Abstract
The most common subtype of lymphoma in the dog is diffuse large B-cell lymphoma (DLBCL). The remaining forms of B-cell lymphoma in dogs are categorized as small-to-intermediate in size and include marginal zone, follicular, mantle cell, and small-cell lymphocytic lymphoma. Marginal zone lymphoma and follicular lymphoma have readily identifiable unique histologic features while other forms of small B-cell lymphoma in the dog are poorly described by histopathology. Forty-seven cases of nodal small B-cell lymphoma identified by flow cytometry (small cell size based on forward scatter) with concurrent histopathology were reviewed. These cases fell into 3 histologic subtypes: marginal zone lymphoma, follicular lymphoma, and a diffuse form of small B-cell lymphoma with consistent features. As a descriptive term, we refer to the latter subtype as diffuse small B-cell lymphoma (DSBCL) until it can be further characterized by gene expression profiling and other molecular tools. Clinical presentation of DSBCL was compared to cases of histologically confirmed DLBCL and clinical follow-up was obtained for 22 of the 27 cases of DSBCL. This subset of diffuse small B-cell lymphoma had an overall median survival of 140 days. The expression of CD21, class II MHC and CD25 by flow cytometry did not differ between DSBCL and the other histologic subtypes of small cell B-cell lymphoma making histopathology the only current method of classification.
B-cell lymphomas account for 60% to 70% of all canine lymphomas, 16,21 with diffuse large B-cell lymphoma (DLBCL) being the most common subtype in both humans and dogs. 16,21,28,30 DLBCL is characterized by large neoplastic B-cells with nuclei >2 times the diameter of a red blood cell and a diffuse histologic growth pattern. 28 Canine DLBCL has a variable clinical course with median survival times reported to be 191 to greater than 300 days with multi-agent chemotherapy. 4,7,27,33 The other reported subtypes of B-cell lymphoma in the dog include marginal zone lymphoma (MZL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and more rarely reported lymphoplasmacytic lymphoma, lymphoblastic B-cell lymphoma, Burkitt-type lymphoma, and small lymphocytic lymphoma (SLL). 16,28,30 B-cell lymphomas other than DLBCL are first delineated by a diffuse or nodular pattern and are composed of small or intermediate-sized lymphocytes with nuclei 1 to 2 times the diameter of a red blood cell. 16,21,28 MZL and FL are well recognized in the dog, and both have a nodular pattern with distinct cellular morphology. 16,28,29 In dogs, splenic MZL is considered an indolent disease of intermediate-sized cells, 13,23,29 but nodal MZL may have a more aggressive clinical course. One third of patients with nodal MZL died due to lymphoma within 6 months despite multi-agent chemotherapy and a low mitotic index. 5
Other forms of non-DLBCL lymphoma are more difficult to identify based on morphology alone. In humans, the distinction between chronic lymphocytic leukemia/small B-cell lymphocytic lymphoma (CLL/SLL) and MCL can be challenging and relies on the expression of several antigens that cannot currently be detected with available antibodies in the dog. 6,34 Cyclin D1 expression is a hallmark of human MCL and not expressed in SLL. 3,10 MCL has been described in dogs as an uncommon nodular splenic tumor. 29 Attempts to identify cyclin D1 expression in canine MCL have not been successful 20,24,29 and diagnosis has relied primarily on morphology. 16,29 SLL has been reported to account for <1% of canine lymphomas in large scale studies, 16 yet B-cell CLL (B-CLL) is a well-recognized disease in dogs, 2,32 and reported to account for 8% of lymphoproliferative disease in dogs. 2 CLL/SLL are considered the same disease in humans defined based on the distribution of disease, either blood and spleen (CLL) or lymph node (SLL). 25 Lymphadenopathy occurs in 45% of dogs with B-CLL suggesting a significant tissue phase of this disease. 2 Therefore, it is possible that SLL is underrecognized in dogs because the diagnosis of neoplasia is made using peripheral blood, and lymph node biopsies are rarely performed in these cases.
Small cell B-cell lymphoma subtypes are much less common than DLBCL, and less information about clinical course is available. For example, it is commonly believed that the descriptor “small cell lymphoma” implies a more indolent disease, 16 but robust data supporting this notion are lacking. In order to identify a cohort of small cell B-cell lymphoma cases for further characterization, we took advantage of a recent study which correlated flow cytometry and histopathology. 33 In that study, we found that B-cell lymphomas with low forward light scatter (FS) properties, roughly reflective of smaller size, were consistently classified as non-DLBCL (marginal zone, follicular, and small cell B-cell lymphomas). In the present study, we identified small B-cell lymphomas by flow cytometry and then obtained prospective or historical histopathology. Here we describe the histologic subtypes of these small B-cell lymphomas using the World Health Organization (WHO) criteria, and identify a histologic form of diffuse small cell B-cell lymphoma that does not fit into established criteria described for dogs. 16,28 Due to the diffuse nature and consistent morphology of small-to-intermediate sized cells lacking other defining features, these cases were provisionally categorized as diffuse small B-cell lymphoma (DSBCL). We further characterized the clinical features and outcomes of this entity.
Materials and Methods
Study Population
In a recent study with both histopathology and flow cytometry from September 2013 to October 2015, no cases of confirmed DLBCL were identified with a FS of under 460. 33 Based on this information, small B-cell lymphomas were defined by flow cytometry to have a median FS of less than 460. We compared large B-cell lymphomas to small B-cell lymphomas by flow cytometry (Figs. 1, 2) using 2 study populations that had available flow cytometry data and concurrent histopathology. Cohort 1 consisted of samples from July 2014 to March 2015 of 41 dogs with a confirmed histopathologic diagnosis of DLBCL; these cases had a median forward scatter of 660 with a single case below 500 (FS = 495). Removing this case, the remaining 40 cases had FS range of 512 to 794. Cohort 2 consisted of samples from October 2013 to May 2018. During this time period, 12% of all nodal B-cell lymphomas (n = 8729) had FS <460. We identified 47 cases in cohort 2 where histopathology had also been performed, or was available as follow-up. Histopathology was performed within 2 months prior to or following flow cytometric analysis. Additionally, flow cytometric parameters for normal B cells in 14 young hound-mix dogs with reactive lymph nodes that were being utilized for a series of gene expression studies were also compared. These samples were processed from August 2016 to November 2017. Finally, for a relative cell size comparison of neoplastic B cells to neutrophils we used data from 100 cases of B-cell chronic lymphocytic lymphoma in peripheral blood in dogs during the same time period (2017) submitted to the laboratory for diagnostic flow cytometry. The neutrophil count was at least 5000 cells/µL in these cases.
