Abstract
An 8-y-old National Show Horse mare was presented for evaluation of pneumonia and laminitis. Harsh bronchovesicular sounds were auscultated throughout both lung fields, and the mare had signs of moderately painful laminitis. Thoracic ultrasonography revealed lung consolidation throughout the dorsal aspect of both lungs, and radiography revealed an extensive diffuse-to-patchy bronchointerstitial lung pattern. The mare’s clinical condition rapidly deteriorated, and euthanasia was elected. On postmortem examination, the lungs, omentum, spleen, liver, adrenal glands, kidneys, and femur contained 0.5–2.5-cm, firm, tan nodules. Histologically, the lungs, spleen, liver, kidneys, adrenal glands, omentum, left eye, and femur were infiltrated by bundles and nests of pleomorphic polygonal-to-spindloid cells intermixed with frequent multinucleate cells. Lymphatic vessels in the affected tissues were frequently distended with tumor emboli. Neoplastic cells were diffusely positive for vimentin, desmin, sarcomeric actin, myoblastic differentiation protein 1, and myogenin, supportive of the diagnosis of rhabdomyosarcoma (RMS), which is a rare neoplasm in horses. Cross-striations were not evident with H&E or phosphotungstic acid–hematoxylin stains. Markedly pleomorphic neoplastic cells, multinucleate cells, and lack of cross-striations suggested the subclassification of pleomorphic RMS.
An 8-y-old National Show Horse mare was presented to the University of Missouri (MU) Veterinary Heath Center Equine Medicine service (Columbia, MO, USA) for evaluation of pneumonia and acute laminitis. The mare had a year-long history of decreased energy and back pain. On presentation, the mare was bright, alert, and responsive, and had a body condition score of 6 of 9 (with 5 of 9 ideal). 12 The rectal temperature was normal, but both the heart (60 bpm) and respiratory (42 brpm) rates were elevated. Harsh bronchovesicular sounds were auscultated bilaterally throughout the chest. Borborygmi were generally diminished. Digital arterial pulses were prominent in all 4 limbs. Signs of laminitic pain included ambulation with a stilted gait, especially evident when turning, and persistent weight-shifting on all limbs (Obel grade 2 of 4). 13 Abnormalities in the CBC included severe leukopenia (1.2 × 109/L; RI: 4.8–10.4 × 109/L) characterized by marked neutropenia (0.3 × 109/L; RI: 2.4–5.8 × 109/L) and lymphopenia (0.9 × 109/L; RI: 1.5–5.2 × 109/L). Regeneration of white blood cells was not evident on repeat testing. Our findings heightened the suspicion of myelophthisis. Prominent lung consolidation was ultrasonographically evident throughout all dorsal lung fields with diffuse, mild B-line artifacts present ventrally on both sides of the chest, which indicated pleural surface changes or irregularities consistent with mild consolidation at the lung surface. Thoracic radiography revealed a severe patchy bronchointerstitial lung pattern. No gross abnormalities were observed during endoscopic examination of the upper airway, and fluid retrieved endoscopically from the tracheal lumen was unremarkable cytologically, with no aerobic or anaerobic bacterial growth.
Medical treatment was pursued, including intravenous fluids (lactated Ringer solution, 30 mL/kg/day), broad-spectrum antimicrobials (potassium penicillin, 22,000 IU/kg IV q6h; gentamicin, 6.6 mg/kg IV q24h; metronidazole, 15 mg/kg PO q8h), anti-inflammatory and analgesic medications (flunixin meglumine, 1.1 mg/kg IV q12h; acetaminophen, 30 mg/kg PO q12h; 2% lidocaine, 0.05 mg/kg/min as a constant rate infusion), steroids (methylprednisolone, 0.05 mg/kg IV once), and distal limb cryotherapy. There was clinical and clinicopathologic deterioration despite treatment, including worsening leukopenia, muscle fasciculations, pyrexia, and worsening laminitis. Euthanasia was elected, and the mare was submitted to the MU Veterinary Medical Diagnostic Laboratory for autopsy.
