Abstract
T-zone lymphoma (TZL) is an indolent, nodal lymphoma that has been clinically characterized in detail in dogs, and T-zone hyperplasia (TZH) is a hyperplastic change in lymph nodes associated with antigen processing. In some cases, histopathological features of TZL and TZH are similar, and are difficult to differentiate by morphology alone. Since there have been few publications characterizing their immunohistochemical profiles, histological, immunohistochemical, and clonality examinations were performed using formalin-fixed paraffin-embedded samples of canine lymph nodes with TZL (14 cases) and canine lymph nodes with TZH associated with nonlymphocytic tumors (10 cases). Immunohistochemically, small- to medium-sized lymphocytes of TZL were immunopositive for CD3, CD5, and HLA-DR, and negative for CD45, FOXP3, and granzyme B (GRB) in all cases. Among these 14 cases, 11 were immunopositive for CD8 and 1 was CD20 positive. Paracortical lymphocytes in TZH were diffusely immunopositive for CD3, CD5, and CD45, with scattered immunopositivity for CD8, HLA-DR, FOXP3, and GRB, and negative for CD20 in all cases. A clonal TCR gene rearrangement was detected in 13/14 TZL and none of the TZH cases. The present study revealed that TZL is a clonal proliferation of monomorphic CD8+CD45-GRB- T cells, while TZH consists of an immunophenotypically heterogenous population of CD45+ T cells that are variably positive for CD8 and FOXP3. These results suggest that canine TZL is a clonal proliferation of naïve or premature cytotoxic T cells. Regarding TZH, variable immunopositivity for cytotoxic and regulatory T-cell antigens may reflect immune responses to a variety of regional neoplastic lesions.
Keywords
T-zone lymphoma (TZL), a histological variant of peripheral T-cell lymphoma (PTCL), is an indolent, nodal lymphoma that has been clinically characterized in detail in dogs.21,22 The diagnosis of TZL is primarily based on histopathology; a paracortical proliferation of neoplastic T cells causing compression of lymphoid follicles is a characteristic findings of TZL. Previous studies using flow cytometry or immunohistochemistry revealed that a characteristic immunophenotype of TZL is an absence of CD45, a pan leukocyte antigen.6,8,18,19 While CD45 negativity in flow cytometry has been proposed as one of the diagnostic criteria of TZL, flow cytometric immunophenotyping lacks morphological and architectural information and its ability to classify and characterize lymphomas is limited. Previous studies reported the expression of CD45 in a subset of TZL and its absence in a high-grade PTCL case, suggesting the potential for a misdiagnosis of TZL in the absence of histopathological examination.13,19 In a study using flow cytometry, TZL expressed higher levels of CD21, class II major histocompatibility complex (MHC), and CD25. 18 T-cell subsets were heterogeneous; CD4+, CD8+, and CD4-CD8- subsets were detected. 18 Limited information is currently available on its immunohistochemical profiles. Therefore, immunophenotyping combined with morphological and architectural features is needed to subtype lymphomas and confirm the diagnosis of TZL.
T-zone hyperplasia (TZH) is a normal reaction of the paracortex in the lymph nodes, in which naïve T cells are stimulated by dendritic cells or antigen-presenting cells that have phagocytosed antigen and traveled to the lymph node sinuses. Stimulated naïve T cells proliferate and mature to become effector T cells, and these effector T cells enter the systemic circulation. Therefore, TZH usually consists of variably sized lymphocytes and dendritic cells, while TZL consists of monomorphic lymphocytes. Since the histopathological features of TZH and TZL are difficult to distinguish from one another in some cases, immunohistochemistry and clonality analysis are needed for an accurate diagnosis.21,22 As TZH is considered a non-specific reactive response to other disease processes, immunophenotypic information on TZH is valuable for understanding immune responses to the disease. However, the immunohistochemical features of T cells in the paracortex of TZH remain unclear. To obtain a more detailed understanding of the pathology of T-zone proliferative lesions, the immunophenotypes of T cells that proliferate in both TZL and TZH need to be identified. Therefore, the aim of the present study was to elucidate and compare the immunohistochemical profiles of TZL and TZH, with a focus on T-cell subsets, using formalin-fixed, paraffin-embedded (FFPE) tissues.
