Abstract
Biliary tract cancers are clinically and genetically heterogeneous cancer type with a worst prognosis among gallbladder adenocarcinoma patients. Systemic therapeutic options for metastatic biliary tract cancers are fewer, and there are limited treatment choices for the patients who progress on first line apart from symptomatic treatment. Thus, a biomarker-guided personalized treatment approach needs to be explored among biliary tract cancer subtypes. We encountered a case of 53-year-old male patient with human epidermal growth factor receptor 2 (HER2, ERBB2) positive metastatic gallbladder cancer, treated with first-line gemcitabine and cisplatin combination–based chemotherapy along with trastuzumab followed by second-line treatment with mFOLFIRINOX. On progression in third line, treated with single agent ado-trastuzumab emtansine targeting human epidermal growth factor receptor 2 and got survival benefit of nearly 6 months. This is the first reported case from India that explored the possibility and impact of ado-trastuzumab emtansine in advanced gallbladder cancer after exhausting standard treatment options. It highlights the possibility of exploring ado-trastuzumab emtansine for treatment resilient, human epidermal growth factor receptor 2-positive and advanced gallbladder adenocarcinoma.
Keywords
Introduction
Biliary tract cancers (BTCs) embody a diverse cluster including intrahepatic cholangiocarcinoma (CCA), perihilar-CCA or Klatskin tumours, extrahepatic-CCA and gallbladder adenocarcinoma (GBC). These heterogeneous cancer types possess diverse demographics, risk factors, clinical presentation, molecular features and treatment opportunities. Systemic therapeutic options for metastatic BTCs are fewer, and in majority of case, gemcitabine plus cisplatin is most commonly used in the first-line treatment; although, there are limited choices for the patients who progress on first line apart from symptomatic treatment. 1
BTCs are clinically and genetically heterogeneous with a worst prognosis among GBC patients; thus, a biomarker-guided personalized treatment approach needs to be explored among BTC subtype. 2 BTC is extremely desmoplastic in nature which limits the diagnostic accuracy of cytological and pathological–based approach; thus, genomic sequencing and molecular subtyping have paved the way for better therapeutic management. 3 Genomic profiling studies in GBC show that amplification or overexpression of erb-b2 receptor tyrosine kinase 2 (ERBB2)/human epidermal growth factor receptor 2 (HER2) can be a major targetable mutation.4,5 HER2 is a well-known therapeutic target in case of breast, gastric and gastroesophageal junction (GEJ) cancer. 6 HER2 overexpression is detected in around 19% of gallbladder tumours, 17% of extrahepatic-CCA, 13% of ampullary carcinomas and 5% of intrahepatic-CCA.1,7 However, studies of HER2-directed therapy for BTCs have been very much restricted to small clinical trials in patient populations unselected for HER2 positivity, in retrospective studies and as an individual case reports.8–10
In this case study, we encountered an HER2-positive, metastatic GBC patient who responded to a series of combination therapies containing trastuzumab, and subsequently to single agent ado-trastuzumab emtansine (T-DM1).
Case presentation
In May 2020, a 53-year-old Indian male patient presented to our tertiary care hospital clinic with a vague abdominal pain along with significant weight loss (Figure 1). He underwent cholecystectomy for cholelithiasis in May 2020, which on histopathological examination revealed moderately differentiated adenocarcinoma invading perimuscular connective tissue on peritoneal side without serosa involvement and with lymph vascular invasion (pT2aNx). Post procedure, he was referred to our tertiary care centre where he was evaluated with positron emission tomography (PET)/computerized tomography (CT) scan which revealed absence of any metabolically active lesion at operative bed site but presence of metastatic disease in terms of cervical, mediastinal, retroperitoneal and pelvic lymphadenopathy. His CA-19.9 level was also raised up to 140.2 U/mL. No loss of mismatch repair protein (MMR) expression was noted by immunohistochemistry (IHC), but Her-2-neu (Ventana Medical systems, Roche, Tuscon, AZ, USA) was showing 3+ positive result in IHC. He was started on chemotherapy (gemcitabine and cisplatin) along with trastuzumab. Repeat imaging after three cycles showed a partial response, so he continued receiving the same regimen and completed eight cycles in November 2020 with a CA-19.9 level of 14 U/mL. Subsequently, he received maintenance trastuzumab with capecitabine for 3 months (November 2020 to February 2021). Repeat imaging in March 2021 showed signs of disease progression in multiple nodes of thorax and abdomen region. Thus, he further received second-line chemotherapy with mFOLFIRINOX for four cycles following which he had progressive disease (Figure 2(a)). At this point, we repeated the biopsy for left supraclavicular lymph node and did broad molecular profiling which showed persistent HER2 overexpression in form of ERBB2 amplification (CNV 8).

