Abstract
The enigmatic pathogenesis of malignant catarrhal fever (MCF) involves dysregulated immune responses in susceptible ruminant species. Economically important outbreaks of MCF are due to 2 of the 10 viruses currently comprising the malignant catarrhal fever virus group: ovine herpesvirus 2 (OvHV-2) and alcelaphine herpesvirus 1 (AlHV-1). Attempts to develop effective vaccines for this group of viruses in the 1970s were sufficiently discouraging that they were temporarily abandoned. This review focuses on recent efforts to understand the pathogenesis of MCF, particularly the sheep-associated form of the disease, with the goal of developing rational control methods, including vaccination. The past 2 decades have seen several advances, including recognition of new members of the MCF virus group, better diagnostic assays, induction of disease by a natural route (aerosol), and clearer understanding of OvHV-2’s shedding patterns by domestic sheep. A consistent theme in experimental studies of OvHV-2 in susceptible species is that there are 2 peaks of OvHV-2 gene expression: a preclinical peak involving the respiratory tract and a second in multiple organ systems leading to clinical disease. Latent and lytic gene expression may coexist in tissues during clinical stages in symptomatic animals.
Keywords
In 1997, we were approached by a bison producer who lost multiple adult animals to malignant catarrhal fever (MCF). 132 As is often the case, the producer’s questions were pragmatic and dealt with the likely source of infection, how the outbreak might be stopped, and whether effective treatment or vaccination was possible. At that time, our group had worked on MCF for several years, relying in part on case material from natural outbreaks of MCF in domestic cattle. 92,93 Diagnosticians and researchers were the beneficiaries of new diagnostic assays to confirm infection in live animals. 4,70 Unfortunately, the news for the producer was bad. The source of infection was probably domestic sheep, but the distance over which transmission was likely to occur was unknown. The producer reported that no sheep were in the immediate vicinity of her herd. Little was known about how sheep shed the virus or the duration of shedding. Since MCF was of modest interest to the cattle industry, funding for basic studies was constrained. No effective treatment existed. In the wake of that owner’s outbreak, it quickly became clear that MCF was of appreciable importance to the commercial bison industry in the United States and Canada. 7,94 The long history and intriguing nature of MCF (Table 1) were small solace to the Colorado producer in the absence of practical solutions from MCF researchers. A total of 27 bison died as a result of the outbreak. Three bison remaining in the herd were butchered. Efforts to raise bison on the property were temporarily abandoned.
Landmarks in the Understanding of Malignant Catarrhal Fever.

AlHV-1, alcelaphine herpesvirus 1; MCF, malignant catarrhal fever; MCFV, malignant catarrhal fever virus; OvHV-2, ovine herpesvirus 2; SA-MCF, sheep-associated malignant catarrhal fever; WA-MCF, wildebeest-associated malignant catarrhal fever.
This review summarizes recent progress in our understanding of sheep-associated MCF. Until the development of molecular and serological assays in the 1980s and early 1990s, 4,8,70 diagnosticians and researchers used the biology of alcelaphine herpesvirus 1 (AlHV-1), much based on the astonishing work of Walter Plowright, 105 –107 as an avatar to predict the likely epidemiology of ovine herpesvirus 2 (OvHV-2). 118 This was logical. With minor exceptions, clinical signs and lesions in susceptible hosts of sheep- and wildebeest-associated MCF were essentially identical. 3,102 An unequivocal diagnosis of MCF required the pathologist to find the right spectrum of lesions. 25,118 Currently, there is renewed interest in vaccination for this group of viruses, a goal long thought out of reach due to discouraging results in the 1970s and 1980s. 32,75,118,125,126 It is now clear that some dead-end host species recover, 92,93,153 corroborating clinical suspicions from the late 1920s and early 1930s that MCF is not invariably fatal. Important differences exist between the ecology of sheep- and wildebeest-associated MCF. The MCF virus (MCFV) group continues to expand and currently comprises at least 10 members (Table 2). Of these, 6 can cause MCF naturally. A seventh causes MCF experimentally in rabbits. The remaining 3 members have yet to be associated with disease.
Members of the MCFV Group.

AlHV-1, alcelaphine herpesvirus 1; AlHV-2, alcelaphine herpesvirus 1; CpHV-2, caprine herpesvirus 2; HipHV-1, hippotragine herpesvirus 1; MCF, malignant catarrhal fever; MCFV, malignant catarrhal fever virus; OvHV-2, ovine herpesvirus 2; WTD, white-tailed deer.
When MCF occurs due to agents other than OvHV-2 and AlHV-1, the spectrum and severity of lesions may differ from the beloved trinity of arteritis, lymphoid proliferation, and mucosal necrosis in the digestive tract, as seen microscopically in domestic cattle. 10 In the past, this was a particular challenge when pathologists examined susceptible animals in mixed-species confinement, particularly in zoological collections and game farms, and found lesions that only approximated those of MCF in domestic cattle. Given the absence of an effective vaccine, it is important to identify which MCFV is involved so that carrier and susceptible hosts can be separated appropriately. 56,76 It is clear that some natural host species, such as goats, can be infected with, and possibly shed, more than 1 MCFV.
While of minor importance for domestic cattle, MCF is economically important in the North American bison industry, 7 for captive and free-ranging cervid species, 44,76,82,90,101,127,148,151 to managers of zoological collections 40,43 and to producers of domesticated species such as Bali cattle (Bos javanicus) and domestic water buffalo (Bubalus bubalis). 28,45,46,84,114,141 Veterinary pathologists working with zoological collections play a useful role by reporting the impact of members of the MCFV group on captive species. 38,76,96,100,127 Such reports provide novel information about the agents involved, the spectrum of lesions induced, and cumulative losses. 35,36
This review focuses on OvHV-2 from the standpoint of diagnostic pathologists. Recently published reviews address other aspects of MCF such as its occurrence in domesticated species in Europe and North America, 52,112 as well as in free-ranging and captive wildlife; 44 its lesions in domesticated and wildlife species; 11 prospects for effective vaccines; 75 diagnostic assays; 22,59,116,144 and molecular virology and pathogenesis. 126 One aspect of the disease we would like to review but cannot is the impact of OvHV-2 on producers in developing countries. Indonesian and Australian colleagues demonstrated what was possible through international cooperation. 25 The importance of sheep-associated MCF in developing agricultural countries may be less a case of terra incognita than terrae notae ac neglectae.
