Abstract
An unusual clinical presentation of lymphoma with vertebral involvement in a dog is reported. A 20-month-old intact female Golden Retriever presented with progressive paraparesis and anorexia. Complete blood count and serum biochemistry profile demonstrated pancytopenia and hypercalcemia. Ventral fusion of the lumbar vertebrae by new bony tissue deposition was evident on X-ray and CT scan. Fine needle aspiration revealed neoplastic lymphoid cells in lymph nodes and bone marrow. Histologically, vertebral bone and osteophytes, liver, bone marrow, kidney, and lymph nodes were diffusely infiltrated by neoplastic, lymphoid cells, with scant cytoplasm and round hyperchromatic nuclei. Polyostotic and medullary T-cell lymphoma with spondylosis was diagnosed. Lymphoma mainly affecting bone is uncommon in the dog. The present case differs from previously described polyostotic lymphomas in clinical signs of the disease, mainly attributable to spondylarthrosis. In addition, lymphomatous proliferation was associated with osteoproductive lesions of the vertebrae.
A 20-month-old intact female Golden Retriever dog presented with a history of dysorexia, lameness of the hind limbs and reluctance to movement, beginning 10 days prior to referral. Lameness progressed to paraparesis. Clinically, poor body condition, tachycardia (160 bpm), dyspnea (100 rpm), and prescapular lymphadenopathy were observed. A complete blood count showed mild macrocytic normochromic anemia (RBC = 5.17 × 106/μl, reference range, 5.70-8.10 and MCV = 73.8 fl, reference range, 61.0-70.0 fl), thrombocytopenia (56 × 103/μl, reference range, 150–450 × 103/μl), neutropenia (2525/μl, reference range, 3500–8700/μl), and lymphocytosis (7272/μl, reference range, 1400–4100/μl). The serum biochemistry profile revealed increased liver-specific serum enzymes (CPK = 187 IU/liter, reference range, 40–150 IU/liter; AST = 151 IU/liter, reference range, 15–40 IU/liter; ALT = 294 IU/liter, reference range, 15–65 IU/liter; ALP = 971 IU/liter, reference range 20–120 IU/liter; GGT = 26.2, reference range, 2–8 IU/liter), an increased albumin/ globulin ratio (A/G = 1.36, reference range, 0.7-1.2) due to decreased globulin level (A = 3 g/dl, reference range, 2.6-3.8 g/dl; G = 2.2 g/dl, reference range, 2.6-4 g/dl), and hypercalcemia (13.9 mg/dl, reference range, 9.6-11.7 mg/ dl). Proteinuria (250 mg/dl, reference range 0–50 mg/dl), increased urinary protein/urinary creatinine ratio (3.5), increased urinary bile acid, and hematuria were shown by urinalysis. Ventral fusion of the lumbar vertebral bodies due to deposition of new bony tissue was evident on X-ray (Fig. 1).and CT scan (Fig. 2). Cytological examination of lymph node fine-needle aspirates, stained with May-Grunwald Giemsa, revealed a population of loose and packed clusters and sheets of medium neoplastic lymphocytes with increased nuclear/cytoplasm ratio, pale, scant cytoplasm, anisocytosis, and anisokaryosis (Fig. 3).
Bone marrow aspiration revealed a population of lymphoid cells, representing approximately 98% of total bone marrow cells, with the same cytological appearance as those observed in lymph nodes. An aspirate from the bone marrow was stained using 6 leukocyte-specific antibodies and analyzed by flow cytometry (Epics XL-MCL). a Measured data were evaluated with the System II (DOS 6.22) counter program. a Flow cytometry identified a single population of cells with low cytoplasm complexity, of moderate size. Lymphoid cells were strongly positive for CD4 (T-helper cells) (89.4%) (Fig. 4), whereas only a small percentage of cells reacted with the antibody against CD3 (T-lymphocytes) (6.8%). Moderate reactivity against CD79α (B-lymphocytes) (22%) was also observed. No positive reactions were observed against CD8 (cytotoxic T-cells), CD34 (hematopoietic stem cells), or CD14 (monocytes). The presence of IgG and IgM on the surface of erythrocytes and platelets was ruled out by flow cytometry.
