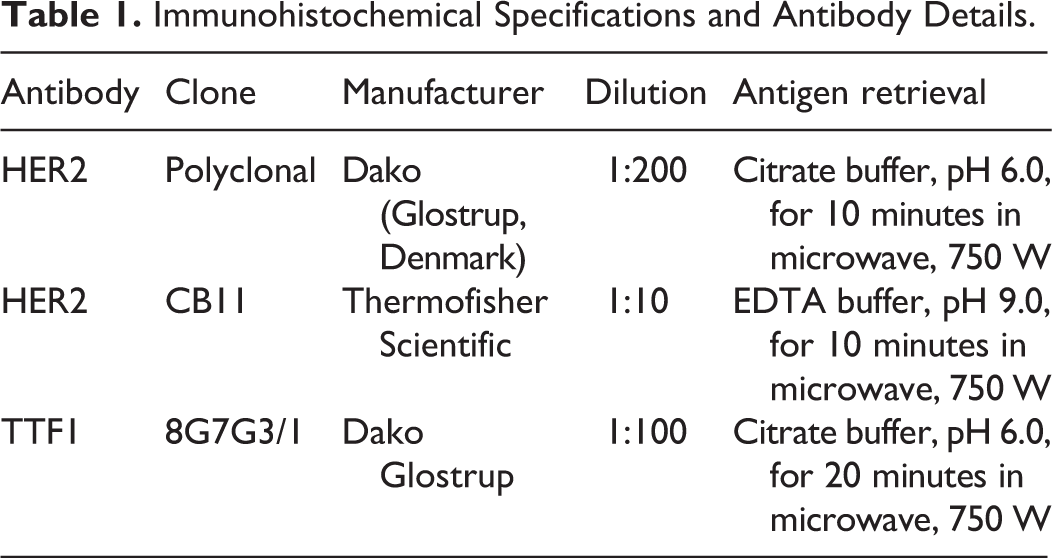
Based on IHC using the polyclonal antibody, HER2 was overexpressed (3+) in 2/13 cases (15%; Fig. 1), equivocal (2+) in 5 cases (38%; Fig. 2), and negative (0 and 1+) in 6 cases (46%). Based on IHC using the CB11 antibody, HER2 was overexpressed in 2 cases (15%), equivocal in 2 cases (15%), and negative in 9 cases (70%). A very high correlation (ρ = 0.97; P < .0001 Spearman test) was detected regarding the expression of HER2 using the 2 different antibodies (polyclonal and CB11). No association was found between HER2 and TTF-1 expression (P = .16 Mann-Whitney).
Pulmonary adenocarcinoma, lung, cat. Immunohistochemistry for HER2.
FISH analyses of HER2 were indeterminate in 2 of 13 cases due to technical reasons caused by necrosis and <20 available nuclei. Three of the 13 cases had HER2 amplification. One of the 3 amplified cases showed >6 HER2 gene copy numbers (7.09 mean copy number/per nucleus); the E1p1.12–p1.11 region produced a mean copy number of 3.19 per nucleus, with a HER2/E1 ratio of 2.22. Two of the 3 amplified cases had HER2 gene copy numbers between 4 and 6 (4.10 and 4.78), E1p1.12–p1.11 copy number of 3.26 and 4.41, and a HER2/E1 ratio of 1.25 and 1.08, respectively. Both cases had 3+ HER2 protein overexpression. Eight of 13 cases were not amplified, with a mean HER2 copy number per nucleus <4, a diploid signal in E1p1.12–p1.11 region, and a HER2/E1 ratio <2. Quality control, performed on 4 of 13 cases, gave similar results. The HER2 gene amplification status was significantly associated with HER2 overexpression (P = .03 Mann Whitney). The results are summarized in Supplemental Table S1.
Lung carcinomas are rare in cats, representing 0.75% of all feline accessions. 5 As there is little information regarding therapeutic predictive markers in feline lung tumors, we investigated the expression of HER2 protein and HER2 gene amplification status in order to identify potential targeted therapies for this tumor. HER2 is a driver oncogene involved in the progression of human breast cancer and its role has been documented in gastric, biliary, colorectal, non–small cell lung and bladder cancers. 14 In humans, oncogenic activation of HER2 can occur with protein overexpression, consequent to an increase in the gene copy number (amplification of the gene) and by genetic mutations that cause molecular alterations in the HER2 receptor. 17
In the present study, HER2 was overexpressed in 15% and amplified in 27% of feline pulmonary carcinomas. Despite the limited number of cases, the percentage of overexpression and amplification is comparable to some rates reported in humans. 12,14,16,17 Overexpression was always matched to the gene amplification status, with statistically significant association (P = .03). This result is comparable with those reported in humans 17 and is biologically interpretable as an overexpression of the protein as a consequence of increased gene copies.
Surprisingly, a HER2-amplified case was negative with IHC, with both antibodies tested. These data may be primarily justified by a loss of reactivity due to preanalytical conditions (overfixation in formalin). Another hypothesis could be HER2 gene amplification not followed by a protein overexpression due to the production of a misformed protein that might have been degraded by the cellular proteasome. Recently, HER2 splice variants have been described, inducing those with a reduced expression of the complete mRNA sequence. 18
HER2 equivocal cases on IHC were never amplified by FISH in the present caseload, demonstrating the reliability efficacy of the HER2 scoring system 20 in the detection of HER2 positive tumors.
In order to support the pulmonary origin of the neoplasms, IHC for TTF-1 was assessed and detected in 58% of feline pulmonary carcinomas. TTF-1 is not constantly expressed in feline pulmonary carcinoma, with a reported prevalence that ranges from 60% to 67%. 5,8 TTF-1 is only weakly expressed by malignant tumors, due to loss of differentiation. 5,10 No specific band was detected by western blot, suggesting that this antibody was not able to specifically recognize TTF-1 in murine, human, and feline tissues. However, it seems to specifically recognize TTF-1 in IHC, both in the present work and in the previous literature. 8,10
In conclusion, we found that the HER2 protein was overexpressed and the HER2 gene was amplified in a subset of feline pulmonary carcinomas, with a good association between protein and gene status. These data are a promising preliminary result, but larger studies are needed to confirm if HER2 is a platform for a prospective targeted therapeutic treatment.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Muscatello_et_al - HER2 Overexpression and Amplification in Feline Pulmonary Carcinoma
Supplemental Material, Combined_supplemental_materials-Muscatello_et_al for HER2 Overexpression and Amplification in Feline Pulmonary Carcinoma by Luisa Vera Muscatello, Enrico Di Oto, Miriam Dignazzi, William J. Murphy, Ilaria Porcellato, Raffaella De Maria, Terje Raudsepp, Maria Pia Foschini, Monica Sforna, Cinzia Benazzi and Barbara Brunetti in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