Ovine Herpesvirus-2 and the MCF Virus Group
The 10 members of the MCFV group belong to the genus Macavirus in the subfamily Gammaherpesvirinae. 27 Gammaherpesviruses have a tropism for T or B lymphocytes, and MCFVs typically infect T cells for part of their life cycle. 91 The MCFV group is related to but distinct from other ruminant gammaherpesviruses, including non-MCF lymphotropic herpesviruses and bovine herpesvirus 4. 60,63 They infect members of the order Artiodactyla in the families Bovidae, Giraffidae, and Cervidae (Table 1). Membership of the MCFV is currently defined by presence of the 15-A antigen epitope, combined with base sequence similarity to conserved regions of the DNA polymerase gene. 63 Five MCFVs are pathogenic in at least 1 species: OvHV-2, 4,8 AlHV-1, 109 alcelaphine herpesvirus 2 (AlHV-2), 88,121 caprine herpesvirus 2 (CpHV-2), 66 and an MCFV in white-tailed deer (Odocoileus virginianus) for which domestic goats may be a carrier host. 56,61 Three members (gemsbok-MCFV, muskox-MCFV, and aoudad-MCFV) are not associated with MCF. 62,149 The remaining virus (hippotragine herpesvirus 1 [HipHV-1]) causes MCF in rabbits experimentally, but naturally transmitted disease has not been reported. 117 It is likely that more members of the MCFV will be identified in coming years, particularly as free-ranging wildlife species and zoological collections are surveyed. In addition to domestic cattle, which can develop MCF due to OvHV-2 and AlHV-1, other species are susceptible to more than 1 MCFV, giving rise to the need for multiplex tests whenever suggestive lesions are found. 22 One reservoir host (domestic goats) can be infected with 2 or possibly 3 MCFVs. 56,66
The genomes of OvHV-2 and AlHV-1 have been sequenced and compared. 30,33,41,138 The products of several open reading frames (ORFs) are used in transmission studies to establish whether latent or lytic infection patterns predominate in animals following challenge at different time points. 21,23,57,59,85,99,123 Expression in tissue of specific viral genes (in the case of OvHV-2, these include ORF9, DNA polymerase; ORF25, major capsid protein; ORF50, R-transactivator inducing latent to productive cycle switch; and ORF73, latency-associated nuclear antigen [LANA] homolog [genome maintenance protein]) is used to establish when productive infection occurs in the course of experimental infection. 21,23,99,139 Polyclonal antibodies to selected gene products have been used immunohistochemically to monitor infection. 139 ORF73 may have epidemiological value due to some variability among OvHV-2 isolates. 126 OvHV-2 codes for interleukin (IL)–10 and members of the Bcl-2 proteins, both of which may be important in susceptible hosts. 50
Permissive cell culture systems exist for 3 MCFVs (AlHV-1, AlHV-2, and HipHV-1). 87,89,109,117 The absence of such a system for OvHV-2 has been a longstanding impediment to research. 44 Experimentally, this was partly bypassed using large volumes of blood or leukocytes from natural cases for experimental transmission. 16,45,78 –80,97,103,104,114,134 The sheep agent can be maintained in lymphocytes derived from cattle and deer with MCF. 118,119,123,128 In this form, the virus has been used to transmit infection to laboratory animals and sheep. 12 –15,45,118,119 Past reports of the isolation of OvHV-2 represent inadvertent contamination of cell cultures with AlHV-1. 122,130 Successful isolation of MCFVs from affected or latently affected animals is achieved by cellular co-cultivation, since the virus is strictly cell associated. 109 Leukocytes from peripheral blood and lymphoreticular tissues are the most common source of samples for attempted isolation. Growth of AlHV-1 in cell culture is associated with the formation of transient syncytia 3 to 21 days after inoculation. 108 Typical intranuclear herpesviral inclusions form in infected cell cultures and, transiently, in the lungs of experimentally infected sheep (D. O’Toole and H. Li, unpublished data). 108,122 Subsequent passage results in better yields of cell-free virus. Viral inclusions are absent in the lesions of susceptible species that die of MCF.
MCFVs are well adapted to their natural host species. Classic MCF is recognized in domestic cattle as fever with oculonasal discharge; panophthalmitis; generalized lymphadenopathy; inflammation and necrosis in alimentary, respiratory, and urinary tracts; dermatitis; neurological signs; and arthritis. 86,102,103,146 The gross and histological lesions are often considered pathognomonic. 118 Increasingly, a definitive diagnosis of MCF depends on a combination of appropriate clinical signs and lesions, as well as detection of MCFV DNA in fresh or fixed tissue. 19,86 The clinical course is short, typically <1 week. 146 Death or euthanasia is the most common outcome. Transmission of OvHV-2 among natural and MCF-susceptible hosts is primarily by contact with nasal secretions, typically when aerosolized. 64,74
There is a spectrum of susceptibility to pathogenic members of the MCFV group. Highly susceptible species include banteng (B. javanicus) and some cervid species, particularly Père David’s deer (Elaphurus davidianus) 98 and white-tailed deer. 43 Intermediate in susceptibility are water buffalo (B. bubalis) and many cervid species. 25,28,45,46 The susceptibility of domestic cattle to MCF, particularly of Bos taurus, is low compared with these species. 73,94 This has been established by transmission studies and by differences in mortality rates when bison and domestic cattle are exposed at the same time to OvHV-2. 73,133 No shedding of infectious virus occurs from clinically affected susceptible hosts. 7
Ovine Herpesvirus-2 and Domestic Sheep ( Ovis aries )
It has been known since the late 1700s that a temporal association exists between exposure to domestic sheep and outbreaks of MCF in cattle. 108 Initial transmission studies relied on large volumes of blood or on suspensions of infected cells from animals with MCF, but this made replicate or comparative studies impossible. Laboratory-maintained lymphocyte populations infected with OvHV-2 that were derived from domestic cattle or deer circumvented the problem. 119,128 A critical advance was the derivation of a DNA clone of OvHV-2, 9 spurred by multiple lines of evidence indicating that domestic sheep harbored an agent similar to AlHV-1. 124 This was followed by the development of a diagnostic polymerase chain reaction (PCR) assay for OvHV-2. 4 Complemented by a competitive inhibition enzyme-linked immunosorbent assay to a conserved epitope of the MCFV group, 68,70 the establishment of OvHV-2–free flocks, and a reliable sources of infectious virus, 60 the stage was set to characterize shedding patterns in domestic sheep.
