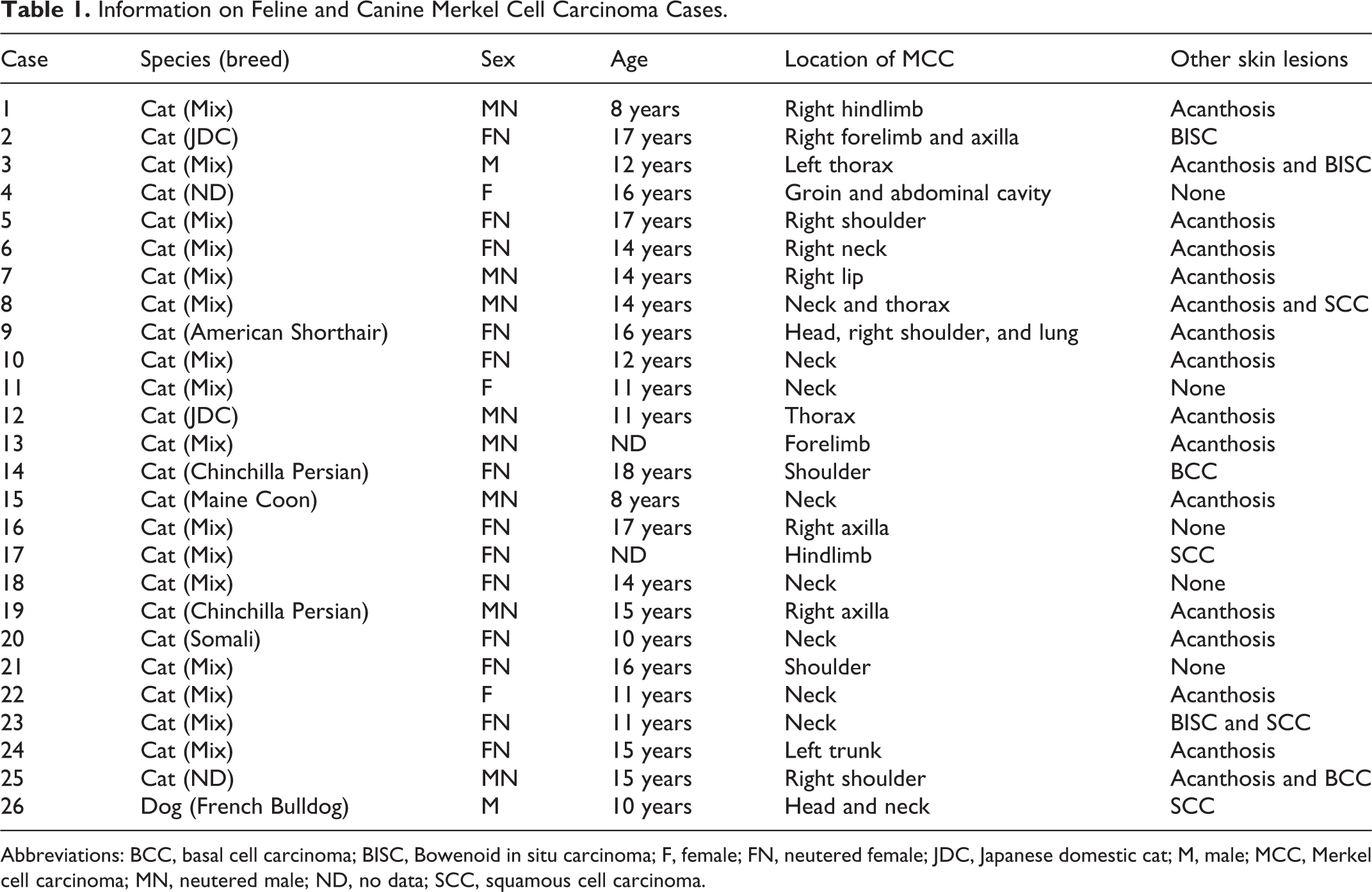
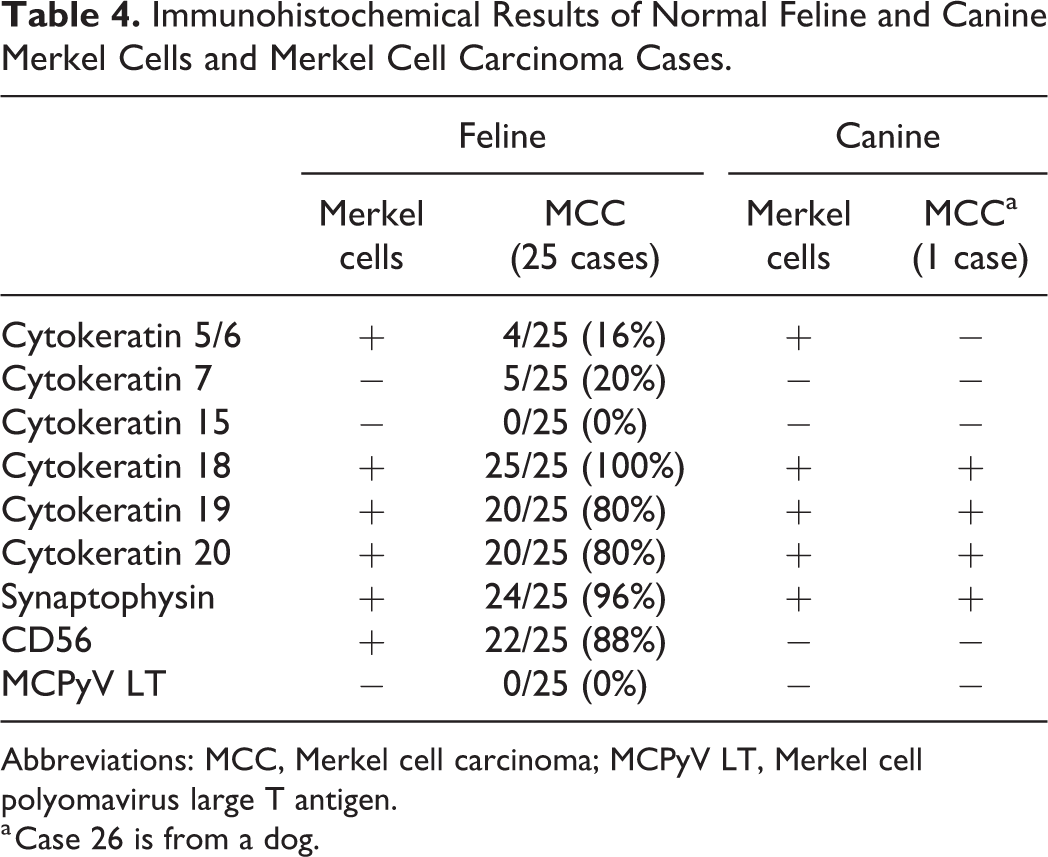
Abstract
Merkel cell carcinoma (MCC) is an aggressive cutaneous neuroendocrine tumor, and most human MCC cases are infected by Merkel cell polyomavirus (MCPyV). However, the underlying pathogeneses of MCC in animals remain unclear. In the present study, newly established cell lines from feline and canine MCC, a MCPyV-positive human MCC cell line, and MCC tissues from 25 cats and 1 dog were examined and compared pathologically. Feline and canine MCCs were composed of tumor cells arranged in trabeculae and solid packets. Twenty out of 25 feline MCC cases (80%) had other proliferative cutaneous lesions, such as carcinoma in situ and squamous cell carcinoma. Among the 25 feline MCC cases, tumor cells were immunopositive for cytokeratins (CKs), including CK5/6 (4/25 cases, 16%), CK7 (5, 20%), CK18 (25, 100%), CK19 (20, 80%), and CK20 (20, 80%). The tumor cells of feline MCC were also immunopositive for synaptophysin (24/25, 96%) and CD56 (22/25, 88%). The tumor cells of canine MCC were immunopositive for CK18, CK19, CK20, and synaptophysin. Cultured feline and canine MCC cells grew in adherent monolayers and exhibited diffuse cytoplasmic immunoreactivity for CKs, whereas human MCC cells grew in suspension and exhibited dot-like cytoplasmic immunoreactivity for CKs. Differences in the distribution of CKs between human and animal MCC may be attributed to cell adhesion propensities. MCPyV genes and antigen were not detected in feline or canine MCC, suggesting a different etiology from human MCC.
Merkel cells are cutaneous touch receptors located in the basal layer of the epidermis. They are distributed in skin regions with high tactile acuity and play a role in light touch responses. 29,30 In animals, including cats and dogs, Merkel cells are also located in sinus hair follicles. 10,37,38 Merkel cells are immunopositive for several epithelial markers, including cytokeratin (CK) 18, CK19, and CK20, and neuroendocrine markers, including synaptophysin, chromogranin, and neuron-specific enolase. 35 Mature Merkel cells are terminally differentiated cells with limited proliferative potential. Pluripotent epidermal stem cells are regarded as the cell of origin during embryonal development as well as for the turnover of mature Merkel cells. 47
