Abstract
Malignant catarrhal fever (MCF) is a sporadic, generally fatal disease caused by gammaherpesviruses in susceptible dead-end hosts. A key pathological process is systemic vasculitis in which productively infected cytotoxic T cells play a major role. Nonetheless, the pathogenesis of MCF vasculitis is not yet clear. We hypothesized that it develops due to an interaction between virus-infected cells and immune cells, and we undertook a retrospective in situ study on the rete mirabile arteries of confirmed ovine gammaherpesvirus-2 (OvHV-2)-associated MCF cases in cattle, buffalo, and bison. Our results suggest that the arteritis develops from an adventitial infiltration of inflammatory cells from the vasa vasorum, and recruitment of leukocytes from the arterial lumen that leads to a superimposed infiltration of the intima and media that can result in chronic changes including neointimal proliferation. We found macrophages and T cells to be the dominant infiltrating cells, and both could proliferate locally. Using RNA in situ hybridization and immunohistology, we showed that the process is accompanied by widespread viral infection, not only in infiltrating leukocytes but also in vascular endothelial cells, medial smooth muscle cells, and adventitial fibroblasts. Our results suggest that OvHV-2-infected T cells, monocytes, and locally proliferating macrophages contribute to the vasculitis in MCF. The initial trigger or insult that leads to leukocyte recruitment and activation is not yet known, but there is evidence that latently infected, activated endothelial cells play a role in this. Activated macrophages might then release the necessary pro-inflammatory mediators and, eventually, induce the characteristic vascular changes.
Malignant catarrhal fever (MCF) is a generally fatal disease in a wide range of susceptible hosts including ungulates of the order Artiodactyla such as cattle, water buffalo, bison, deer, antelope, elk, and reindeer. 25,37,42,56 The disease occasionally affects pigs 4,32 and can be experimentally induced in rabbits, guinea pigs, and hamsters. 5,7,8,13,21 The causative agents of MCF are herpesviruses of the subfamily Gammaherpesvirinae, genus Macavirus, of which at least 6 are pathogenic under natural conditions. Among these, ovine gammaherpesvirus-2 (OvHV-2) is the most relevant globally. It is endemic in sheep and causes sheep-associated MCF in various ruminant species, 26 and is the virus reported in association with MCF outbreaks in pigs. 4,32 Alcelaphine gammaherpesvirus 1 (AlHV-1) is endemic in wildebeest and causes significant outbreaks of wildebeest-associated MCF of domestic cattle in sub-Saharan Africa. 42 Caprine gammaherpesvirus-2 (CpHV-2) is endemic in domestic goats and has been reported in MCF in 2 deer species and in a domestic water buffalo in Switzerland. 10,12,23,29 White-tailed deer and red brocket deer are susceptible to MCFV-WTD (malignant catarrhal fever virus–white-tailed deer) that is carried by domestic goats. Ibex MCF virus (Ibex-MCFV) has been identified in bongo and anoa with MCF and is carried by the nubian ibex. 17 Alcelaphine herpesvirus 2 (AlHV-2) is carried by hartebeest and topi and has been reported as a cause of MCF in barbary red deer and experimentally in bison. 24,25
MCF viruses are carried by the above-listed reservoir species (so-called “host species”). These γ-herpesviruses have evolved with their host species, where infection generally does not cause disease, though recent studies based on natural cases and experimental infection have confirmed that sheep can develop OvHV-2-associated MCF. 27,50,51 Lambs usually become infected soon after birth, via horizontal transmission. 2,56 Natural transmission studies in sheep suggested that white blood cells are the first cells to carry infectious virus after infection, 19 and experimental infection studies identified lytically infected lower airway epithelial cells as the main source of infectious virus early (7 days) after infection. 66 OvHV-2-infected healthy lambs shed infectious virus intermittently, mainly through nasal secretions, but also via the alimentary and urogenital tracts. 15,19,28 Infected animals generally develop a persistent latent infection, with white blood cells as the reservoir. 20,67 Further studies identified the infected leukocytes as CD4+ and CD8+ T cells. 36,58 Viral reactivation can then result in transmission to susceptible species. 37,56
When MCF viruses are horizontally transmitted to susceptible hosts, MCF can develop. In natural infections of cattle, an incubation period of 3 weeks to 5 months has been suggested, after which the ensuing lymphoid proliferation and infiltration with widespread vascular and epithelial lesions can result in the generally fatal multisystemic disease, with different clinical forms. 30,31,65,68 MCF in cattle is usually sporadic and mainly affects single animals; 56 however, serious outbreaks have been reported in cattle and more often in certain species of deer and in bison. 1,6,53 Bison appear to be particularly susceptible to infection, which is reflected in higher numbers and rapid death of affected animals. 6,42,45 Susceptible hosts do not shed the virus, at which point the viral cycle terminates. 68 It is believed that in these, disease is the result of direct virus-cell interactions or a dysregulated immune-mediated response against infected cells. 61
The diagnosis of MCF is based on clinical signs, histopathological features, and the detection of viral DNA by PCR. 56 In affected species, the main gross findings are erosions and ulcerations in the upper respiratory tract and gastrointestinal tract, keratoconjunctivitis, and lymphadenomegaly. 31,42,56,68 It has been shown that the viral load increases slowly in the early phases of the disease; after the onset of clinical signs, high viral loads are measured. 4,6 This is thought to be the result of high viral replication or the proliferation of virus-infected T cells, which can in turn lead to the observed lymphocytic hyperplasia. 35,52,65,68 Histological examination shows that the mucosal lesions (ie, the necrosis of the respiratory, alimentary, and urinary epithelium) are the consequence of a systemic vasculitis, which also involves vessels in other tissues such as the brain. 30,42 The vasculitis is a key pathological process of MCF in all affected species. Arteries are predominantly affected, exhibiting mononuclear leukocyte infiltration of the media and adventitia. In addition, myocyte necrosis in the tunica media, endothelial cell hyperplasia, intimal thickening, and necrotizing vasculitis have been described. 37,42,53,68 The most in-depth description of MCF vasculitis results from an ultrastructural study published in 1980 and undertaken on rete mirabile arteries of experimentally infected calves. It identified the infiltrating mononuclear cells as lymphocytes and lymphoblasts, with fewer monocytes and macrophages. 30 Later immunohistological examinations showed a dominance of T cells, with abundant activated cytotoxic and fewer helper T cells, and a substantial proportion of macrophages, whereas the numbers of other leukocytes were negligible. 39,40 The vasculitis is thought to be the consequence of immune dysregulation that results in accumulation of the lymphocytes. 39,40,56,61,69 A recent study using RNA in situ hybridization confirmed productive OvHV-2 infection of lymphocytes in the lesions. 49
The OvHV-2 genome has been cloned and sequenced and the functions of a number of genes assigned. Many of the genes have direct homologues in other herpesviruses whereas some are unique to OvHV-2. 18,64 Like all γ-herpesviruses, OvHV-2 has a productive replication cycle in which most virus genes are expressed and viral particles are produced, as well as a latency phase where a restricted set of genes are expressed. 55 The genes expressed during latency have been defined and include ORF73 and Ov2.5. ORF73 encodes a nuclear protein (oLANA) that is a homologue of the KSHV LANA protein, and Ov2.5 encodes ovIL-10 that is a functional homologue of cellular IL-10. 3,22 These latency-associated genes are also expressed during the productive cycle and so act as markers for all OvHV-2-infected cells.