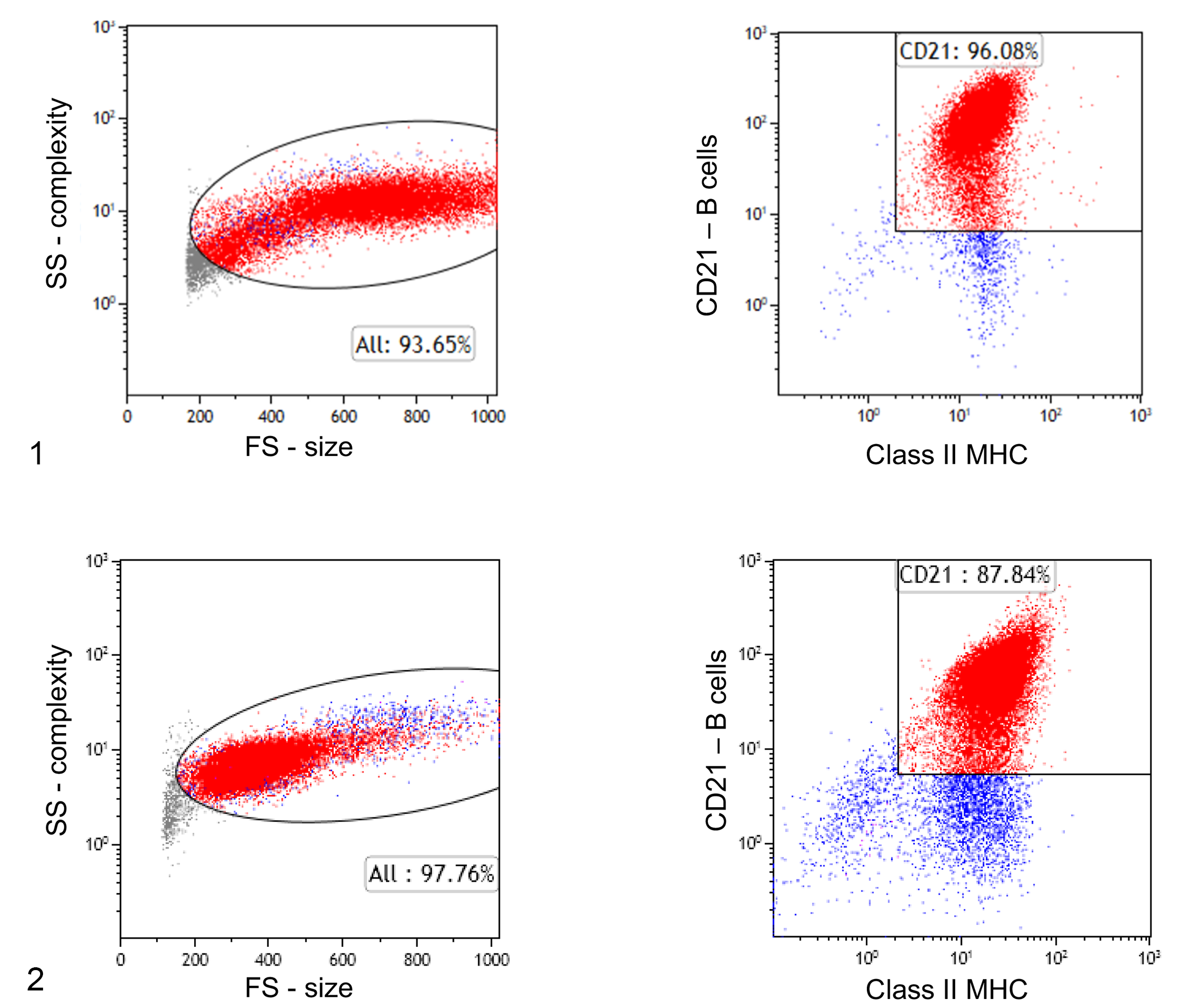
Flow cytometric features of large and small B-cell lymphoma subtypes, lymph nodes, dogs.
Flow Cytometric Criteria
Flow cytometry was performed as previously described, 22 at the Colorado State University Clinical Hematopathology (CSU-CH) Laboratory; antibodies are listed in Table 1. Lymph node aspirates in this study (88 cases and 14 normal; total = 102) were analyzed by flow cytometry; 84 samples submitted prior to March 2017 were stained with a panel of antibodies listed in Table 1 (panel 1) and the remaining 18 samples submitted after March of 2017 were stained with panel 2. For neutrophil size comparison (100 peripheral blood samples), 30 samples were stained with panel 1 and 70 samples were stained with panel 2. Diagnostic criteria for lymphoma included (1) >60% of the total lymph node population expressed CD21 by flow cytometry, based on previous criteria for lymphoma/leukemia diagnosis in dogs; 18,31 or (2) histopathologic confirmation of lymphoma. Flow cytometric parameters for the different B-cell subtypes were compared. Parameters included %CD5 T cells, %CD21 B cells, neoplastic B-cell size (FS), B-cell median fluorescence intensity (MFI) of class II MHC, CD21 and CD5, and the % of B cells expressing CD25.
Flow Cytometry Panel of Antibodies for Dogs in This Studya.

a Unless otherwise noted, all antibodies were purchased from AbD Serotec. Clones are as follows: CD45 = YKIX716.13, CD18 = YFC118.3 (human CD18), CD4 = YKIX302.9, CD8 = YCATE 55.9, CD5 = YKIX322.3, CD21 = CA2.1D6, CD22 = RFB4 (human CD22, purchased from AbCam), CD3 = CA17.2A12, CD14 = TUK4 (human), class II MHC = YKIX334.2, CD34 = 1H6, CD25 = P2A10 (purchased from eBiosciences).
Cytomorphology, Immunohistochemistry, and Histopathologic Classification
Biopsies from the 41 confirmed DLBCLs (cohort 1) were part of a clinical trial as previously described. 9 Biopsy and flow cytometry were performed simultaneously on all 41 cases. Immunohistochemistry was performed for all cases as previously described 9 and were diagnosed by WHO classification independently by 2 pathologists (VDx Veterinary Diagnostics and Preclinical studies, KLH).
Biopsy samples from the 47 nodal cases with small B cells as determined by flow cytometry (cohort 2) were either solicited from diagnostic laboratories when histology had already been performed, or collected in 10% neutral-buffered formalin and processed for paraffin embedding through the CSU Veterinary Diagnostic Laboratory. Tissue samples were cut at 3 to 5 µm thickness and stained with hematoxylin and eosin. Immunohistochemistry was performed on all cases with anti-CD3 to identify T cells (monoclonal mouse anti-human CD3, LN10 clone; Leica Biosystems Newcastle Ltd) and PAX5 (monoclonal mouse anti-human Pax5, DAK-Pax5 clone; Dako North America Inc) to identify B-cells. Two cases were additionally evaluated by Ki-67 (monoclonal mouse anti-human Ki-67, K-2 clone; Leica Biosystems Newcastle Ltd).
Biopsies of small B-cell lymphomas were reviewed by 3 pathologists (KLH, EJE, and LJH) to come to a consensus diagnosis based on WHO criteria. 28 Diagnoses included DLBCL, MZL, FL, and DSBCL as a descriptive term for this consistent subtype. The histologic pattern, cellular morphology, and mitotic count (MC) were obtained for all DSBCL cases. MC was determined by counting the number of mitotic figures in 12 consecutive high-powered fields (hpfs, 400× magnification) with an ocular FN 20/40× = 2.37 mm2 standardized area. 12 If there were at least 10% of neoplastic cells that were large (nuclei ≥ 2 × RBC) with a diffuse pattern the case was categorized at DLBCL.