Clinical timeline of case report.
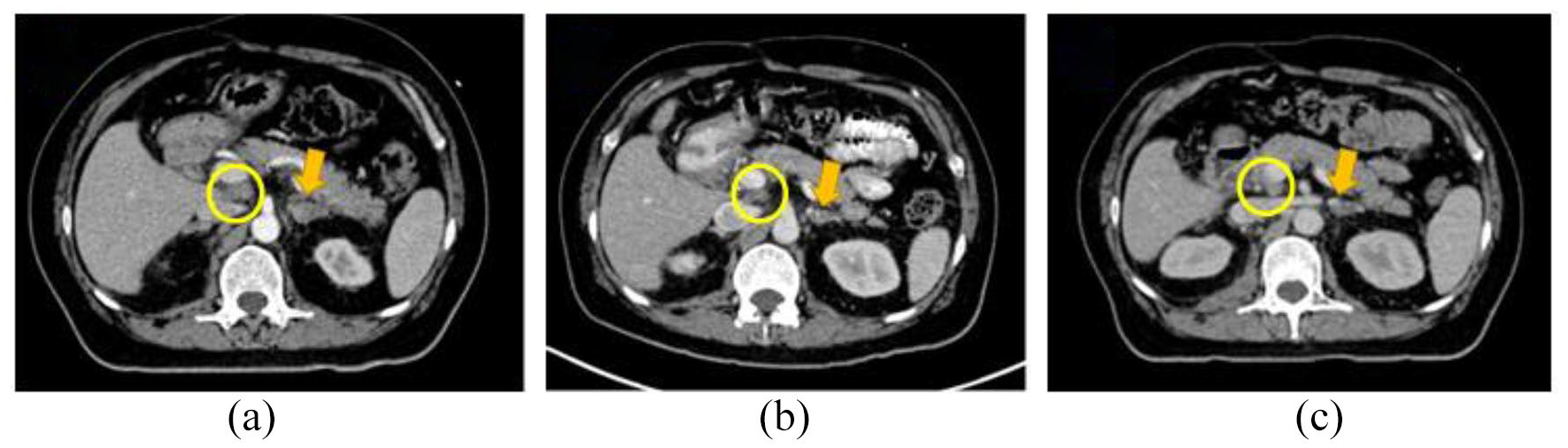
CECT images of the patient. (a) Before starting T-DM1. Left adrenal metastasis (yellow arrow) and portocaval lymph node (yellow circle). (b) After three cycles of T-DM1 showing a partial response. (c) Sustained response after six cycles of T-DM1.
After extensive multidisciplinary team (MDT) discussion, we elected to continue targeting HER2 and started patient on single agent T-DM1 (dose 160 mg q3w). Imaging done after three cycles showed a partial response (Figure 2(b)). He further received six cycles with a sustained response (Figure 2(c)). However, in January 2022, he had clinical worsening and imaging reports showed disease progression. Patient was then kept on best supportive and hospice care treatment. In order to publish this report, written informed consent was obtained from patient’s legal guardian for disclosure of images and clinical details.