So far, despite extensive research on the molecular aspects of the disease and its variable clinical presentation, the pathogenesis of MCF and in particular of the vasculitis is still poorly understood. We hypothesized that the vasculitis develops due to an interaction between virus-infected cells and immune cells in the susceptible species. This led us to undertake a retrospective in situ study on MCF vasculitis, making use of confirmed OvHV-2-associated natural MCF cases in cattle, water buffalo, and bison. We focused on the arteries in the rete mirabile as these have previously been shown to exhibit changes representative of the vascular lesions in other organs. 30,45 Aiming to gain further knowledge on the pathological processes underlying and maintaining the vasculitis in MCF, we investigated the inflammatory and remodeling processes in the context of virus latency using mRNA for ovIL-10 and oLANA protein as the markers.
Material and Methods
Animals and Tissues
The study was performed on 35 OvHV-2-associated MCF cases (26 cattle of various breeds, 7 water buffalo, 2 bison). Animals ranged from 6 months to 12 years in age (2.67 ± 2.45 years); 33 were female and 3 were male (Supplemental Table S1). Cases had been retrieved from the database (2000–2015) of the Institute of Veterinary Pathology, Vetsuisse Faculty Zurich, and the Institute of Animal Pathology, Vetsuisse Faculty Berne, where animals had undergone a full diagnostic post mortem examination upon the owners’ request.
The diagnosis of MCF and the involvement of OvHV-2 as the causative agent was confirmed on the basis of the gross and histopathological findings, coupled with the detection of OvHV-2 by quantitative polymerase chain reaction (qPCR) on EDTA (ethylenediaminetetraacetic acid) blood samples (Institute of Virology, Vetsuisse Faculty, University of Zurich), and/or qPCR (see below) on formalin-fixed, paraffin-embedded tissue specimens as part of the present study. Furthermore, Ov2.5 mRNA (coding for OvHV-2 viral IL-10) and OvHV-2 latency-associated nuclear antigen (oLANA) protein expression were demonstrated within tissue sections of rete mirabile arteries by RNA in situ hybridization (RNA-ISH) and immunohistochemistry, respectively (see below).
From all animals, the paraffin blocks containing samples of the rete mirabile were retrieved. The tissue had been fixed in 10% buffered formalin for at least 24 hours, followed by trimming and routine paraffin wax embedding.
Quantitative Polymerase Chain Reaction for OvHV-2
The qPCR for OvHV-2 served to confirm the presence of the virus in the diseased animals, using previously reported primer and probe sequences. 63 DNA was extracted from 20 μm-thick paraffin sections of lung, lymph nodes, and/or spleen. Briefly, after deparaffination in a series of xylene and ethanol (100%), tissue lysis was performed by proteinase K digestion at 56 °C for 15 to 18 hours, followed by incubation in a heating block at 90 °C for 1 hour to reverse formalin crosslinking. For DNA extraction, the QIAamp DNA FFPE Tissue Kit (Qiagen) was used, following the manufacturer’s instructions. The DNA was stored at −20 °C until further use.
The qPCR was performed on an Applied Biosystems 7500 fast real-time PCR system using 96-well plates. The reaction volume (20 μl) consisted of 10 µl of Taqman Mastermix 2× (Thermo Fischer Scientific), each 1 µl of forward and reverse primer (10 μM) and 1 µl of probe (5 μM) for OvHV-2 and 12s-ribosomal DNA, 5 µl of nuclease-free water and 2 µl of template DNA (50 ng/μl). Primers and probes were identical to those in a previous publication. 65 Detection of the 12s-ribosomal DNA internal genome served to normalize for DNA variability and contaminants. Thus, the exact viral copy number was normalized and determined at a detection limit ranging from 2 × 107 to 2 × 100 copies/2 μl. The RT-PCR was run with the following cycling conditions: (1) 20 seconds at 95 °C, (2) 3 seconds at 95 °C, (3) 30 seconds at 56 °C, 40 cycles between steps 2 and 3. The data were collected during step 4 of the RT-PCR program.
Histology, Immunohistochemistry, and Immunofluorescence
Consecutive sections (4–5 μm) were prepared from the rete mirabile blocks. They were stained with hematoxylin and eosin and elastica stain for elastic fibers, and subjected to immunohistochemistry, immunofluorescence, and RNA-ISH.
Immunohistochemistry served to characterize the infiltrating leukocytes, that is, T cells (CD3+), B cells (CD20+), macrophages (calprotectin+ and/or Iba-1+), as well as to assess cell death (cleaved caspase 3+) and proliferative processes (Ki67+) in the vessel walls, to further assess the muscle layer (α-smooth muscle actin (α-SMA)+), and to detect viral antigen in the lesions. The latter used a custom-made polyclonal rabbit antibody against the latency-associated nuclear antigen (oLANA), encoded by the ORF73 of OvHV-2. 3,18 Immunolabeling was carried out in a Dako autostainer (Dako), using the horseradish peroxidase method. Antibodies, antigen retrieval and detection methods are listed in Supplemental Table S2. Briefly, after deparaffinization, antigen retrieval for immunohistology was performed for all antigens except for α-SMA, by incubation of the slides with citrate buffer (pH 6) or EDTA buffer (pH 9) at 98 °C for 20 minutes in a pressure cooker. For immunofluorescence, all slides were incubated with EDTA buffer.
For immunohistochemistry, after incubation with the primary antibodies for 60 minutes at room temperature (RT), endogenous peroxidase was blocked by incubation with peroxidase blocking solution (Dako, S2023) for 10 minutes at RT. This was followed by incubation with the matching secondary antibodies and the appropriate detection kits (Supplemental Table S2). Sections were washed with phosphate-buffered saline (pH 8) between each incubation step. Finally, sections were counterstained with hematoxylin for 20 seconds and mounted. Slides incubated with nonimmune serum from the species in which the primary antibody was raised instead of the primary antibodies served as negative controls. A formalin-fixed, paraffin-embedded cell pellet of BJ2586 cells (permanently OvHV-2-infected bovine large granulocyte lymphocyte cell line) served as a positive control for the anti-LANA antibody. 67 Sections from a bovine lymph node served as positive control for cleaved caspase-3, Ki-67, CD3, CD20, Iba-1, and calprotectin, and sections from a rete mirabile of a non-MCF cow as positive control for α-SMA. Sections of the unaltered rete mirabile of non-MCF cattle served as tissue negative controls for Ki-67, CD3, CD20, Iba-1, and anti-OvHV-2 LANA antibodies, and to assess the amount of leukocytes present in the absence of MCF.
For immunofluorescence, endogenous peroxidase was blocked prior to incubation with the first primary antibody overnight or 1 hour at RT, followed by 1 hour incubation with the respective green fluorescence labelled antibody, incubation with the second primary antibody for 1 hour at RT, and 1 hour incubation with the respective red fluorescence labelled antibody (Supplemental Table S2). The final incubation was with DAPI (4′,6-diamidino-2-phenylindole, Novus Biologicals; 1:10 000 in PBS), for 15 minutes at RT. After that, sections were washed twice with distilled water, air dried, and a coverslip placed with FluoreGuard mounting medium (Biosystems).