Cytology was available for 6 of the cases and was reviewed by 2 clinical pathologists (PRA and EDR).
Clinical Outcome
Within cohort 2, 27 cases of small B-cell lymphoma were not histologically consistent with DLBCL, FL, or MZL or the nodular form of MCL and were categorized as DSBCL. For those 27 cases, we sought to further describe the histologic characteristics and evaluate clinical outcome. We obtained medical records from 22/27 patients and reviewed them from the time of diagnosis to death or loss to follow-up. Clinical data including signalment (age at presentation, breed, sex) and presence of hyperglobulinemia, lymphocytosis, and anemia were obtained from the sample submission form at the time of presentation and compared to the 41 cases of confirmed DLBCL.
Additional data were extracted from the medical records and included date of diagnosis, date of treatment initiation, treatment protocol, documentation of response, documentation of relapse or progression of disease, CBC abnormalities, presence of hypercalcemia or hyperglobulinemia, hepatomegaly/splenomegaly based on ultrasound, involvement of the rectum, date of death or last contact, and cause of death.
Statistical Analysis
Clinical presentation (sex, lymphocytosis, hyperglobulinemia, and anemia) was compared between DSBCL and DLBCL using Fisher’s exact test. The flow cytometric parameters and age for the B-cell lymphoma subtypes did not pass normality tests resulting in the use of nonparametric statistical tests. The difference in age were compared using the Mann-Whitney test. Flow cytometric features between small B-cell lymphomas were compared using Kruskal-Wallis test followed by the Dunn’s test for multiple comparisons.
Lymphoma-specific survival time (LSST) was calculated from treatment start date to death date due to lymphoma-specific reasons. Patients that died for reasons other than lymphoma or were lost to follow-up were not included in this statistic (n = 11). The overall median survival time (MST) was calculated from the treatment start date to death date. Patients were censored at the last date reported to be alive if they were alive at the time of analysis or lost to follow-up (5 patients censored). The Kaplan-Meier method was used to calculate the LSST and overall MST distribution.
A Pearson correlation table was used to compare flow cytometric features of small B-cell lymphoma subtypes (%B cells, B-cell size, MFI of B-cell class II MHC, CD21, CD5, and CD25% expression of B cells) and mitotic count with overall survival and response time for small B-cell lymphoma cases with follow-up data. Response was classified as complete remission, partial remission, stable disease, no response, or progressive disease. Response was defined at the discretion of the referring veterinarian’s documentation and was confirmed for a duration of at least 28 days. 26 Treatment was divided into 2 groups: prednisone and chlorambucil (includes 1 dog that was treated with prenisone alone) and multi-agent chemotherapy. Multi-agent chemotherapy included dogs treated with CHOP, or a variation of one or more of the following: COP, L-asparagine, mitoxantrone, lomustine (CCNU), and anti-survivin.
Results
Histologic and Flow Cytometric Diagnosis
Of the 47 cases of small B-cell lymphoma, 43% could be classified according to WHO criteria. These included 9 cases of MZL, 9 cases of FL, and 2 cases of DLBCL. Representative histopathologic features are shown in Figures 3 to 5. The presence of small numbers of DLBCL cases indicates that the forward scatter properties are not a perfect predictor of histologic cell size. By histopathology, the majority of cases of small B-cell lymphoma were DSBCL (27/47, 57%), which were consistently characterized by a diffuse growth pattern of small-to-intermediate sized B cells (Figs. 6–8); nuclei were 1 to 1.5 times red blood cell diameter, round to indented, with condensed chromatin, and indistinct nucleoli. The cells had scant to moderate, homogeneous, eosinophilic cytoplasm. The mitotic rate for DSBCL varied but in general was moderate to high (5–10 mitotic figures per single high-powered field, 400×). Median MC was 61 (range 23–196) per 2.4 mm2 (equivalent to 12 FN 20/40× fields). Ki-67 immunohistochemistry was performed in 2 cases (MC 66 and 61). The percentage of neoplastic cells that expressed Ki-67 was 30% to 40% in one case and 60% to 70% in the other case. In both cases, Ki-67 expression was disseminated across the lymph node with minor patchiness; there were no overt proliferation centers evident by HE staining or with Ki-67 immunohistochemistry (Figs. 9, 10). The neoplastic cells showed diffuse immunoreactivity for PAX5, and there was evenly dispersed scattered T cells with cytoplasmic immunoreactivity for CD3 (non-neoplastic T cells; data not shown). Additionally, there were frequent histiocytes containing globular eosinophilic cytoplasmic material (Figs. 7, 8). Moderate numbers of tingible-body macrophages were often present. When the capsule was present it was consistently invaded with the small-to-intermediate sized neoplastic lymphocytes.
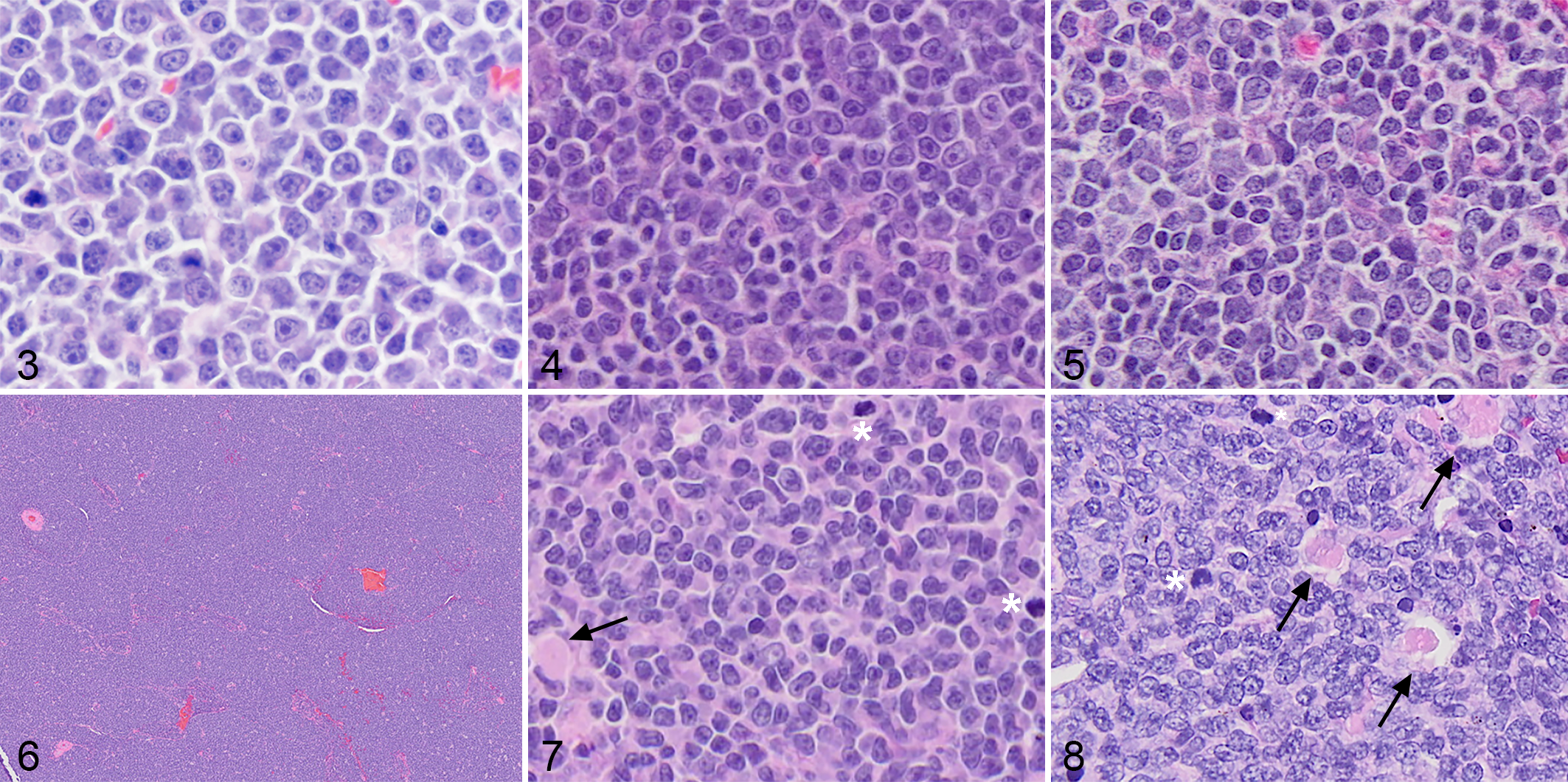
B-cell lymphoma, lymph node, dog.