Materials and Methods
Case Selection
Canine cases diagnosed as TZL or TZH were retrieved from the database of the Laboratory of Veterinary Pathology, Graduate School of Agricultural and Life Sciences, the University of Tokyo between 2012 and 2021. Eighteen TZL cases were identified, and 4 were excluded due to difficulties with detailed immunophenotyping because of poor sample quality; therefore, 14 cases were ultimately examined in the present study. For TZH, 63 cases were identified, and the 10 most recently diagnosed cases were included, all of which were regional lymph nodes resected together with a neoplastic lesion, including liposarcoma, mammary adenoma, mammary benign mixed tumor, mammary carcinoma, melanoma, osteosarcoma, and squamous cell carcinoma. Metastatic neoplasia was not detected in the lymph nodes.
Histopathology
All samples were fixed in 10% neutral buffered formalin solution, processed routinely, and embedded in paraffin wax. Four-micrometer-thick paraffin sections were deparaffinized, rehydrated through a graded series of alcohol, and stained with hematoxylin and eosin. The mitotic count was assessed by manually counting the number of mitotic figures in 10 consecutive high-power fields (HPF; 1.96 mm2) from a randomly selected field using a microscope with a 40× objective and ocular of FN 20 mm, and then multiplying by 1.17. 9 The maximum number of mitotic figures per single HPF (0.196 mm2) was also evaluated by examining the whole slides manually.
Immunohistochemistry
Details on primary antibodies and antigen retrieval procedures are summarized in Table 1. Following antigen retrieval, endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. Sections were blocked with 8% skim milk in Tris-buffered saline (TBS) at 37°C for 40 minutes, and then incubated with the primary antibodies at 4°C overnight. After washing with TBS, each secondary antibody was applied. In the Dako Envision+ System (Dako, Tokyo, Japan), sections were incubated with an anti-rabbit or anti-mouse hydrogen peroxidase-labeled secondary antibody polymer at 37°C for 40 minutes. In the Ventana OptiView DAB Universal Kit (Ventana Medical Systems, Tucson, USA), sections were incubated with Linker-HQ at 37°C for 8 minutes and then incubated with Multimer-HRP at 37°C for 8 minutes. To detect CD8, 17 sections were incubated with biotin-conjugated anti-rat immunoglobulin G (1:500; Dako) at 37°C for 40 minutes and then with horseradish peroxidase-conjugated streptavidin (1:400; Dako) at 37°C for 40 minutes. Labeled complexes were visualized with 3,3’ -diaminobenzidine chromogen and 0.03% hydrogen peroxide in Tris-HCl buffer and then counterstained with hematoxylin. Canine activated lymph nodes were used as positive and negative tissue controls, and negative antibody controls were performed by applying an irrelevant antibody and buffer instead of the primary antibodies. 15