RNA In Situ Hybridization
RNA-ISH was performed using the RNAscope ISH method (Advanced Cell Diagnostics) and the automated RNAscope 2.5 Detection Reagent Kit (Brown) according to the manufacturer’s protocol. All cases were first tested for the suitability of the tissue (RNA preservation and quality) with an oligoprobe for Bos taurus peptidylprolyl isomerase B (PPIB). Those yielding good PPIB signals were then subjected to RNA-ISH for Ov2.5 mRNA (coding for OvHV-2 viral IL-10; Genbank NC_007646.1). 18 Briefly, sections were heated to 60 °C for 1 hour and subsequently deparaffinized. Permeabilization was achieved by incubating the section in pretreatment solution 1 (RNAscope Hydrogen Peroxide) for 10 minutes at RT, followed by boiling in RNAscope 1× Target Retrieval Reagents solution at 100 °C for 15 minutes and washing in distilled water and ethanol. After digestion with RNAscope Protease Plus for 30 minutes at 40 °C, sections were hybridized with the oligoprobes at 40 °C in a humidity control tray for 2 hours (HybEZ Oven, ACD Advanced Cell Diagnostics). Thereafter a serial amplification with different amplifying solutions (AMP1, AMP2, AMP3, AMP4: alternating 15 minutes and 30 minutes at 40 °C) was performed. Between each incubation step, slides were washed with washing buffer. They were subsequently incubated with AMP5, AMP6, and DAB at RT for 30 and 15 minutes, respectively. Gill’s hematoxylin served to counterstain the sections which were then dehydrated with graded alcohol and xylene and coverslipped. A formalin-fixed, paraffin-embedded pellet of OvHV-2-infected BJ2586 cells served as a positive control. The negative control was consecutive sections incubated accordingly but without including the hybridization step.
Results
MCF Arteritis Is Variable in Extent and Severity, Culminating in Transmural Inflammation With Fibrinoid Necrosis
The rete mirabile of all cases contained arteries with mononuclear cell infiltration of the tunica adventitia that formed a rim around each individual artery and also surrounded the vasa vasorum (Fig. 1). In 7/35 cases (20%), all arteries on the section were affected, otherwise the proportion of affected arteries varied and was in one case as low as 15%; the average was 64%. In 28/35 cases (80%), a proportion of arteries (6% to 100%; average: 40%) also exhibited focal to multifocal infiltration of the media; this was frequently a continuum of the adventitial infiltrate (Figs. 1, 2). In addition, in 26 cases (74%), affected arteries often (affecting 2% to 96% of arteries, average: 33%) had activated endothelial cells and leukocytes that were located immediately beneath the endothelium (Figs. 3, 4); these were also found as part of an infiltrate stretching from the endothelium into the tunica media (Fig. 4). In 19 cases (54%), a proportion of arteries (2% to 95%, average: 28%) also exhibited segmental transmural infiltrations (Figs. 4, 5). The infiltrates in the media led to focal thickening of the wall (Fig. 6) and were sometimes associated with disruption of the media and/or degeneration of smooth muscle cells (Figs. 5a, 5b, and 6). In 10 cases (29%), individual affected vessels exhibited necrosis of the intima, sometimes stretching through to the adventitia, with replacement of the tissue by amorphous eosinophilic material that contained cellular debris and pyknotic nuclei (Fig. 7). In all cases, a proportion of infiltrating leukocytes within arterial walls and the adventitia were found to undergo apoptosis (cleaved caspase 3+; Fig. 8); of these, several were identified as T cells (Supplemental Figure S1). The described features were observed in the rete mirabile of all examined species (cattle, buffalo, bison).
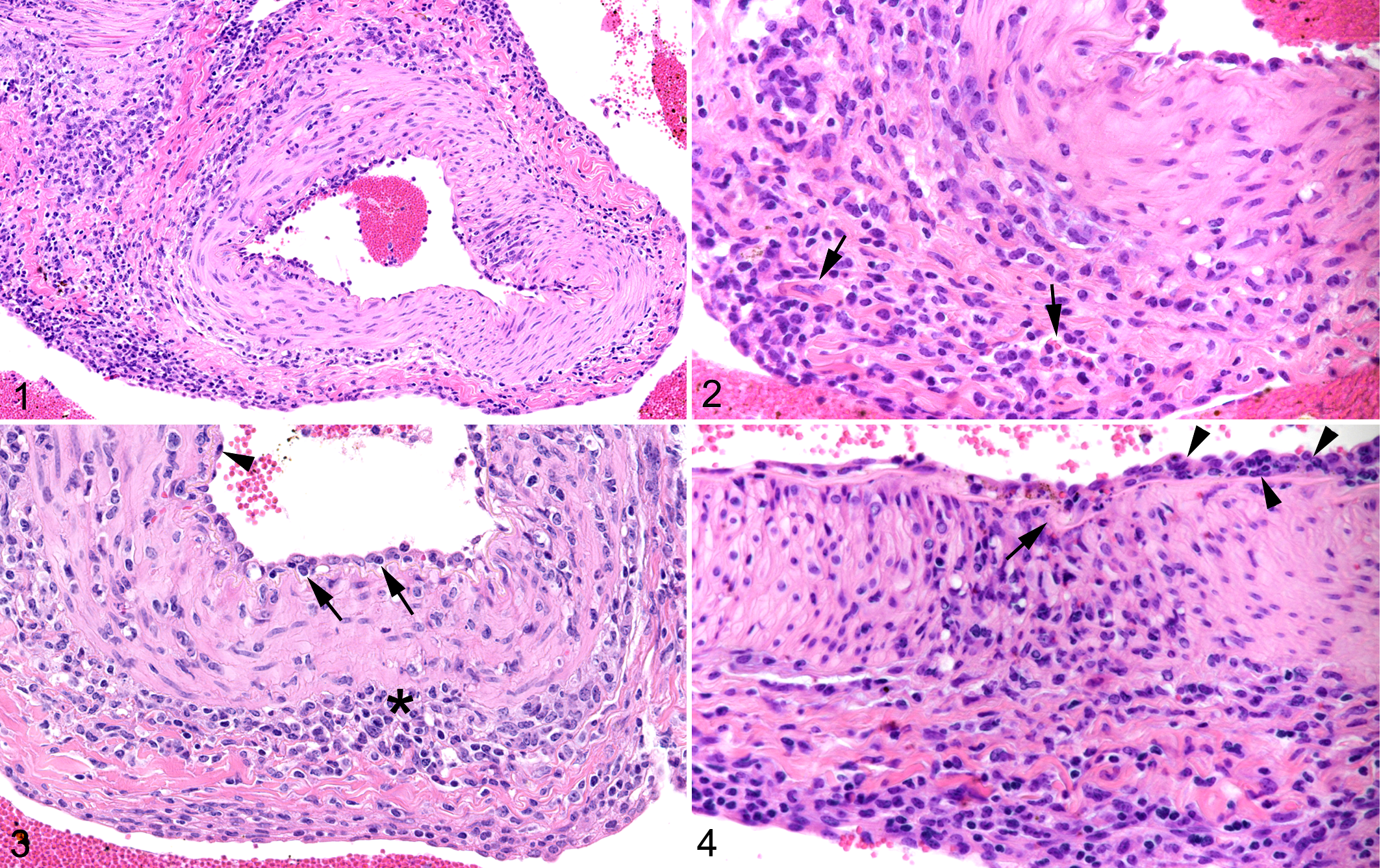
Malignant catarrhal fever, rete mirabile arteries, cow, case 15. Hematoxylin and eosin.