Merkel cell carcinoma (MCC) is a rare and highly aggressive tumor in humans. 35 Exposure to ultraviolet light and infection by Merkel cell polyomavirus (MCPyV) are associated with the development of human MCC. 12,35 The large T (LT) antigen of MCPyV is detected in the tumor cells of approximately 80% of human MCC cases. 17 In these cases, the MCPyV gene is integrated into the host genome and the viral LT antigen promotes cell division by suppressing the host retinoblastoma (RB) gene. 35 Although Merkel cells and MCC cells share morphological and immunohistochemical features, the origin of MCC does not appear to be mature Merkel cells because MCC lesions are located in the dermis and/or subcutis, while mature Merkel cells are present in the epidermis. 6,45 Furthermore, immunohistochemistry (IHC) for CK20, a marker of Merkel cells and MCC, show different cytoplasmic staining patterns: diffuse staining in Merkel cells and dot-like staining in MCC cells. Previous studies suggested that epidermal stem cells and dermal fibroblasts are the cells of origin of MCPyV-negative MCC and MCPV-positive MCC, respectively. 45
In animals, MCC has mostly been examined in cats; however, there have been a few case reports of MCC in dogs and a cow. 4,5,10,13,14,20,24,36,44,46 A previous study showed that MCC has a predilection for older cats (median age, 14 years) and is a highly aggressive tumor associated with a poor prognosis. 44 Lesions of feline MCC are most commonly found in the head and neck, and its histopathological features are similar to those of MCC in humans and other animal species. Feline MCC cells are also immunopositive for CK18, CK20, synaptophysin, and p63. Cats with MCC often have other proliferative diseases of the skin, such as epidermal dysplasia, squamous cell carcinoma (SCC), and basal cell carcinoma (BCC). 4,44 Since a larger number of MCC cases has been reported in cats than in other animal species, cats appear to be more susceptible to developing MCC.