The results yielded several surprises. Virtually all lambs were born free of infection and became infected after 2 months of age under natural flock conditions. 72 The kinetics of viral shedding from sheep using quantitative real-time PCR indicated that adolescent lambs (6–9 months old) shed virus more frequently and intensively than adults. 71,72 Uninfected sheep remained susceptible to infection as adults. 72 The lower respiratory tract was the primary viral target in sheep following aerosol challenge. 57 Challenge by intravenous or intraperitoneal routes did not establish infection, presumably because OvHV-2 inefficiently infects mononuclear-macrophage cell populations. 57 Unlike wildebeest infected with AlHV-1, 106 no transplacental infection occurred in sheep, and infection via colostrum or milk appeared to be rare. 59 Lifelong infection was established in most sheep by 1 year of age. 64 It was assumed for many years, by extrapolation from AlHV-1, that the main risk period for susceptible host species was at or shortly after lambing. 118 The results of some studies support this conclusion. 5 Outbreaks of MCF in susceptible species were most likely when exposure involved large groups of sheep, particularly those 6 to 9 months old age. 65 It has been suggested that OvHV-2 undergoes mutation to become more infectious for susceptible species. 44 Newborn lambs are susceptible to aerosol infection, but the rarity with which infection occurs immediately after birth is probably due to limited viral shedding by ewes, combined with inefficient infection. 64,71 An intriguing observation is the detection of OvHV-2 in semen and reproductive glands of rams; 47 whether this has epidemiological significance remains to be established. 112
Infectious virus harvested and pooled from nasal secretions of multiple sheep during shedding episodes is used to infect carrier and susceptible species. 55,57,58,69,140 Initial lytic replication of OvHV-2 in sheep occurs in alveolar epithelium, primarily type II pneumocytes. 139 The process by which lymphocytes become latently infected during the course of infection is unclear; cell tropism switching analogous to that of Epstein-Barr virus may be involved. 21,139 Viral replication is localized to the respiratory tract, predominantly in turbinates during periods of shedding. 23,139 Replication in nasal epithelium and viral shedding with the formation of herpesvirus-like particles is brief (<24 hours), 23 suggesting a single cycle of replication occurs at this site. 74 A key difference between sheep and susceptible host species such as bison and rabbits is the higher level of expression of multiple immune-related genes during initial pulmonary viral infection. A 10- to 70-fold increase in the transcription of 16 immune response genes occurs in sheep during the pulmonary phase. 57 Viral replication in lung in the natural host leading to latent infection is presumably controlled by a combination of innate, cell-mediated, and humoral responses.
A proportion of lambs challenged by aerosol develop subclinical bronchointerstitial pneumonia with degeneration, loss and hyperplasia of terminal bronchiolar cells, and intra-alveolar fibrin exudation within a week of infection (Figs. 1, 2). 69 Histological lesions persist for at least 2 weeks after the challenge. Viral replication, as evidenced by transcripts of ORF25, occurs in lung within a week of infection (Fig. 3). Lytic infection does not occur in other tissues during this period. 57 Lambs exposed to high doses of OvHV-2 develop an MCF-like syndrome. This is unlike AlHV-1–infected wildebeest calves, in which an MCF-like disease has not been described. Affected lambs are febrile, with mucopurulent nasal discharge and lesions restricted to respiratory and upper digestive tracts. 69 Whether a similar syndrome occurs naturally in flocks is debatable. Fetal lambs that were challenged intravenously with infected leukocytes developed an MCF-like syndrome with lymphoproliferation and panarteritis. 15 Systemic vasculitis is sporadically recognized as a syndrome in commercial flocks, raising the suspicion of ovine MCF. In such outbreaks, the distribution of vascular lesions is systemic, 42,113 unlike intranasally challenged sheep in which arteritis is restricted to the lungs. 69 It is unclear whether these episodes represent ovine MCF, since diagnosticians lack a robust immunochemical method to co-localize OvHV-2 antigens with lesions in formalin-fixed material. Most domesticated sheep are infected with OvHV-2 during their first year of life. 71,72 The detection of the virus in sheep with MCF-like lesions is not compelling evidence for a causative role. Detection of OvHV-2–encoded proteins such as ORF25 in lesional tissue would be more persuasive.
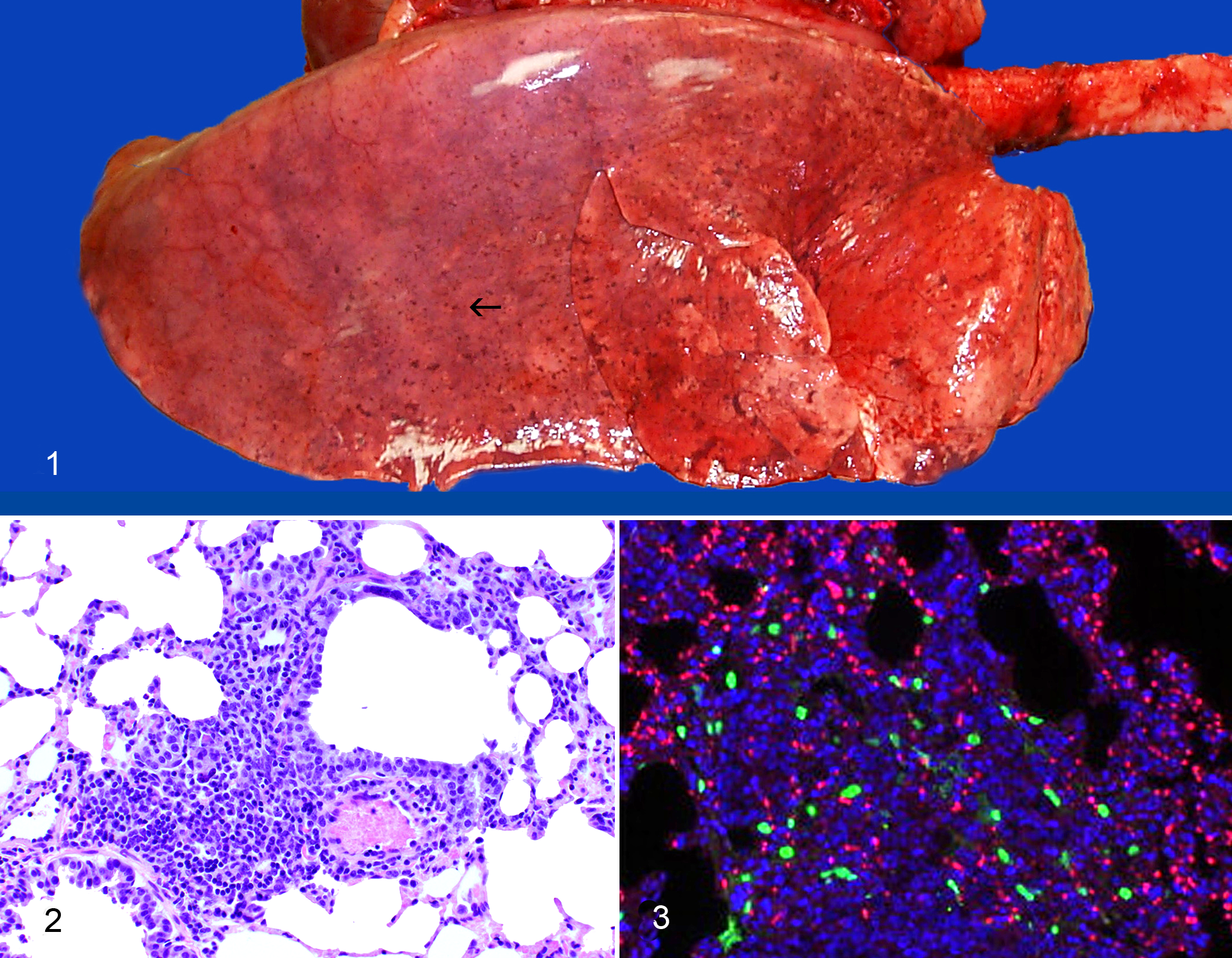
Three other small ruminant species can be productively infected with OvHV-2: bighorn sheep (Ovis canadensis), domestic goats (Capra hircus), and mouflon (Ovis orientalis). Although the data are limited, mouflon—considered an ancestor of domestic sheep—appear to be particularly productive sources of OvHV-2.