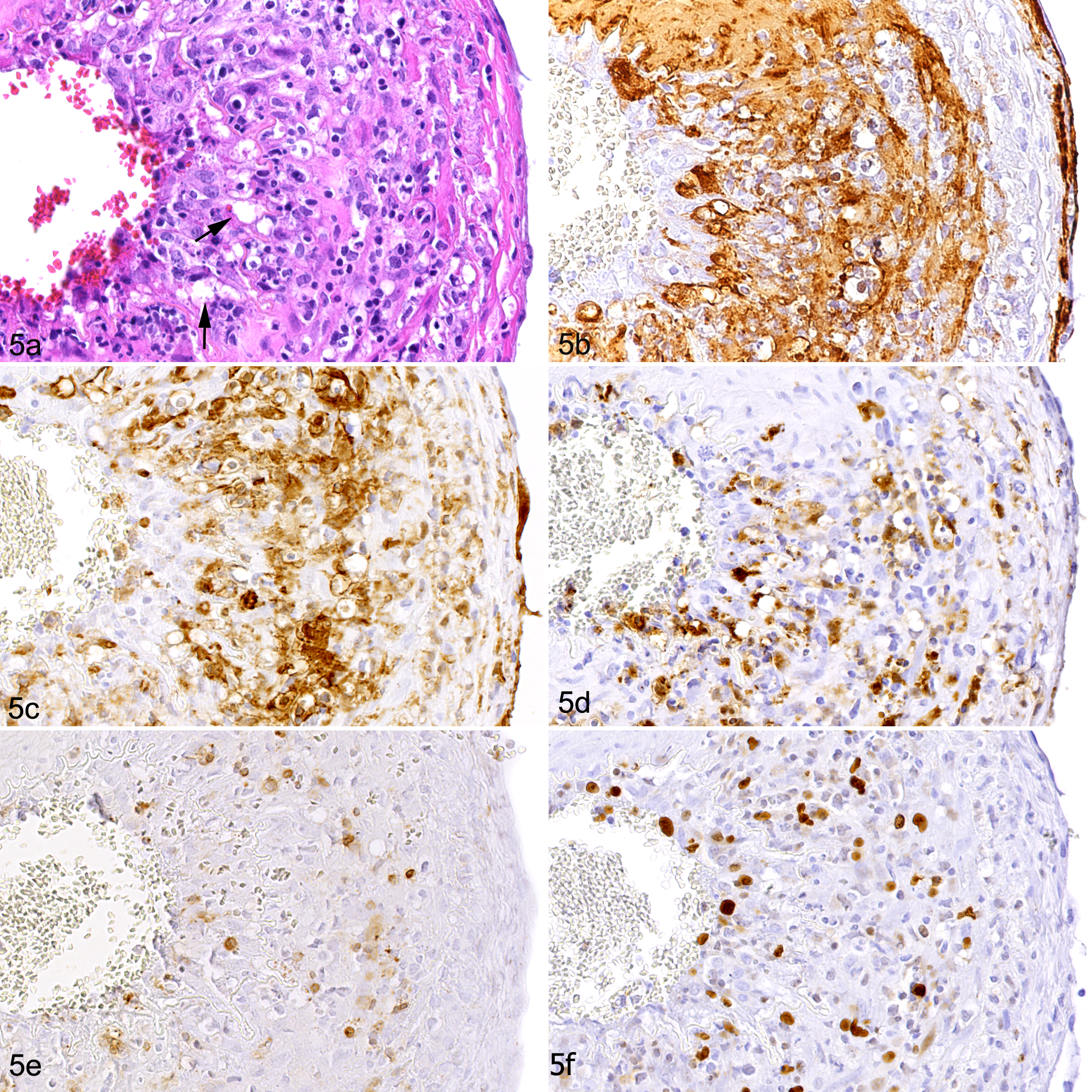
Malignant catarrhal fever, rete mirabile arteries, water buffalo, case 18. (a) Transmural infiltration with multifocal vacuolation of the media (arrows). Hematoxylin and eosin. (b) Within areas of vacuolation of the media, the smooth muscle cell arrangement is disrupted. Immunohistochemistry (IHC) for α-SMA. (c) The inflammatory infiltrate is dominated by Iba-1-positive macrophages. IHC. (d) A large proportion of the infiltrating macrophages are calprotectin-positive, implying they are recently blood-derived. IH. (e) CD3+ T cells are far less numerous. IHC. (f) A substantial proportion of infiltrating cells are immunolabeled for Ki-67, indicating they are proliferating. IHC.
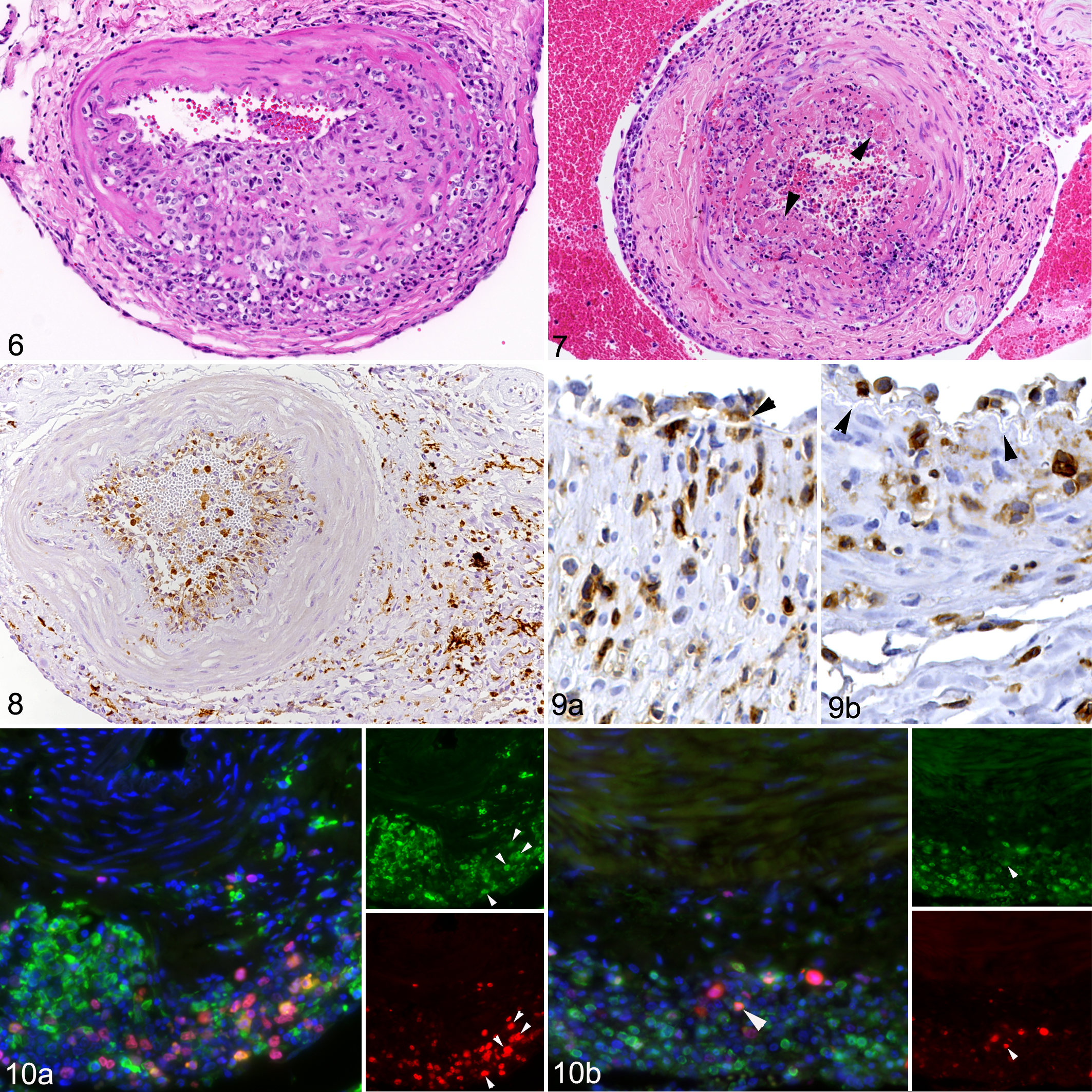
Malignant catarrhal fever, rete mirabile arteries, cattle (Figs. 6–9) and water buffalo (Fig. 10).