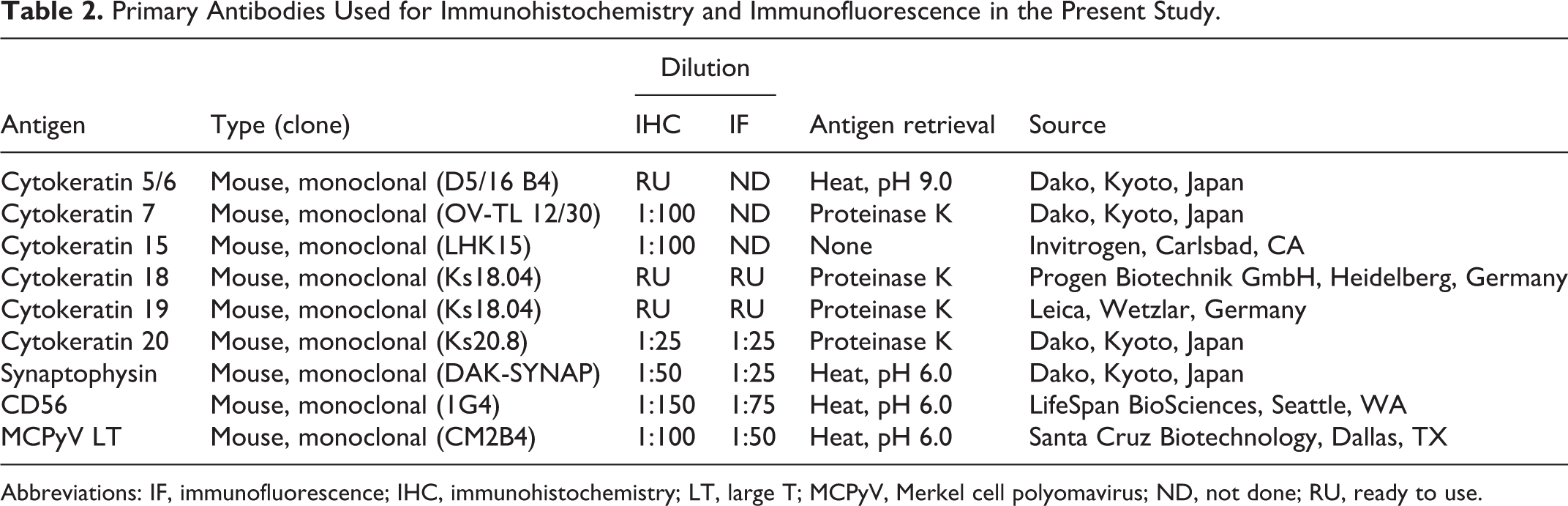
In the present study, to elucidate the underlying causes and cell of origin of feline MCC, novel cell lines of feline and canine MCC were established and their cytological features and MCPyV gene integration were compared with those of a MCPyV-positive human MCC cell line. The expression of the MCPyV LT antigen was examined in these cell lines and also in MCC tissues collected from 25 cats and 1 dog. Furthermore, the CK profiles and other markers of Merkel cells and MCC were immunohistochemically examined.
Materials and Methods
Samples
Biopsy samples from 25 cats and 1 dog that were histopathologically diagnosed with MCC were examined (Table 1). The samples were from the archive of the Laboratory of Veterinary Pathology, the University of Tokyo, and Diagnostic Laboratory, Patho-Labo, between 2008 and 2020. Some of the cases were included our previous studies: case 5, Dohata et al, 10 and cases 3, 9, and 13 to 24, Sumi et al 44 Necropsy was performed on 1 cat (case 4) and 1 dog (case 26). Histological slides were examined by 3 veterinary pathologists approved by the Japanese College of Veterinary Pathologists (JKC, AS, and KU). To examine normal Merkel cells, normal skin tissues with sinus hairs were collected from 1 cat (face and carpus) and 1 dog (face) through routine necropsies performed at the institution. To establish MCC cell lines, cell cultures were performed using fresh tumor tissue samples from 1 cat (case 9) and 1 dog (case 26). A MCPyV gene-integrated human MCC cell line (MKL-1) was commercially obtained (Merck, Darmstadt, Germany).
Information on Feline and Canine Merkel Cell Carcinoma Cases.
Abbreviations: BCC, basal cell carcinoma; BISC, Bowenoid in situ carcinoma; F, female; FN, neutered female; JDC, Japanese domestic cat; M, male; MCC, Merkel cell carcinoma; MN, neutered male; ND, no data; SCC, squamous cell carcinoma.
Histopathology and IHC
Specimens were routinely fixed in 10% phosphate-buffered formalin solution, embedded in paraffin, sectioned at a thickness of 4 μm, and stained with hematoxylin and eosin (HE). The mitotic count was calculated as the sum of mitoses in 10 consecutive high-power fields (HPF; diameter of the field of view = 0.55 mm; HPF = 2.37 mm2; 40× magnification; Olympus CX31 microscope) starting from the most mitotically active areas. 31 Only fields with representative tumor cells were assessed. After counting mitoses in the first HPF, if no mitoses were found in the next 3 consecutive HPF, the process was repeated by moving to another mitotically active area.
IHC was performed on normal skin and MCC tissues using the primary antibodies listed in Table 2. Deparaffinized tissue sections were immersed in 10% hydrogen peroxide (H2O2) in methanol at room temperature for 5 minutes and then incubated in 8% skim milk in Tris-buffered saline (TBS) at 37 °C for 40 minutes to avoid nonspecific reactions. Each tissue section was then treated with a primary antibody at 4 °C overnight. An anti-mouse IgG polymer labeled with horseradish peroxidase (Envision, Agilent Technology) was applied at 37 °C for 40 minutes and sections were then rinsed with TBS. Reactions were visualized with 0.05% 3,3′-diaminobenzidine and 0.03% H2O2 in Tris–hydrochloric acid buffer, followed by a counterstain with Mayer’s hematoxylin (Muto Pure Chemicals). Semiquantitative scores included 4 categories: −, no positive cells; +, 1% to 30% positive cells; ++, 31% to 60% positive cells; +++, >60% positive cells.
Primary Antibodies Used for Immunohistochemistry and Immunofluorescence in the Present Study.
Abbreviations: IF, immunofluorescence; IHC, immunohistochemistry; LT, large T; MCPyV, Merkel cell polyomavirus; ND, not done; RU, ready to use.
