Abstract
A newly described onygenalean fungus, Emydomyces testavorans, has been isolated from ulcerative shell and skin lesions of freshwater aquatic chelonians. To investigate the shell lesions associated with infection and determine if any lesional features were unique to E. testavorans, tissues from turtles housed in zoological institutions (n = 45) in the United States and free-living turtles (n = 5) submitted for diagnostic biopsy or necropsy were examined. Free-living turtles were from geographically distinct habitats in Florida (n = 1) and Washington (n = 4) at the time of sampling. Histologic shell sections were evaluated for the presence or absence of specific lesional features. Infection with E. testavorans was evaluated in all cases by screening GMS (Grocott-Gomori’s methenamine silver)-stained histologic sections for the presence of morphologically consistent fungi and by quantitative PCR (polymerase chain reaction) on representative frozen tissue or formalin-fixed paraffin-embedded sections. Additionally, culture was performed for 15 cases with available fresh/frozen tissue. In total, there were 17 PCR-confirmed E. testavorans cases, 29 cases with morphologically consistent fungi on GMS-stained sections, and 21 cases of shell lesions without histologic or molecular evidence of E. testavorans infection. Epithelial inclusion cysts, defined as cystic structures within the dermis lined by keratinized stratified squamous epithelium and containing necrotic bone and keratin debris, were significantly (P < .01) associated with E. testavorans infection. Other significantly associated shell lesions included squamous metaplasia, hyperkeratosis, inflammation, and osteonecrosis (P < .05). This study identified characteristic shell lesions associated with E. testavorans infection. Further studies to prove causality are needed.
Several fungi in the genera Nannizziopsis, Paranannizziopsis, and Ophidiomyces have been identified as primary reptile pathogens that can impact in situ conservation efforts, most notably Ophidiomyces ophiodiicola. 2,3,6 These keratinophilic fungi belong to the order Onygenales and many are known to exist as environmental saprophytes. 3,23,25 Nannizziopsis sp. infection in lizards results in ulcerative dermatitis that may progress to localized granulomatous cellulitis and osteomyelitis. 5,18,28 In many cases, infections are of such severity that the animal is euthanized or dies due to complications. O. ophiodiicola infection has been reported in a variety of snake species, including the endangered Eastern massasauga (Sistrurus catenatus catenatus), in which fungal infection causes severe facial deformities. 2 Rare cases progress to systemic mycotic infection. 20 O. ophiodiicola infection has also been associated with population decline in the timber rattlesnake (Crotalus horridus), which is endangered in portions of its range. 6,14 The current understanding of the epidemiology and pathogenesis of onygenalean fungal infections in reptiles is limited, although one study has suggested that cutaneous damage enhances, but is not required for, disease development. 18 There are few reports of onygenalean fungi isolated from chelonians. Fungi identified as Chrysosporium have been isolated from the cloaca and eggs of sea turtles but without report of associated disease, and Aphanoascella galapagosensis has been isolated from a Galapagos tortoise (Chelonoidis nigra microphyes) with carapace keratitis. 8,19,27
Shell disease is a common cause of morbidity and mortality in free-living chelonians and those under human care. 4,9,21 Septicemic cutaneous ulcerative disease (SCUD) is an example of shell disease in aquatic turtles, in which bacterial dermatitis and osteomyelitis develop secondary to superficial trauma and poor water quality. 4,11 In addition to bacteria, common causes of shell lesions include traumatic injury and metabolic/nutritional imbalances, although often the cause is unknown. 4,21 A newly described onygenalean fungus, Emydomyces testavorans, has been isolated from unusual ulcerative shell lesions of freshwater aquatic turtles. 29 This retrospective descriptive study was initiated to describe the shell lesions associated with E. testavorans infection.
Materials and Methods
Study Subjects
Study subjects (cases) included were submitted for diagnostic necropsy or biopsy between 1996 and 2019 to the Zoological Pathology Program (ZPP) at the University of Illinois Urbana-Champaign (n = 48) and the Aquatic, Amphibian, and Reptile Pathology Program at the University of Florida (n = 2). Archived cases were identified by conducting a search of the ZPP laboratory database (VADDS v9.0) using the keywords “carapace” and “plastron” and limiting to freshwater aquatic and semiaquatic turtle species. Cases from freshwater turtles for which there were both documented shell lesions and formalin-fixed, paraffin-embedded (FFPE) lesional shell tissue available for histologic examination and PCR (polymerase chain reaction) were considered for inclusion. Final inclusion was further limited to cases in which histologic shell sections included keratin layer, epidermis, dermis, and shell (dermal) bone. Cases in which only partial shell sections were available, for example, consisting of superficial keratin or necrotic debris, were excluded. Where individual turtles were associated with multiple submissions, such as when a shell biopsy was submitted prior to death and necropsy, histopathologic and E. testavorans detection data were collated so that each case used in final analyses represented a single turtle. Selected turtles were free-living from the states of Florida and Washington or were managed under human care in either zoological collections or as pets in the United States.
Histopathologic Evaluation
Formalin-fixed tissues were processed for histopathology at the University of Illinois Veterinary Diagnostic Laboratory (n = 48) and the Veterinary Histology Laboratory at the University of Florida (n = 2). As chelonian shells contain both bone and keratin, formalin-fixed shell sections at the University of Illinois were pretreated sequentially with both a commercially available decalcifying agent containing 15% formic acid (DeltaFORM, Delta Medical, Inc) and a keratin softening solution (3% HCl and 9% Tween80 in aqueous solution). Shell sections at the University of Florida were decalcified in in 0.5 M EDTA, pH 8.0, for ∼72 hours. Tissues were then processed routinely and stained with hematoxylin and eosin (HE). One representative shell slide from each case was also stained with Grocott-Gomori’s methenamine silver (GMS) to enhance detection of fungi and evaluation of fungal morphology within lesions.