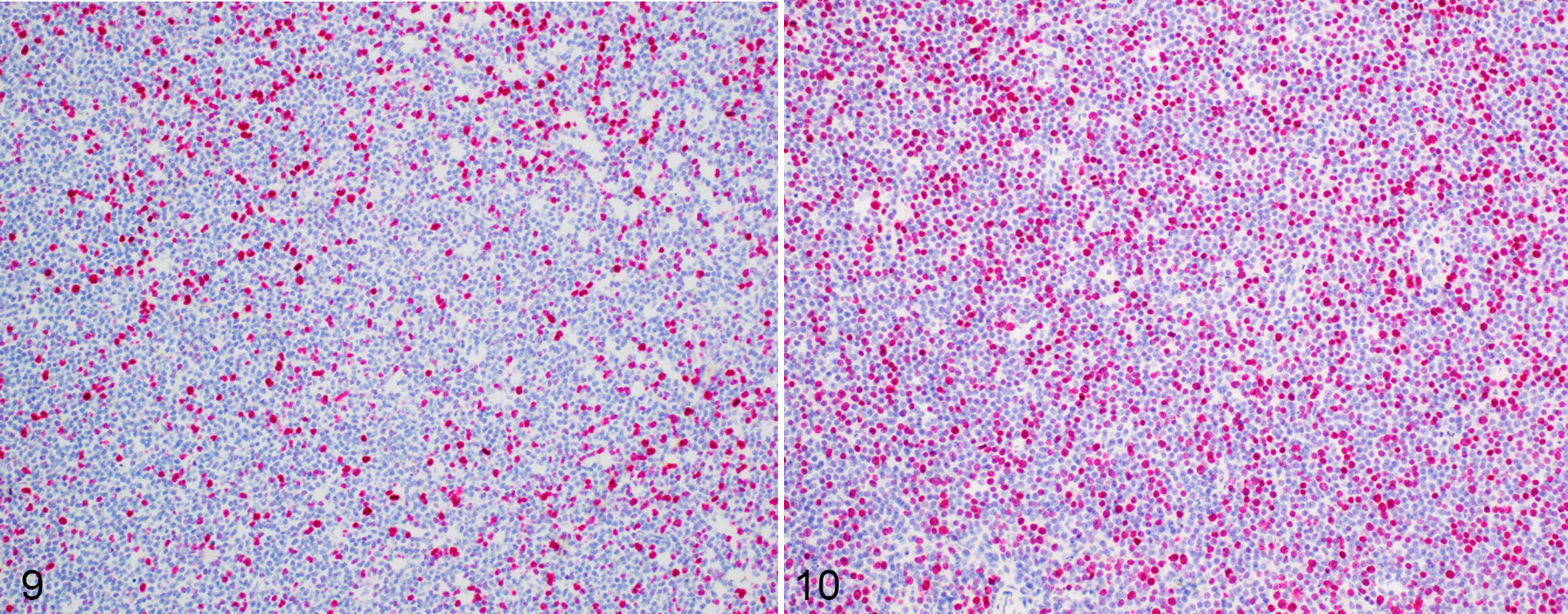
Diffuse small B-cell lymphoma (DSBCL), lymph node, dog. Immunohistochemistry for Ki-67.
The 2 cases diagnosed as DLBCL that were “small” by flow cytometry, had a diffuse pattern, and neoplastic cells were overall considered large with prominent nucleoli. These cases were diagnosed as DLBCL because at least 10% of the neoplastic cells had nuclei >2× the size of a red blood cells. Both cases also had areas containing smaller lymphocytes.
Patient Characteristics
The signalment and clinical presentation of DSBCL cases are summarized in Table 2 and compared to the 41 cases of DLBCL (cohort 1). The median age for DLBCL was 9 years with a range of 4 to 13. The median age for dogs with DSBCL was 11 years (range of 7–14), which was significantly different (P = .0039) from DLBCL (cohort 1 only). The most common breeds in the small B-cell subtype were a mix of small and large breed dogs; breeds representing more than one case of DSBCL dogs included English bulldog (4/27), West Highland white terrier (2/24), Shih Tzu (2/24), and Labrador retriever (2/24). The majority of the dogs were mixed breed (11/27). Lymphocytosis was significantly more common in DSBCL (cohort 2) compared with DLBCL (cohort 1) (27.3% vs 2.9%, respectively; P = .017). None of the other parameters evaluated (anemia, hyperglobulinemia, age) were different between subtypes (Table 2).
Patient Characteristics for Dogs With Small B-Cell Lymphomas (Cohort 2) Compared to Dogs With Diffuse Large B-Cell Lymphoma (Cohort 1).