Macrophages and T Cells Dominate the Arteritis
In all cases and all affected vessels, macrophages (Iba-1+) and, often in lesser proportions, T cells (CD3+) were the principal infiltrating cells (Fig. 5c–e). B cells (CD20+) were either absent or rare in the inflammatory infiltrates. Both T cells and macrophages were observed immediately beneath the endothelium in the vascular wall (Fig. 9). A large proportion of the macrophages in the infiltrate were immunolabeled for calprotectin (Figs. 5d, 9a). In cases with predominantly adventitial inflammation, calprotectin-positive macrophages were not abundant in the infiltrates. Numerous cells within the wall infiltrates were Ki67-positive (Fig. 5f). Double immunofluorescence staining identified Ki67-positive cells as both Iba-1+ macrophages and, to a lesser extent, CD3+ T cells (Fig. 10).
Chronic Arterial Lesions Are Characterized by Muscular Hypertrophy and Intimal Hyperplasia
Thickening of the arterial wall was not only seen in association with the inflammatory infiltrates in the media (Fig. 6), but also due to overall thickening of the muscle layer, or more frequently (ie, in individual vessels of 11 cases, 31%) due to a focal to circular, cushion-like thickening of the intima, leading to luminal protrusion and narrowing of the vessel lumen (Figs. 11, 12). These cushions were located between the endothelial cell layer and the internal elastic lamina and were composed of loosely arranged smooth muscle cells (Figs. 11a, 12a). Embedded between the smooth muscle cells were abundant macrophages (Fig. 11b) and a few intermingled T cells (Fig. 11c); several infiltrating cells were Ki67+ (Fig. 12d). Within these areas, the internal elastic lamina appeared split and dissociated (Fig. 12). In 4 cases (cases 5, 17, 20, and 21), the lumen of one or more arteries was completely occluded by the intimal hyperplasia. Also, hyperplasia of the vasa vasorum suggesting neoangiogenesis was sometimes seen (Fig. 13). In 3 animals (cases 5, 20, and 29), all rete mirabile arteries exhibited the described chronic changes, and these vessels only exhibited minimal inflammatory cell infiltration. In other animals, the chronic changes were only observed in a proportion of arteries.
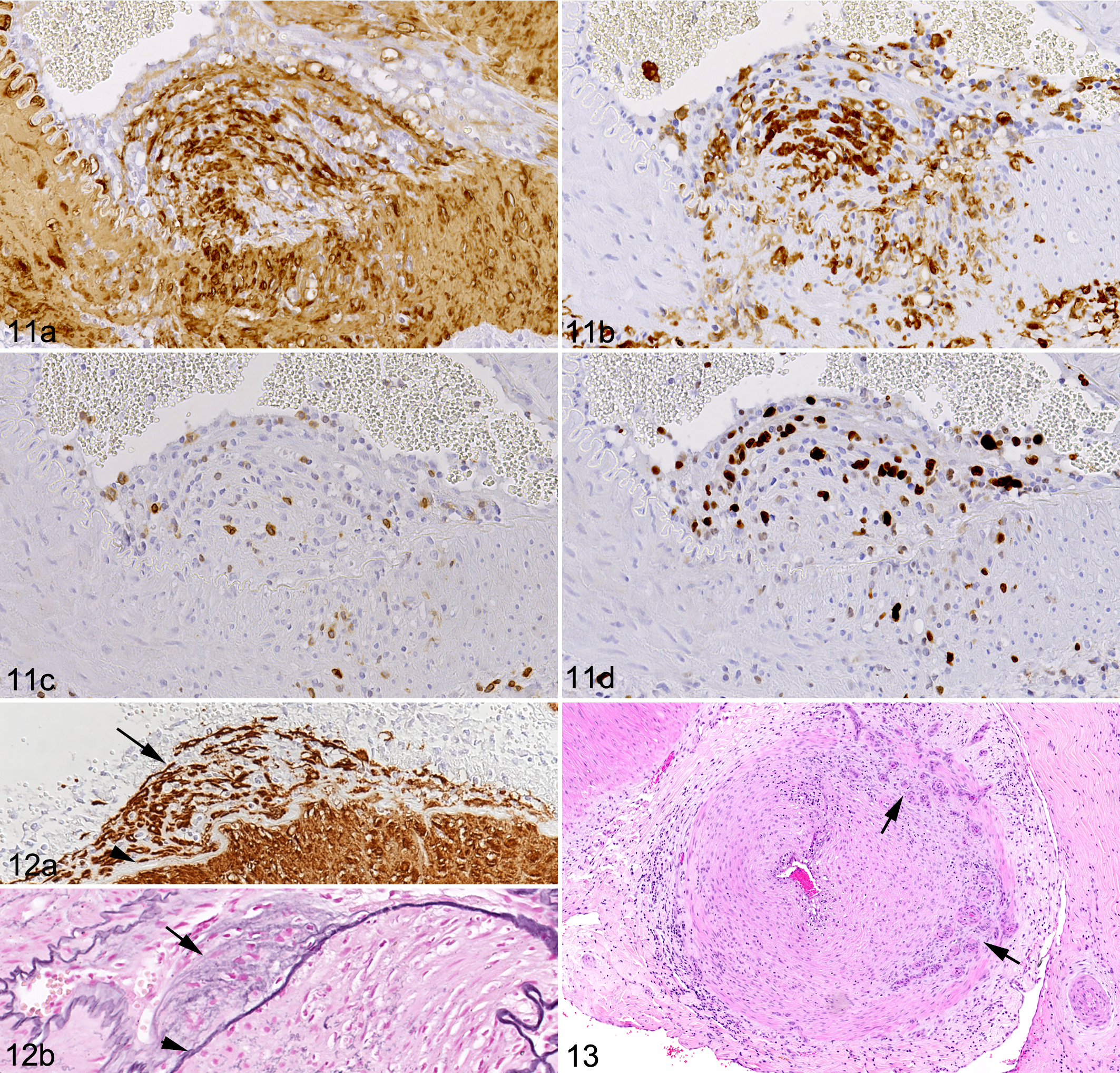
Malignant catarrhal fever, rete mirabile arteries, buffalos.
MCF Arteritis Is Associated With the Presence of oLANA Protein and vIL-10 Transcription
Both oLANA protein and ovIL-10 mRNA expression were detected in the rete mirabile arteries, with and without inflammatory changes. Immunolabeling for oLANA was widespread (Figs. 14 –20). It was seen in leukocytes in the vessel lumen, closely apposed or attached to the endothelium, and within the inflammatory infiltrates in the arterial walls (Figs. 17 –20); double IF showed that both monocytes/macrophages (Iba-1+) and T cells (CD3+) in the infiltrate were infected (Figs. 18, 19). oLANA immunolabeling was also present in endothelial cells and smooth muscle cells, mainly within the cytoplasm, but occasionally also in the nucleus (Figs. 14 –17, 20). In the chronic cases with scant inflammation, faint oLANA labeling was seen in scattered endothelial cells and smooth muscle cells in the tunica media. In the adventitia, oLANA was labeled in infiltrating cells, in endothelial cells and smooth muscle cells of the vasa vasorum, and in fibroblasts (Fig. 14). Viral IL-10 mRNA showed the same expression pattern by RNA-ISH, though the number of positive cells was substantially lower (Figs. 21, 22). It was also detected in occasional inflammatory cells and fibroblasts in the adventitia (Fig. 22).