Signalment information, gross necropsy findings, and gross images, as available, were examined and recorded for each submission. From necropsy submissions, sections from various organs were also reviewed histologically to identify any underlying or systemic disease processes. Shell tissues were evaluated for any morphologic changes to the epidermis, dermis, and bone, and lesions were identified descriptively with respect to process and potential cause. If any potentially infectious organisms were detected in HE-stained sections, their morphology and location (ie, whether along the leading edge of lesions or along the surface of the shell [superficial]) were recorded.
During the course of preliminary histologic evaluation, unusual shell lesions were detected in a subset of cases. These lesions consisted of cystic structures within the dermis and/or bone lined by stratified squamous keratinizing epithelium and containing abundant keratin, necrotic debris, and fragments of necrotic bone. For the purposes of analysis, these lesions were termed “epithelial inclusion cysts.”
To allow for statistical comparison between lesional features associated with E. testavorans infection and those due to other causes, shell sections were specifically evaluated for the strict presence or absence of epithelial inclusion cysts, ulceration, hyperkeratosis, squamous metaplasia of the scute epidermis, necrosis of shell (dermal) bone (osteonecrosis), and inflammation of any nature.
Fungal Detection
The presence or absence of E. testavorans within shell tissues was determined using 3 different modalities. For all cases (n = 50), GMS-stained sections were evaluated for the presence or absence of fungi, fungal morphology, and location of fungal elements (lesional or superficial). Hyphae that were 1 to 4 µm diameter with regular septations and acute to right-angle branching were considered morphologically consistent with E. testavorans. 29 Cases having fungi consistent with E. testavorans were recorded as a positive detection by GMS.
Fresh or frozen lesional tissue were available from a subset of cases (n = 15; cases 1–12, 14, 16, 17) including 5 free-living turtles and 10 turtles from zoological institutions. Culture of E. testavorans was performed using previously published methods and selective media. 29 The specific identity of all cultured fungal isolates was confirmed via PCR and sequence analysis of the internal transcribed spacer (ITS) region according to previously published methods. 29 For fresh or frozen tissues from which fungal culture was unsuccessful, the same ITS PCR and sequence analysis was performed directly on tissue DNA extracts.
For the remaining cases (n = 35), only FFPE shell was available for molecular testing. Because the ITS PCR targets a 630 bp fragment and formalin fixation degrades DNA, a qPCR targeting an 83 base-pair segment of the E. testavorans 28S rRNA gene was developed to test for E. testavorans in these FFPE samples. Using DNA alignments generated for previous phylogenetic analyses of E. testavorans and related onygenalean fungi 29 a custom TaqMan MGB (Applied Biosystems) probe-based qPCR assay was designed. The assay had an analytical sensitivity of 9.2 copies of target sequence per microliter, a reaction efficiency of 94.1%, and a coefficient of determination (r 2) of 0.997. Specificity of the assay was confirmed by testing available DNA extracts of onygenalean fungal isolates (Aphanoascus fulvescens [telomorph of Chrysosporium keratinophilum], Coccidioides immitis, Ophidiomyces ophiodiicola, and Nannizziopsis arthrosporioides). To test the ability of this assay to identify E. testavorans DNA in FFPE samples, all samples (n = 50) were tested, including the 15 with previous positive culture and or ITS PCR identification. DNA was extracted from one representative block of FFPE shell tissue according to previously published protocols. 24 DNA extracts were tested in triplicate via qPCR, and samples for which target DNA was amplified with a mean Ct value ≤37 were considered positive for E. testavorans.
Statistical Analyses
Data analysis was performed using R Statistic Software v3.6.0. Univariate and multivariate logistic regression models were used to evaluate associations between E. testavorans infection and specific lesion characteristics. Manual backward stepwise regression was used to sequentially eliminate variables using information-theoretic model selection and the Akaike information criterion (AIC). Biologically meaningful pairwise interactions were assessed between model variables. As multiple detection methods were utilized, 2 sets of analyses were performed with different criteria for confirmation of E. testavorans infection. In the first analysis, all cases in which morphologically consistent hyphae were apparent in GMS-stained tissue sections were considered to have confirmed E. testavorans infection. In the second and more stringent set of analyses, only cases in which E. testavorans was detected via PCR and/or culture were considered to have confirmed infection. For both sets of analyses, significance of association between lesional features and E. testavorans infection was determined at the P < .05 level.