Cell Cultivation
Fresh tumor tissues were minced and washed in phosphate-buffered saline (PBS; pH 7.4) with 1% penicillin-streptomycin (FUJIFILM Wako Pure Chemical Corporation). After discarding the supernatant, tissues were digested with 0.25% crude trypsin (BD Difco) in medium at 4 °C for 18 hours. In the cell culture, Roswell Park Memorial Institute (RPMI) 1640 medium (Invitrogen) was used for canine MCC cells and Dulbecco’s Modified Eagle’s Medium (DMEM; Thermo Fisher Scientific) for feline MCC cells. After centrifugation (1000 rpm), cell pellets were resuspended in the corresponding medium supplemented with 10% heat-inactivated fetal bovine serum (FBS; GE Healthcare Life Sciences) and 1% penicillin-streptomycin, and cells were filtered through a cell strainer (70 μm; Falcon) to obtain a single cell suspension. Suspended cells were seeded on 60-mm Petri dishes (NuncTM delta surface; Thermo Fisher Scientific) and incubated at 37 °C in an atmosphere of 5% CO2 and 95% air. After several subcultures, cells were cloned by a serial dilution method using a 96-well plate. A single layer of cloned cells was removed and seeded on a 60-mm Petri dish with the corresponding medium supplemented with 10% FBS. Cell passages were performed once every 3 to 4 days. The canine MCC cell line obtained was designated as FB-MCC01 and the feline MCC cell line as AS-MCC01. MKL-1 cells were cultured in RPMI 1640 medium according to the manufacturer’s instructions.
Growth Analyses
The growth curve was assessed by seeding 1.0 × 104 AS-MCC01 cells at the 21st passage and FB-MCC01 cells at the 66th passage in 24-well multiplates and maintaining cells in medium at 37 °C for 14 days. The number of cells in each well was counted 1, 2, 4, 6, 8, 10, 12, and 14 days after the initiation of the cell culture. Cell counting was done in quadruplicate. The cell population doubling time was calculated with the following equation: Doubling time (hours) = T log(2)/log(Ne/Nb), where T is the duration (hours) of the culture, Nb is the cell number at the beginning of the incubation, and Ne is the cell number at the end of the incubation.
Chromosome Analyses
Exponentially growing FB-MCC01 cells at the 84th passage and AS-MCC01 cells at the 22nd passage were treated with colcemid (KaryoMax; Invitrogen) at 37 °C for 1 hour in an atmosphere of 5% CO2 and 95% air. Cells were harvested by trypsinization, suspended in 5 ml of a hypotonic solution (0.075 M KCl) for 30 minutes, and fixed in 1 ml of Carnoy’s fixative solution (methanol: acetic acid = 3:1) for 5 minutes. The cell suspension was dropped onto cleaned glass slides and chromosomes were stained with Giemsa solution for 10 minutes. Two hundred metaphases were counted under a light microscope.
Immunofluorescence
FB-MCC01 cells and AS-MCC01 cells were seeded on 8-well slide chambers (Watson) with the corresponding medium and incubated for 3 to 4 days until cells had grown to form a single layer. MKL-1 cells were suspended in medium and centrifuged onto slides using CytoFuge 2 Cytocentrifuge (StatSpin Technologies). Cells were fixed with 4% paraformaldehyde at room temperature for 15 minutes. To detect intracellular antigens, cell membrane permeabilization was performed by incubating cells with 0.25% Triton X-100 for 10 minutes. After rinsing with PBS, cells were treated with 1% bovine serum albumin (BSA) in PBS with Tween 20 (PBST) to block nonspecific reactions. Primary antibodies for CK18, CK19, CK20, synaptophysin, CD56, and the MCPyV LT antigen were applied for immunofluorescence analyses (Table 2). Primary antibodies were diluted with BSA in PBST and applied to cells at 4 °C overnight. Cells were subsequently incubated with secondary antibodies at room temperature for 1 hour, counterstained with DAPI (Vectashield1; Vector Laboratory), and then observed under a Zeiss LSM 700 laser scanning confocal microscope (Carl Zeiss Meditec).
Polymerase Chain Reaction (PCR) Analysis
To detect MCPyV genes, specific PCR was performed on FB-MCC01, AS-MCC01, and MKL-1 cells. 12 DNA was extracted using the DNeasy Blood and Tissue kit (QIAGEN) according to the manufacturer’s instructions and used as a template for the PCR analysis. Information on the primers used in the present study is shown in Table 3. 12 Amplification was performed using TaKaRa Ex Taq Hot Start Version (Takara). The total reaction volume was 25 µl containing 1 µl of extracted DNA, 0.5 µl of 10 μM forward primer, and 0.5 µl of 10 μM reverse primer. The cycling protocol for MCPyV-specific PCR included a 3-minute incubation at 94 °C, followed by 31 cycles each of 94 °C for 45 seconds, 58 °C for 30 seconds, 72 °C for 45 seconds, and 72 °C for 15 minutes. A negative control was included by adding 1 µl of ultrapure water (FUJIFILM Wako Pure Chemical Corporation) instead of DNA extracts. All PCR products were mixed with a DNA loading buffer (Excellgen), and electrophoresed on a 2% agarose gel. The molecular imager ChemiDoc XRS+ System (Bio-Rad Laboratories) was used to visualize bands.
PCR Primers Used to Detect Merkel Cell Polyomavirus.

Abbreviation: LT, large T.
