Abstract
Two cases of sheep-associated malignant catarrhal fever (MCF) in pigs were diagnosed on a small farm in New York, and in Kentucky. In both cases, the initial diagnosis was based on histopathological changes representing typical lymphoproliferative vasculitis in multiple tissues of the affected pigs. Ovine herpesvirus 2 (OvHV-2) DNA was detected by polymerase chain reaction in the tissues of affected pigs in both cases. The amplified sequences were identical between the clinically affected pigs and the OvHV-2-infected sheep. Additional virological and bacteriological examination showed that the affected pigs were negative for agents that cause Aujeszky's disease, classical swine fever, porcine enterovirus, and rabies. An antibody against a conserved epitope among MCF viruses was detected in 1 clinically affected pig and 2 unaffected cohort pigs, as well as in all the associated sheep. To the authors' knowledge, this is the first report of porcine MCF in North America, and suggests that OvHV-2 is associated with clinical MCF in pigs.
Malignant catarrhal fever (MCF) is a frequently fatal, generalized herpesviral disease primarily affecting cattle, bison, deer, and many other species of wild ruminants. It is characterized by high fever, copious nasal discharge, corneal opacity, generalized lymphadenopathy, lymphopenia, inflammation and necrosis of mucosal surfaces, and severe vasculitis. 16 Although several other newly identified MCF viruses can cause the disease in certain species, 4,5,9,14 Alcelaphine herpesvirus 1 (AlHV-1) and Ovine herpesvirus 2 (OvHV-2) are, to date, the 2 major forms of the disease, and both forms are clinically and pathologically indistinguishable. 16 Alcelaphine herpesvirus 1 causes the African or “wildebeest-associated” form, and wildebeest are the reservoir for the virus; OvHV-2 causes the “sheep-associated” form, and sheep are the natural host. In the United States, MCF has been reported in cattle, bison, cervids, and other wild ruminants. 5 The majority of the cases have been associated with OvHV-2 originating from sheep. 3 Although OvHV-2 has not been grown in cell culture, OvHV-2 DNA can be detected in peripheral blood leukocytes and tissues of infected animals by molecular assays, such as polymerase chain reaction (PCR). 2
Malignant catarrhal fever has been reported in pigs in Europe where the etiologic agent, OvHV-2, was detected by PCR in affected animals in Norway 12 and Switzerland. 1 In those European cases, typical MCF histologic findings included lymphoproliferative vasculitis with meningitis, catarrhal bronchopneumonia, bronchitis, bronchiolitis, and edema. 1 Since the early 1900s, porcine MCF has been recognized clinically in Norway, 15 Germany, 8 Italy, 13 Switzerland (Pohlenz J, Bertschinger HU, Koch W: 1974, A malignant catarrhal fever-like syndrome in sows. Proceedings of Third International Pig Veterinary Society Congress, Lyon, France 15:1–3), and Sweden, 6 without confirmation of the causative agents. In the current report, the first 2 cases of systemic MCF in pigs in the United States are described. The presumptive diagnosis of MCF was made based on histologic changes, particularly lymphoproliferative vasculitis, and OvHV-2 DNA was detected by PCR procedures in the clinically affected pigs. To the authors' knowledge, these are the first confirmed cases of porcine MCF in the United States and outside the European continent.