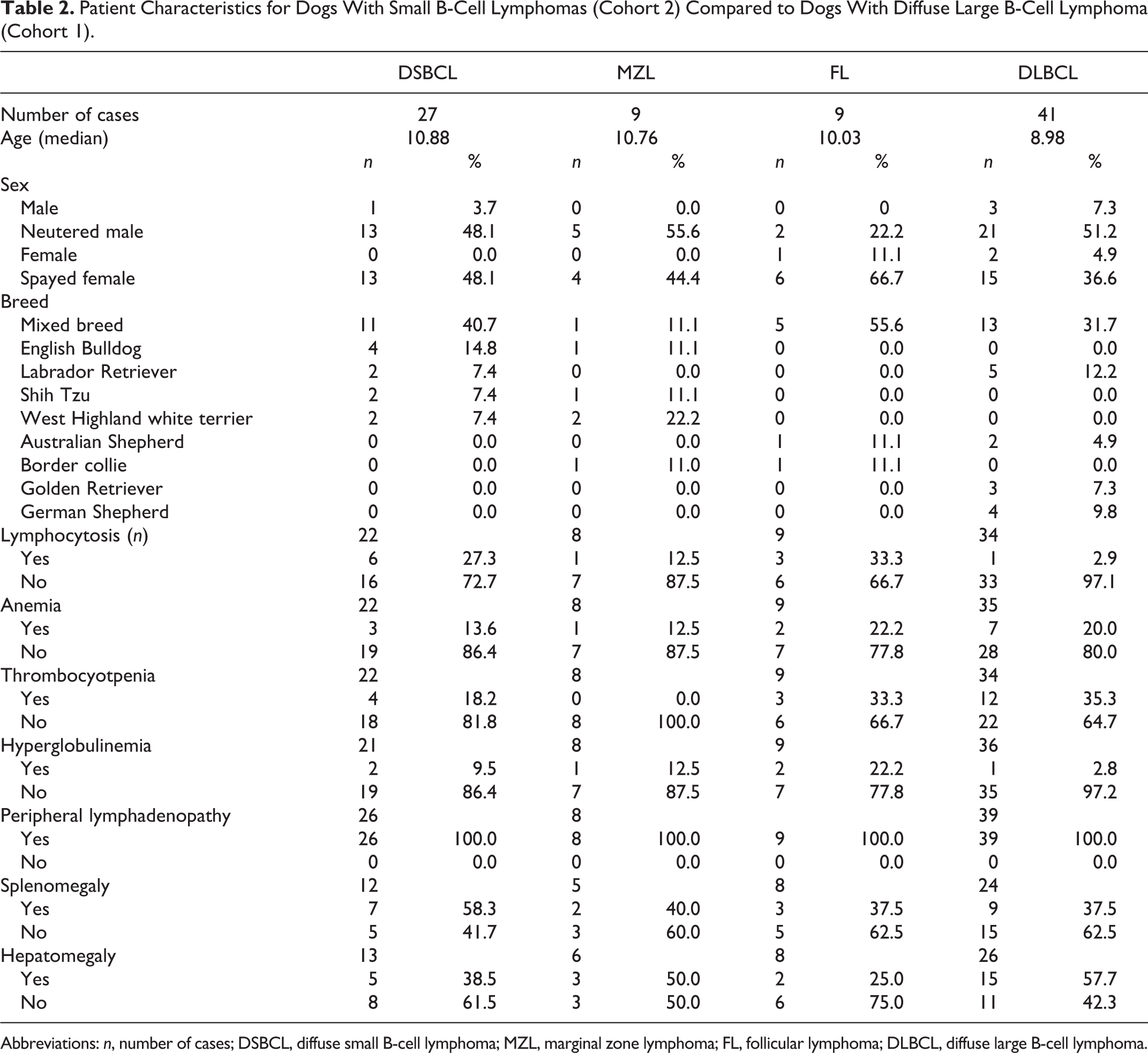
Abbreviations: n, number of cases; DSBCL, diffuse small B-cell lymphoma; MZL, marginal zone lymphoma; FL, follicular lymphoma; DLBCL, diffuse large B-cell lymphoma.
Flow Cytometric Features
The size as determined by FS for all subtypes and normal B cells is summarized in Figure 11. The median FS of the B-cell population for DSBCL, FL, and MZL was 379, 364, and 396, respectively (cohort 2), whereas the median FS of the B-cell population for DLBCL was 660 (cohort 1 only). It should be noted that the range in size of FL and MZL may be falsely low since we selectively included cases with FS <460; therefore, FL and MZL may have a wider FS size range than identified in this study. Similarly, clinical progression in these subtypes might also be biased due to cell size selection, and clinical follow-up was not pursued for FL or MZL. As a size comparison, the mean forward scatter of neutrophils by flow cytometry in the peripheral blood of dogs with a lymphocytosis was 620 (SD 61). This indicates that by flow cytometry dogs with histologically confirmed DLBCL, on average, had neoplastic B cells larger than a neutrophil, whereas dogs with small B-cell lymphomas identified by flow cytometry, had neoplastic B cells that were smaller than the size of an average neutrophil (0.74× the size of a neutrophil) based on FS. There was no significant difference in the size of any of the small B-cell subtypes nor were there significant differences in expression of CD21, class II MHC or CD25 between the small B-cell subtypes. There were also no significant differences in the % of normal T cells in any of the subtypes of small B-cell lymphoma. Therefore, flow cytometry was unable to differentiate the small B-cell subtypes identified by histopathology.
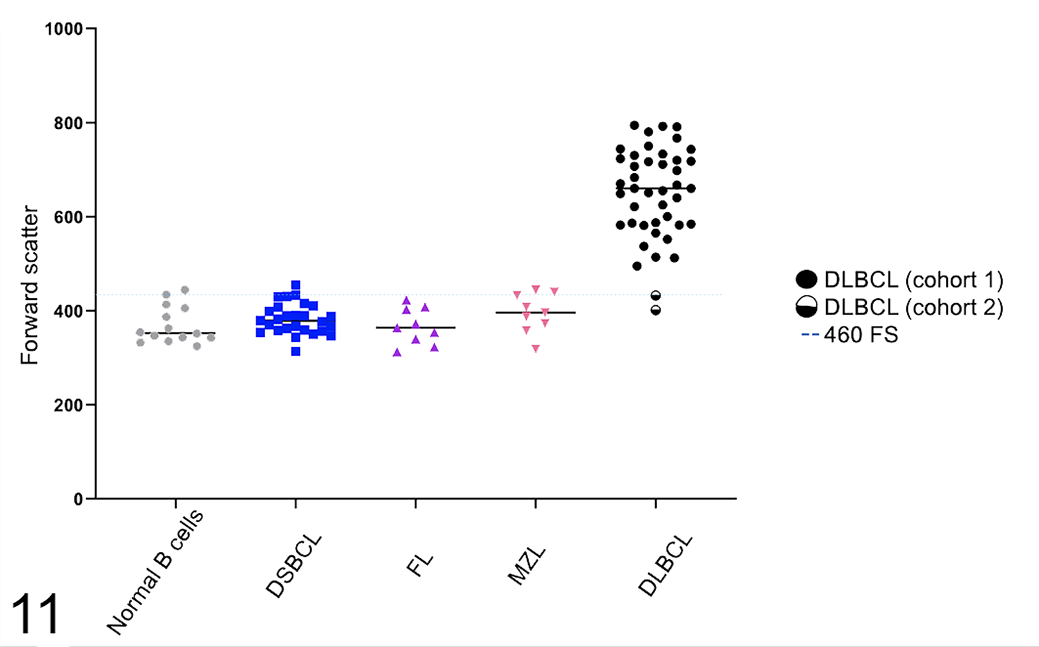
Flow cytometric cell size (FS) for B-cell subtypes of canine nodal lymphoma subtypes. Cell size determination is based on forward scatter characteristics. The size of confirmed DLBCLs (black circle) was significantly greater than that of all small B-cell subtypes (FL, MZL, DSBCL) and normal B cells. The horizontal line shows the cutpoint of 460 for determination of small size.