On postmortem examination, all lung lobes contained 0.5–2.5-cm, irregular, firm, tan nodules (Fig. 1A). The omentum, spleen, liver, adrenal glands, and kidneys contained similar pinpoint-to-3-cm, tan, flat-to-raised, semi-solid to firm nodules. Tracheobronchial and intra-abdominal lymph nodes were enlarged up to 8 × 5 × 4 cm. On cut section, the left femur contained poorly demarcated, pale-tan regions surrounded by dark-red rims throughout the medulla (Fig. 1B). Tissues were fixed in 10% neutral-buffered formalin and processed routinely; 4-µm sections were stained with H&E and phosphotungstic acid–hematoxylin (PTAH). Immunohistochemistry (IHC) was performed to reach a definitive diagnosis (Table 1). All of the antibodies used have been previously validated for use in horses.7,15 A heat-induced method was performed for antigen retrieval using a retrieval solution (Diva Decloaker; Biocare Medical), except for the pancytokeratin IHC for which proteinase K was used.
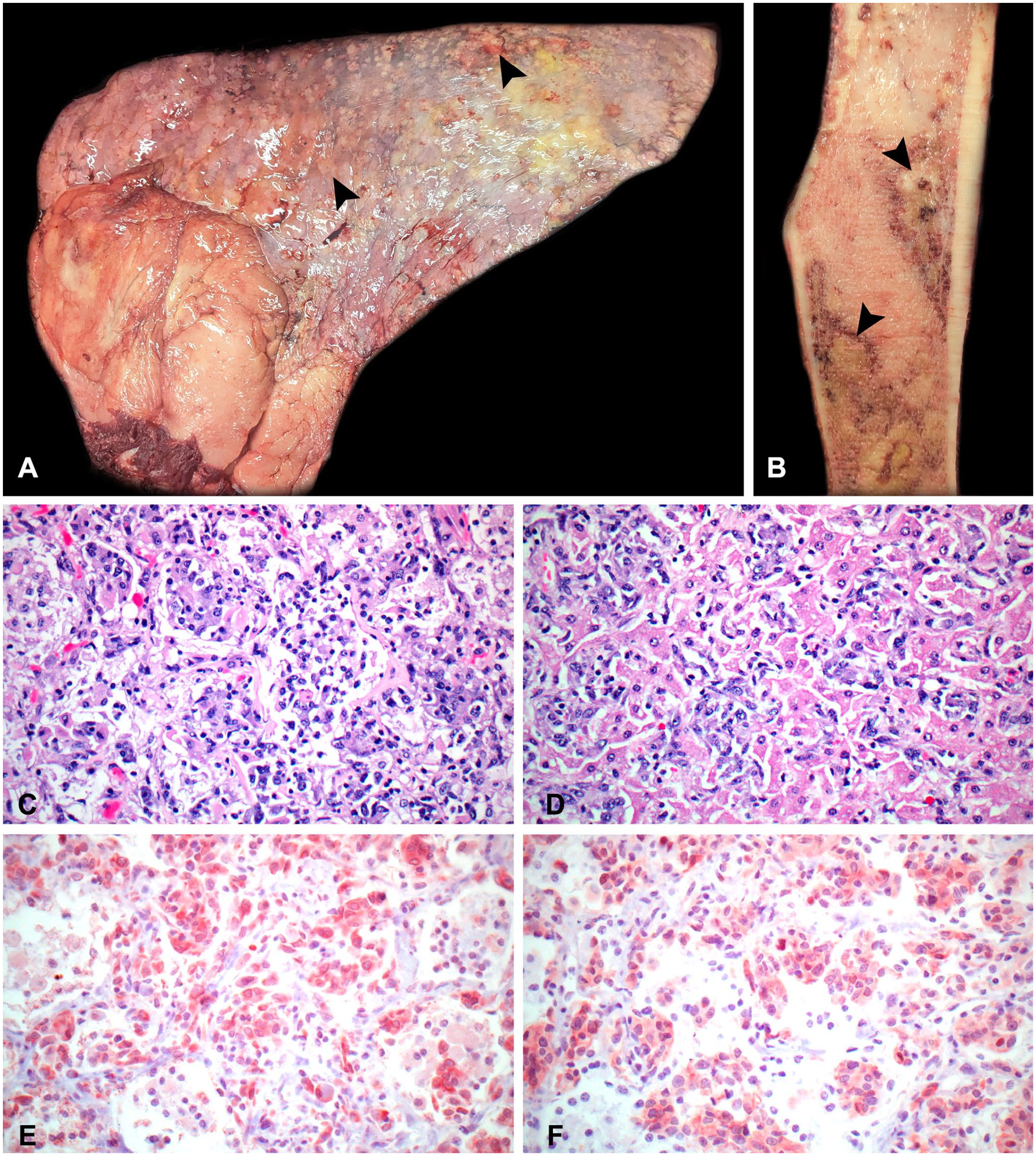
Disseminated pleomorphic rhabdomyosarcoma in a horse.