Xenotransplantation Into Mice
Each cell line was prepared in 200 µl of Hank’s balanced salt solution (Invitrogen), including AS-MCC01 (69th passage, 1.0 × 106 cells), FB-MCC01 (85th passage, 1.0 × 106 cells), and MKL-1 (2.0 × 107 cells). Each cell suspension was subcutaneously injected into the lower flank of 3 female C.B-17/Icr-scid/scidJcl (SCID) mice (total of 9 mice; CLEA Japan). Animals were euthanized under isoflurane 2 months after the inoculation, and tumor tissues were collected at necropsy, immediately fixed in 10% neutral-buffered formalin, and embedded in paraffin. Specimens were sectioned at a thickness of 4 µm and stained with HE. The mitotic count was calculated in the same manner as described above. IHC was performed using primary antibodies against CK18, CK19, CK20, synaptophysin, CD56, and MCPyV LT (Table 2). Experiments involving animals were approved by the Animal Care and Use Committee of the Graduate School of Agricultural and Life Sciences, the University of Tokyo (Approval #P17-017).
Results
IHC of Feline and Canine Merkel Cells
Feline and canine Merkel cells were detected in the isthmus of sinus hair follicles, attached to a thick hyaline membrane of the outer root sheath, and often had a vacuolar cytoplasm (Figs. 1, 2). Immunohistochemically, feline and canine Merkel cells in sinus hair follicles were positive for CK5/6, CK18, CK19, CK20, and synaptophysin (Fig. 3), and negative for CK7 and CK15 (Table 4). Feline Merkel cells were also positive for CD56, whereas canine Merkel cells were not. Immunoreactivity for CK20 was specifically restricted to Merkel cells in the hair follicles and epidermis. Besides Merkel cells, the epithelial cells of apocrine glands were immunopositive for CK18 and CK19, and the keratinocytes of the outer root sheath and epidermis were immunopositive for CK5/6. Peripheral nerve fibers surrounding sinus hair follicles were also positive for synaptophysin and CD56 (Fig. 3). Merkel cells that were immunopositive for these markers were also located in the basal layer of the epidermis as single cells or in clusters (ie, touch domes).
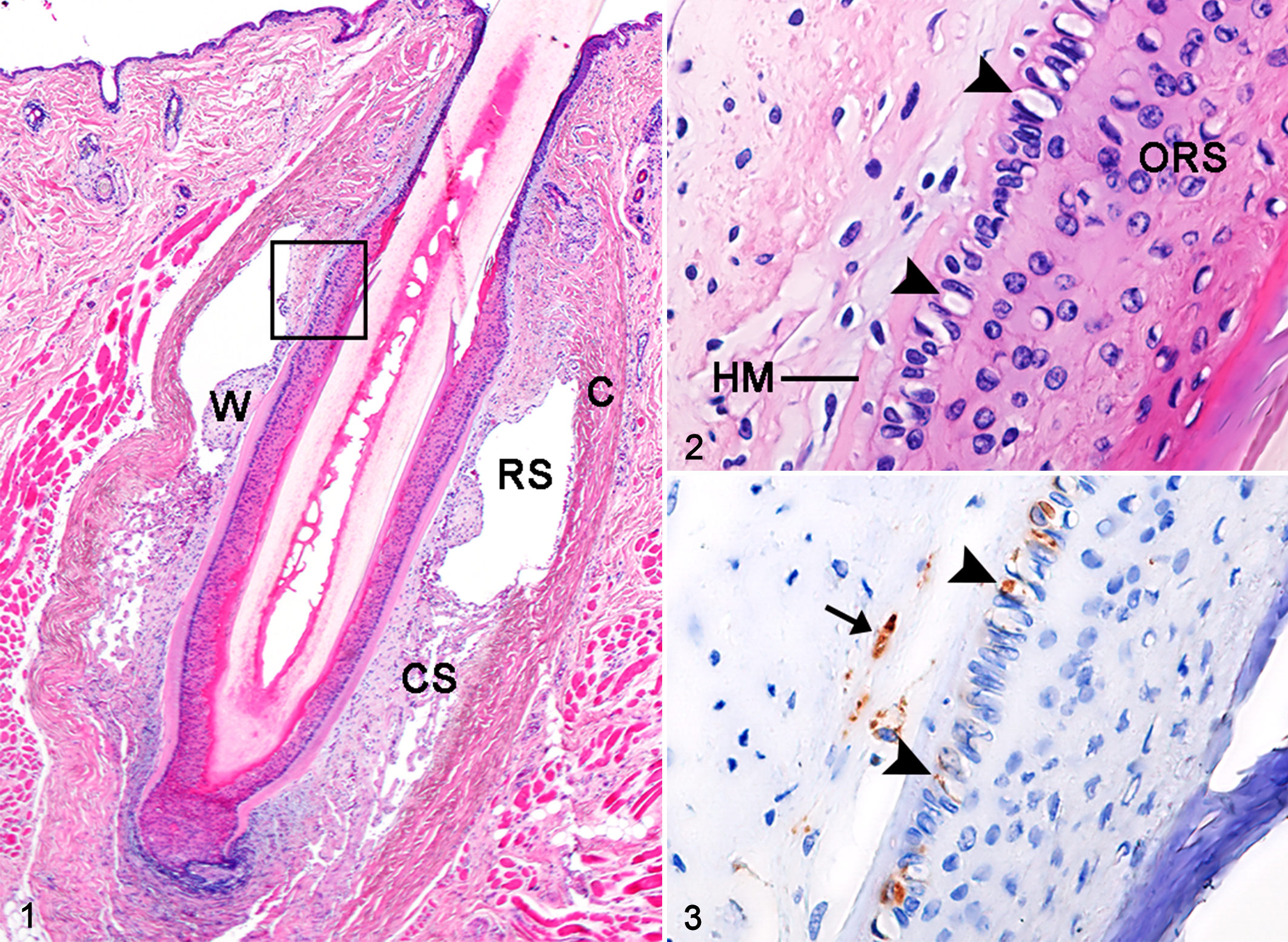
Normal facial skin, cat.