Following the owner's decision, the dog was euthanized and necropsied. Grayish-white confluent masses were found in the spinal cord, heart, liver, kidney, and adrenal glands, together with diffuse lymphadenomegaly. Osteophytes were also present on the ventral surface of lumbar vertebral bodies, causing bone ossification of the spinal cord. Samples from several organs were taken, immediately fixed in 10% neutral buffered formalin, routinely processed, and paraffin-embedded for histological examination. Histologically, both vertebral bodies and osteophytes were diffusely infiltrated by neoplastic lymphoid cells (Fig. 5), with scant cytoplasm, round hyperchromatic nuclei with single prominent nucleoli, which also invaded the periosteum and infiltrated the muscles. Neoplastic invasion of vertebral bone was associated with multifocal bone lysis and poorly organized reactive bone. Mitotic activity was of 2–3 mitotic figures per high power field. The bone marrow was almost completely replaced by neoplastic cells, with only rare megakaryocytes remaining (Fig. 6). Foci of neoplastic bone invasion were mostly separated from bone marrow, and in some cases neoplastic cells infiltrated the adjacent bone marrow cavity. Mediastinal and iliac lymph nodes were also diffusely infiltrated by sheet of the same lymphoid population, obliterating the node architecture. Liver, kidney, adrenal glands, heart, and duodenal submucosa were also infiltrated by multifocal to coalescent sheet of neoplastic