Abstract
Mycobacteriosis is infrequently reported in free-ranging sea turtles. Nontuberculous Mycobacterium haemophilum was identified as the causative agent of disseminated mycobacteriosis in a juvenile leatherback turtle (Dermochelys coriacea) that was found stranded on the Atlantic coast of Florida. Disseminated granulomatous inflammation was identified histologically, most notably affecting the nervous system. Identification of mycobacterial infection was based on cytologic, molecular, histologic, and microbiologic methods. Among stranded sea turtles received for diagnostic evaluation from the Atlantic and Gulf of Mexico coasts of the United States between 2004 and 2015, the diagnosis of mycobacteriosis was overrepresented in stranded oceanic-phase juveniles compared with larger size classes, which suggests potential differences in susceptibility or exposure among different life phases in this region. We describe M. haemophilum in a sea turtle, which contributes to the knowledge of diseases of small juvenile sea turtles, an especially cryptic life phase of the leatherback turtle.
Sea turtles have complex life histories that span from nesting beaches to the open ocean. Following hatching and a frenzied swim to deep water, juveniles of most well-studied species spend the initial period of their lives in surface-pelagic habitat and later recruit into areas closer to shore where they mature. In contrast, leatherback turtles (Dermochelys coriacea) and olive ridley turtles (syn. Pacific ridley turtle, Lepidochelys olivacea) largely occur farther from shore in continental shelf and pelagic waters.2,7 In North America and other regions, smaller juveniles of all species (including those ~10–25 cm in carapace length) are primarily found at greater distances from shore, and thus are encountered as beach-cast strandings less frequently than other size classes. Consequently, there is a paucity of information available on diseases that affect small juvenile sea turtles in the wild.
As in other animals, mycobacteria are well-recognized potential pathogens of reptiles and have been reported in various species, primarily in captive individuals. 14 Of the few published reports of mycobacteriosis in sea turtles, most have been in captive-reared animals.3,8 Mycobacterium chelonae was isolated from osteoarthritis in a wild Kemp’s ridley (Lepidochelys kempii) after 4 months of captivity for rehabilitation. 9 Disseminated mycobacteriosis, also identified as M. chelonae, was described in a stranded loggerhead turtle (Caretta caretta) found on the Adriatic coast of Italy. 17 To our knowledge, the latter case is the only published report of mycobacteriosis in a free-ranging sea turtle that was not associated with rehabilitation or captivity.
A 20.3-cm (straight carapace length, SCL) female juvenile leatherback turtle was found stranded in February 2013 on the Atlantic coast of Florida (26.365597, –80.068247). The SCL measurement was from the nuchal notch to the tip of the pygal. The turtle was moribund and was transported to a rehabilitation center where it died within 48 h. A routine postmortem examination was conducted. The turtle was emaciated, and the gastrointestinal tract was devoid of digesta. The cranial aspect of the left lung was effaced by granulomatous inflammation. There also was abundant opaque mucoid material within the small intestine, and multifocal ulceration of the proximal colon that affected ~10% of the mucosa. Cytology of an enteric swab revealed marked heterophilic and histiocytic enteritis with macrophages containing multiple negative-staining bacilli (Fig. 1A) that were positive with Ziehl–Neelsen acid-fast stain. All other organs were unremarkable grossly. Tissues were fixed in 10% neutral buffered formalin and processed for histology by routine methods. Sections were stained with hematoxylin and eosin and using the Fite acid-fast method.