MCF in American Bison ( Bison bison )
MCF affects plains and wood subspecies of Bison bison, in addition to wisent (European bison; Bison bonatus). 7,131,132,136 Bison with MCF due to OvHV-2 are generally found depressed and isolated from herdmates. 7,94,95,131,132 More commonly, affected bison are found dead or dying, due to the large size of bison pastures and the infrequency with which herds are checked. Owners become adept during the course of large outbreaks at recognizing more subtle clinical features such as feed refusal, oculonasal discharge, conjunctival hyphema, fever, dysentery, hematuria, and stranguria. The florid oculonasal lesions typical of most cattle with acute MCF tend to be milder in bison, perhaps because of a shorter clinical course, typically <1 to 4 days. 94,132
Mortality rates due to MCF among bison herds can be high, in some instances approaching 100% when animals are stressed or crowded in feedlots. 18 Presumably this reflects the low doses of OvHV-2 that are capable of establishing infection (5 × 103 OvHV-2 DNA copies). 37 In 1 documented outbreak, 825 feedlot bison (51.2% of the exposed group) died following exposure to sheep. 73 More typical was the experience in 1 feedlot, which had annual mortalities (of all causes) of 3.9% to 9.4% over several years. In the year when the impact of MCF was quantified, >75% of deaths were due to MCF. 94 A puzzling feature of OvHV-2–induced MCF is the long distances (up to 5 km) over which infection is transmissible. These estimates are based on natural outbreaks, such as when sheep feedlots are established adjacent to established bison operations that had no prior history of MCF. 65 The importance of aerosol transmission and the peculiar sensitivity of bison to infection were underscored in a Canadian outbreak where 45 of 163 bison died following exposure for <24 hours to sheep in a sale barn. 7 Losses began 50 days later and peaked at 60 to 70 days, with the final death occurring 7 months after the exposure. It was noteworthy that only the bison exposed to sheep in the sale barn developed disease; none of their unexposed bison herdmates did so. Experimentally, clinical signs develop at >28 days after nebulization. 21 Bison with specific major histocompatibility complex (MHC) class II haplotypes appear more resistant to MCF. 142,143
The predominant form of MCF in bison is acute clinical disease. Chronic and subclinical forms occur but are rare. 37,94,131 As in other OvHV-2–susceptible species, viral replication occurs initially in the lower respiratory tract and peaks at 7 to 12 days after intranasal challenge (Fig. 4). Disseminated lobular foci of interstitial pneumonia affect all pulmonary lobes following intranasal challenge, involving 1% to 2% of parenchyma. Unlike sheep, this stage of infection is not associated with a marked change in the local or regional immune gene expression, which may explain subsequent progression to disease. 21 In natural outbreaks, the preclinical pneumonia is either inconspicuous or masked by aspiration pneumonia by the time clinical disease supervenes. 94 Histological lesions are evident in tissues at 16 to 18 days postinoculation (PI). The onset of MCF coincides with a steep increase in OvHV-2 DNA copy number in leukocytes, peaking at 26 to 35 days PI. 21
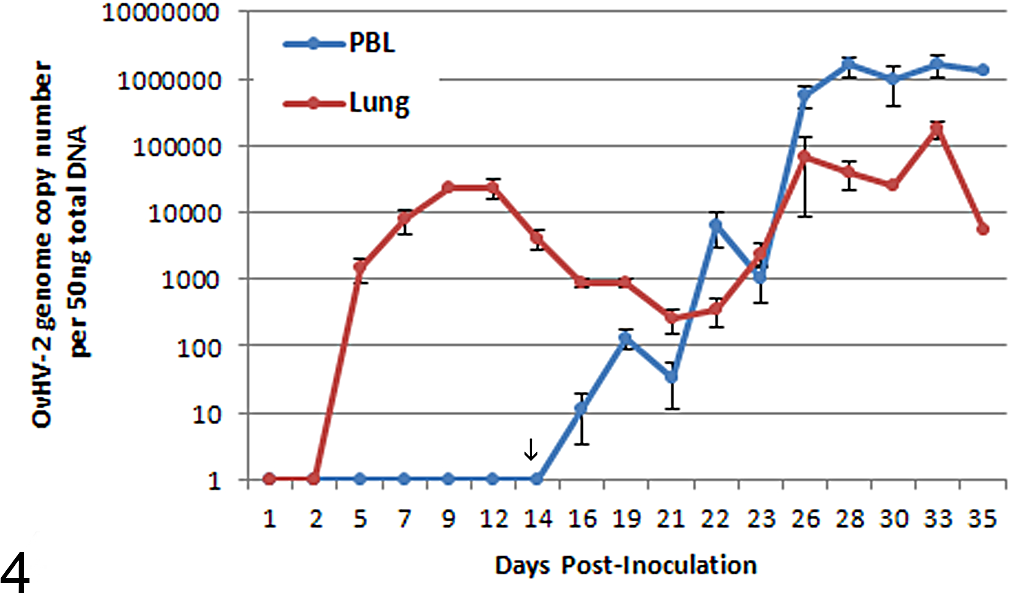
Ovine herpesvirus 2 (OvHV-2) copy number in lung (red) and peripheral blood leukocytes (PBLs) (blue) in bison following experimental challenge. There is a transient peak in lung at 9 to 11 days postinoculation, followed by a steady rise after 14 days in PBLs (arrow). Low challenge doses extend the period of time for the PBL rise in copy number by several weeks (experimentally) or possibly months (natural outbreaks).