Cytologic Features of Small B-Cell Lymphomas
DLBCL is characterized by an expansion of intermediate to large-sized lymphocytes that are generally 15 to 20 µm in diameter with a round nucleus, dispersed chromatin, 1 to 3 prominent nucleoli, and mildly expanded deeply basophilic cytoplasm (Fig. 12, cohort 1).
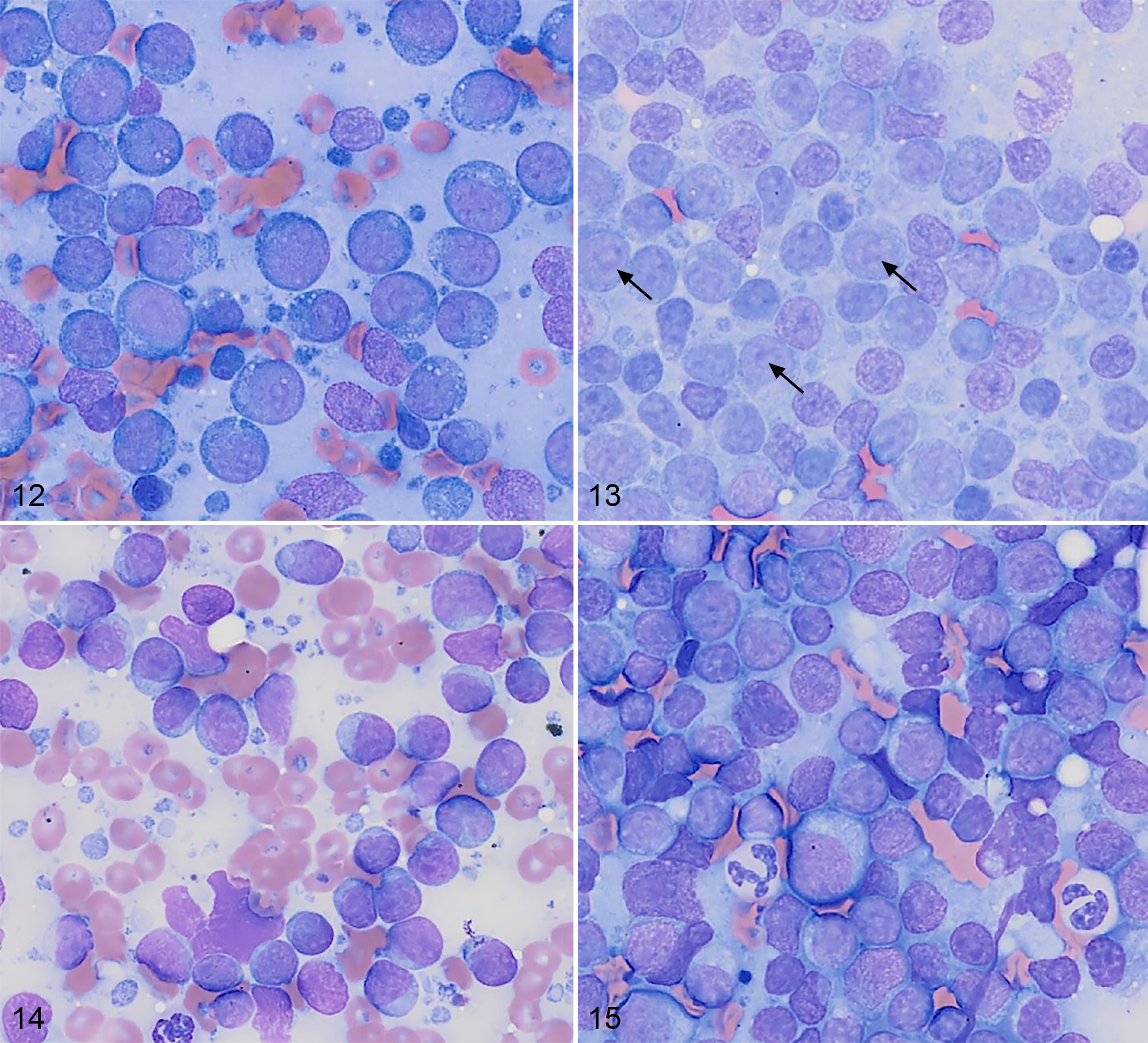
B-cell lymphoma, lymph node, dog. Cytologic preparations of fine-needle aspirates. Wright Giemsa
MZL samples had an expanded population of intermediate-sized lymphocytes, approximately 15 µm in diameter with mild anisocytosis, a round nucleus, fine chromatin, often one large central nucleolus, and moderately expanded pale blue cytoplasm (Fig. 13, cohort 2). The MZL samples examined in this study displayed heterogeneity, with some regions of the sample having small to moderate numbers of residual small lymphocytes.
Four DSBCL cases were reviewed cytologically (Figs. 14, 15, cohort 2). These cases had similar cytomorphology, and the samples appeared largely effaced by neoplastic lymphocytes. These lymphocytes were generally small-to-intermediate in size, with the majority of cells 12 to 15 µm in diameter, and similar in size to slightly smaller than a neutrophil. While overall anisocytosis and anisokaryosis appeared mild, small numbers of larger cells (15–20 µm in diameter) were present. Nuclei were round to slightly indented with coarse chromatin and generally lacked prominent nucleoli. However, small numbers of cells had a large central nucleolus, similar to MZL morphology, and in cases where these cells were expanded, some fields had an MZL appearance. DSBCL lymphocytes had mildly to moderately expanded lightly basophilic cytoplasm. The cells had a mitotic count ranging from 0 to 3 mitotic figures per five 500× magnification fields. There were small to moderate numbers of cytophagic macrophages and small numbers of residual small mature lymphocytes.
Clinical Outcome for Diffuse Small B-Cell Lymphoma
Clinical outcome was evaluated in 22 patients with DSBCL. Dogs were treated with a variety of chemotherapy protocols and many with more than one protocol. Six dogs were treated with only prednisone with or without chlorambucil, and the remaining 16 dogs were treated with a multi-agent chemotherapy protocol.
The median lymphoma-specific survival time (LSST) for DSBCL from time of diagnosis was 105 days for 11 dogs. The median overall survival for dogs (n = 22) with DSBCL was 140 days, range 2 to 464 days (Fig. 16). The treatment protocols were too variable to compare treatment with survival. Response and survival time are summarized in Table 3.