cells. The spleen was very congested with extramedullary hematopoiesis, but neoplastic involvement was not evident. Additional sections were processed for immunohistochemistry and tested with the polyclonal rabbit anti-canine CD3 b and the monoclonal mouse antihuman CD79α, c but only weak immunostaining was observed in some cells. A diagnosis of polyostotic and medullary T-cell lymphoma associated with spondylosis was made.
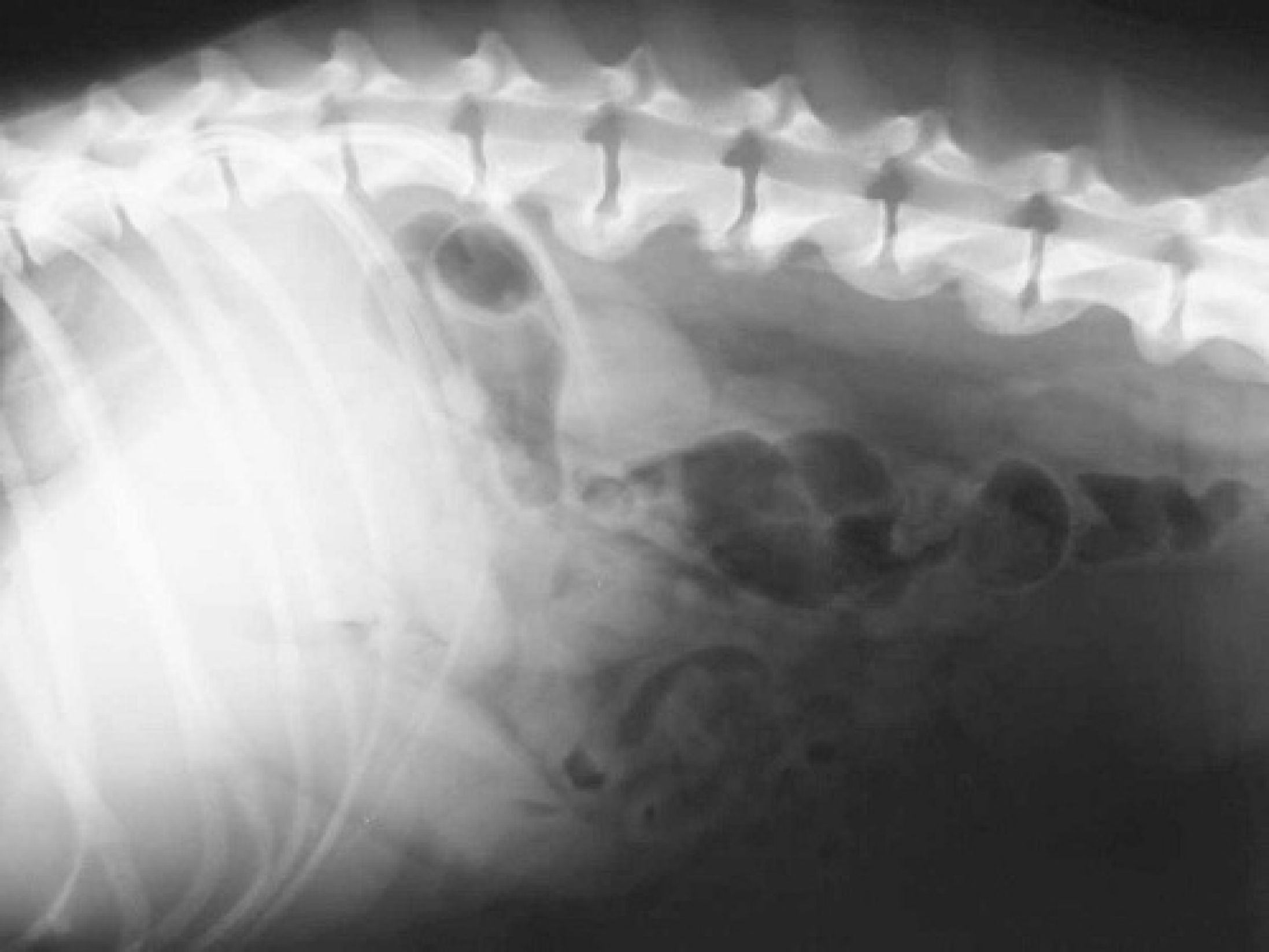
Lateral view radiograph of lumbar vertebrae of dog with lymphoma. Note the osteophytes on the ventral surface of lumbar vertebral bodies.
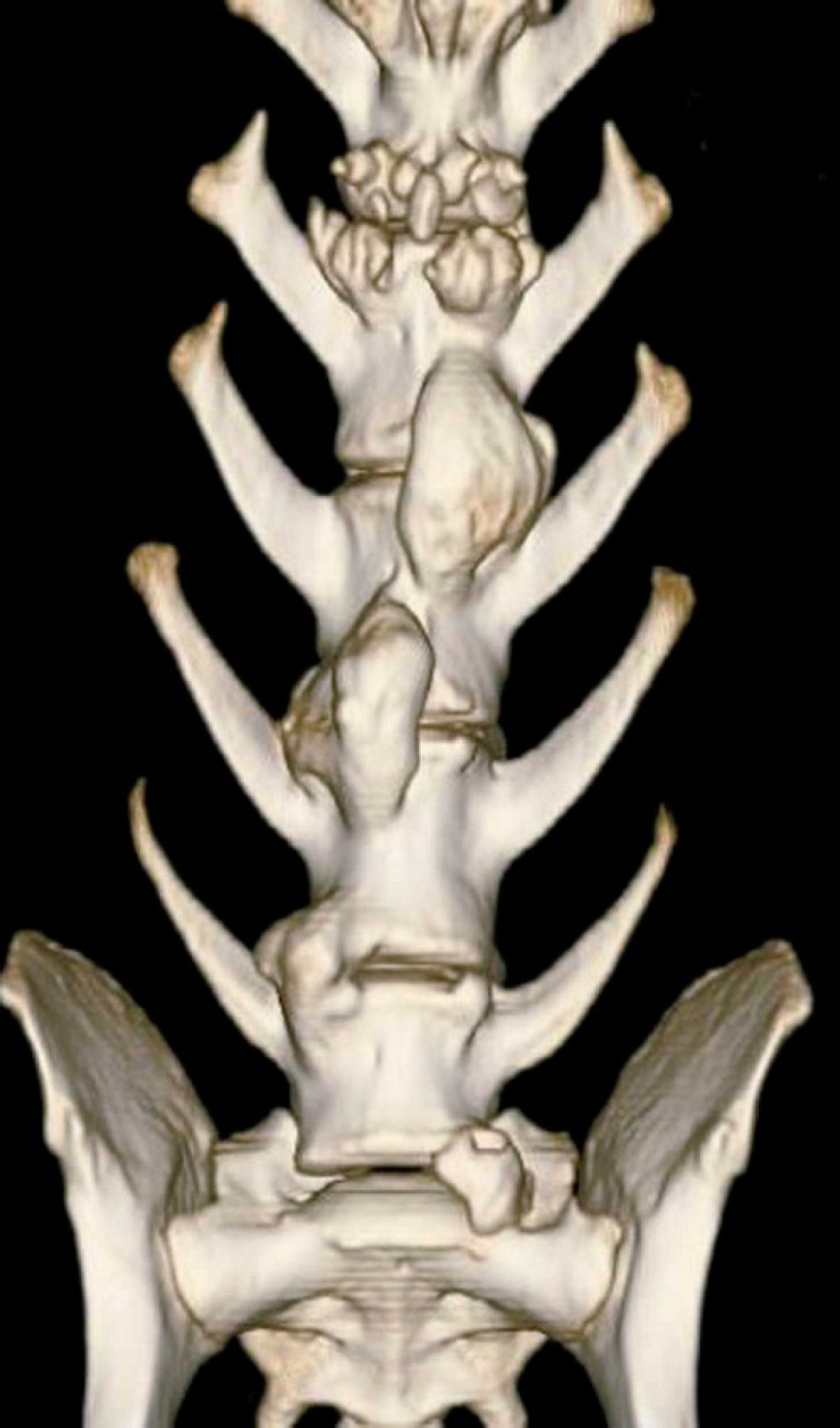
Computed tomography of lumbar spinal cord. Osteoproductive lesions causing ventral ossification of the vertebral bodies are evident.
Malignant lymphoma may occur as a primary tumor of bone in humans and dogs, but most frequently bone involvement occurs in association with multicentric lymphoma in dogs, cats, and cattle. 14 Lymphoma tends to infiltrate and replace bone marrow, but bone destruction is rarely identified radiographically or as a cause of clinical signs. 1,6 There are few published reports about dogs of lymphoma where lytic bone lesions are the prominent feature of the disease. These cases are perhaps best referred to as lymphoma mainly affecting bone rather than as primary lymphoma of bone. 14 Polyostotic lymphomas have been described in young dogs ranging in age from 15 weeks to 5 years 3,7,9,13 and in children. 