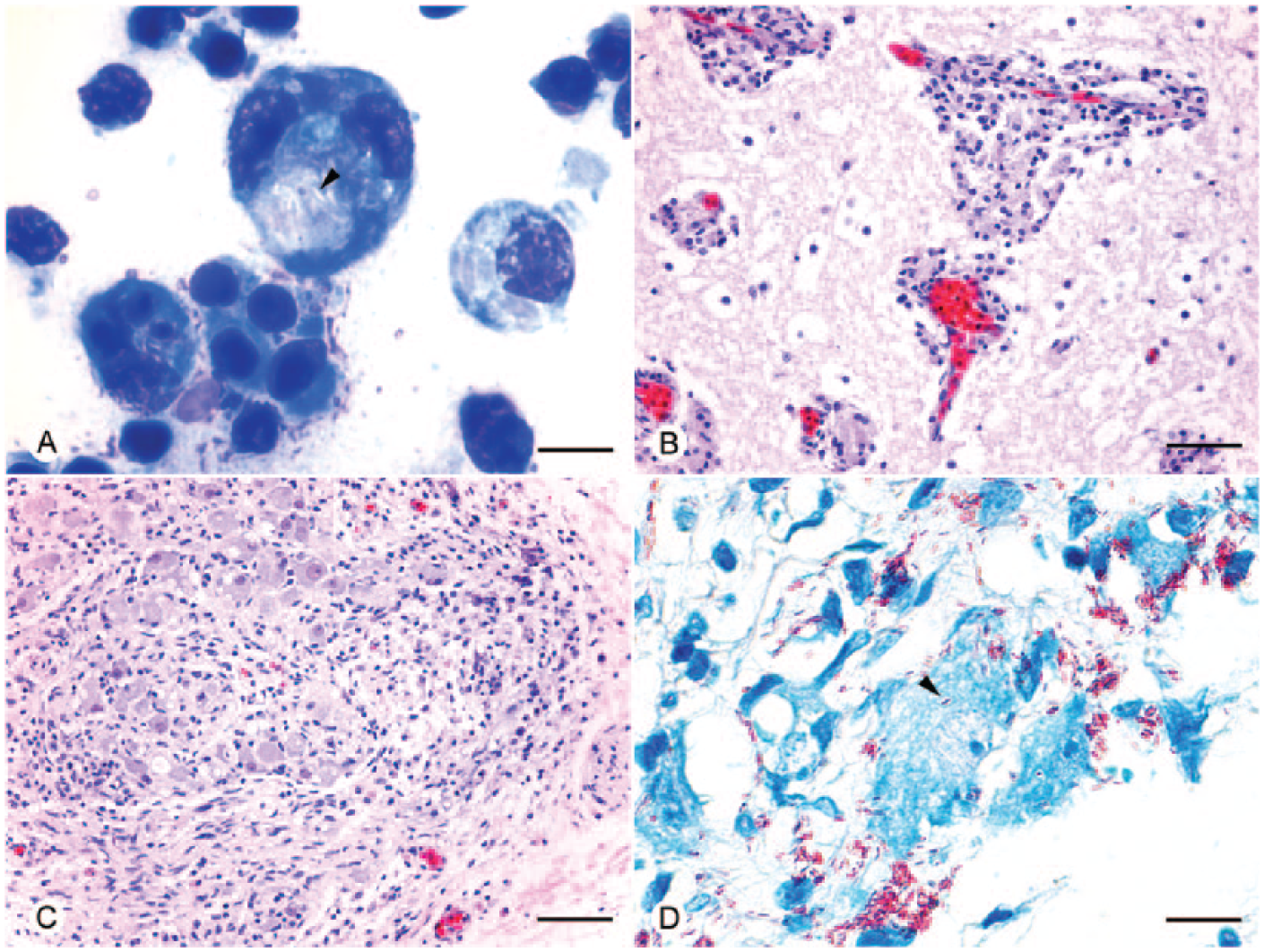
Histologic and cytologic findings in a wild juvenile leatherback sea turtle (Dermochelys coriacea).
The predominant histologic finding was disseminated granulomatous inflammation within most examined tissues, including the nervous system, lungs, liver, spleen, kidneys, small intestine, pancreas, thymus, and bone marrow. Lymphocytes, macrophages, and fewer heterophils expanded the meninges and formed perivascular cuffs throughout the brain and spinal cord (Fig. 1B). Cranial and spinal nerves, ganglia, and peripheral nerves were similarly affected (Fig. 1C). Intracellular acid-fast bacilli were present within macrophages in all lesions, as well as within circulating monocytes. Organisms were especially numerous within inflamed ganglia and also appeared to be within neurons and possibly Schwann cells and nerve fibers (Fig. 1D). The chronic pulmonary granuloma contained septate, branching fungal hyphae with parallel walls (ascomycete species). The predominant organisms within the colonic ulcers were non–acid-fast, gram-negative bacilli, although intrahistiocytic acid-fast organisms were also observed.