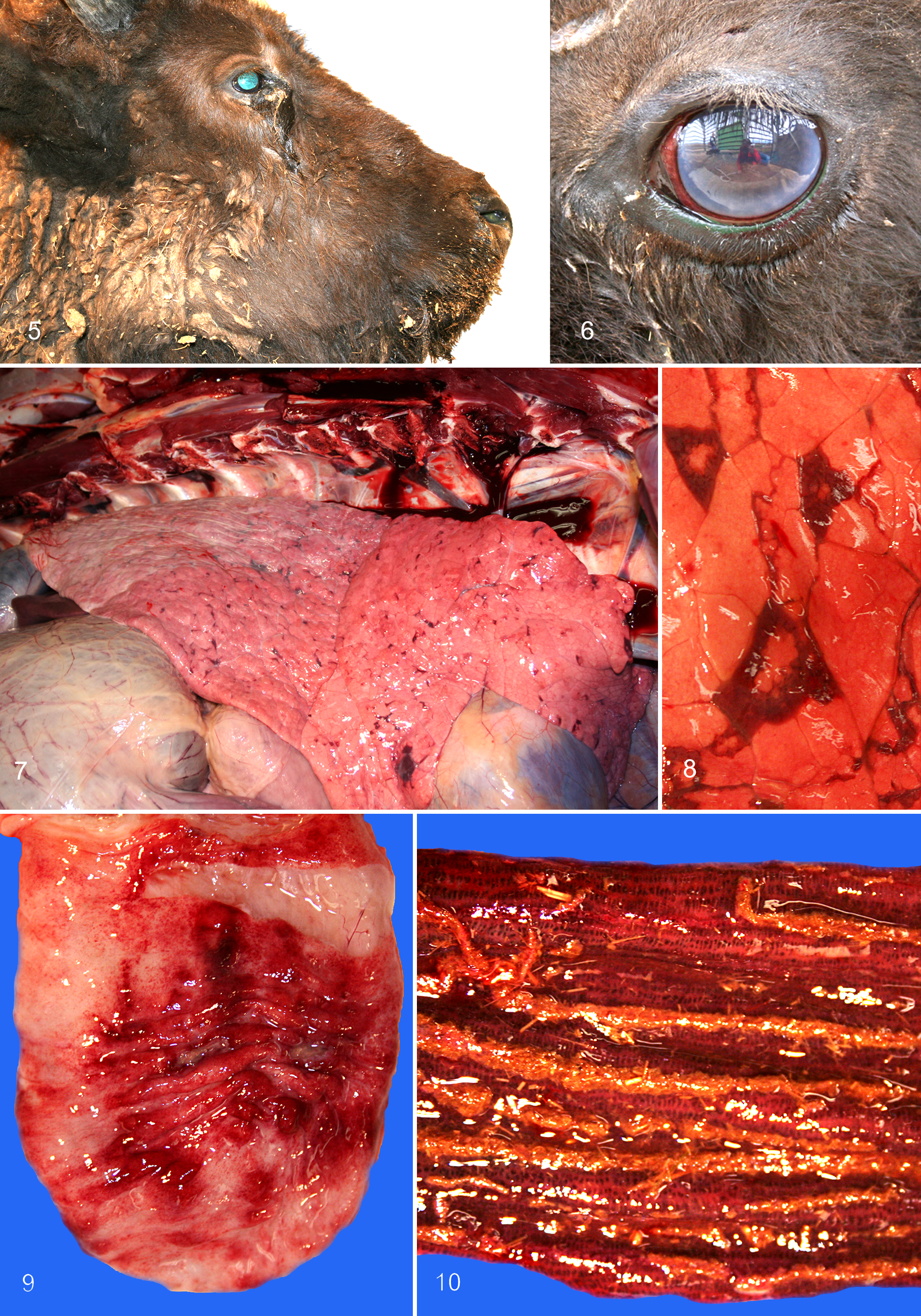
ORF25 transcripts are an index of lytic viral gene expression and are detectable in multiple tissues as clinical signs develop following challenge in bison, in contrast to sheep, wherein ORF25 transcripts are restricted to the lungs during the initial replication. 21,57 Once clinical signs supervene, OvHV-2 is detectable in multiple tissues in addition to lung. 21,50 Characteristic gross lesions in terminally affected bison are oculonasal discharge, hemorrhagic cystitis, necrohemorrhagic typhlocolitis, erosive-ulcerative rhinitis, stomatitis, pharyngitis, laryngitis, and esophagitis (Figs. 5–10). Once owners learn to identify hemorrhagic cystitis, many become proficient using this feature to make a tentative gross diagnosis in field necropsies. Ulcerations are generally present in forestomachs and abomasum. 94 Generalized lymph node hyperplasia is present, but rarely is it as pronounced as in cattle. Some bison abort during the clinical course, presumably due to fever and toxemia. Bison terminally ill with MCF are often attacked and wounded by herdmates. Traumatic lesions due to goring are relatively common at necropsy in bison dying of MCF and may be incorrectly assumed to be the sole cause of illness.
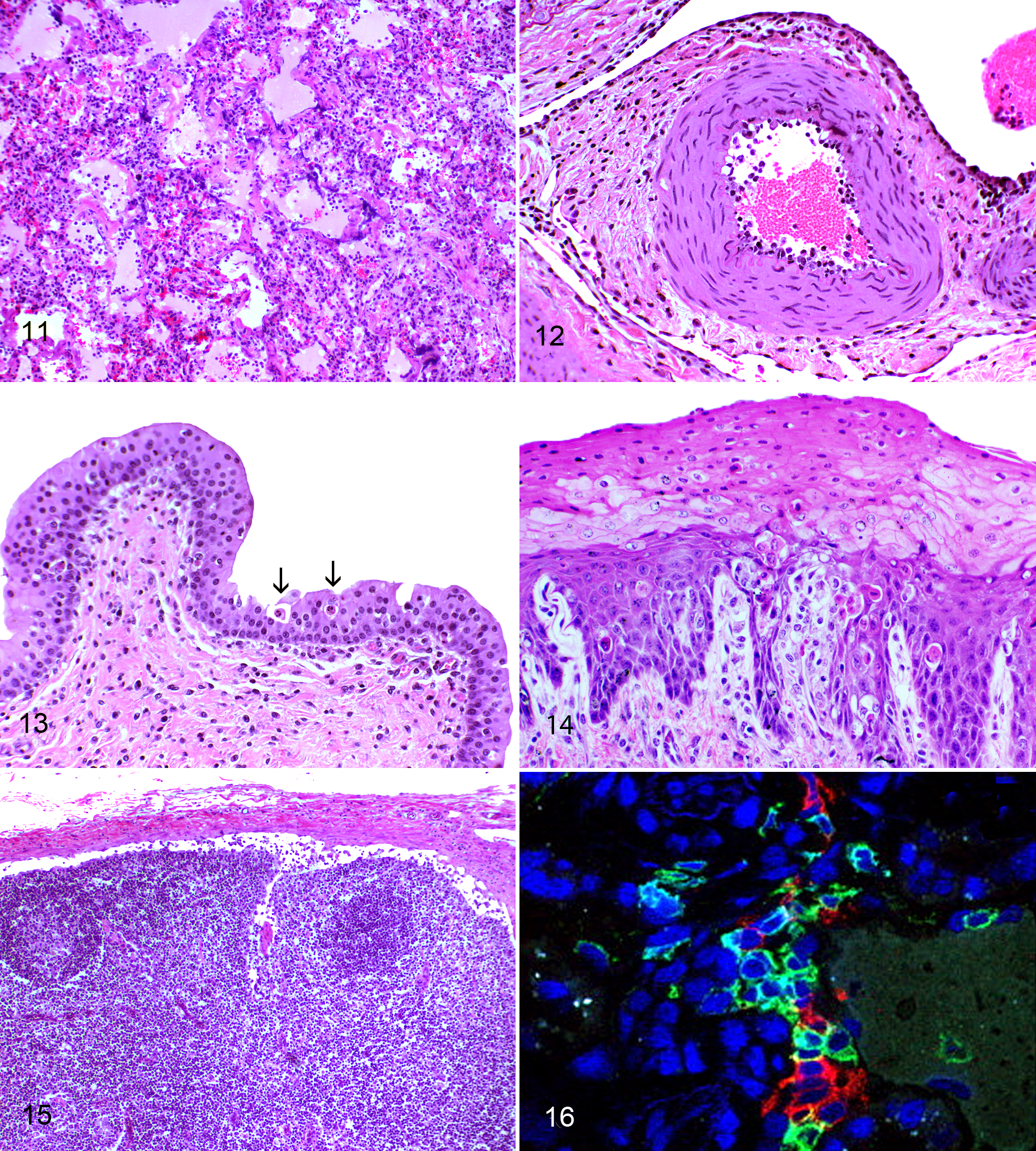
Histological lesions comprise arteritis-phlebitis of medium-caliber vessels, hyperplasia of parafollicular areas of lymph nodes, and apoptosis and/or necrosis in skin, digestive tract, and urinary tracts (Figs. 11–14). Degeneration of urothelium in renal calyces, ureters, bladder, and urethra is a consistently helpful diagnostic feature in fresh carcasses. In some animals, lymph nodes are devoid of lesions, apart from fibrin deposition in sinusoids and around medullary veins (Fig. 15). Arteritis-phlebitis is less florid than in cattle, with minimal or no fibrinoid necrosis in tunica media detected even after multiple tissues are examined. 132 A grading system was developed for arterial lesions in MCF; 92 most bison have grade 1 arteritis with pronounced endarteritis. 94,95,132 Arteritis-phlebitis affecting the carotid rete, which is almost invariant in cattle with acute or chronic MCF, is absent in some bison. The surprisingly modest histological lesions of MCF may be masked by rapid autolysis due to high fever, summer heat, or insulation by winter pelage and heavy body fat. In such situations, it can be difficult to evaluate the digestive tract for lesions. The presence of hemorrhagic cystitis is a helpful clue that MCF may be involved. Some degree of nonsuppurative meningoencephalitis with mild multifocal gliosis is present. Multifocal myocardial necrosis, presumably due to exertional myopathy, is common in bison with clinical signs of longer duration (>3 days). There is generally a correlation between the duration of clinical signs, on one hand, and the severity of lesions and OvHV-2 copy number, on the other. Despite the pansystemic nature of MCF in bison, some tissues are negative for OvHV-2. 132 Sampling at least 2 tissues for the detection of OvHV-2 DNA, ideally lymphoid and nonlymphoid tissue, increases the likelihood of corroborating a morphological diagnosis of MCF, particularly when fixed tissues are used. 18,19,94,132
A proportion of clinically healthy bison has detectable OvHV-2 DNA in peripheral blood, suggesting subclinical or latent infection. 21,94 Typically, copy number is low and such bison are only intermittently positive. An experimental attempt to define a dose of OvHV-2 that established subclinical infection was unsuccessful. 37 Lower challenge doses of OvHV-2 resulted in longer incubation periods for MCF. Once a threshold level of viral DNA was reached in peripheral blood, infection progressed to acute clinical disease at a relatively constant rate. 37 Intravenous challenge of bison with OvHV-2 did not induce MCF. 21