17 The present case differs from previously described polyostotic lymphomas in 2 regards. First, clinical signs of the disease were mainly attributable to spondylarthrosis. Second, lymphomatous proliferation was associated with osteoproductive lesions of the vertebrae. In all previous cases, clinical presentation included pathological fractures associated with multiple osteolytic lesions, particularly affecting the long bones. 3,7,9,13 In the present case, as well as in previously reported ones, the polyostotic distribution of lesions was compatible with hemolymphatic spread. 11 Several of the hematological and biochemical alterations observed were related to the diffuse spread of neoplastic cells. Pancytopenia was probably due to replacement of the normal marrow cells by the lymphoma. Despite this, anemia was mild, presumably due to intense splenic extramedullary hematopoiesis. A variety of immune-mediated responses, such us immune-mediated thrombocytopenia and hemolytic anemia, may be seen in dogs with lymphoma. 5 In this case, the hypothesis of immune-mediated anemia and thrombocytopenia was ruled out by the lack of expression of IgG and IgM antibodies on the surface of erythrocytes and platelets. The increased liver-specific serum enzyme activity and serum iron levels were considered secondary to hepatic neoplastic invasion, associated with necrosis and altered blood circulation. Impaired hepatic function was also highlighted by decreased cholesterol serum and increased urinary bile acid.