Immunohistochemical Results of Normal Feline and Canine Merkel Cells and Merkel Cell Carcinoma Cases.
Abbreviations: MCC, Merkel cell carcinoma; MCPyV LT, Merkel cell polyomavirus large T antigen.
a Case 26 is from a dog.
Histological Features of Feline and Canine MCC
In all MCC cases (25 cats and 1 dog), neoplastic lesions were located in the dermis and/or subcutis. In 7 feline cases, tumors invaded the skeletal muscle. All feline MCC cases exhibited infiltrative growth that was characterized by multifocal nests (19/25, Fig. 4) or diffuse infiltration (6/25, Fig. 5) of tumor cells. Tumor cells were arranged in trabeculae (3/25, Fig. 6), solid packets (8/25, Figs. 7, 8), or a mixture of trabeculae and solid packets (14/25) separated by a thin fibrous stroma. Tumor cells were polygonal in shape and had a moderate amount of amphophilic cytoplasm and a round nucleus with dispersed chromatin. Nuclei were often compressed and hyperchromatic in the periphery of neoplastic foci. In 10/25 feline cases (40%), tumor cells showed a vacuolar cytoplasm at the basal side (Fig. 8). The mean mitotic count was 35.3 per 10 HPF (median, 31.0; range, 10.5–74.9). Besides MCC, other skin lesions were found in 20/25 feline MCC cases (80%; Table 1). These lesions were located adjacent to the MCC lesion in 14 cases, distant from the MCC lesion in 3 cases, and in both locations in 3 cases. Acanthosis with mild dysplastic changes was observed in 12 cases, characterized by irregular thickening, cytoplasmic vacuolation, and large, dense keratohyalin granules. 33 Bowenoid in situ carcinoma was observed in 3 cases, characterized by focal proliferation of keratinocytes involving the epidermis and hair follicle infundibulum. Cells with a dark shrunken nucleus surrounded by a clear halo (ie, koilocytes) and cells exhibiting moderate to severe dysplastic changes, such as irregular cell arrangement, anisokaryosis, and loss of nuclear polarity were observed (Fig. 9). SCC (Fig. 10) and BCC were observed in 3 and 2 cases, respectively.
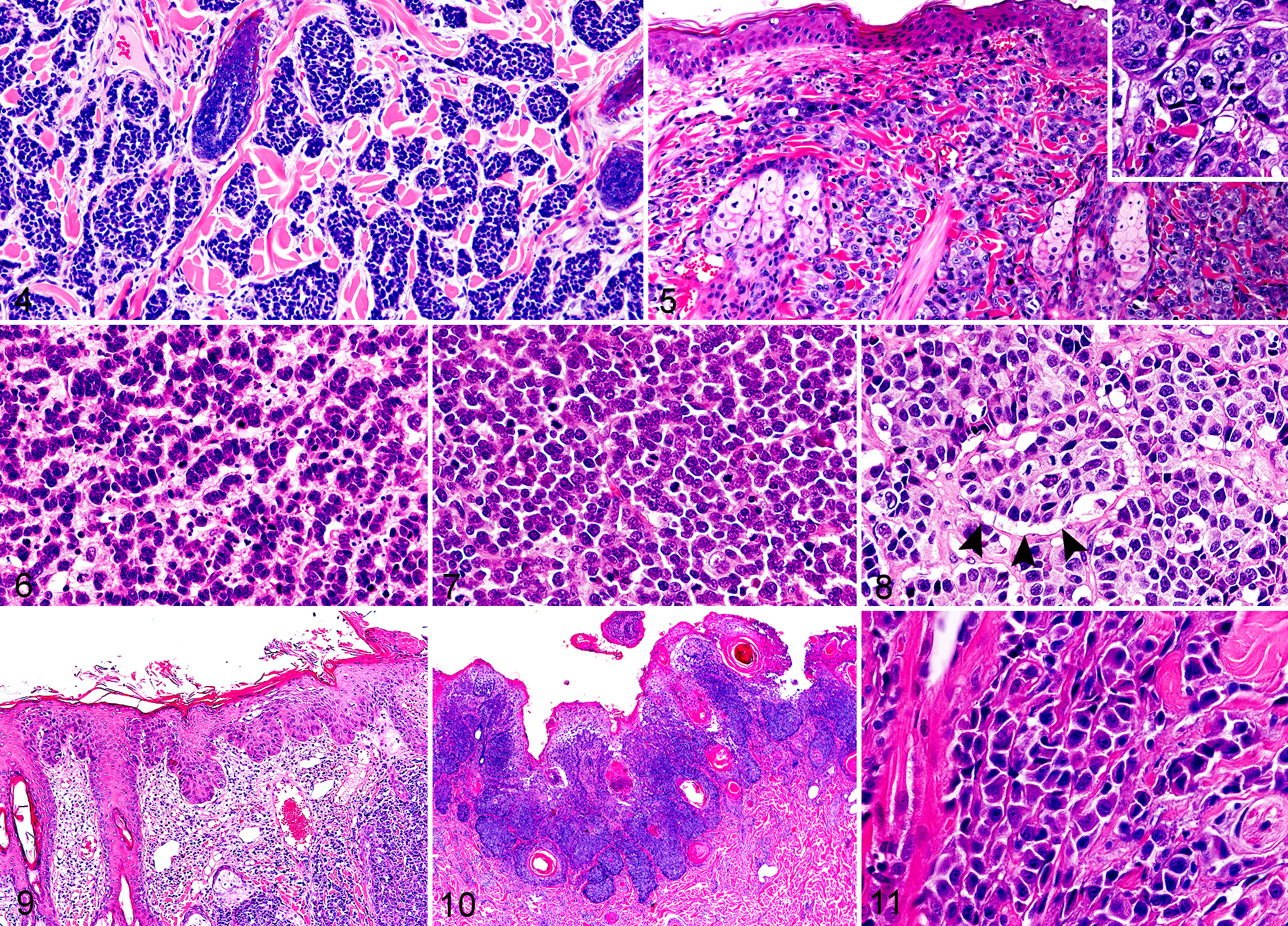
Merkel cell carcinoma, skin, cat. Hematoxylin and eosin.