Results
Study Animals
In total, 62 submissions met criteria for inclusion in this study. Submissions consisted of 24 biopsies and 38 necropsies from 50 individual turtles (Table 1). Nine turtles had multiple submissions, either biopsy and necropsy (cases 7, 16, 17, 26, 28, 39, 47) or multiple biopsies only (cases 12, 15). Ten cases only had a single biopsy submission (cases 1–4, 13, 20, 21, 25, 26, 34). Turtles were taxonomically diverse spanning 20 genera and 27 species: Actinemys marmorata (n = 5), Apalone ferox (n = 1), Apalone spinifera (n = 2), Chelus fimbriatus (n = 3), Chrysemys picta (n = 3), Clemmys guttata (n = 2), Emydura macquarii (n = 1), Emydura subglobosa (n = 5), Geoemyda spengleri (n = 3), Graptemys oculifera (n = 2), Heosemys spinosa (n = 1), Hydromedusa tectifera (n = 2), Kinosternon scorpioides (n = 1), Malaclemys terrapin (n = 1), Phrynops hilarii (n = 1), Platemys platycephala (n = 1), Platysternon megacephalum (n = 1), Podocnemis expansa (n = 1), Podocnemis unifilis (n = 4), Podocnemis vogli (n = 3), Pseudemys floridana (n = 1), Pseudemys nelsoni (n = 1), Pseudemys rubriventris (n = 1), Terrapene carolina (n = 1), Terrapene ornata (n = 1), Trachemys decorata (n = 1), and Trachemys scripta (n = 1). Sex distribution of study turtles was 22 females, 18 males, and 10 turtles of unspecified or undetermined sex. There were 39 adults, 10 juveniles, and 1 turtle of unspecified age. At the time of sampling, 5 turtles were free-living within the states of Washington (cases 1–4) and Florida (case 6) in the United States of America. Turtles from Washington State had been previously housed in a zoological facility as part of a “head-start” program before being released into the wild.
Signalment, Detection of Emydomyces Testavorans, and Associated Histologic Lesions in Turtles Examined by Histopathology in this Study.
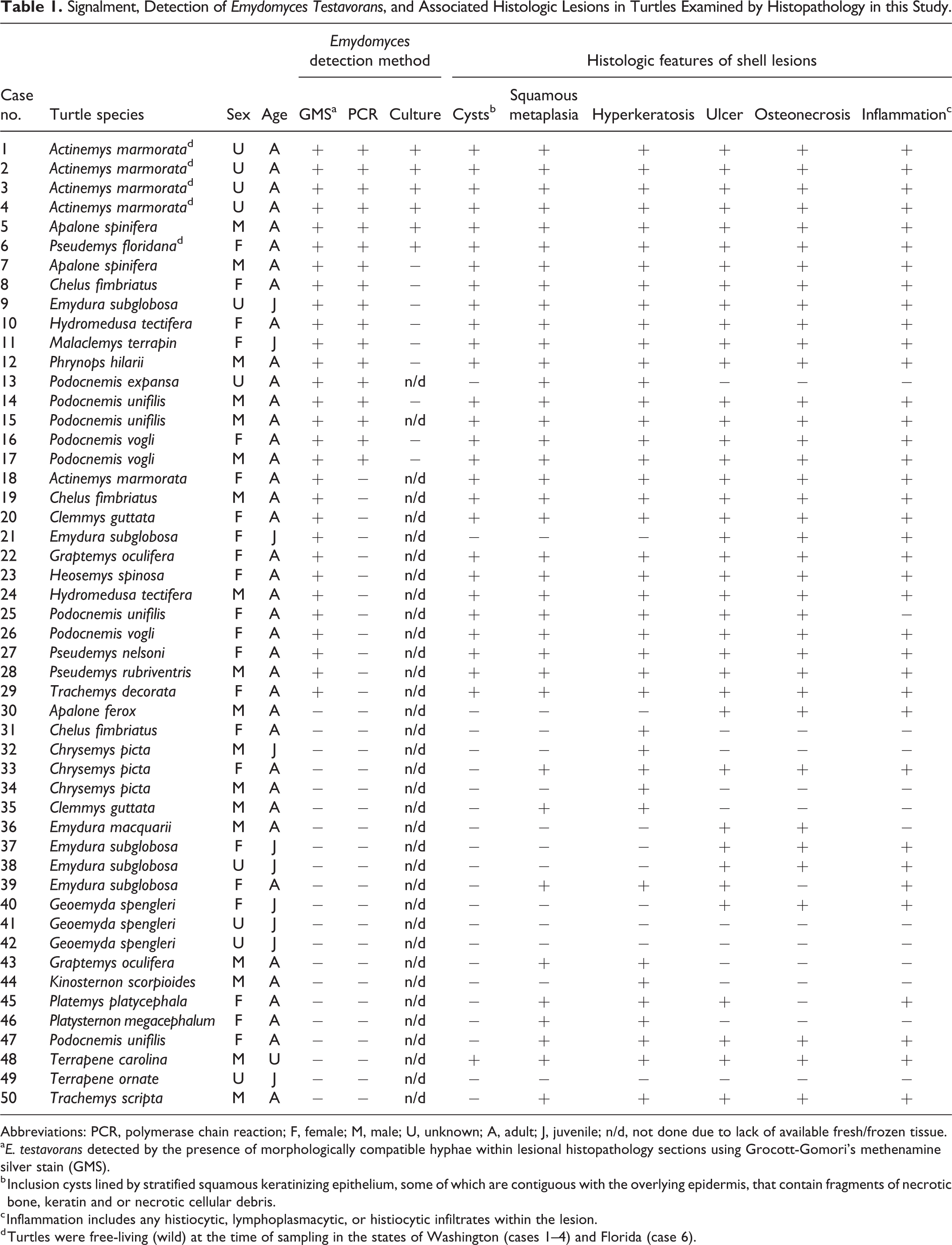
Abbreviations: PCR, polymerase chain reaction; F, female; M, male; U, unknown; A, adult; J, juvenile; n/d, not done due to lack of available fresh/frozen tissue.
a E. testavorans detected by the presence of morphologically compatible hyphae within lesional histopathology sections using Grocott-Gomori’s methenamine silver stain (GMS).
b Inclusion cysts lined by stratified squamous keratinizing epithelium, some of which are contiguous with the overlying epidermis, that contain fragments of necrotic bone, keratin and or necrotic cellular debris.
c Inflammation includes any histiocytic, lymphoplasmacytic, or histiocytic infiltrates within the lesion.
d Turtles were free-living (wild) at the time of sampling in the states of Washington (cases 1–4) and Florida (case 6).