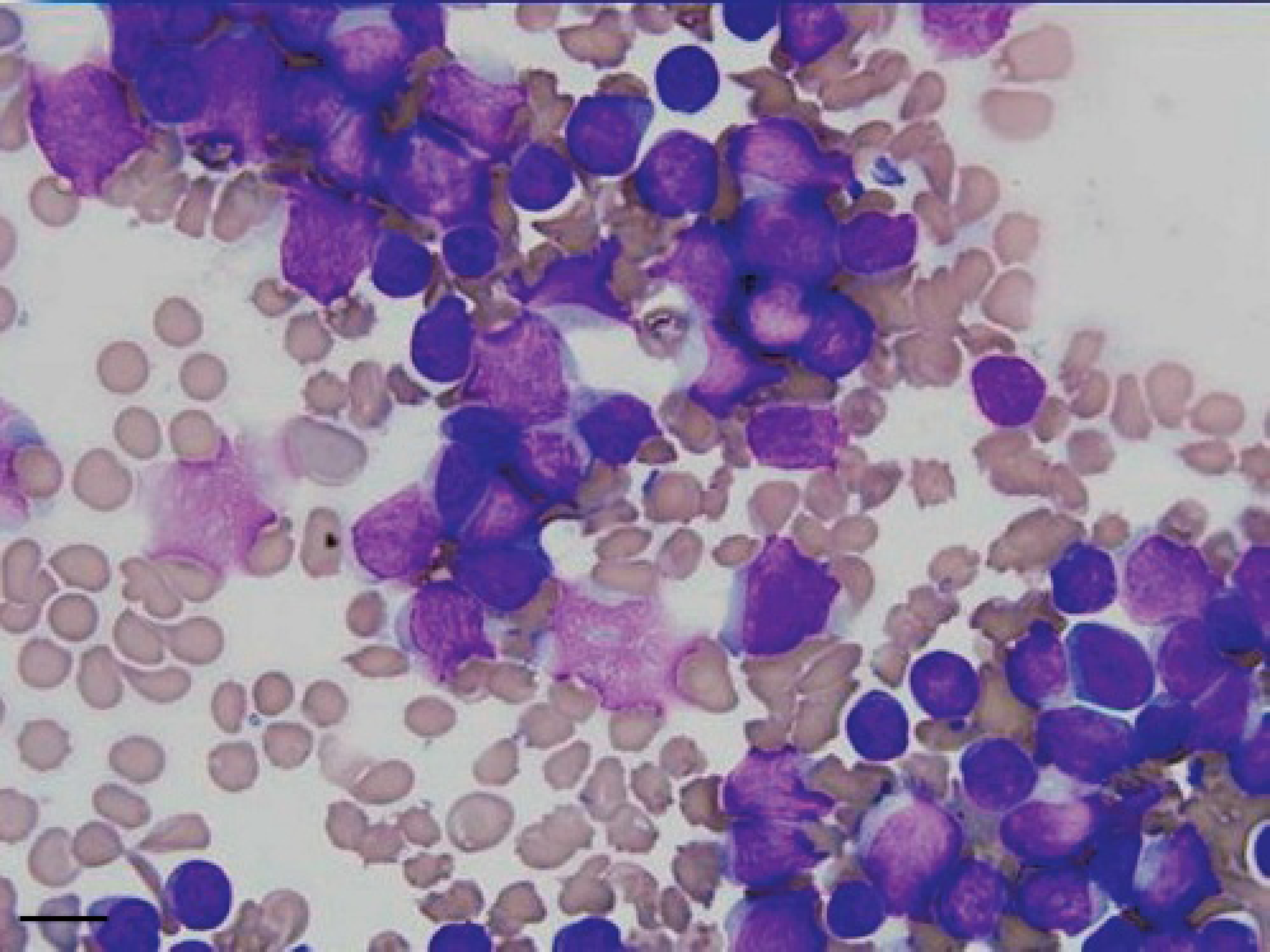
Cytological appearance of fine-needle aspirate from bone marrow, showing a population of lymphoid neoplastic cells with increased nuclear/cytoplasm ratio, pale scant cytoplasm, anisocytosis, and anisokaryosis (MGG, Bar = 5 μm).