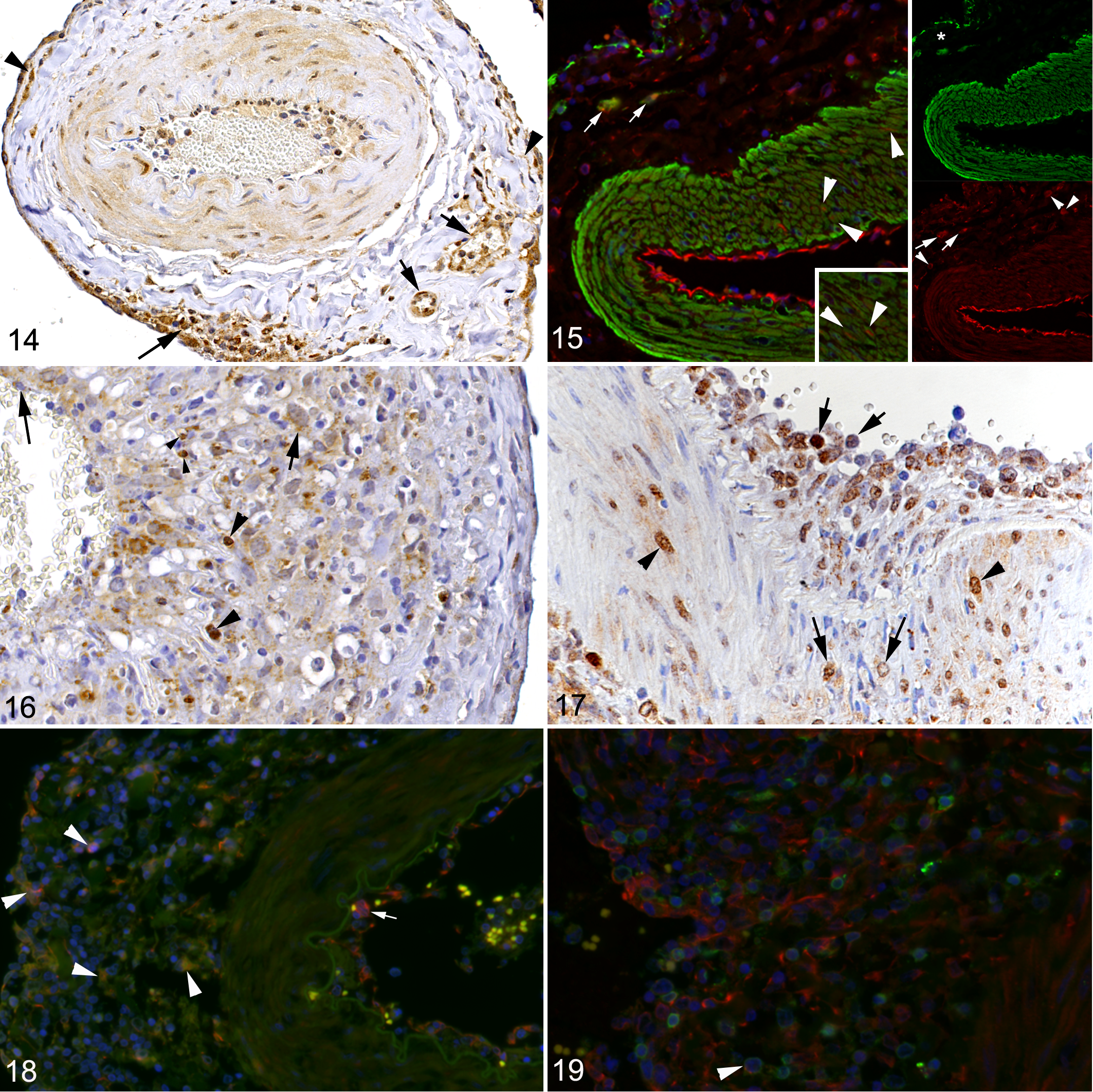
Malignant catarrhal fever, rete mirabile arteries.
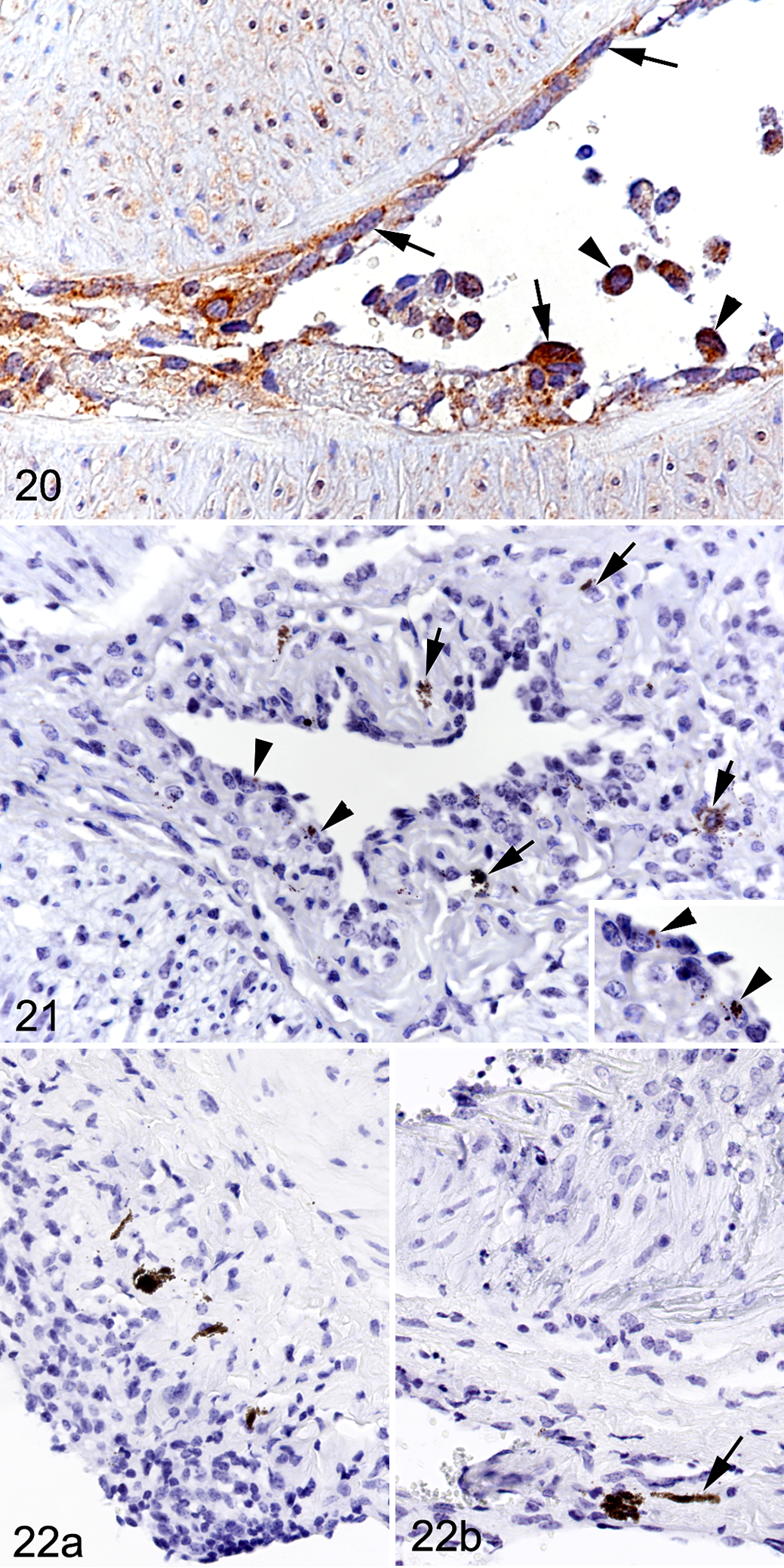
Malignant catarrhal fever, rete mirabile arteries, cattle.