Pathologic Evaluation
Thirty-two cases had gross reports available for review and gross shell lesions were documented in 31 of 32 (97%) cases at necropsy. In most cases (n = 24), external lesions were described as multifocal ulcerations affecting carapacial and/or plastronal scutes. Additional reported gross lesions included smooth pitted depressions typical of previously healed damage (cases 6, 11, 31, 35), discoloration and flaking of scute keratin (cases 6, 23, 47), increased pliability of the shell (cases 6, 11, 41), thickening and exudation along plastronal scute margins (cases 11, 19), and traumatic perforation (case 44).
In 7 cases (cases 5, 7, 11, 16, 17, 18, 26), there were firm to hard, expansile, nodular masses within the shell that could not be detected externally but were apparent following exposure of the internal coelomic surface (Fig. 1). Nodules displaced the coelomic membrane, distorted the internal contour of the shell, and compressed coelomic viscera, but were not noted to penetrate into the coelomic cavity. Nodules were cystic with central tan to gray-brown caseous to granular material, and in some cases communicated with surface ulcerations (Fig. 2).
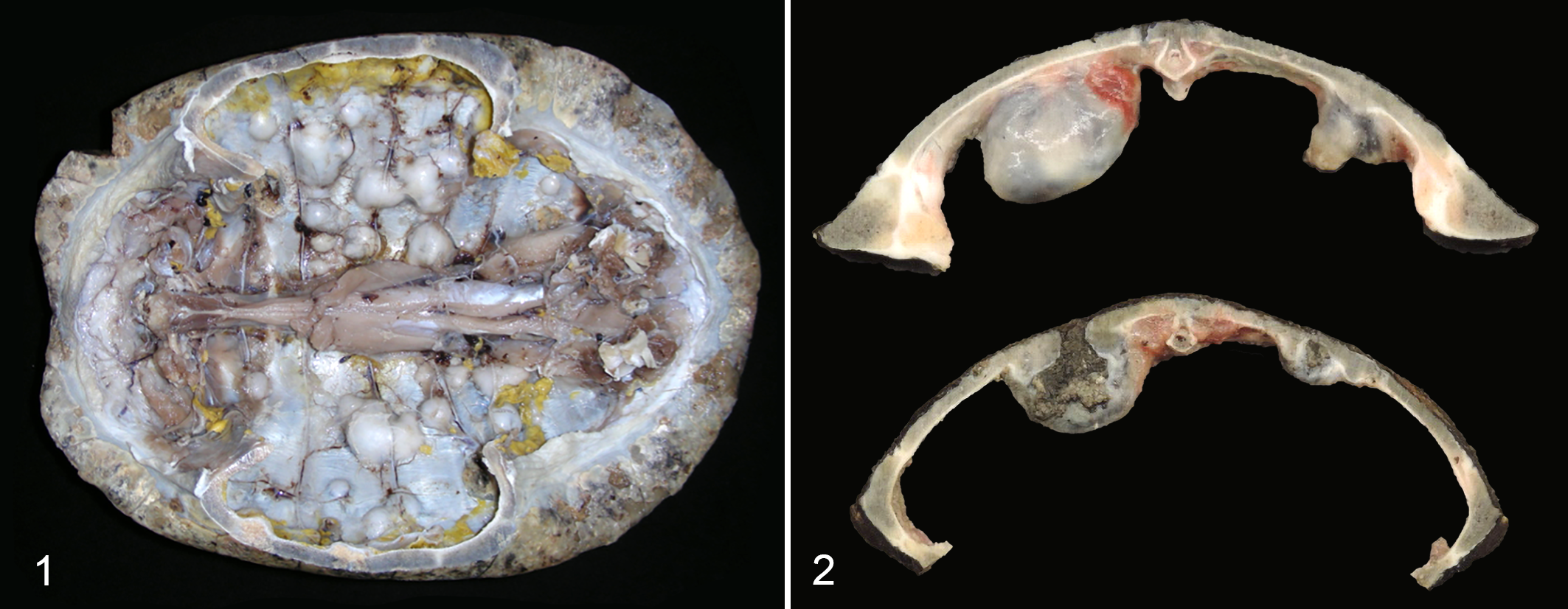
Emydomyces testavorans infection, shell, turtles.
Results for histopathologic evaluation and fungal detection are presented for each case in Table 1. Histologically, normal chelonian shells are composed of a thick layer of compact acellular surface β-keratin, a layer of pseudostratified columnar epithelial cells, a thin layer of superficial dermal connective tissue, and full thickness shell bone that ranges from compact to cancellous depending on the species and location on the shell (Fig. 3). Epithelial inclusion cysts were detected in 28 of 50 individuals (56%, cases 1–12, 14–20, 22–29, 48), including the 7 cases with grossly evident nodular lesions and in 27 of the 29 (93%) cases with E. testavorans–consistent fungi. Histologically, cysts were multilocular intradermal and intraosseous structures and were associated with loss of shell bone (Fig. 4). Cysts were lined by well-differentiated stratified squamous epithelium (squamous metaplasia) with orderly maturation and gradual keratinization; lining epithelium was contiguous with the overlying epidermis in sections where cyst lumens communicated with shell ulcers. Cyst lumens contained large amounts of lamellated and fragmented keratin, fragments of necrotic bone, and necrotic cellular debris (Fig. 5). Except for one individual (case 48), all turtles with inclusion cysts also had hyphae morphologically consistent with E. testavorans, as detected by GMS, within squamous epithelium and among keratin debris (Fig. 6). In the remaining case (case 48), there were lesional intrahistiocytic acid-fast bacteria (consistent with Mycobacterium spp.), but fungi were not detected by PCR or on GMS stains. In many cases with inclusion cysts (n = 21, cases 1–7, 10, 11, 14–20, 23, 25–27) bacteria ranging from pure cocci to mixed populations were also noted within lesional necrotic debris, but not within the cyst epithelium.