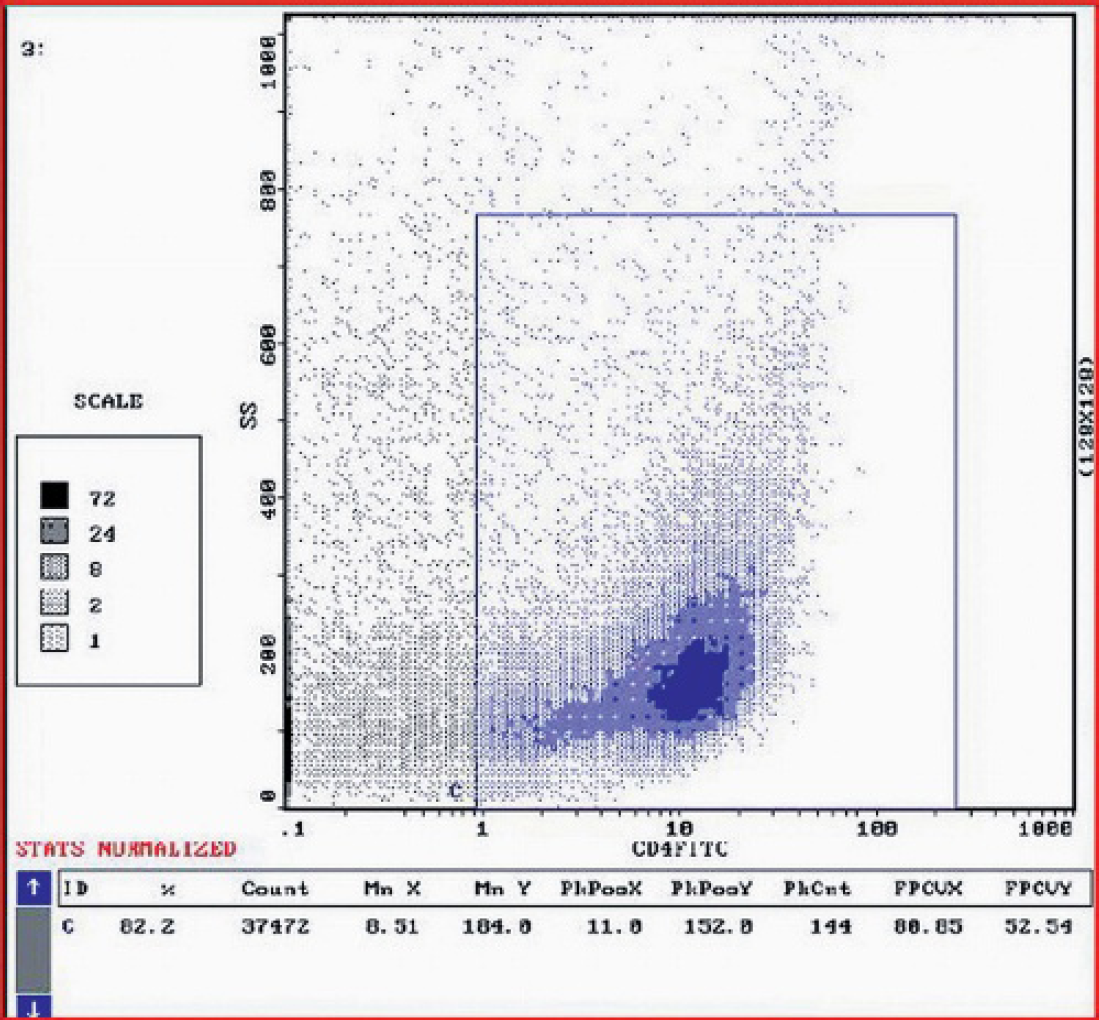
Flow cytometry of bone marrow aspirate revealed a single population of cells with low cytoplasm complexity (side scatter), of moderate size (forward scatter) strongly positive for CD4 (T-helper cells). Data evaluated by System II (DOS 6.22) counter program.