The pathogenesis of MCF in bison, as in other species, remains unresolved. No viral particles are detectable ultrastructurally, and the expression of viral antigens in tissue is limited. It is generally assumed that the small fraction of infected lymphocytes induces proliferation and deregulation of uninfected cells. The latter may induce lesions through nonspecific cytotoxicity. 122 Interestingly, suppression of lymphoid hyperplasia did not prevent fatal MCF in rabbits. 14 There is little indication that B lymphocytes play a significant role in preventing or exacerbating disease, notwithstanding their presence in lesions. 91 In our experience, occlusive thrombosis is rare in acute MCF in bison and is unlikely to explain extensive mucosal necrosis. Immunophenotyping studies of lesions in bison with clinical MCF demonstrated the predominance of CD8+ and CD3+ cells (bison) and CD8+ and CD4+ cells (cattle). 91 Inflammatory infiltrates contain a mixture of CD4+/perforin-αβ T cells (T regulatory or T helper cells), cytotoxic γδ T cells of the innate immune system, B cells (CD79a+), and macrophages (Fig. 16). Most infected cells coexpressed CD3+ and CD8+ in 1 study of MCF vascular lesions. 135 The notion that CD8+ cells are selectively infected is supported by studies in rabbits and calves following experimental infection with AlHV-1. 29,99
MCF in Domestic Cattle
The clinical signs and lesions of classic MCF in domestic cattle (Bos taurus; Bos indicus) are described in detail elsewhere. 10,78 –80,108 Some of the neglected older literature contains astute clinical observations about clinical signs and their progression over time. 83 MCF outbreaks in herds are typically of low morbidity and high mortality, generally but not exclusively affecting younger cattle (8–24 months). 18,92,104,147 Outbreaks with clinical signs in >5% of individuals are the exception rather than the rule. The natural incubation period for cattle is generally stated to be 2 to 10 weeks but can be as long as 9 months. 108 In a recent outbreak following exposure of cattle to sheep in an enclosed space at a state fair, the mean number of days to death due to MCF was 71 with a range of 46 to 139. 86 The incubation period is shorter when cattle are challenged with infected blood from acutely infected animals, typically around 30 days. 80,102,103 Of the various clinical forms of the disease, the “head and eye” form is the most common and characteristic. While terms for other clinical forms of the disease (peracute, alimentary, neurological, cutaneous) are entrenched and likely ineradicable, 108 oculonasal and oral lesions are almost invariably present in individual cattle with acute MCF. They will be found when looked for. Skin lesions, when present, can be a useful indication of the presence of MCF.
Abrupt onset fever (>41°C), depression, keratoconjunctivitis, lymphadenopathy, oral ulcers, and rhinitis are typical. 146 MCF is sufficiently characteristic in cattle that, once clinicians have seen a laboratory-confirmed case, they are unlikely to misdiagnose a subsequent outbreak. Clinicians unfamiliar with MCF often suspect mucosal disease or bovine rhinotracheitis first. Until it was considered eradicated worldwide, rinderpest was important to exclude as a possibility in endemic areas. 108 Other considerations are bluetongue, epizootic hemorrhagic disease, vesicular stomatitis, foot and mouth disease, and photosensitization. 10,108 The clinical course in most symptomatic cattle generally results in death or euthanasia after 2 to 18 days. Disseminated arteritis-phlebitis, diffuse lymphoid hyperplasia, and ulcerative lesions in digestive, respiratory, and urinary tracts are characteristic. 10 Lesions occur elsewhere, including in brain, retina, uvea, cornea, synovia, and skin. Interstitial lymphocytic nephritis is generally present and may be sufficiently severe to be identified grossly. 10 Useful tissues for morphological confirmation of MCF are carotid rete or, in bulls, pampiniform plexus. A diagnosis of MCF should be reconsidered in cattle if no arteritis is present.