The histological findings of canine MCC were similar to those of feline MCC with the mixed trabecular and solid pattern (Fig. 11). The mitotic count was 9.36 per 10 HPF. SCC was also observed in the skin adjacent to the MCC lesion (Table 1).
Immunohistochemical Features of Feline and Canine MCC
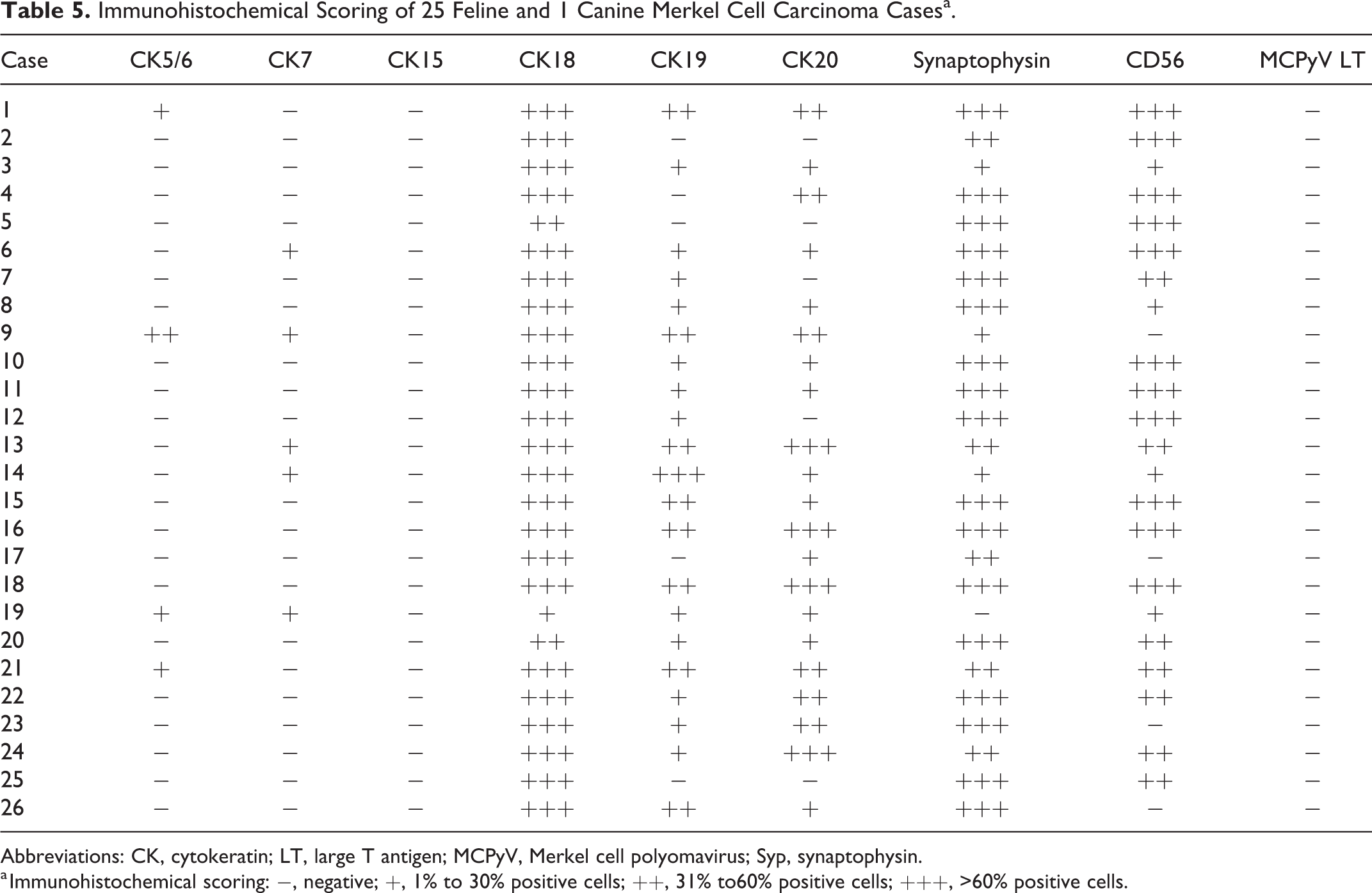
The results of IHC for 26 MCC cases are summarized in Table 4. Among the 25 feline MCC cases, tumor cells were immunopositive for CK5/6 (in 4 cases, 16%), CK7 (in 5 cases, 20%), CK18 (in all cases, 100%), CK19 (in 20 cases, 80%), and CK20 (in 20 cases, 80%). The tumor cells of feline MCC were also immunopositive for synaptophysin (in 24 cases, 96%) and CD56 (in 22 cases, 88%). The diffuse cytoplasmic immunolabeling with these markers was observed in tumor cells (Figs. 12–16). All feline MCC cases were negative for the MCPyV LT antigen.