Primary antibodies used for immunohistochemical staining in the diagnosis of pleomorphic rhabdomyosarcoma in a horse.
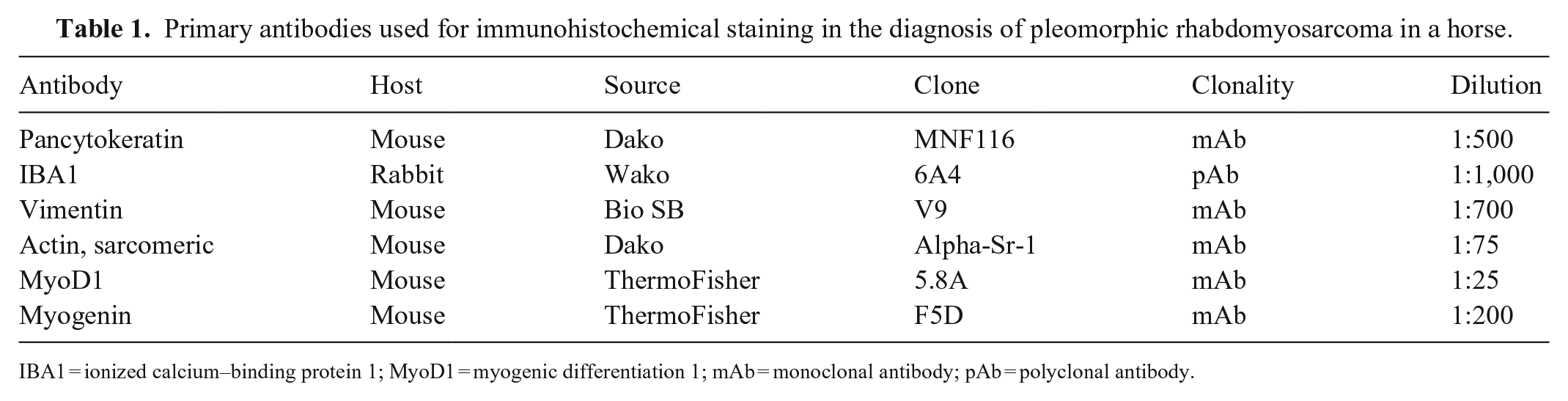
IBA1 = ionized calcium–binding protein 1; MyoD1 = myogenic differentiation 1; mAb = monoclonal antibody; pAb = polyclonal antibody.