In the New York case, a 2-year-old, mixed-breed, castrated, male pig, weighing 295 kg, was presented to the Cornell University Hospital for Animals (Ithaca, NY) with depression, inappetence of up to 2 days duration, and pyrexia (39.7°C). The pig lived on an animal rescue farm with 2 other adult pigs, on a farm with 3 adult sheep, 4 ducks, and 2 chickens. The pigs all lived next to the sheep and were able to have nose-to-nose contact. No other animals were showing any signs of disease on the premises before or during the pig's illness. On examination, the pig was depressed and recumbent, but it had a normal gait and was able to rise. Cardiovascular status was normal, but there was an exaggerated abdominal press associated with end-expiration. There were no adventitial lung sounds. Abdominal borborygmi were absent, and feces were scant, hard, and mucus-covered. Mild corneal edema was present bilaterally, and pupils were miotic, but there were no corneal erosions. Thoracic radiographs and urine and fecal analyses were unremarkable. Hematologic evaluation revealed moderately reactive lymphocytes (resolved in 5 days). Throughout the 7 days of hospitalization, the pig's rectal temperature ranged from 39.1°C to 39.8°C. The degree of pyrexia was not correlated with the severity of clinical signs. Antipyretic and antibiotic therapy failed to alter clinical signs. On day 4 in the hospital, the pig was abruptly more lethargic, and there was an onset of a crusting rhinitis with mild stertor. On day 6 in the hospital, the pig developed mild ataxia (central vestibular) that progressed over a period of 48 hr to severe and violent balance loss to either side. When recumbent, the pig had spastic limb and trunk movements that were either the result of seizures or unsuccessful attempts at righting. Attempts to obtain cerebrospinal fluid were unsuccessful. Corneal edema became severe, and uveitis was apparent. The rhinitis progressed to nonpatency with open-mouthed breathing and severe distress. The pig was euthanized on the morning of day 8.
In the Kentucky case, an adult pregnant sow of unknown breed, weighing approximately 80 kg, was presented for necropsy after previously being treated for pneumonia 3 months before. The animal's rectal temperature was 41.1°C for 2 days. The pig was housed with 2 other adult pigs and 3 adult sheep in the same barn but were not kept together in the same pens. Length of contact was unknown. The barn is a high-school agricultural facility. The animal was treated with flunixin meglumine a and isoflupredone acetate. b The pig died the third day after presentation. Other pigs in the group had similar signs but did not die. In both cases (New York and Kentucky), the affected animals were housed with several sheep.
A complete postmortem examination was performed on both pigs, and only a small number of macroscopic lesions were observed. Both animals were in good body condition. In the New York case, the kidneys were diffusely pale, and numerous petechial hemorrhages were scattered across the cortical surface and, on section, through the cortex and medulla. A focal renal cyst, filled with a clear, straw-colored fluid was found incidentally in the right kidney. In addition, there were numerous small (<5 mm) erosions on the mucosal surface of the stomach. There were multifocal small areas of saponification in the mesenteric fat immediately surrounding the pancreas. The meninges were mildly congested. The corneas were markedly opaque, and the conjunctivae were congested. No gross findings were identified in lungs, liver, heart, or gastrointestinal tract. In the Kentucky case, the only macroscopic lesions were found in the lungs and pancreas. The diaphragmatic lobes were, bilaterally, mildly firm and dark red; the pancreas was severely hemorrhagic. The sow was pregnant with 10 piglets. The fetuses did not demonstrate any gross abnormalities and appeared viable.
Routine tissue samples were collected and fixed in 10% (w/v) buffered formalin for 4 days. Representative samples were prepared for histopathology. Samples were progressively dehydrated in ethanol, cleared with xylene, and embedded in paraffin wax according to standard laboratory practice. Sections (6 μm) were stained with hematoxylin and eosin (HE) for routine histopathologic examination by light microscopy. Both cases revealed similar histopathologic changes. There was moderate to severe lymphocytic and histiocytic vasculitis with mural fibrinoid necrosis in the lungs, liver, heart, gastrointestinal tract, and meninges. Lesions were most severe in the kidneys, where there was severe, multifocal, interstitial, nonsuppurative nephritis (Fig. 1) associated with fibrinoid necrosis of the walls of most large arteries examined (Fig. 2). In the Kentucky case, severe blastic lymphoid infiltrates expanded the renal cortical interstitium with multifocal vasculitis. Similar moderate blastic lymphoid infiltrates were observed in the adrenal cortex, particularly in the zona glomerulosa and the intestinal submucosa. The cerebrum and cerebellum showed mild to moderate perivascular and meningeal nonsuppurative inflammation and minimal nonsuppurative vasculitis. Vasculitis was characterized as minimal, transmural infiltration by mononuclear cells with minimal karyorrhectic debris. In addition, infrequent intranuclear inclusion bodies were observed in both cases; affected cell contained prominent intranuclear inclusions that were rimmed by peripherally arranged chromatin (Fig. 3). Ocular lesions consisted of severe corneal edema with fibrinous exudates in the anterior and posterior chambers. No ocular lesions were observed in the Kentucky sow.