By both immunohistochemistry and flow-cytometry, the expression of CD79α and CD3 was moderate and scarce, respectively, making it difficult to classify the lymphoma as of B- or T-cell type. However, strong CD4 expression was consistent with a T-cell immunophenotype. Due to the negativity of cells for CD34, expressed by noncommitted hematopoietic stem cells together with strong CD4 expression, indicative of a thymocyte differentiation stage, the neoplasm was not considered as a primary bone marrow lymphoma. In the dog, CD4 is also expressed by neutrophils and monocytes, but these were rarely observed among bone marrow cells in the present case. Thus, CD4 positivity was concluded to be attributable to neoplastic lymphoid cells. Interestingly, this case expressed the combination of B- (CD79α) and T-cell markers (CD4), with totals exceeding 100%. The flow cytometry results are tentatively explained on the one hand by partial loss of expression of CD3 antigen by T-cells, and on the other by coexpression of T- and B-cell antigens by a fraction of the neoplastic cells. T-cell lymphomas with loss of expression of the CD3 molecule, as well as aberrant coexpression of B-and T-cell antigens, have been described in both human and canine lymphoid neoplasia. 2,12,16
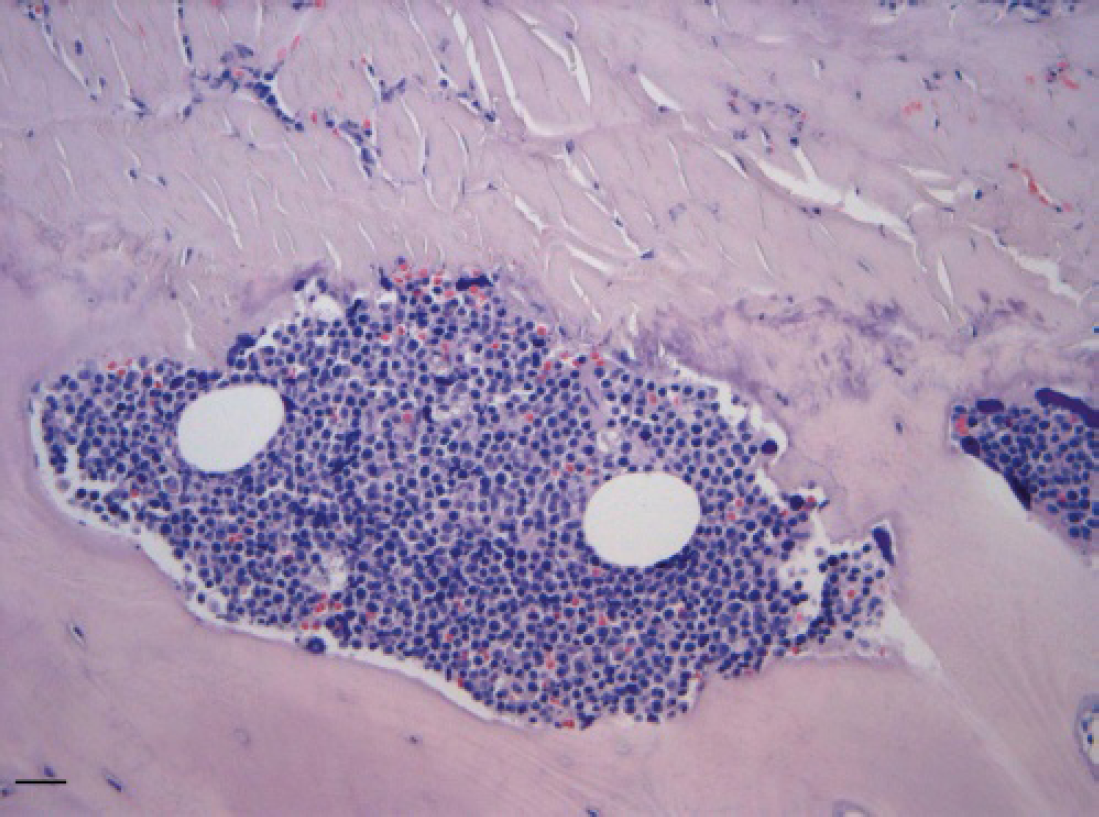
Histologically, both vertebral bodies, and osteophytes were diffusely infiltrated by neoplastic lymphoid cells (HE, Bar = 25 μm).