Histologically, the lungs (Fig. 1C), spleen, liver (Fig. 1D), kidneys, adrenal glands, omentum, and left eye had a poorly demarcated unencapsulated invasive neoplasm composed of bundles and nests of polygonal-to-spindloid cells supported by a thin fibrous stroma. Neoplastic cells were 14–35-µm diameter with mild-to-moderate amounts of eosinophilic cytoplasm and variably distinct borders. Nuclei were round-to-oval and euchromatic with finely stippled chromatin and 1 or 2 prominent nucleoli. Frequent multinucleate cells were scattered throughout the tumor. Anisocytosis and anisokaryosis were marked. The mitotic count was 22 in ten 400× fields (2.37 mm2). The neoplastic cell population effaced marrow spaces in the left femur. Lymphatics, veins, and occasionally arteries in the liver, lungs, kidneys, adrenal glands, and left eye were frequently distended with tumor emboli. The lamellar architecture of the hoof was moderately disrupted by elongation of secondary epidermal lamellae at the axial aspect of each primary epidermal lamella and the formation of excessive cap horn over the tips of primary dermal lamellae. These changes were consistent with chronic laminitis. There was no evidence of neoplasia in the sections of hooves examined. Due to severe pleomorphism of the tumor cells, severe effacement of all tissues by the tumor, and lack of obvious skeletal muscle involvement, the origin of the tumor could not be determined.
Immunohistochemically, the neoplastic cells were negative for cytokeratin and had moderately intense red-brown intracytoplasmic immunolabeling for vimentin, suggesting mesenchymal origin. Scattered tumor-associated macrophages and multinucleate giant cells had strong red-brown intracytoplasmic immunolabeling for ionized calcium–binding protein 1 (IBA1). Neoplastic cells were diffusely negative for IBA1, thus ruling out histiocytic sarcoma. Neoplastic cells had moderately intense to strong red-brown intracytoplasmic immunolabeling for desmin, suggesting that the tumor was of muscle origin. Furthermore, the neoplastic cells had strong red-brown intracytoplasmic immunolabeling for sarcomeric actin, moderately intense red-brown intracytoplasmic immunolabeling for myoblastic differentiation protein 1 (MyoD1; Fig. 1E), and patchy weak red-brown membranous to moderately intense intracytoplasmic immunolabeling for myogenin (Fig. 1F). Cross-striations were not observed in neoplastic cells in H&E- or PTAH-stained preparations. Based on histology and IHC, rhabdomyosarcoma (RMS) was diagnosed.
RMSs are malignant mesenchymal neoplasms arising from primitive mesenchyme that differentiates into skeletal muscle. 3 RMS is rarely encountered in domestic animals, especially horses.1 –3,14,17 RMSs can affect newborn to geriatric-aged horses,3,5 and no sex or breed predilections are reported. 1 RMS in horses can be invasive with metastatic potential. 3 RMSs in veterinary medicine are classified as embryonal, alveolar, or pleomorphic.2,3,8,19 Whereas the human classification scheme is correlated with prognosis, data and clinical outcome information is limited for horses due to the rarity of RMS and lack of grading criteria. 3
Embryonal RMS has 2 variants, including a myotubular variant and a rhabdomyoblastic variant. 2 The myotubular variant has multinucleate strap cells that frequently have cross-striations. 2 The rhabdomyoblastic variant has small round cells intermixed with large rhabdomyoblasts that rarely have cross-striations. 2 In horses, embryonal RMS has been reported in the abdominal wall, 3 tongue, 3 right shoulder,1,3 buccal wall, 3 and heart. 3
Botryoid RMS is a type of embryonal RMS that has characteristic grape-like masses most commonly arising from the submucosa of the urinary bladder. 2 Botryoid embryonal RMS have been reported in the uterus 16 and urinary bladder 18 of horses.
Alveolar RMS is reported in human adolescents and adults, and has a distinct alveolar pattern composed of packets of poorly differentiated small round cells separated by fibrous septa. 2 Alveolar RMS in horses has been reported to occur most typically in the extremities.5,9,10,14
Pleomorphic RMS is rare and the least common subclass in domestic animals. 2 This type of RMS is commonly reported in adult animals and humans, typically arises on the extremities, 5 and is comprised of haphazardly arranged polygonal-to-spindloid cells with marked pleomorphism, bizarre mitotic figures, and the absence of features associated with embryonic and alveolar RMS.2,17 In horses, pleomorphic RMSs have been reported in the tongue, 11 masseter muscle, 3 thoracic cavity, 10 and abdominal wall. 10 Pleomorphic RMS can be misdiagnosed as an anaplastic sarcoma or poorly differentiated sarcoma, 2 thus highlighting the importance of immunohistochemical differentiation. Defining features that included markedly pleomorphic polygonal-to-spindloid cells, multinucleate cells, and lack of cross-striations with positive immunohistochemical staining for muscle markers warranted the diagnosis of pleomorphic RMS in our case.