Discussion
In India and Chile, GBCs are very common, with a higher incidence of cholelithiasis. 11 India, in particular, is a region with higher prevalence for gallbladder cancer contributing to around 10% of the worldwide load. In Indian context, the rate is higher in North, North-East, Central and Eastern region while it is less prevalent in South and West India. 12
This sporadic tumour has not been studied well, and ‘Precision oncology’ becomes practicable once the genomic landscape of GBC is well explored. However, limited exertions have been carried out to understand the genomic alterations present in this sporadic tumour and to advance guidelines for the clinical practice of precision oncology in GBC. 13 Even though several trials have been tried to assess the efficacy of targeted drugs as monotherapy or as combination drugs, no targeted therapeutic regime has yet been approved for advanced GBC treatment. 1 Although, the percentage of GBC patients gaining benefit from targeted therapy is sparse, especially in case of country like India. This is most likely due to the lack of availability of translational research and also because of affordability issues in terms of expensive treatment. The present case study primarily aims to explore the usage of targeted antibody-drug conjugate (ADC) in HER2 overexpressed GBC patient. Gemcitabine and cisplatin combination (GemCis) is used as the standard chemotherapy treatment since the conduct of pivotal ABC-02 trial which showed higher efficacy and survival outcomes of GemCis combination over gemcitabine monotherapy. 14 This is also consistent with latest ICMR (Indian Council of Medical Research) guideline for the management of GBC, of the note that there is no standard second-line therapy for advance GBC globally as well as in India. Hence, we went out to move towards the usage of T-DM1, if feasible. 15
Although, the clinical conclusion in case of advanced BTCs still remains inferior, with around 13%–26% overall response rate towards GemCis chemotherapy and less than 10% 5-year survival rates.14,16 While as per the randomized phase III ABC-06 study, in the second-line setting treatment with fluorouracil, leucovorin and oxaliplatin showed significant benefit in comparison with symptomatic treatment, with a median survival of 6.2 and 5.3 months, respectively. 17 Despite these treatments, overall survival (OS) in advanced stages remains of about 1 year only.
Next-generation sequencing (NGS) revealed that the three clinical subtypes of BTCs possess significant molecular differences in ERBB2 gene, and it is most prevalent in case of GBC (12%–19%). 18
Considering the lower incidence rate of BTCs, the clinical subtyping of subgroups based on overexpression or mutation of ERBB2 is sparse and the clinical practice with HER2-directed therapy has been largely anecdotal. Trastuzumab-directed treatment in HER2+ BTC is being evaluated in a phase II trial (ClinicalTrials.gov identifier number (National Clinical Trial (NCT)) 03613168). The results of a phase II trial on T-DM1 in BTCs are still awaited (NCT02999672).
A recent basket trial presented an objective response in 4 (36%) out of 11 patients with HER2 amplified or mutant BTC patients treated with the combination of trastuzumab and pertuzumab–directed anti-HER2 therapy. 19 We report here a GBC patient with HER2 overexpression who attained a sustained partial response towards the HER2-directed ADC T-DM1 and showed good survival outcome in comparison with the traditional therapy. With the availability of T-DM1 biosimilar in India, it becomes a cost-effective option for patients exhausting standard treatment line.
Conclusion
To the best of the authors’ knowledge, this is the first reported case from India that explored the possibility and impact of T-DM1 in advanced gallbladder cancer after exhausting standard treatment options. In one case reported from basket study, the patient had extrahepatic cholangiocarcinoma and had metastatic recurrence within 3 months of starting adjuvant chemotherapy, which was further treated with T-DM1. Patient had shown durable progression-free survival (PFS) of 8.6 months with this first-line T-DM1 usage. In our case, T-DM1 was used in third-line and patient still got clinical and radiological benefit of nearly 6 months which supports a clinically meaningful benefit in real world. This case highlights the possibility of exploring T-DM1 for treatment resilient, HER2-positive and advanced GBC.
Footnotes
Acknowledgements
The authors thank hospital staff, patient and his guardian for their support in providing necessary details.
Author contributions
All authors meet the International Committee of Medical Journal Editors (ICMJE) authorship criteria. V.L. provided medical care and contributed to the preparation of the manuscript. V.T. and P.M designed the manuscript outline, collected and analysed data and drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors received financial support for publication of manuscript from Zydus Lifesciences Ltd.
Ethical approval
Institution does not require ethical approval for reporting individual cases or case series. However, authors are accountable for all aspects of the work including full data access, integrity of the data and the accuracy of the data analysis.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