Antibodies and methods used for immunohistochemistry.
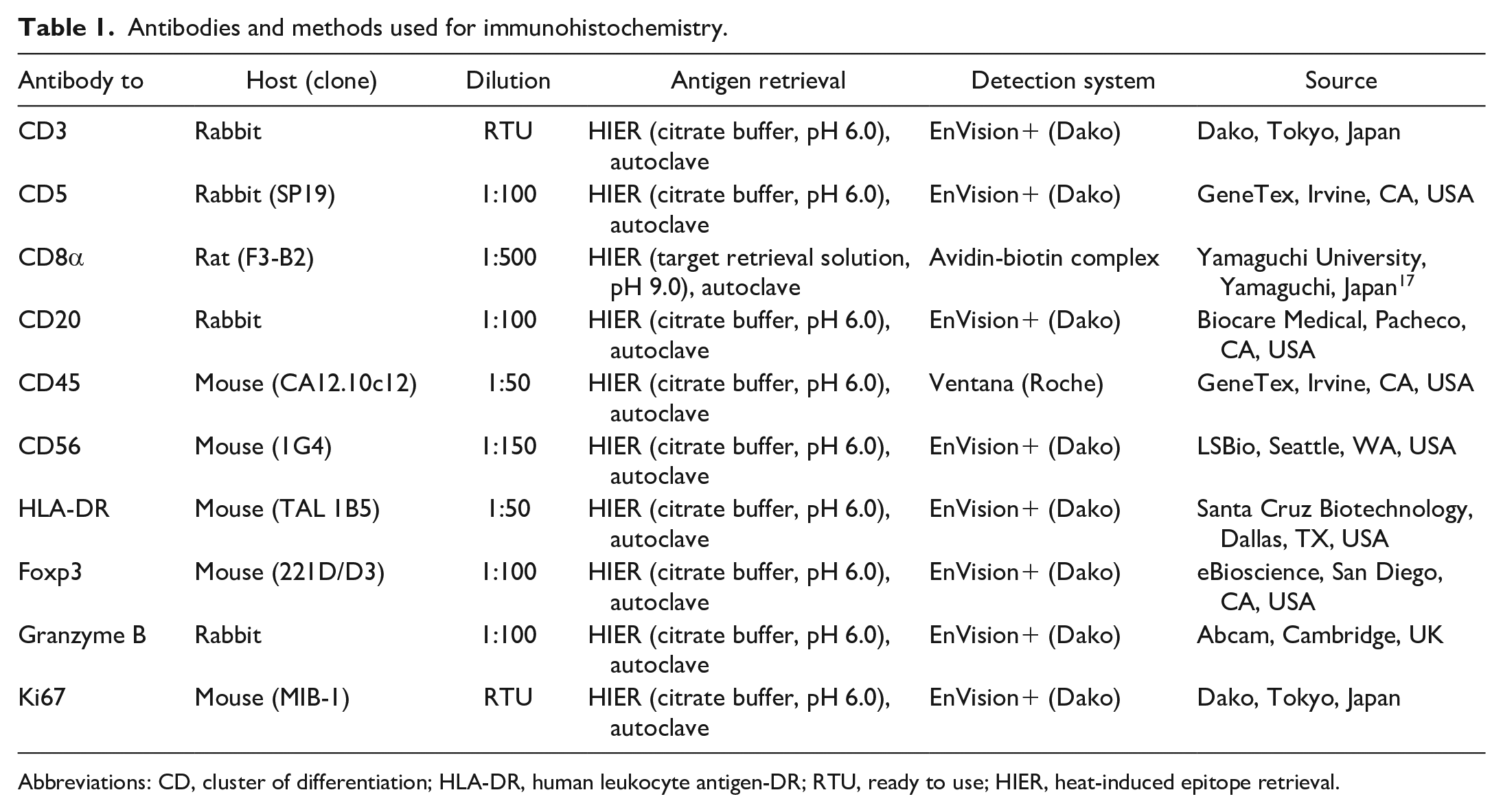
Abbreviations: CD, cluster of differentiation; HLA-DR, human leukocyte antigen-DR; RTU, ready to use; HIER, heat-induced epitope retrieval.
Immunohistochemical staining was evaluated by 2 veterinary pathologists accredited by the Japanese College of Veterinary Pathologists (KU and JKC). All slides were evaluated in the paracortical areas with the highest immunopositivity using a semiquantitative assessment (<5%, 5%-24%, 25%-49%, 50%-74%, and 75%-100% positive cells). Cases were excluded from evaluations if the internal positive controls were negative. TZL cases were considered to be positive when more than 75% of neoplastic cells were positive. Ki67 labeling was examined at a medium magnification to select paracortical areas with the strongest proliferative activity. Within these areas, 5 fields at a high magnification (40× objective, 0.04 mm2) were photographed. The numbers of Ki67-positive and Ki67-negative nuclei were counted in each image using ImageJ software (National Institutes of Health, Bethesda, MD, USA), and the Ki67 index was calculated as the mean percentage of Ki67-positive nuclei. Six cases of TZL were excluded from the Ki67 index assessment because the Ki67 index of these cases was 0%, possibly due to poor sample quality such as suboptimal fixation or oxidation of tissues.
Clonality Analysis
To detect the clonal rearrangement of TCRγ genes, multiplex PCR analyses of all cases were performed. Genomic DNA was extracted from FFPE tissues using the QIAamp DNA FFPE Tissue Kit (Qiagen, Venlo, The Netherlands). Previously reported primer sequences and PCR conditions for the TCRγ genes were used.5,24 PCR products were analyzed using 5% to 20% gradient polyacrylamide gel electrophoresis (ATTO, Tokyo, Japan), and PCR analyses were run in duplicate.
Diagnostic Criteria
All cases were reviewed, and final diagnoses were made based on histopathological examination and immunohistochemistry for CD3 by 2 veterinary pathologists (KU and JKC), separately. The diagnosis of TZL was based on the paracortical proliferation of monomorphic T cells with compression of lymphoid follicles and immunohistochemical positivity for CD3.21,22 Positive clonality analysis is one of the distinguishing features of TZL, but one case which was negative for clonality was also diagnosed as TZL because it had histological and immunohistochemical features consistent with TZL. The diagnosis of TZH was based on expansion of the paracortex with a heterogeneous population of large and small lymphocytes, and immunohistochemistry for CD3 to confirm the mixture of T cells and dendritic cells.