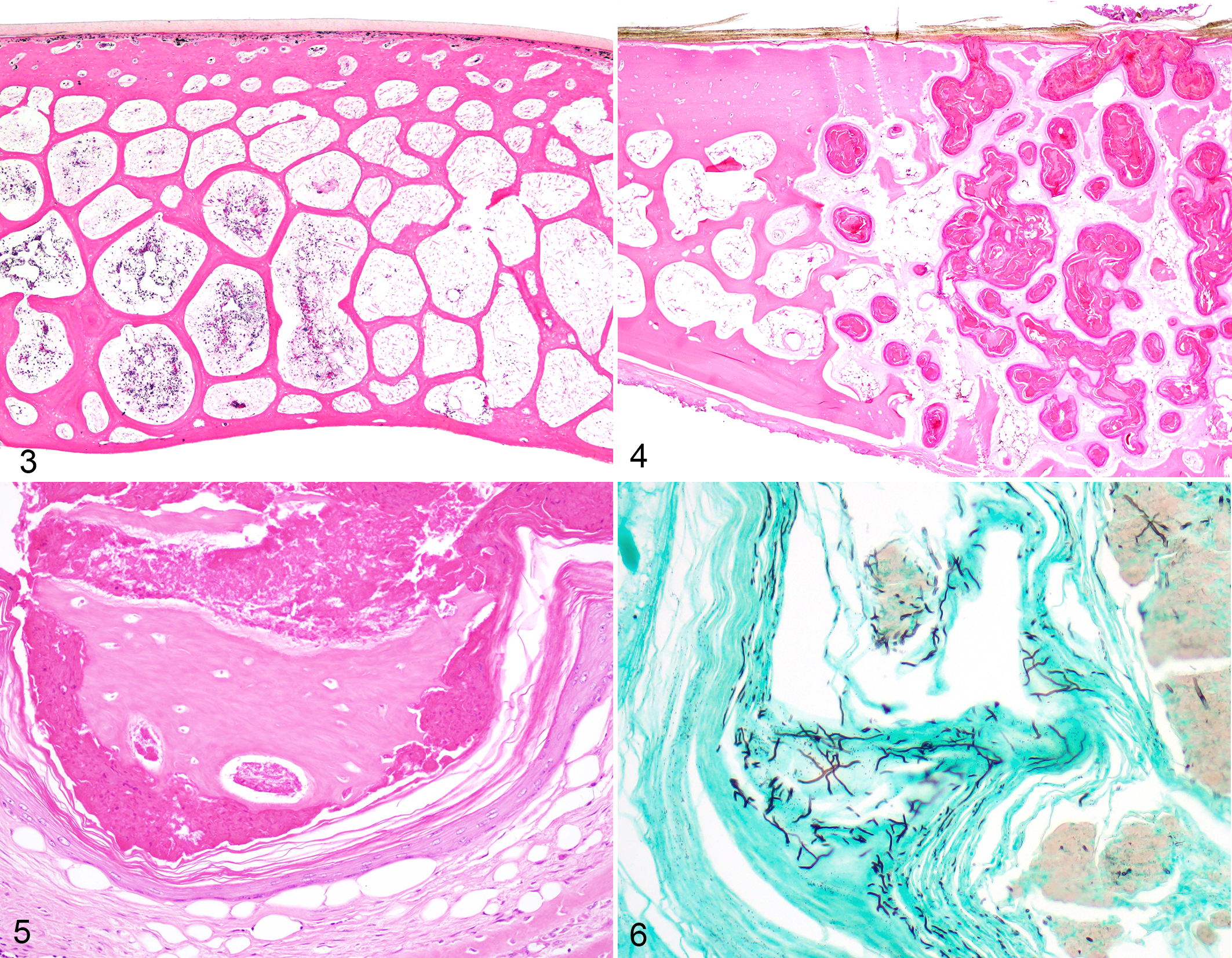
Normal carapace, Chrysemys picta, case 33. The normal chelonian shell has superficial keratin, a thin band of dermal connective tissue, and shell bone. Hematoxylin and eosin (HE).
In cases with inclusion cysts, concurrent inflammation was minimal and characterized by necrotic heterophils within cyst lumens and few scattered heterophils, macrophages, multinucleated giant cells, lymphocytes, and/or plasma cells. In 10 cases (cases 12, 13, 15, 18, 19, 20, 23, 25, 28, 29) no inflammatory cells were present although in 3 of these cases (cases 13, 20, 25) evaluated submissions were from biopsy material. There were no distinct granulomas.
Fungi were not detected within coelomic organs. Skin lesions were noted in 6 necropsy submissions with fungal shell lesions (cases 5, 8, 10, 19, 24, 26). Morphologically consistent fungi were only noted in the skin in 2 cases (cases 8, 26). Other skin lesions included coagulative necrosis associated with bacterial sepsis (case 24) and ulcerative dermatitis associated with bacteria (cases 5, 19) or 10 to 20 µm diameter fungi not consistent with E. testavorans (case 10).