The morphological hallmark of MCF in cattle is severe lymphocytic arteritis-periarteritis with necrosis of tunica media. 10 Distinctive large lymphocytes are present, mixed with small lymphocytes and plasmacytes. They consist predominantly of T lymphocytes, with smaller numbers of B lymphocytes, monocytes-macrophages, and neutrophils. 122 Outbreaks involve direct or indirect exposure to sheep. Atypically large outbreaks have occurred when lamb and cattle feedlots were adjacent to each other. In such situations, producers may accept sporadic annual losses due to MCF as the cost of business. With the availability of serological and molecular diagnostic assays, it is clear that a fraction of acutely affected cattle recover. 92,93,153 Cattle surviving acute MCF generally have chronic lesions in medium-caliber arteries and in cornea. 92 Vascular lesions consist of proliferative arteriopathy with varying degrees of recanalization. Ocular lesions consist of anterior or posterior synechiae and fibrosis in corneal substantia propria. Perforating corneal ulcers are common, resulting in entrapment of the iris in the healing cornea. As in bison, there is evidence that subclinical and/or latent infection occurs in cattle, representing an intriguing, poorly understood epidemiological aspect of MCF. 18,68 Experimentally induced aerosol transmission of cattle is possible, but the high doses required militate against using cattle in large-scale studies at present. 140 Preclinical events in cattle at the molecular and morphological level remain poorly defined. The significance of finding low levels of OvHV-2 in peripheral leukocytes of healthy cattle is unclear. 18,111
MCF in Water (Swamp) Buffalo (B. bubalis)
Reports of MCF due to OvHV-2 are sparse in this species (reviewed in Hoffmann et al 46 ), but water buffalo are likely to be highly susceptible given the absence of disease in co-housed domestic cattle. 25 Clinical signs and lesions are similar to those in cattle, with the exception that fibrinoid arteritis is uncommon according to some but not all reports, 141 oral and esophageal lesions are rare, keratitis is relatively mild, 141 and epicarditis-myocarditis is generally present, commonly with myocardial necrosis. 25,44,45,141 Recovery occurs but is likely to be rare. 141
MCF in Cervid Species
MCF has been reported in at least 13 species of deer. 35,43,44,53,54,77,100,115,126,133 As the captive deer industry expanded in New Zealand, Scotland, and elsewhere, MCF became important due to its high morbidity and cumulative mortality. 82 The number of species in which “typical” lesions of MCF occur and MCFV has been conclusively demonstrated by molecular means is limited. Many reports of MCF in the family Cervidae predate the availability of specific molecular assays in the late 1980s to early 1990s. 43 Some cervid species, particularly white-tailed deer and Père David’s deer (E. davidianus), are highly susceptible. 11,53,77,98,100 Moose (Alces alces) and elk (Cervus canadensis) appear to be moderately susceptible. Fallow deer (Dama dama) are relatively resistant. 115,122 Sheep-associated MCF in cervid species is characteristically acute, resulting in death within 1 to 3 days. 44 Clinical signs resemble those in bison: depression, separation from herdmates, reluctance to move when approached, and neurological signs such as convulsions. 44 Gross lesions are hemorrhagic with a prominent gastrointestinal component. 44 Other lesions include congestion and petechial or ecchymotic hemorrhage in multiple tissues, including skeletal muscle, and lymphadenopathy. As in bison, affected deer are often found terminally ill or dead. The proportion of affected animals in captive herds may be high. Vascular lesions affect multiple organs and consist of lymphocytic vasculitis and perivasculitis, fibrinoid necrosis of tunica media, and varying degrees of thrombosis. 11 Lymph nodes exhibit lymphoid hyperplasia in paracortical and interfollicular areas. Diagnostic considerations in deer with MCF-like signs include clostridial enteritis, epizootic hemorrhagic disease/bluetongue, adenoviral infection, and yersiniosis. 122 Surveys of healthy free-ranging and semi-domesticated cervid populations for evidence of MCFV infection, while limited, indicate that subclinical infection is relatively rare. 6,26,36 A distinctive form of chronic MCF with granulomatous hyperplastic cutaneous lesions with folliculitis is reported in several species, particularly sika deer (Cervus nippon). 20,35,51 OvHV-2 or CpHV-2 has been found in individual cases. In addition to OvHV-2, which is responsible for most outbreaks of MCF in deer, CpHV-2, AlHV-1, and MCFV–white-tailed deer (WTD) can also cause disease. 51,77
MCF in Domestic Pigs (Sus scrofa)
One surprise in the past 15 years was the recognition that pigs are infected sporadically with OvHV-2 and develop MCF. 1,2,81,150 An MCF-like syndrome was recognized recurrently in pigs in European countries as early as 1901, but confirmation was not possible before the early 1990s. 1 Pigs are similar to cattle in presenting with a head and eye form of MCF. Gross lesions tend to be minimal in acutely affected animals. 39,55 In addition to typical disseminated arteritis affecting medium-caliber vessels, there is an unusually florid nonpurulent meningoencephalitis, possibly in response to high viral loads at that site. 1 Lesions in meninges may be sufficiently severe to be recognized grossly. Pulmonary edema and bronchopneumonia may be prominent and account for the principal presenting sign of respiratory distress. 1,137 Abortions and stillbirths were a feature of one outbreak. 39 The proportion of affected pigs tends to be small and the contact with sheep unusually close. Mild and chronic forms of an MCF-like disease may be seen concurrently. 2 MCF has been induced experimentally by aerosol challenge with OvHV-2, requiring relatively high concentrations of virus. 55 Detection of ORF25 and ORF73 in tissues suggests that OvHV-2 lytic gene expression occurs as clinical disease develops. 55
MCF in Domestic Goats (Capra aegagrus hircus)
Goats are natural hosts of CpHV-2 and can be infected asymptomatically with OvHV-2. 17,66,67 There is recent evidence that goats harbor MCFV-WTD, the agent responsible for some outbreaks of MCF in white-tailed deer. 56 An MCF-like disease has been seen in domestic goats based on the presence of generalized arteritis and detectable OvHV-2. 34,48,145 In most goats, infection with OvHV-2 is asymptomatic. It is not clear whether the MCF-like syndrome in goats reflects unusually high challenge with OvHV-2 or is a peculiar response to infection, or if OvHV-2 DNA is unrelated to the presence of lesions.