Results
The age, sex, breed, and location of the lymph nodes in each case are summarized in Supplemental Table S1. Histopathological, immunohistochemical, and clonality results are summarized in Table 2.
Histopathological, immunohistochemical and clonality results of dogs with T-zone lymphoma and T-zone hyperplasia.
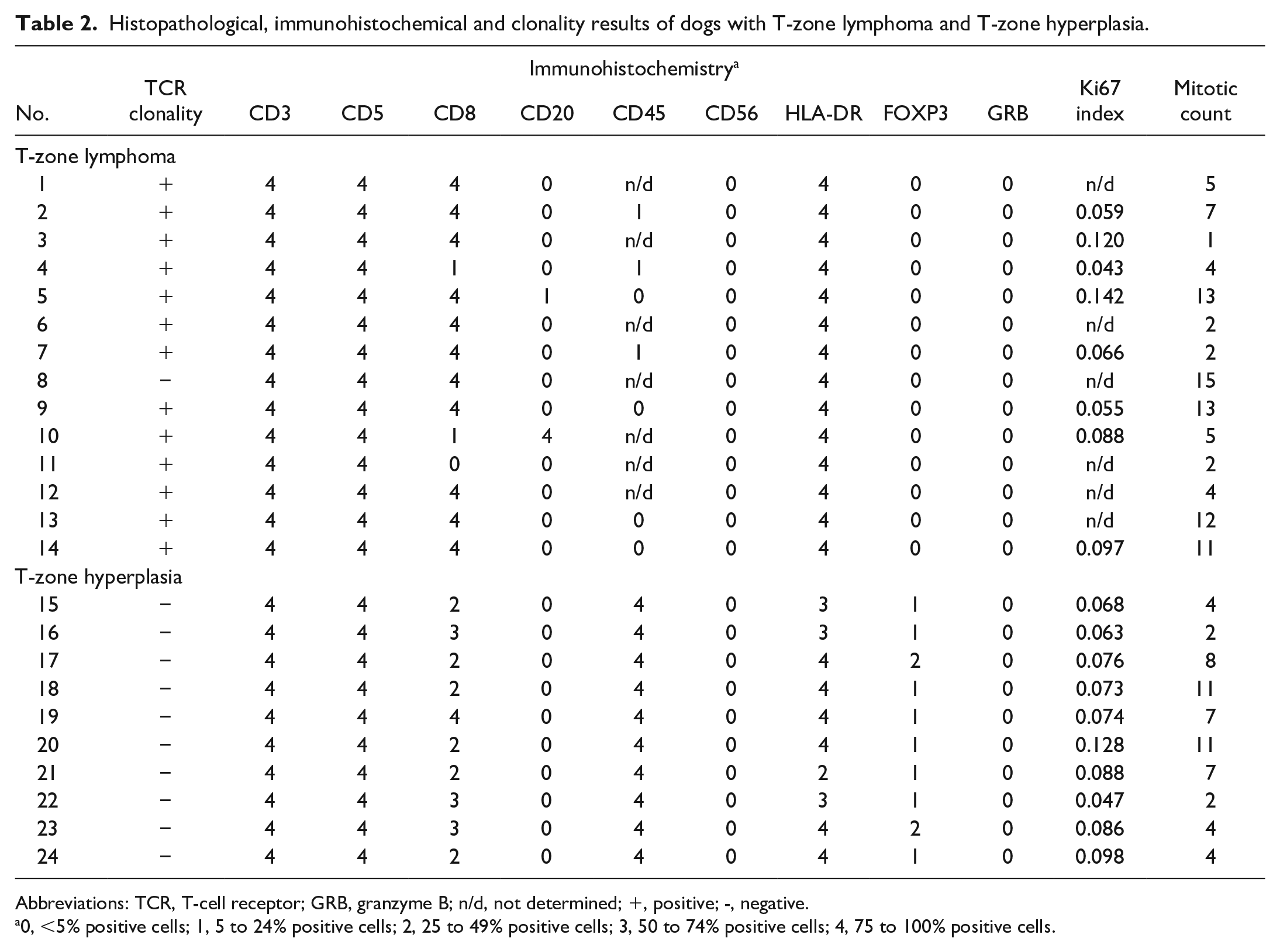
Abbreviations: TCR, T-cell receptor; GRB, granzyme B; n/d, not determined; +, positive; -, negative.
0, <5% positive cells; 1, 5 to 24% positive cells; 2, 25 to 49% positive cells; 3, 50 to 74% positive cells; 4, 75 to 100% positive cells.