In cases for which inclusion cysts were not observed, morphologic changes were within the spectrum of shell lesions previously reported for aquatic turtles. These included acute to chronic bacterial infections (n = 8, cases 30, 32, 33, 39, 40, 43, 47, 50) characterized by ulceration, necrosis, and predominantly heterophilic inflammation. With increasing chronicity, additional changes indicative of repair such as fibrosis, bony remodeling, squamous metaplasia, and hyperkeratosis were noted. In 2 cases (cases 34, 44), lesions were restricted to the stratum corneum and consisted of hyperkeratosis, with superficial layers of scute keratin colonized by mixed organisms including mixed bacteria, yeast, and fungal hyphae not morphologically consistent with E. testavorans. In one case (case 38), there was ulceration and osteonecrosis associated with fungal hyphae not morphologically consistent with E. testavorans. Three additional cases (cases 36, 37, 45) had acute ulcerative and necrotizing lesions where an infectious etiology was suspected, but organisms were not detected within the examined shell sections.
Noninfectious causes of shell lesions included fibrous osteodystrophy resulting from renal or nutritional secondary hyperparathyroidism (cases 41, 42, 49), healed defects resulting from previous damage of unknown etiology (cases 31, 35), and acute traumatic injury (case 46).
Fungal Detection
On GMS-stained sections, fungal elements morphologically consistent with E. testavorans were identified in 29 of 50 cases (58%, cases 1–29) within the squamous epithelium lining inclusion cysts and among keratin debris (Fig. 6). Arthroconidia were not noted. In addition to lesional E. testavorans hyphae, one case also had yeast (case 22) and another (case 28) had a second form of hyphae that were aseptate, larger than E. testavorans (up to 25 µm diameter), and adherent to superficial scute keratin but not present within cysts. These superficial fungi were interpreted as environmental contamination. Similar superficial environmental hyphae or yeasts were also noted in 6 cases lacking inclusion cysts or E. testavorans hyphae (cases 34, 35, 37, 38, 44, 46). All but 2 cases (cases 13, 21), which had morphologically consistent hyphae also had epithelial inclusion cysts. Both cases 13 and 21 were submissions from biopsies comprised of small sections of shell tissue.
E. testavorans was confirmed via PCR and/or qPCR in 17 cases (cases 1–17). ITS PCR and sequencing were positive in fresh or frozen tissues for E. testavorans in 15 cases (cases 1–12, 14, 16, 17). qPCR on FFPE tissues was positive in only 4 cases (cases 11, 12, 13, 15) including 2 cases with morphologically consistent fungi but from which culture and ITS PCR was negative (cases 11, 12). For 13 cases (cases 1–10, 14, 16, 17) in which E. testavorans was detected via ITS PCR and/or culture from fresh or frozen tissue, the organism could not be detected via qPCR of FFPE tissue. Fungal culture isolated E. testavorans in 6 of 15 (40%) cases (cases 1–6). Identity of fungal isolates was confirmed with ITS PCR and sequencing. In one case (case 8), fungal culture yielded growth of Fusarium sp.; however, E. testavorans was detected via PCR of lesional tissue. For all cases in which E. testavorans was detected via molecular modalities and/or culture, morphologically consistent hyphae were also detected in GMS-stained sections of lesional tissue.
Statistical Analyses
A summary of univariate logistic regression models is presented in Table 2. There was no significant association between age and infection. The results of univariate analyses indicated that multiple histologic features were significantly associated with E. testavorans infection. When infection was defined by the presence of hyphae in GMS sections, inclusion cysts (P < .0001), squamous metaplasia (P = .0011), hyperkeratosis (P = .0105), ulceration (P = .0035), osteonecrosis (P = .0011), and inflammation (P = .0016) were significantly associated with E. testavorans infection. Using the more stringent molecular (PCR) detection criterion, only inclusion cysts (P = .0023), osteonecrosis (P = .0428), and inflammation (P = .0428) were significantly associated with E. testavorans infection. The best fit final multivariant logistic regression model contained only inclusion cysts irrespective of how infection was defined. Because there were larger numbers of Actinemys marmorata than other species within the exposed group, to ensure results were not influenced by this bias in our study population, significance of inclusion cysts was confirmed using a multivariate model that excluded this species.
Univariate Logistic Regression Models for the Association of E. Testavorans Infection Detected via the Presence of Hyphae in GMS Stained Sections or PCR Confirmation and Presence of Shell Lesionsa.