Small, unfixed samples of kidney, lung, liver, and lymph nodes were stored at 4°C and submitted without delay to the New York State Animal Health Diagnostic Center (Ithaca, NY) for virologic and bacteriologic examination. In the Kentucky case, similar samples were taken and submitted to the Kentucky Livestock Disease and Diagnostic Center (Lexington, KY) for bacterial and viral testing. The virologic and bacteriologic examination in both cases showed that both of the affected pigs were negative for agents that cause Aujeszky disease, classical swine fever, porcine enterovirus, and rabies. Spleen, lymph node, and lung tissues from the affected pig, and peripheral blood samples from 2 normal cohort pigs and 3 sheep in the New York case, were submitted to the MCF Research Laboratory of the Animal Disease Research Unit of the U.S. Department of Agriculture, Agricultural Research Service (Washington State University, Pullman, WA) for the detection of MCF viral DNA and antibody. The identification of MCF viral DNA from the tissues of the affected pig in the Kentucky case was carried out in the Kentucky Livestock Disease and Diagnostic Center, and the detection of MCF viral antibody was carried out at the MCF Research Laboratory. The protocols for OvHV-2-specific nested PCR were the same in both laboratories using primers targeting a portion of open reading frame 75, a gene coding a tegument protein. 2,11 In the New York case, the PCR amplified a 238-bp DNA fragment from all 3 tissue samples (spleen, lymph node, and lung) of the clinically affected pig. The PCR also amplified the DNA fragment from the peripheral blood samples of the 3 New York sheep. Peripheral blood samples from both clinically normal New York pigs (one of which was positive for MCF viral antibody) were negative by OvHV-2 PCR. By realtime PCR, 7,17 targeting the same region of open reading frame 75, OvHV-2 DNA levels in the spleen, lymph node, and lung were detected as 89,500, 119,000, and 382,000 copies per 2 μg of tissue DNA, respectively. In the Kentucky case, a 238-bp DNA fragment was also amplified by nested PCR from the tissue samples (intestine and the pooled tissues) of the clinically affected pig. The amplicons from both clinically affected pigs and sheep were cloned and sequenced. The sequence identities matched the OvHV-2 sequence in GenBank.
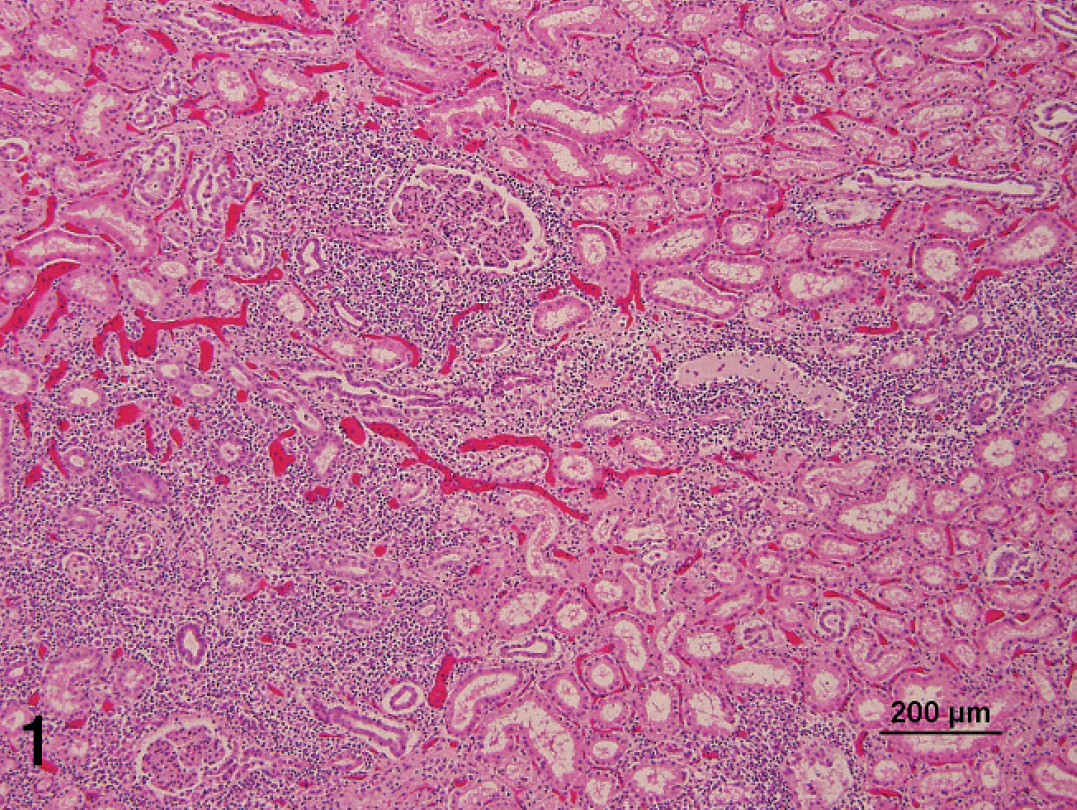
Kidney; severe, multifocal, interstitial, nonsuppurative nephritis. Hematoxylin and eosin. Bar = 200 μm.