Goats are asymptomatically infected with CpHV-2 with patterns of transmission similar to that of OvHV-2 in flocks. 67 Disease due to CpHV-2 has been reported in sika deer (C. nippon), roe deer (Capreolus capreolus), moose, white-tailed deer, water buffalo (B. bubalis), and pronghorn (Antilocapra americana). 60 Many animals present with weight loss and alopecia with serocellular crusting of the head, neck, thorax, abdomen, and limbs. Euthanasia is often required. Severe chronic lichenoid granulomatous lymphocytic dermatitis and destructive folliculitis with giant cell formation are present. Internal lesions are granulomatous and associated with lymphocytic fibrinoid vasculitis in brain, leptomeninges, lymph nodes, adrenal glands, and liver. 20,35,51
MCF in Laboratory Rabbits (Oryctolagus cuniculus)
The use of large ruminants for transmission and pathogenesis of MCF is ideal, but there are limitations due to high animal care costs, small size of test groups, and range of available immunological reagents. Laboratory rabbits, in addition to other laboratory species, 49,120 have been used for aerosol studies with OvHV-2 due to the similarity of lesions to those in naturally affected ruminant species. 24,58 MCF can also be induced in rabbits by inoculation of lymphoblastoid cells derived from clinically affected animals, cell suspensions from MCFV-infected lymph nodes, or from other tissues such as spleen or cornea, 13 as well as by inoculation with AlHV-1. 8,31 MCF has been induced using OvHV-2 aerosols derived from sheep and with bacterial artificial chromosome-derived AlHV-1. 31 Following intranasal challenge, replication occurs in lung at 11 days postinoculation (PI). 58 As in bison, a second peak in viral gene expression coincides with the onset of clinical disease, which in rabbits is at 20 to 30 days PI. 30,58 OvHV-2 replication and dissemination and immunological responses resemble those in bison. Symptomatic rabbits have well-developed postmortem lesions of splenomegaly, generalized lymphadenopathy, and multifocal intrahepatic foci. Histologically, there is diffuse lymphadenitis, with progression to granulomatous inflammation in more severe lesions. Lymphoid necrosis is a feature in gut-associated lymphoid aggregates. Pulmonary lesions consist of interstitial pneumonia with phlebitis-arteritis. Hepatic lesions comprise cholangitis, hepatic necrosis, portal vasculitis, and portal hepatitis extending into periportal hepatic parenchyma. As in lymph nodes, larger lesions in liver are distinctly granulomatous. It is sometimes stated that disease induced by OvHV-2 and AlHV-1 in rabbits is undistinguishable from MCF in naturally susceptible species. 29,126 The granulomatous character of OvHV-2–induced lesions following nebulization is atypical of acute MCF in domestic cattle and bison in our experience. Vasculitis in affected rabbits tends to be modest and limited in distribution. Rabbits resemble natural hosts in the minimal amount of detectable viral DNA in tissue. 8 It is unclear the extent to which the absence of the full spectrum of MCF lesions as seen in susceptible ruminants lessens the value of rabbits as laboratory models. The initially promising results from vaccination trials using AlHV-1 in the 1980s could not be repeated in cattle. 110 Not surprisingly, there are distinct differences in rabbits between the lesion profile following nebulization with OvHV-2 and intravenous challenge with OvHV-2–infected lymphocytes. Both models result in extensive necrosis. 3,129 The predominant inflammatory cell component comprises CD8+ T lymphocytes. It is assumed that only a small fraction of T lymphocytes in lesions are infected with OvHV-2, as in AlHV-1-infected rabbits. 8 Depending on the route of inoculation and the agent used, a latent or a lytic viral gene expression profile occurs in terminally affected rabbits. 29
Conclusion
It is de rigueur when writing a review like this to conclude with a ringing declaration that more research is needed, in the hope that someone will cut the researchers a check. Indeed, we wish to live so long that we will someday encounter an abstract stating that exactly the right amount of research has now been done! But despite its recognition for >300 years, more research is needed for the MCF disease complex. Much emphasis has been placed on the wildlife-livestock disease interface in recent years, with the implication that problematic diseases generally flow from wildlife to livestock. The flow is in the opposite direction with OvHV-2. As a result, the recognition of MCF in wildlife populations often occurs when captive animals are in close proximity to sheep.
At times, it has seemed that the biology of the MCFV group is, to paraphrase Haldane, not just queerer than we suppose, but queerer than we can suppose. As suggested in the introduction, 2 issues are paramount for managers of MCF-susceptible species. One is whether the disease can be effectively controlled, ideally through vaccination. Early efforts were discouraging since little protection was afforded. Even in animals that survived the initial challenge, the duration of immunity was brief. 32,110,126 Recombinant bacterial artificial chromosomes containing the genome of pathogenic AlHV-1 can induce MCF in rabbits 30 and are used to establish the role in infection of individual genes. There have been promising results using an intranasal challenge model and an attenuated AlHV-1 vaccine candidate, with the goal of evoking protective mucosal responses. 125 If, as seems likely, there is a promising AlHV-1 vaccine candidate, it make sense to see whether it will protect bison to challenge with OvHV-2.
The other issue is the spatial distance over which disease is transmitted. Traditional recommendations for spatial separation between sheep and susceptible species are difficult to apply given the long distances over which OvHV-2 can be transmitted. 65 In special situations, such as on islands, mandating spatial separation between sheep and susceptible species is a reasonable approach. 122 Establishment of OvHV-2–free flocks is possible in locations like petting zoos. 60 Unfortunately, there is no personal incentive for owners of commercial flocks to create and maintain OvHV-2–free flocks. A recurrent issue is the introduction of flocks onto land adjacent to bison, resulting in heavy continuing losses with no legal or regulatory recourse for owners. Three avenues that beckon are the development of effective vaccines, 30 identification of genetic resistance factors, 143 and better definition of safe distances between carrier and susceptible host species. Diagnosticians now have effective tools to establish a diagnosis of MCF before and after death. 116,134 For researchers, the central enigma of the disease remains its pathogenesis, specifically how immune dysregulation is triggered. A long dominant assumption was that a small fraction of lymphocytes infected by OvHV-2 or AlHV-1 coordinated an unregulated inflammatory response by uninfected lymphocytes. 8,12 As the capability of detecting OvHV-2 in tissue and the proportion of detectably infected lymphocytes grows, there is a possibility that infected lymphocytes directly mediate lesions. 21,126 In a recent study of AlHV-1–induced MCF, it was estimated that approximately 10% of CD8+ T cells were infected, suggesting that these cells replicate intensively or eliminated uninfected cells. 29
The importance of lifelong latent or subclinical infection in MCF-susceptible species is unclear. This likely occurs in domestic cattle and in some wildlife species, including bison, moose, reindeer (Rangifer tarandus), roe deer (C. capreolus), and red deer (Cervus elaphus). The sporadic occurrence of MCF in herds that had no recent contact with domestic sheep may be due to reactivation of infection, in addition to prolonged incubation.
Time will tell whether efforts to develop effective vaccines for MCFVs bear fruit. If a candidate product is developed, a separate issue is whether a market exists for its use. That bridge will have to be burnt when we get to it. An AlHV-1–based vaccine that protects both cattle (from AlHV-1) and bison as well as other susceptible species (from OvHV-2) might be economically viable.
Footnotes
Acknowledgements
This article is respectfully dedicated to bison owners who continue to work with bison researchers to ensure that useful information is derived from outbreaks of MCF, and to the research animals that died in the course of the experimental studies. We thank colleagues Drs Naomi Taus and Danielle Nelson for permission to republish images from their work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