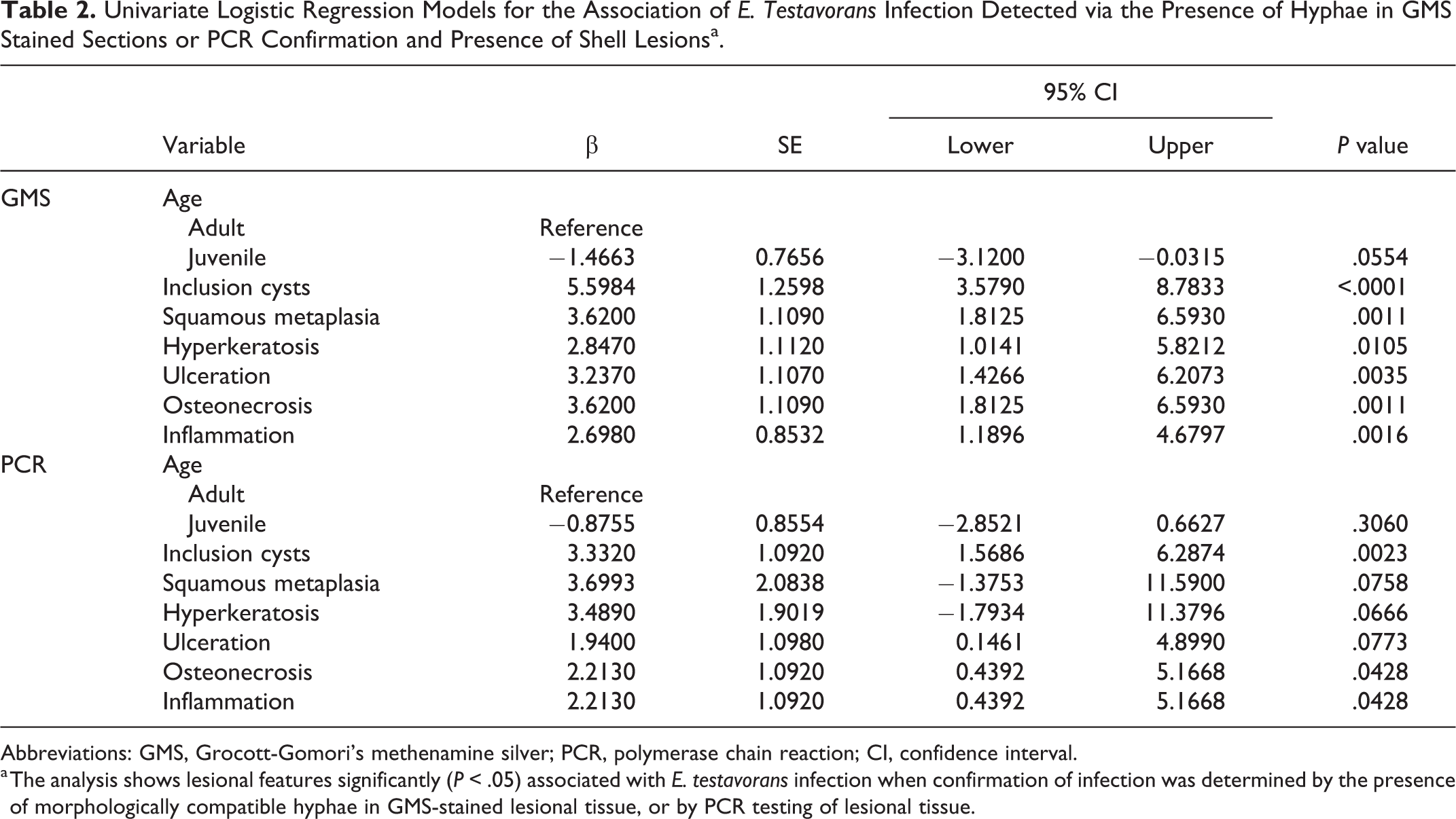
Abbreviations: GMS, Grocott-Gomori’s methenamine silver; PCR, polymerase chain reaction; CI, confidence interval.
a The analysis shows lesional features significantly (P < .05) associated with E. testavorans infection when confirmation of infection was determined by the presence of morphologically compatible hyphae in GMS-stained lesional tissue, or by PCR testing of lesional tissue.
Discussion
Emydomyces testavorans is a newly described onygenalean fungus that has been isolated from shell lesions of freshwater aquatic turtles. 29 While anecdotal evidence has suggested that infection may be associated with unique shell lesions, namely, epithelial inclusion cysts, the purpose of this descriptive retrospective study was to describe the shell lesions associated with the fungus. In the present analysis, epithelial inclusion cysts were indeed the most striking and consistent lesions significantly associated with E. testavorans infection within all multivariate models. Although 2 cases with confirmed infection did not present with inclusion cysts, both cases were biopsy submissions. While containing all shell layers, sections were small and fragmented, which may have precluded identification of inclusion cyst architecture. It is also notable that one turtle (case 48) had inclusion cysts in the absence of confirmed E. testavorans infection. Lesions in this turtle were associated with mycobacterial infection, and thus epithelial inclusion cysts are highly suggestive of, though not necessarily pathognomonic for, E. testavorans infection in chelonians.
Shell disease is a significant cause of morbidity and mortality in aquatic chelonians. 4,21 Similar cysts lined by thick cornified epithelium with luminal keratin, necrotic bone, and cellular debris have been reported previously in river cooters (Pseudemys concinna) and yellow-bellied turtles (Trachemys scripta). 7,16 In these cases, cysts were presumed to be a chronic lesion and indicative of a healing process with cysts walling off and expelling the necrotic bone. However, cysts were less extensive and less numerous than those noted herein. In the current study, cysts were the defining characteristic and did not appear to be part of a continuum. Similar cysts were also noted in desert tortoises as part of a disease presumed associated with dyskeratosis. 10 Although fungi were described within lesions in both case series, they were presumed contaminants and a definitive cause was not confirmed. Whether these represent previous, unrecognized cases of E. testavorans infection is unknown. Isolation of E. testavorans is enhanced by selective media containing β-keratin but remains difficult as evidenced by a 40% success rate in this study. 29 Growth of E. testavorans is slow and bacterial overgrowth is common. Although in our case series, hyphae morphologically consistent with E. testavorans were typically easy to identify on GMS-stained sections, fungal morphology was similar to that of other environmental fungi and thus their presence may have been previously discounted. In the cases described herein, molecular techniques helped identify the fungi.
Reports of fungal causes for shell infections are rare. An Aldabra tortoise (Aldabrachelys gigantea) with deep flaking lesions and necrotic bone had brown-pigmented fungi within keratin and PCR sequence consistent with Exophiala oligosperma. 26 Purpureocillium lilacinum (formerly Paecilomyces lilacinus) was cultured from shell and systemic lesions from Fly River turtles (Carettochelys insculpta). 13 Fusarium incarnatum (formerly F. semitectum) was cultured from scute lesions on Texas tortoises (Gopherus berlandieri) and Hermann’s tortoises (Testudo hermanni). 17,22 Finally, an onygenalean fungus, Aphanoascella galapagosensis, has been isolated from a Galapagos tortoise (Chelonoidis nigra microphyes) with carapace keratitis. 27 Inclusion cysts were not described in any of these cases.