Kidney; large blood vessel with vasculitis and transmural fibrinoid necrosis. Hematoxylin and eosin. Bar = 50 μm.

Kidney; large blood vessel with severe vasculitis and 2 intranuclear inclusion bodies (arrows). Hematoxylin and eosin. Bar = 50 μm.
Serum or plasma samples collected from the animals in both cases were tested for MCF viral antibody by competitive inhibition enzyme-linked immunosorbent assay. 10 In the New York case, serum samples from the clinically affected pig, 1 of 2 unaffected cohort pigs, and 3 normal sheep were seropositive. In the Kentucky case, 1 of 2 unaffected cohort pigs and 3 Kentucky clinically normal sheep were positive, whereas no serum sample was available from the clinically affected pig.
To the authors' knowledge, these are the first confirmed U.S. cases of MCF in pigs. The clinical history and histologic features, particularly the lymphoproliferative nature of lesions and vasculitis, were similar to that described in the cases of porcine MCF from Europe. 1,12 The data from the serology test, PCR, and sequence analysis confirmed an association between the disease in the pigs and OvHV-2.
Porcine MCF is a very rare, poorly documented disease of swine, and the differential diagnoses considered in the current case included Aujeszky's disease, classical swine fever, porcine enterovirus infection, and rabies. There is a paucity of reported cases of porcine MCF in the veterinary literature, although a few cases were recently reported from European countries. 1 Reasons could be the limited availability of molecular diagnostic assays and the possibility that many cases may have simply been unrecognized. The low frequency of MCF cases in pig populations may also be due to host susceptibility to the disease, although that has not been determined. A significant difference in host susceptibilities to OvHV-2-induced MCF between bison and cattle revealed how vulnerable bison are to the disease compared with cattle, even under the same sheep exposure conditions. On the other hand, pigs and sheep usually do not share close environments, which might explain the infrequent presentation of MCF in pigs. In the current cases, it seems likely that sheep were the source of infection, especially because both source farms also housed sheep that were positive for OvHV-2. Despite a low rate of porcine MCF in North America, it is important to keep in mind that contact between pigs and sheep should be discouraged to minimize any unnecessary losses due to MCF.
Acknowledgements. This work was partly supported by the U.S. Department of Agriculture-Agricultural Research Service (USDA-ARS; CWU 5348–32000–024–00D). The authors thank the laboratory staff at the New York State Animal Health Diagnostic Center at Cornell University, and the Kentucky Livestock Disease and Diagnostic Center at the University of Kentucky, for technical assistance. The authors also thank Janice Keller, Lori Fuller, and Shirley Elias at the Animal Disease Research Unit, USDA-ARS, and Washington State University for technical assistance, and Duncan Russell for assistance with the figures.
Footnotes
a.
Banamine® Injectable Solution, Schering-Plough, Summit, NJ.
b.
Predef® 2× Sterile Aqueous Suspension, Pfizer, Groton, CT.