DNA was extracted from a frozen sample of spinal cord using a commercial extraction kit. a The partial RNA polymerase B subunit (RpoB) and 16S-23S rRNA gene spacer (IGS) regions were amplified and sequenced using previously described methods.15,20,21 Bidirectional sequencing of amplicons was performed using automated DNA sequencers, b and results were compared with sequences available in GenBank. Nucleotide sequences for both genetic targets (GenBank accessions KM230419-22) were obtained from the spinal cord, and were 100% homologous with M. haemophilum (GenBank accessions DQ851570.1 and NR_104888.1).
Mycobacterium haemophilum has an unusual growth requirement for iron, often provided as blood, hemoglobin, or hemin. 16 In light of sequence-based detection of M. haemophilum directly from spinal cord, a sample of spinal cord was collected from the frozen carcass using sterile technique and was inoculated onto blood agar and colistin and nalidixic acid (CNA) agar plates, c rather than more typical media used for other mycobacterial species, and incubated at 27.6°C as described previously. 5 Subculture onto CNA agar yielded pinpoint colonies composed of acid-fast bacilli after 3 months of growth. DNA was extracted from colonies, and polymerase chain reaction (PCR) amplification and sequencing was performed as had been done for the spinal cord sample. The RpoB and IGS sequences were identical to those amplified from the spinal cord.
Diagnostic case submissions and diagnoses for sea turtles examined by one of the authors (BA Stacy) from 2004 through 2015 were reviewed to identify previous cases of mycobacteriosis in stranded sea turtles found on the Gulf of Mexico and Atlantic coasts of the United States. All turtles in which the lung, liver, and spleen were examined by histology were considered because mycobacterial lesions are typically observed in 1 or more of these organs in reptiles. 14 Cases were excluded if infection occurred during rehabilitation and was not clearly present at the time of stranding. Five cases with lesions and acid-fast bacilli consistent with mycobacteriosis were identified that met these criteria, 4 of which (2 Kemp’s ridley turtles; 1 hawksbill sea turtle, Eretmochelys imbricata; and the leatherback of our report) were <25 cm SCL. These 4 cases accounted for 12% of 33 submissions of turtles of this size as compared to only 1 of 228 (0.4%) of turtles >25 cm. Mycobacterium was only identified to species—using the same methods reported herein—for the larger turtle, a 68-cm (SCL) loggerhead (Caretta caretta) with severe pneumonia infected with M. chelonae.
Our report describes M. haemophilum in a sea turtle and a case of neurologic involvement in a reptile. Two cases have been described in snakes, including granulomatous pneumonia in a royal python (Python regius) and disseminated infection in a trinket snake (Elaphe frenata).11,25 In humans, M. haemophilum causes disease primarily in immunocompromised individuals and rarely in healthy children, and is mainly associated with cutaneous infections. 16 Infections involve the extremities primarily, which is attributed to lower optimal temperature for growth of M. haemophilum. 16 Similarly, the lower body temperatures of ectotherms may contribute to disseminated infections observed in reptiles and fish.24,25 Central nervous system infection is relatively rare in humans, 1 and has also been reported in zebrafish (Danio rerio). 24 Involvement of peripheral nerves was an interesting aspect of infection in the leatherback turtle and is similar to infections by Mycobacterium leprae, which is closely related to M. haemophilum. 16 Rare cases of M. haemophilum infection of peripheral nerves resembling leprosy have been reported in humans.4,13
The leatherback described in our report had multiple indications of ill health, including emaciation and concurrent fungal pneumonia, suggesting that infection most likely was opportunistic. An underlying cause of debilitation was not identified. Juvenile sea turtles undoubtedly face many challenges related to environmental conditions, fulfilling energetic needs, predator avoidance, and other threats. Stranding occurred during the winter, thus low ambient temperature also could have been a factor if this turtle was within colder inshore waters for a prolonged period. Leatherbacks <100 cm are thought to occur only in waters warmer than 26°C. 6 A buoy ~25 km south of the location where the turtle was found recorded water temperatures as low as 11.4°C during the months prior to stranding (Station LKWF1-8722670, http://www.ndbc.noaa.gov/, accessed September 11, 2015).