Discussion
MCF is a sporadic disease of ruminants with high mortality. One of its pathological hallmarks is a vasculitis, which is predominantly a mononuclear arteritis, with variable additional features, such as necrosis, intimal hyperplasia, and obliterative changes. 30,59,68 In sheep, the host species of OvHV-2, the virus targets T cells, 36,58 where it predominantly persists in its latent form. 67 However, in susceptible species, animals with MCF also carry OvHV-2 in blood lymphocytes, which are not only latently 35 infected but are at least in a proportion also productively infected. 67 Productively infected lymphocytes, and specifically cytotoxic CD8+ T cells, have also been detected in and around the lesions of vasculitis, 41,49,61 and there is evidence of abortive infection of perivascular fibroblasts associated with the vasculitis. 41 The present detailed study on rete mirabile arteries in MCF aimed to shed more light on the pathogenesis of MCF arteritis and focused on infiltrating cells and the presence of OvHV-2-encoded ovIL-10 mRNA and oLANA that are expressed during latency as well as the productive cycle. 67
A Relevant Role for Macrophages in MCF Arteritis
Previous studies have shown that besides lymphocytes, macrophages and to a low extent neutrophils and plasma cells are present in MCF vasculitis. 29,30,39 Overall, macrophages were not considered to represent a relevant leukocyte population in the infiltrates. 1,61 The results of the present study suggest the opposite, showing that macrophages are consistently abundant in MCF vasculitis, outnumbering or at least equaling the T cell population; other leukocytes were not as numerous. This suggests that macrophages, in addition to T cells, could play an important role in the development and progression of MCF vasculitis. Both cell types and in particular the macrophages were found to proliferate locally, as shown by their Ki67 expression. This proliferation likely reflects an autocrine effect of the cytokine and growth factor release by the leukocytes, and thereby a consequence of the continuous inflammatory stimulus. At the same time, we found evidence of ongoing monocyte recruitment into the lesions, as a proportion of infiltrating macrophages was found to express calprotectin, a marker of monocytes and recently blood-derived macrophages. 14
The leukocyte infiltrates in the media were sometimes accompanied by disruption of the media and/or degeneration of smooth muscle cells. The damage might be the result of macrophage infiltration, as activated macrophages can release reactive oxygen species and matrix metalloproteinases that could destroy the media and digest the internal elastic lamina. 57,60
Arteritis in the Rete Mirabile Is Consistent With an Intense “Outside-In” Vasculitis and an “Inside-Out” Inflammatory Infiltration
Medium-sized and larger arteries, like those in the rete mirabile, are multilayered vessels that are surrounded by the adventitia which contains a delicate microvasculature, the vasa vasorum. The latter supply oxygen and nutrients to the vessel wall and are the first site of response to arterial injury, providing access for inflammatory cells. Due to this vessel architecture, 2 different pathways of arterial inflammation have been suggested for arteritis in humans, a traditional concept known as “inside-out” and the newer paradigm of an “outside-in” pathway. 33,38
Similar to other authors who reported on the features of the inflammatory processes in the rete mirabile in MCF, 30 we observed that the infiltration is generally most intense in or even restricted to the tunica adventitia of the arteries, with only focal and frequently less substantial infiltration of the tunica media; the latter often appeared to be a continuum of the adventitial infiltrate. This provides strong evidence that the inflammatory cells are recruited to the vessel through the vasa vasorum and would be consistent with the “outside-in” pathway. 33,38 In humans, under inflammatory conditions, macrophages in particular are recruited through this microvasculature; this is also suggested in our study, considering the large proportion of macrophages among the infiltrating cells. 14 The “inside-out” inflammatory process, however, is triggered by injured endothelial cells. These express or upregulate surface adhesion molecules and inflammatory mediators that participate in monocyte homing to the endothelium and eventual transmigration into the media. 33 In the case of MCF arteritis it appears likely that monocytes are recruited into the adventitia from the blood via the vasa vasorum, differentiate into macrophages, become activated, and may secrete inflammatory cytokines that result in further inflammatory cell recruitment. With subsequent infiltration of the media, mediators released from the macrophages might lead to activation of the arterial endothelial cells, which could then trigger the inside-out inflammatory process that we also observed in a proportion of affected vessels. Indeed, in these vessels, the endothelial cells appeared activated and there was evidence of leukocyte recruitment, since intravascular leukocytes were found attached to and immediately beneath the endothelium and within the media.
Arteritis in the Rete Mirabile Can Culminate in Necrotizing Inflammation or Progress Toward Arterial Remodeling
In addition to the inflammatory cell infiltration, arteries were occasionally found to exhibit more pronounced changes. In some cases where the arteries showed different degrees of inflammatory infiltration, individual affected vessels exhibited extensive damage to the wall, represented by circumferential accumulation of fibrin at the intima-media junction, overlain by large numbers of intact and degenerating inflammatory cells at the luminal surface. Among the infiltrating cells were neutrophils that were mainly degenerate. The MCF literature reports such fulminant changes as a finding of variable frequency and has interpreted these as fibrinoid necrotizing vasculitis. 1,30,37,59 Since the latter is generally seen as a consequence of antigen-antibody complex deposition, this pathogenesis has indeed been considered for MCF vasculitis. 30 However, there has neither been convincing evidence of virus-specific immune complexes in vessel walls in MCF, 48 nor have ultrastructural studies of MCF vasculitis found any evidence of immune complex deposition in the lesions. 30,44 Instead, it is possible that the necrosis is a secondary event, resulting, for example, from the release of reactive oxygen species by infiltrating macrophages, 60 or from ischemia, as a result of an increase in wall thickness due to the inflammatory infiltrate and consequently an insufficient oxygen supply through the vasa vasorum. 38
We also observed proliferative changes in some affected arteries, features that have been reported in MCF cases with prolonged clinical disease or after clinical recovery, and that are considered as chronic changes. 43,44,59 We found the proliferative changes in individual affected vessels, usually in the form of focal intimal hyperplasia; in a few cases, they were the only observed type of vascular lesion. While we cannot determine the time point of infection in our cohort of natural cases, the latter group of animals might have passed an earlier peak of the disease and only exhibited the remnants of the original inflammatory processes at the time of necropsy. 43,44,59 The proliferative changes were associated with a variable degree of focal or circumferential luminal narrowing, as a consequence of subendothelial accumulations of spindle-shaped cells that were mostly α-SMA-positive. The latter were associated with disarrangement or splitting of the internal elastic lamina and were intermingled with macrophages and a few T cells that seemed to proliferate locally. Subendothelial accumulations of spindle-shaped cells have previously been reported in recovered MCF cases, 43,44 and in the literature they are also referred to as neointimal proliferation as they are located immediately beneath the endothelium and luminal to the internal elastic lamina. The cells represent smooth muscle cells of the tunica media that have migrated to the intima, or myofibroblasts originating from adventitial fibroblasts that have acquired α-SMA expression due to a phenotype switch and have migrated to the intima. 33,38