In humans, dogs, and horses, RMS routinely immunolabels for vimentin, desmin, and sarcomeric actin.2,3 Myogenin and MyoD1 are myogenic transcriptional regulatory proteins associated with myogenesis.2,4,15,17 These proteins are found in fetal muscle and typically are no longer expressed after birth.1,4,8 Immunohistochemical markers for these proteins have proven to be useful for the diagnosis for RMS.4,8 Studies of human RMS have determined myogenin to be highly useful in conjunction with histology to diagnose specific subtypes of RMS; however, this feature has not been replicated in domestic animals.4,8
Histologic subtypes are important for guiding prognosis and therapeutic strategies in humans along with patient demographics, tumor location, tumor spread, and excisional margins. 8 This prognostic information is not available for dogs and horses, and criteria for grading equine RMS have not been developed due to a limited number of case reports.2,3,6 A study of 8 horses found that solitary neoplasms >10-cm diameter were more likely to be infiltrative and more difficult to excise surgically. 3 Pleomorphism of neoplastic cells did not appear to be predictive of tumor behavior or prognosis with surgical treatment. 3 However, high mitotic count appears to be associated with more infiltrative and metastatic behavior, 3 similar to previous reports.5,9,10 Our case had a high mitotic count (≤22 in 2.37 mm2) with dissemination to multiple organs.
Evidence of hematogenous and lymphatic tumor emboli3,5 and metastasis to the lungs, 10 thoracic wall, 10 pericardium, 10 ureter, 16 abdominal wall,10,18 omentum, 10 spleen, 10 internal iliac lymph nodes, 9 and mesenteric lymph nodes 16 has been reported in horses with various subtypes of RMS, but to our knowledge, disseminated pleomorphic RMS extensively involving multiple organs throughout the body, including the eyes and bone marrow, has not been reported previously in horses. We retrieved no cases of disseminated pleomorphic RMS diagnosed in horses in a search of Google, PubMed, CABI Direct, Web of Science, and Scopus, using the search terms “disseminated rhabdomyosarcoma” and “pleomorphic rhabdomyosarcoma”, suggesting that this condition has not been reported in horses.
Clinical presentations described in horses vary depending on tumor location. Cases of RMS arising from external skeletal muscle or the tongue often appeared as grossly visible swellings, 1 slowly growing masses,3,5 lameness, 5 or the tumor was found incidentally with no reported clinical signs. 3 A case of RMS arising within the peritoneum had colic signs, edema, and ascites. 3 A case of botryoid RMS arising from the urinary bladder had hematuria and stranguria with a large mass protruding from the vagina. 18 Cases with RMS arising from the thoracic aspect of the diaphragm or with metastasis involving the lungs had respiratory distress. 10 Our case had signs of pneumonia and laminitis with no clinical evidence of abdominal disease despite widespread involvement of abdominal organs.
The tissue of origin could not be determined in our case due to the lack of neoplasia in the skeletal muscles and heart, and dissemination of similar tumor nodules throughout multiple organs. In dogs and humans, there are reports of RMS occurring in tissue other than skeletal muscle, and it is hypothesized that myogenic transformation of cells may be involved. 20 It is unclear if the laminitis in our case is related to tumor metastasis as there were no tumor emboli in the examined sections; however, inflammation in response to the tumor may have contributed. Our case highlights the importance of considering RMS as a differential for disseminated neoplasms or pleomorphic spindle-cell tumors in the horse, as well as considering neoplasia as a differential in cases of pneumonia.
Footnotes
Acknowledgements
We thank Karen Clifford for her help creating the figures.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research or authorship of this article. Support for publication was provided by the Kent Tomazi Memorial Research Fund in Veterinary Medicine.