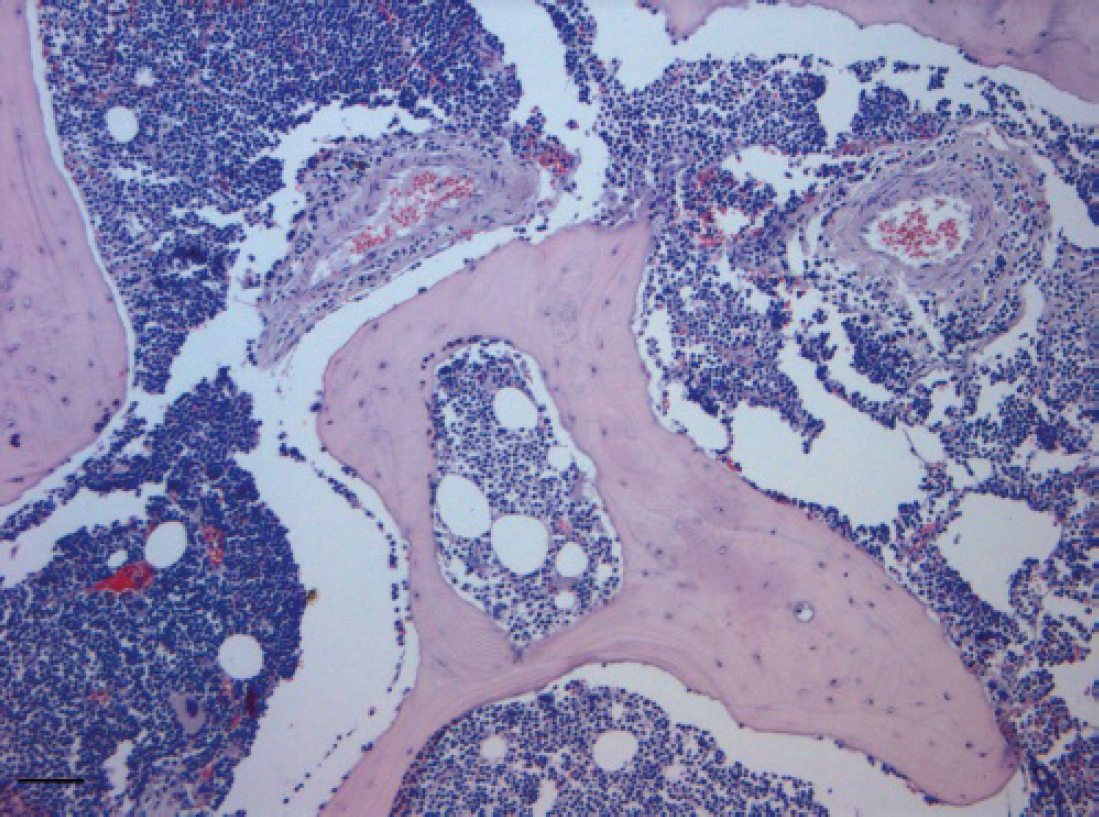
The bone marrow was almost completely replaced by neoplastic cells, with only rare megakaryocytes remaining (HE, Bar = 100 μm).
In the canine polyostotic lymphomas reported so far, neoplastic cells showed features of immature lymphocytes, but the immunophenotype was rarely defined. In one case immunohistochemistry failed to define the immunophenotype, since tumor cells did not label positively for any of the T and B lymphocyte markers. 7 In a recent report of a polyostotic lymphoma with vertebral involvement in a dog, immunohistochemical stains indicated a T-cell immunophenotype. 6 Peripheral T-cell lymphoma with acquired systemic osteosclerosis has been described in a human patient. 4 Osteosclerosis was interpreted as a paraneoplastic syndrome, due to secretion of an osteoblast-activating factor by the T-cell lymphoma. In contrast, in B-cell lymphomas and in multiple myelomas, there is secretion of osteoclast-activating factors, 4 which may be responsible for the osteolytic lesions observed in the polyostotic lymphomas previously described. In the present case, hypercalcemia was considered a paraneoplastic feature, mainly associated with CD4 T-cell lymphomas. 10 Secondary hyperparathyroidism was excluded because of the low serum level of parathormone (<0.05 pmol/liter, reference range 0.10-1.10 pmol/liter). Hypercalcemia is one of the most common paraneoplastic syndromes in dogs with lymphoma, occurring in approximately 10%-40% of clinical cases. 5,8 Clinical manifestations of hypercalcemia vary between dogs that are asymptomatic and those with severe systemic illness. Clinical signs of hypercalcemia are usually renal, resulting in polyuria/polydipsia and potentially prerenal azotemia, gastrointestinal or neuromuscular in nature. 8 Although there is still no agreement on the mechanisms of hypercalcemia in canine lymphoma, in most cases it is thought to occur as a result of the production by neoplastic cells of a parathormone-like protein called PTH-related protein. 8,15 In polyostotic lymphoma with extensive neoplastic osteolysis, hypercalcemia has also been attributed to marked bone resorption. 11
Footnotes
a.
Beckman Coulter, Miami, FL.
b.
Dr. P. F. Moore, School of Veterinary Medicine, University of California, Davis, Davis, CA.
c.
DAKO, Carpinteria, CA.