Histopathology
TZL
Histopathologically, TZL cases were characterized by diffuse proliferation of small to medium-sized (1 to 1.5 × red blood cells), monomorphic lymphocytes in the paracortical area with compression of lymphoid follicles (Fig. 1). These lymphocytes had a moderate amount of lightly stained cytoplasm and ovoid, densely stained nuclei. The maximum number of mitotic figures per single HPF ranged from 1 to 4. The mitotic count ranged between 1 and 15 (average: 6.8). A small number of lymphoid follicles with germinal centers were noted in 7/14 cases. The medullary sinuses were mildly to severely dilated and filled with a moderate number of small- to medium-sized lymphocytes, red blood cells, and/or macrophages in 11/14 cases.
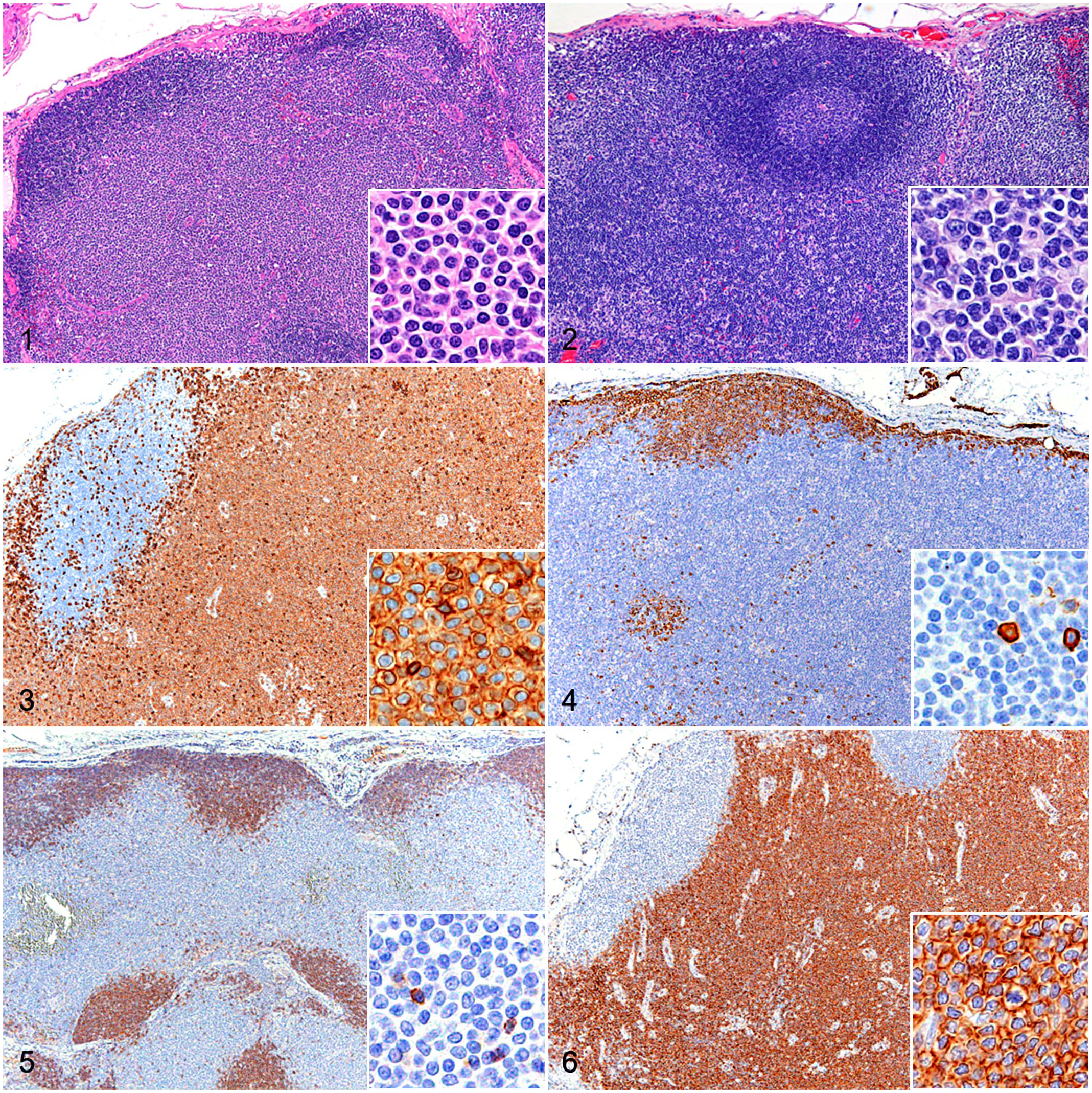
Lymph node, dog. Insets: higher magnification. Hematoxylin and eosin. Figure 1 . T-zone lymphoma (TZL). Paracortical proliferation of neoplastic lymphocytes with the compression of lymphoid follicles. Neoplastic lymphocytes have lightly stained cytoplasm with small, densely stained nuclei. Figure 2. T-zone hyperplasia. Expansion of the paracortex surrounding lymphoid follicles with a distinct germinal center. Paracortical lymphocytes have nuclei of various sizes. Figures 3–6. TZL, lymph node, dog. Insets: higher magnification. Immunohistochemistry. Figure 3 . Diffuse, membranous, and cytoplasmic immunolabeling for CD3 in neoplastic lymphocytes. Figure 4 . Neoplastic lymphocytes are not immunolabeled for CD20. However, lymphocytes in lymphoid follicles and lymphocytes that are scattered in the paracortex have membranous immunolabeling for CD20. Figure 5 . Neoplastic lymphocytes are not immunolabeled for CD45. However, lymphocytes in lymphoid follicles and lymphocytes that are scattered in the paracortex have membranous immunolabeling for CD45. Figure 6 . Diffuse, membranous, and cytoplasmic immunolabeling for CD8 in neoplastic lymphocytes.