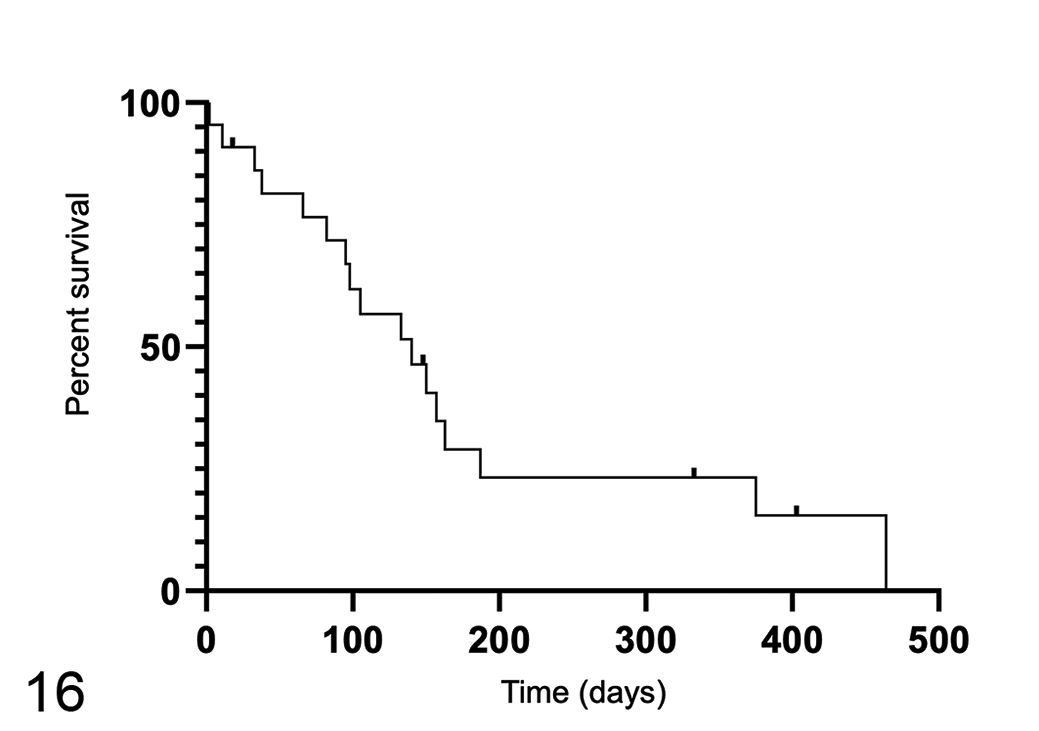
The overall survival times for 22 dogs with diffuse small B-cell lymphoma. The median overall survival for dogs with diffuse small B-cell lymphoma is 140 days. Ticks indicate censored data.
Response of 22 Dogs With Diffuse Small B-Cell Lymphoma Treated With Prednisone and Chlorambucil Versus Multi-Agent Chemotherapy.
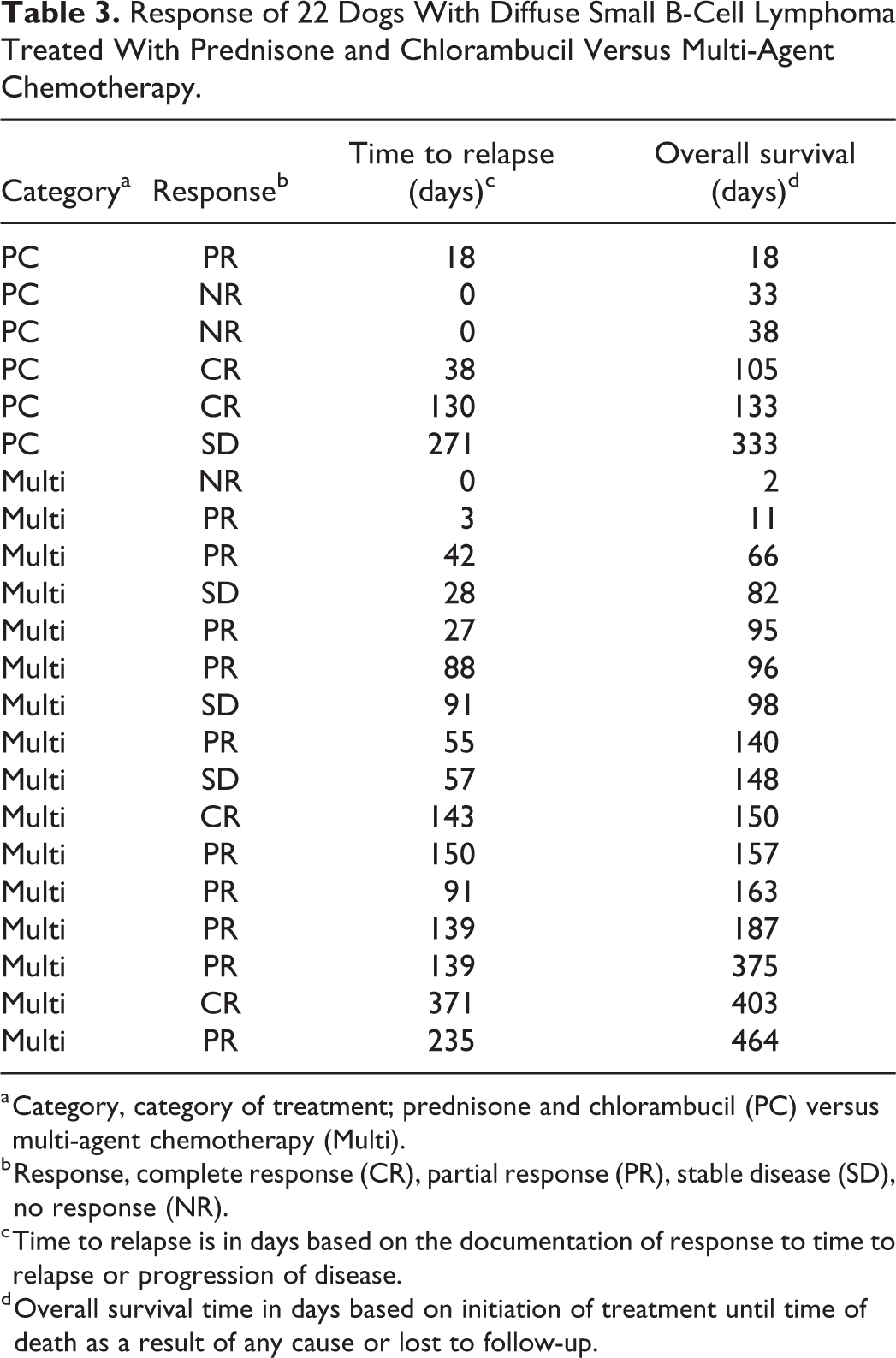
a Category, category of treatment; prednisone and chlorambucil (PC) versus multi-agent chemotherapy (Multi).
b Response, complete response (CR), partial response (PR), stable disease (SD), no response (NR).
c Time to relapse is in days based on the documentation of response to time to relapse or progression of disease.
d Overall survival time in days based on initiation of treatment until time of death as a result of any cause or lost to follow-up.