The present study also associated squamous metaplasia, hyperkeratosis, osteonecrosis, and inflammation with E. testavorans infection in univariate models when infection was determined by GMS staining. Despite a statistically significant association with E. testavorans, these features were also present in cases of shell lesions due to other infectious and noninfectious causes and were deemed nonspecific. Ulceration, squamous metaplasia, and hyperkeratosis were not significant when considering the more stringent criterion of PCR-based detection. A variety of gross lesions were also described in affected shells including ulceration, pitting, discoloration, flaking, and increased shell pliability. These lesions were noted in both infected and noninfected cases. Flaking is likely associated with the histologic finding of hyperkeratosis. Gross description of these lesions was obtained from reports and could not be independently assessed for this study. To avoid bias, gross lesions were not included in the statistical analysis.
The 2 sets of statistical analyses were conducted due to limits on fungal detection imposed by the retrospective nature of this study. Only FFPE sections were available for PCR testing from some turtles. Both formalin fixation and formic acid decalcification necessary for histologic processing of turtle shells are known to fragment nucleic acids, 1,12 resulting in lower yields of amplifiable DNA. A sensitive real-time quantitative PCR (qPCR) was developed to specifically detect minute quantities of E. testavorans. However, positive detection was limited to only 4 cases, 3 of which (cases 12, 13, 15) were small(er) biopsy submissions, processed after only a short time in formic acid, presumably with less resultant DNA degradation. Thus, qPCR, or indeed any molecular-based testing including in situ hybridization, may result in false negatives when applied to FFPE shell tissues. Conversely, GMS-based confirmation of infection may result in false positives, as many fungi can have similar morphologic features in histologic section. Irrespective of the criteria for a positive case, inclusion cysts were significantly associated with E. testavorans infection, and their presence should prompt a thorough search for the fungus.
This study was able to demonstrate an association between E. testavorans infection and specific shell lesions, but the retrospective nature has limitations and causation remains to be determined. Temporal association with initial infection is unknown and it is not yet known how lesions develop over time in this infection. Given the general lack of understanding of inflammatory responses and wound healing in turtle shells and the number of previous case series describing shell disease of unknown cause in other chelonians, 7,10,16 following the lesion progression over time may help with future investigations into shell diseases in other populations and with other pathogens. Additionally, because other bacteria and fungi were noted in some of these cases, it is uncertain whether E. testavorans can cause disease on its own or is facilitated by other risk factors or co-pathogens. In experimental Nannizziopsis dermatitidis infections, previous trauma or abrasions facilitated infection but was not required. 18 Similarly, experimental studies of O. ophiodiicola have confirmed the pathogenic nature of this related fungus in the absence of previous trauma, though lesions developed at a higher rate in abraded inoculation sites. 15 Further research into the pathogenesis of E. testavorans infection and general studies into wound healing in the turtle shell are needed.
The present study found that E. testavorans infection is associated with the development of cystic structures within the shells of freshwater aquatic turtles that are lined by keratinized stratified squamous epithelium and contain necrotic bone and keratin debris. This study limited the analysis to shell lesions as this was the site where the fungus was identified histologically and its presence was confirmed by PCR or culture. Prospective studies to assess potential involvement of other keratinizing epithelium (eg, skin, nails) are needed. The International Union for Conservation of Nature Red List (www.iucnredlist.org) currently classifies 104 species of aquatic and semi-aquatic chelonians as vulnerable or endangered. Of the species with confirmed E. testavorans infection in the present study, three are listed as endangered (Actinemys marmorata, Clemmys guttata, and Heosemys spinosa), and two are listed as vulnerable (Malaclemys terrapin and Podocnemis unifilis). In addition to cases from turtles housed under human care, E. testavorans and associated lesions were identified in free-living chelonians from both the west and east coasts of the United States, suggesting a potentially broad geographic distribution. Although this fungus has only recently been described, it may represent a significant, and previously overlooked, disease threat to both free-living turtles and those under human care.
Footnotes
Acknowledgements
We thank the Washington Department of Fish and Wildlife (WDFW), John G. Shedd Aquarium, Chicago Zoological Society’s Brookfield Zoo, Lincoln Park Zoo, South Florida Wildlife Center, and Cedar Key Nature Coast Biological Station for providing source biomaterials used in this study. We thank WDFW Biologists Stefanie Bergh, Carly Wickman, Emily Butler, Brian Murphie and WDFW Herpetologist Lisa Hallock. We thank the pathologists and residents of the University of Illinois Zoological Pathology Program for diagnostic assistance with these cases and the Histology Laboratory of the University of Illinois Veterinary Diagnostic Laboratory and the University of Florida College of Veterinary Medicine for excellent technical assistance. We thank Anne Kimmerlein and Gretchen Anchor for assistance with the statistical analysis. Financial support for portions of this study was generously provided by the Morris Animal Foundation through a Fellowship Training Grant (D16ZO-414). This study has not been reviewed or endorsed by the Foundation, and the views expressed in such publication or presentation do not necessarily reflect the views of the Foundation, its officers, directors, affiliates or agents.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for portions of this study was generously provided by the Morris Animal Foundation through a Fellowship Training Grant (D16ZO-414).