TZH
Paracortical lymphocytes were heterogenous and composed of small to large lymphocytes (Fig. 2). The maximum number of mitotic figures per single HPF was 1 or 2. The mitotic count ranged between 2 and 11 (average: 6.0). Lymphoid follicles with distinct germinal centers were noted in 6/10 cases, while compression of lymphoid follicles in some parts was observed in 3/10 cases. The medullary sinuses were mildly to severely dilated and filled with macrophages, neutrophils, eosinophils, and/or red blood cells in all cases.
Immunohistochemistry and Clonality of TCR Gene Rearrangement
TZL
Neoplastic lymphocytes were diffusely positive for CD3 (Fig. 3) and CD5 in all cases, negative for CD20 in 13/14 cases (Fig. 4) and weakly immunopositive for CD20 in 1 case, and negative for CD56 in all cases. For the evaluation of CD45, 7 cases were excluded due to the absence of labeling of positive internal controls, which was suspected to be due to poor tissue preservation. In all 7 of the remaining cases, neoplastic cells were negative (Fig. 5). Among these 14 cases, 11 were diffusely positive for CD8 (Fig. 6), and the remaining 3 were variably positive for CD8. All cases were diffusely positive for HLA-DR and negative for FOXP3 and granzyme B. The mean Ki67 index was 8.4%. A clonal TCR gene rearrangement was detected in 13/14 cases (93%).
TZH
In all cases, paracortical lymphocytes were diffusely positive for CD3 (Fig. 7), CD5, and CD45 (Fig. 8), and negative for CD20 and CD56. They were variably positive for CD8 (Fig. 9), HLA-DR, and FOXP3 (Fig. 10), with only a few scattered cells showing immunopositivity for granzyme B. The mean Ki67 index was 8.0%. In the clonality analysis, all cases were negative.