The observed changes are consistent with vascular remodeling processes and have several features in common with the lesions in chronic arteritis in humans, for example, giant cell arteritis. 57 Similar to MCF arteritis, giant cell arteritis is associated with the recruitment of CD8+ T cells and monocytes into the arterial wall. The following scenario is suggested: through the release of IFN-γ the T cells can recruit monocytes as well as induce the observed apoptosis of cells in the media via cytotoxic molecules such as granzyme B and perforin. 40 Monocytes differentiate into macrophages, which release cytokines that perpetuate the inflammatory response, and mediators such as reactive oxygen species and MMPs that further damage the media. Also, cytokine-activated endothelial cells can recruit more inflammatory cells into the vascular wall. Then, growth factors released by macrophages and smooth muscle cells come into action; PDGF can induce proliferation and migration of these cells, leading to intimal hyperplasia, and VEGF can induce neoangiogenesis, 57 which could contribute to local hyperplasia of vasa vasorum. Interestingly, there is evidence of such an effect from other γ-herpesviruses: Epstein-Barr virus infection has been associated with pulmonary vascular remodeling in human idiopathic pulmonary fibrosis, and a similar effect was seen in CD1 mice infected with murine γ-herpesvirus (MHV)-68. 9
Broad Expression of oLANA and ovIL-10 mRNA in MCF Vasculitis Suggest a Role of the Virus in the Pathogenesis
Affected arteries exhibited variable, but consistent OvHV-2 oLANA expression in endothelial cells and smooth muscle cells in the vascular wall and in both intravascular and infiltrating monocytes/macrophages and T cells. This indicates a vascular target cell spectrum in MCF that is broader than anticipated based on previous studies, though these detected lytic infection, which may indeed be restricted to CD8+ T cells. 40,41,49,61 oLANA was also found in occasional endothelial cells in vessels without inflammatory changes, indicating more widespread viral latency in the endothelium. LANA is a nuclear protein that is encoded by ORF73 and expressed during viral latency as well as during the productive cycle and so will be present in all infected cells. 18,55,67 The LANA of Kaposi’s sarcoma herpesvirus (KSV) is a homologue with sequence similarity to OvHV-2 oLANA. KSHV LANA is a multifunctional protein that is responsible for persistence of the viral episome in mammalian cells. It also enhances survival and proliferation of infected cells, and seems to have an immunomodulating function, damping the IFN response and slowing MHC I recognition. 34,70,71 It is therefore likely, by inference, that oLANA has similar functions during OvHV-2 infection. A role for the AlHV-1 LANA in MCF has been shown, at least with regard to the proliferation of T cells. 47 Unexpectedly, we observed more intense cytoplasmic than nuclear staining for OvHV-2 LANA, which suggests that, similar to KSV LANA, the protein has cytoplasmic isoforms. Cytoplasmic KSHV LANA may be involved in viral activation, with transition from latency to the lytic cycle. 34,71 This would be consistent with a recent RNA-ISH study that detected lytic viral infection in lymphocytes in MCF vasculitis. 49 Similarly, OvHV-2 oLANA transcription has previously been reported in tissues of bison with MCF, though alongside ORF25 transcription which showed that lytic infection was taking place. 11
We also examined the rete mirabile for the transcription of ov2.5, a latency-associated gene that encodes for ovIL-10, a protein similar to ovine interleukin IL-10 that has retained its function such as the inhibition of macrophage function. 22 Using RNA-ISH, we found ovIL-10 transcription in a similar set of cells in MCF vasculitis as viral LANA, that is, vascular endothelial cells and myofibroblasts or smooth muscle cells as well as infiltrating leukocytes (macrophages and lymphocytes), the latter predominantly in the tunica adventitia. We also saw it occasionally in endothelial cells and adventitial fibroblasts of unaffected arteries.
Viral interleukins, acquired during coevolution with their host species and encoding for orthologues of cellular interleukins, provide a viral strategy to deregulate the host immune system. 46,62 ovIL-10 has a direct immunosuppressive effect on macrophages, inhibiting the release of proinflammatory mediators. 22 The principal action of cellular IL-10 is to modulate immune responses and to protect from their damage. 54 By hijacking cIL-10, OvHV-2 might evade an immune response; its presence in MCF vasculitis could suggest that it is also associated with modulation of macrophage function that might be crucial for their interaction with equally infected (CD8+) T cells.
OvHV-2 infection (as defined by oLANA and ovIL-10 expression) was mainly found in intact cells without degenerative changes, and only rarely in cells that exhibited features of apoptosis. Similarly, a recent study detecting lytic infection did not describe damage of infected cells. 49 This suggests that functional or phenotypic changes potentially induced by the virus in infected cells are not associated with direct cytopathic effects. The immune response to the virus may play a role here; this is indicated by the fact that MHV-68 can infect vascular smooth muscle cells and cause arteritis in immunodeficient mice. 70
The key question regarding the pathogenesis of MCF vasculitis is therefore what sets off the inflammatory cell recruitment in the first place? Whether this is the activation of endothelial cells and circulating T cells or monocytes by (latent) OvHV-2 infection remains to be elucidated. Also, the effect of OvHV-2 infection on other adventitial cells, such as fibrobasts or medial smooth muscle cells, which were both found to be latently infected, warrants further investigations. 41 Adventitial fibroblasts are activated under pathological conditions and undergo phenotypic changes that include proliferation, differentiation into myofibroblasts and migration to the intima, increased production of extracellular matrix proteins, and release of factors (ie, reactive oxygen species) that directly or indirectly affect vascular function and structure. In this context, α-SMA expression is triggered by a complex microenvironment that enhances fibroblast differentiation into a myofibroblast phenotype where TGF-β induces this phenotypic switch. 16
The literature has provided evidence that the development of MCF is a consequence of failure of the immune system to maintain a balance between restricted growth and uncoordinated multiplication of infected T cells with unrestricted killer activity. However, our results suggest that T cells are not the only leukocytes that are latently infected, and that monocytes and locally proliferating macrophages are equally infected and also important for the development of the vasculitis, as they act as regulators in recruitment of other cells with their cytokine cascade and can simultaneously cause vessel wall damage. What remains to be identified is the initial trigger that leads to their activation. There is at least morphological in situ evidence that latently infected, activated endothelial cells play a role in this. Activated macrophages might then release the necessary pro-inflammatory mediators and, eventually, induce the characteristic vascular changes.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Suara_Martinez_et_al - Sheep-Associated Malignant Catarrhal Fever: Role of Latent Virus and Macrophages in Vasculitis
Supplemental Material, Combined_supplemental_materials-Suara_Martinez_et_al for Sheep-Associated Malignant Catarrhal Fever: Role of Latent Virus and Macrophages in Vasculitis by Helena Saura-Martinez, Mohammed Al-Saadi, James P. Stewart and Anja Kipar in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to the technical staff in the Histology Laboratory, Institute of Veterinary Pathology, Vetsuisse Faculty, University of Zurich (IVPZ), for excellent technical support. We would also like to thank Drs. Martina Dettwiler and Corinne Gurtner at the Institute of Animal Pathology, Vetsuisse Faculty Berne, for their support with access to the Institute’s archives, and Dr Udo Hetzel from the IVPZ, for his support toward the double IF analysis. Mohammed Al-Saadi has been supported by a PhD scholarship from the Ministry of Higher Education and Scientific Research in Iraq (MOHESR).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