There were 4 dogs with a complete response to treatment with a duration of 38, 130, 143, and 371 days. Two of those dogs were treated with CHOP multi-agent chemotherapy. The first dog received CHOP which was adjusted to COP because of renal failure with a response of 143 days and overall survival 150 days. The other dog completed the CHOP protocol with a response of 371 days and overall survival of 403 days. There was one dog that began with prednisone and chlorambucil and had a complete response for 38 days but then was adjusted to doxorubicin when she was no longer responding to prednisone/chlorambucil alone. This dog had an overall survival of 105 days. The last dog with a complete response was treated with prednisone and chlorambucil alone with a response of 130 days and overall survival of 133 days. There were 4 dogs with a survival time of over 300 days (range 333–464). Two of these dogs were English bulldogs, one was a Shih Tzu, and the last dog was a mixed breed. A mild lymphocytosis developed in the mixed breed dog 2 months after diagnosis (9100 lymphocytes/μl) and this dog was treated with prednisone. The other dogs were treated with multi-agent chemotherapy and lymphocytosis never developed.
None of the parameters examined (mitotic count, B-cell size, MFI of CD21, MHC class II, CD25 expression) were correlated with response time or survival. Interestingly, 5/22 dogs had documented rectal prolapse or cobblestone appearance to the rectum. Rectal involvement is possible in these cases; however, histopathology was not performed in any case to confirm.
Discussion
We have described the clinical presentation and flow cytometric features for a unique, consistent subtype of B-cell lymphoma with a diffuse growth pattern and small-to-intermediate sized B cells. We have provisionally designated this entity diffuse small B-cell lymphoma (DSBCL).
DSBCL differs in several ways from the most common form of B-cell lymphoma, DLBCL. Histologically, DLBCL is characterized by large B cells (nuclei > 2× RBC) with a diffuse growth pattern and a prominent nucleolus or multiple nucleoli. This size difference is also observed by flow cytometry. The average overall cell size of DLBCL, as reflected in the FS value, is similar to or greater than that of a neutrophil, whereas the median FS value for DSBCL is 0.74× that of a neutrophil. Similarly, by histopathology, DSBCL were small to intermediate sized cells (nuclei = 1–1.5 × RBC) and nucleoli were indistinct. Clinical characteristics that differed between the 2 subtypes include an older average age seen in DSBCL and a significantly higher proportion of cases presenting with lymphocytosis in DSBCL; lymphoyctosis was present in 27% of dogs with DSBCL compared to 3% in DLBCL. DSBCL may have a poorer outcome than DLBCL, based on the reported survival times of the latter disease, 4,11,15,33 but a larger follow-up study would be required to thoroughly assess this possibility.
Given the diffuse growth pattern, DSBCL is histologically distinct from nodular small cell lymphomas, FL, MZL, and splenic MCL, which have been described in both dogs and humans. DSBCL cellular morphologic features overlap most closely with human CLL/SLL and diffuse MCL. We therefore considered whether DSBCL was consistent with either of these lymphoma subtypes.
In humans, MCL is often disseminated with an aggressive clinical behavior and diffuse histologic pattern. 1,14 Human MCL has several described variants including blastoid, small cell, marginal zone-like, and pleomorphic, making the distinction between small mature B-cell subtypes challenging. Human SLL is also characterized by a diffuse pattern. It can have areas of pallor giving a pseudofollicular pattern. The areas of pallor are proliferation centers with larger cells consisting of prolymphocytes and paraimmunoblasts with prominent nucleoli. 17 Another typical feature of SLL in humans is a low mitotic count; however, histologically aggressive SLLs do occur in humans and are recognized by large confluent proliferation centers and >2.4 mitoses within the proliferation centers. 8 SLL cells typically have a round nucleus with clumped chromatin and indistinct nucleoli. SLL can also have cleaved, folded, or plasmacytic morphology making MCL a prominent differential. 19
In this study, DSBCL was composed of monotonous cells with a diffuse pattern. In 2 cases with Ki-67 evaluation there was disseminated expression lacking evidence of distinct proliferation centers. Similarly, areas of pallor or a pseudofollicular pattern were not apparent in DSBCL cases. The mitotic count in the DSBCL tumors in this study was moderate to high (median MC of 61 within a 2.37 mm2 standardized area). Furthermore, the overall median survival time and LSST of dogs with DSBCL was 140 and 105 days, respectively, suggesting an aggressive clinical course. Therefore, in many respects, DSBCL is more similar to human MCL than to CLL/SLL. We cannot draw this conclusion definitively, however, without additional diagnostic information. In humans, identification of the translocation of cyclin D1 [t(11;14)(q13;q32)] is often the defining feature of MCL diagnosis. 25 Cyclin D expression by immunohistochemistry has been unsuccessful in other reports of MCL in dogs, and may not be a relevant marker for canine MCL. 20,24,29 Furthermore, MCL and SLL in humans express CD5, and can often be distinguished by differential expression of a variety of proteins. These include expression of CD23 and CD10 in SLL but not MCL. 25 DSBCL cells do not express CD5, and the expression of other antigens is either not possible because of a lack of antibodies, or has not been performed yet. Therefore, we leave open the possibility that DSBCL is a unique canine entity, which requires further characterization using gene expression profiling and mutational analysis. This entity may be similar to what has previous been described in dogs as small B-cell lymphoma–not otherwise specified, which also showed a mitotic activity that was suggestive of aggressive disease. 16
Cell size, as approximated by forward light scatter measured by flow cytometry, was useful for discovering these rare forms of small cell B-cell lymphoma. Ninety-six percent of the cases with B cells below an FS value of 460 (0.74× the FS of an average neutrophil) were histologically described as DSBCL, FL, or MZL. However, the remaining 4% were diagnosed with DLBCL by histopathology, meaning that flow cytometry is an imperfect tool for ruling out DLBCL. Importantly, no immunophenotypic features, including expression of class II MHC, CD21, CD5, and CD25 could be used to distinguish any of the B-cell lymphoma subtypes. A prior study from our laboratory reported the same finding. 33 Therefore, at present, histopathology is necessary for subclassification of small/intermediate sized B-cell lymphomas.
It is a common understanding among veterinary oncologists that “small cell B-cell lymphoma,” often characterized as such by cytology, is an indolent disease, and this understanding affects treatment. Our findings here should change these assumptions. All measurements of size (cytology, histopathology, flow cytometry) described DSBCL cells as small, but overall survival was 140 days in dogs in this study, which is inconsistent with an indolent clinical course.
Conclusions
Diffuse small B-cell lymphoma is a distinct entity from other lymphoma subtypes described in the dog. It exhibits a consistent and unique histologic pattern and cellular morphology which is not easily correlated with an already established WHO classification lymphoma subtype. The disease appears to have an aggressive clinical course. Because it cannot be distinguished from other B-cell lymphomas by flow cytometry features, histopathology is required for a definitive diagnosis.
Footnotes
Acknowledgements
We would like to thank Aratana Therapeutics for collaboration in obtaining the diagnostic samples used for cohort 1 in this study, and John Peauroi and VDx Veterinary Diagnostics and Preclinical studies for performing the original histology and immunohistochemistry for cohort 1. We would also like to thank Todd Bass for performing histology and immunohistochemistry on samples from cohort 2.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