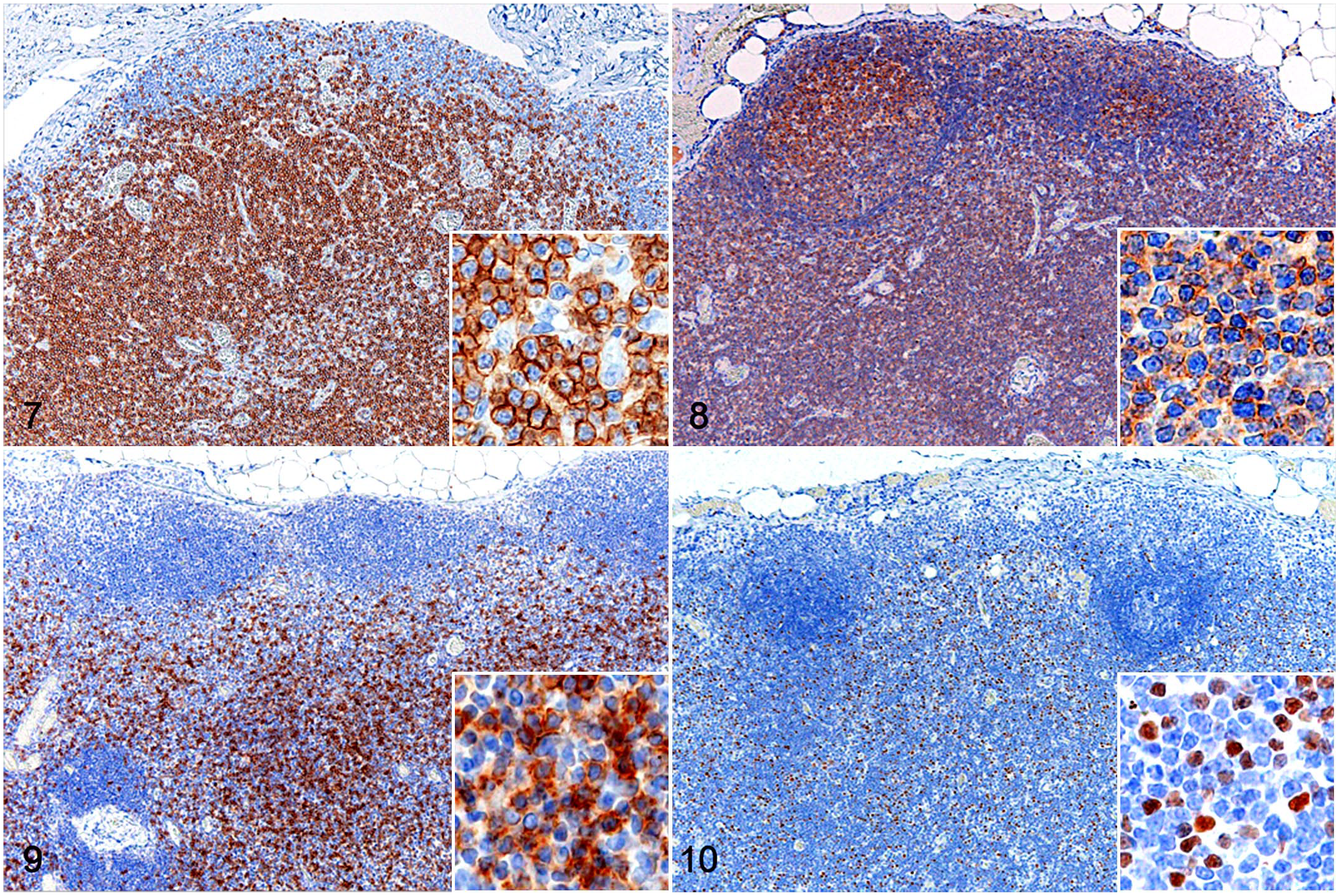
T-zone hyperplasia, lymph node, dog. Insets: higher magnification. Immunohistochemistry. Figure 7 . Diffuse, membranous, and cytoplasmic immunolabeling for CD3 in paracortical lymphocytes. Figure 8 . Diffuse membranous immunolabeling for CD45 in lymphocytes in lymphoid follicles and the paracortex. Figure 9 . Scattered membranous immunolabeling for CD8 in paracortical lymphocytes. Figure 10 . Scattered nuclear immunolabeling for FOXP3 in paracortical lymphocytes.
Discussion
Canine TZL is an indolent, nodal lymphoma that can be histologically difficult to distinguish from TZH. Recent studies revealed that the loss of CD45 expression is one of the immunophenotypic features of canine TZL.8,18,21,22 In the present study, all TZL cases were also negative for CD45, while paracortical lymphocytes in TZH cases were consistently immunopositive, indicating that immunohistochemistry for CD45 is a useful diagnostic modality that facilitates the histopathological discrimination of TZL and TZH. However, a previous study reported 2 cases of CD45+ TZL 19 ; thus, it is important to make a diagnosis of TZL comprehensively based on histopathological examination, immunohistochemistry, and, if needed, clonality analysis. In addition, the present study revealed the diffuse proliferation of CD8+ T cells in canine TZL and frequent appearance of FOXP3+ T cells in canine TZH, suggesting that these immunohistochemical features may be also helpful to distinguish TZL and TZH. Ki67 index did not differ significantly between TZL and TZH.
The results suggest that clonality analysis is a useful method for differentiating TZL from TZH. However, one TZL case was negative for clonality, possibly due to poor sample quality as immunohistochemistry for CD45 and Ki67 also didn’t work in this case. Other possible reasons for a lack of detectable clonality are insufficient coverage of primers, primer site mutations, or chromosomal aberrations.