Overrepresentation of stranded small juvenile sea turtles with mycobacteriosis among diagnostic cases is interesting and requires further investigation. Nonrandomized sampling, which is often inherent to stranding-based studies, and a relatively low sample size of examined turtles <25 cm are acknowledged caveats and potential sources of bias. Nonetheless, disparity in diagnosis among size classes is suggestive of possible differences in susceptibility or exposure, at least within the Gulf of Mexico and western Atlantic Ocean. No cases of mycobacteriosis were reported in a small number (n ≤ 10) of juvenile turtles <25 cm in a larger review of postmortem findings in stranded turtles found in the Canary Islands. 19
Mycobacterial disease in reptiles is generally attributed to predisposing factors. 14 The overrepresentation of mycobacteriosis in captive reptiles may reflect both husbandry-related disorders as well as the far greater numbers of captive animals examined by diagnosticians. Virtually nothing is known at this time about the epizootiology of mycobacterial infections in free-ranging reptiles. In the sea turtle cases discussed herein, we do not know whether infection occurred within pelagic habitat or after turtles became debilitated closer to shore prior to stranding. On the Atlantic coast of the United States, strandings are most representative of morbidity and mortality occurring closer to shore; predominantly larger juveniles and subadults are normally found in these areas. 10 If mycobacteriosis in sea turtles was simply a matter of nearshore exposure among susceptible individuals regardless of size and maturity, it is expected that more cases would be encountered among larger stranded turtles, which have various conditions that may predispose to mycobacterial infections, such as emaciation, hypothermia, traumatic injuries, and various chronic diseases.
Mycobacterium haemophilum is not well studied in the environment. Although it is hypothesized to be present in aquatic biofilm reservoirs, many culture-based studies have failed to identify it because of its specific nutrient requirements for growth. 16 The current observation of infection in a wild sea turtle and in a previous human case following injury from contact with coral indicates that this organism occurs in the marine environment. 22 The public health threat posed by mycobacteria is most recognizable among well-known diseases such as tuberculosis and leprosy. However, reports such as those described above outline the importance of atypical mycobacterial species. There is increasing evidence to suggest that water aids in the transmission of these organisms, and several reports implicate mycobacterial infection from exposure to natural water sources.12,18,22,23
Specific identification of Mycobacterium species associated with disease in wild sea turtles is important to understanding infections in free-ranging animals. We were unable to identify organisms in most of the earlier cases because of lack of suitable materials for culture or PCR. Without prior knowledge from molecular methods of identification of the species and specific media growth requirements, culture likely would have been unsuccessful. In light of the observations included in this report, we recommend that frozen samples of major organs (including spleen and nervous system) and any granulomatous lesions be collected routinely during postmortem examination of small juvenile sea turtles, especially those that are found weak or underweight.
Footnotes
Acknowledgements
We thank Linda Archer and Natalie Steckler for assistance with molecular testing, and Rebecca Richardson, Annie Barbe, Robert Bowden, and Cindy Muralles for their help with microbiologic culture. We are grateful to the Florida Fish and Wildlife Conservation Commission, participants of the Sea Turtle Stranding and Salvage Network, and the Gumbo Limbo Nature Center for providing animal response and care.
Author’s contributions
K Donnelly contributed to conception and design of the manuscript; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. TB Waltzek and JFX Wellehan Jr contributed to conception and design of the manuscript, and contributed to acquisition, analysis, and interpretation of data. NI Stacy contributed to analysis and interpretation of data. M Chadam contributed to acquisition of data. BA Stacy contributed to acquisition, analysis, and interpretation of data, and drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
DNeasy blood and tissue kit, Qiagen Inc., Valencia, CA.
b.
ABI 3130, Applied Biosystems Inc., Foster City, CA.
c.
Hardy Diagnostics, Santa Maria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