In a previous study using flow cytometry, neoplastic lymphocytes of canine TZL showed heterogeneous immunopositivity for CD4 and CD8; that is, CD4+, CD8+, and CD4-CD8- subsets were included in canine TZL cases. 18 In the present study, CD4 expression in canine TZL could not be investigated due to lack of a commercially available antibody to CD4; however, nodal CD4+CD8+ lymphoma is extremely rare in dogs. 6 Furthermore, it has been reported that canine TZL was consistently immunopositive for class II MHC and CD25, suggesting that TZL arises from activated effector T cells. 18 In the present study, 11/14 (79%) of TZL cases were immunopositive for CD8, indicating that most canine cases of TZL were neoplasms of CD8+ cytotoxic T cells. In addition, TZL cases were consistently negative for granzyme B, which is one of the effector molecules of cytotoxic T cells, and appeared to have a similar immunophenotype to naïve CD8+ T cells which usually do not express the effector molecules. Therefore, the cell of origin of canine CD8+ TZL may be naïve or premature CD8+ T cells. On the other hand, all TZL cases in the present study were positive for HLA-DR which is one of the class II MHC antigens. Although high expression of class II MHC is considered to be a characteristic immunophenotype of activated T cells,7,12 the functions of class II MHC in T cells have not yet been elucidated in detail. Therefore, further studies using gene expression analysis are warranted to clarify the activated state of neoplastic T cells. In humans, the expression of cytotoxic molecules in nodal PTCL-NOS is associated with a poor prognosis. 1 The absence of granzyme B in CD8+ TZL in dogs may be related to its good prognosis.
In the present study, one TZL case was weakly immunopositive for CD20. CD20 expression is uncommon in T-cell lymphomas, but reported in a subset of T-cell lymphoma, including canine TZL. 10 In humans, it has been reported that a subset of circulating normal T cells express CD20, 20 and some CD20+ T-cell lymphomas are considered to be neoplastic transformation of normal CD20+ T cells. 14 These normal CD20+ T cells in humans are predominantly CD8+CD45+HLA-DR-20 and are different from the immunophenotype of canine TZL in the present study (CD8+CD45-HLA-DR+). Therefore, it is still unclear whether CD20 expression in canine TZL is aberrant or reflecting the immunophenotype of the cell of origin. In a monkey study, CD20 expression in lymph node T cells was increased after stimulation by a mitogen and interleukin-2, 11 indicating that CD20 expression, similar to class II MHC, may also be related to the activation status of T cells in dogs.
TZH is a hyperplastic lesion of lymph nodes associated with antigen processing. In the present study, the lymph nodes of all TZH cases were in the region of neoplastic lesions; therefore, the population of hyperplastic T cells appears to reflect an immune response to the tumors. CD8+ cytotoxic T cells play an important role in tumor immunity, recognizing tumor antigens presented by tumor class I MHC molecules and inducing tumor cell death. 23 Infiltrating CD8+ cytotoxic T cells have been associated with the prognosis of canine tumors, including oral melanomas and mammary carcinomas.4,25 In the present study, paracortical lymphocytes were variably immunopositive for CD8, and in 4 cases, more than 50% of paracortical lymphocytes expressed CD8. Previous studies using flow cytometry reported that the number of CD4+ T cells was higher than that of CD8+ T cells in normal canine lymph nodes.3,16 Therefore, CD8+ T-cell numbers are considered to be elevated in TZH in association with immune responses to tumors. FOXP3+ regulatory T cells play an important role in tumor immunity, particularly in relation to immune evasion by tumors; tumors secrete molecules that create a microenvironment that increases or activates regulatory T cells, which suppresses effector T-cell or CD8+ T-cell responses. 23 Increases in FOXP3+ T cells in the lymph nodes of dogs with tumors were previously demonstrated using flow cytometry. 2 Therefore, FOXP3+ T cells in TZH cases may be induced under the microenvironment created by the primary disease.
In conclusion, the present study revealed that most TZL cases were characterized by the proliferation of monomorphic CD8+CD45- T cells, while TZH consisted of an immunophenotypically heterogenous population of CD45+ T cells that were variably positive for CD8 or FOXP3. In addition to the histopathological examination and clonality analysis, these immunophenotypic features are helpful in differentiating TZL from TZH in dogs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221102599 – Supplemental material for Nodal T-zone lymphoma and T-zone hyperplasia in dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858221102599 for Nodal T-zone lymphoma and T-zone hyperplasia in dogs by Kazuhiro Kojima, James K. Chambers, Takuya Mizuno and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