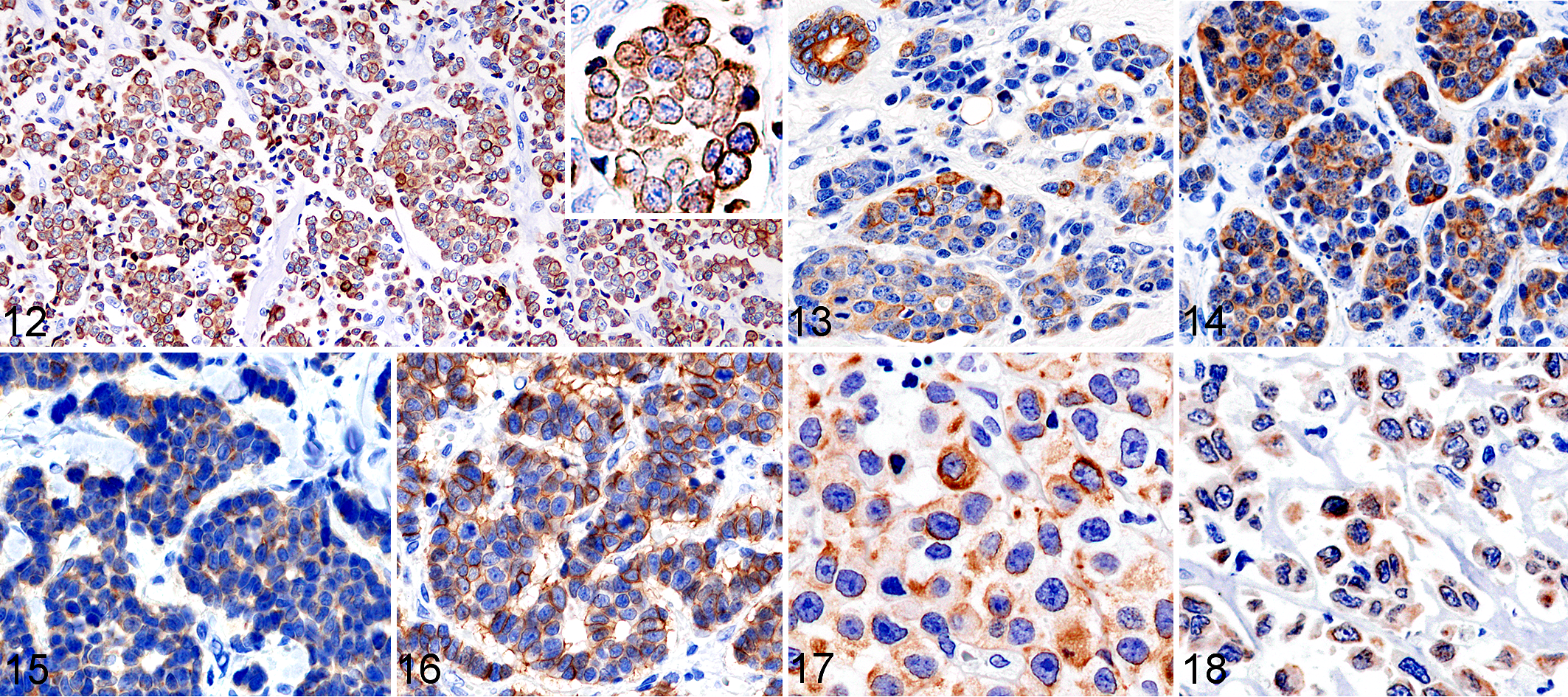
Merkel cell carcinoma, skin, cat. Immunohistochemistry. Tumor cells show cytoplasmic immunolabeling for cytokeratin (CK) 20 (Fig. 12, case 13; inset, higher magnification), for CK19 (Fig. 13, case 1), for CK18 (Fig. 14, case 1), for synaptophysin (Fig. 15, case 12) and for CD56 (Fig.16, case 12). Apocrine luminal cells also show immunolabeling (Fig 13, upper left).
In semiquantitative scoring, shown in Table 5, CK20 immunolabeling was low (score +) in 10/25 feline cases and moderate or high (++ or +++) in 20/25. CK19 immunolabeling was low (score +) in 12/25 feline cases and moderate or high (++ or +++) in 8/25. In contrast, CK18 immunolabeling was low or moderate (score + or ++) in only 2/25 feline cases and high (score +++) in 22/25. The CK positivity of cells in the peripheral area of tumor nests that invaded the surrounding tissues had a tendency to be higher than that of cells in the center area.
Immunohistochemical Scoring of 25 Feline and 1 Canine Merkel Cell Carcinoma Casesa.
Abbreviations: CK, cytokeratin; LT, large T antigen; MCPyV, Merkel cell polyomavirus; Syp, synaptophysin.
a Immunohistochemical scoring: −, negative; +, 1% to 30% positive cells; ++, 31% to60% positive cells; +++, >60% positive cells.
The canine MCC case was positive for CK18, CK19, CK20, and synaptophysin with a diffuse cytoplasmic pattern (Figs. 17, 18) and negative for CK5/6, CK7, CK15, CD56, and the MCPyV LT antigen (Table 4).
Comparison of Feline, Canine, and Human MCC Cell Lines In Vitro
Feline (AS-MCC01) and canine (FB-MCC01) MCC cell lines were established. The results of an in vitro analysis are shown in Table 6. The population doubling times of AS-MCC01 and FB-MCC01 were 42.8 and 36.3 hours, respectively (Supplemental Fig. S1). The modal chromosome numbers of AS-MCC01 and FB-MCC01 were 38 to 70.5 (mean 50.5) and 80 to 112 (mean 88.3), respectively. Cultured AS-MCC01 and FB-MCC01 cells were an adherent monolayer of polygonal- or spindle-shaped cells with thin processes (Figs. 19, 20). The nucleus was round with single or multiple prominent nucleoli and showed anisokaryosis. Multinucleated cells were often observed. The human MCC cell line (MKL-1) formed clusters of small round cells suspended in the culture medium (Fig. 21). The nucleus was round with a single or multiple prominent nucleoli. Immunofluorescence for CK18 and CK20 revealed diffuse cytoplasmic immunoreactivity in AS-MCC01 and FB-MCC01 cells (Figs. 22, 23), and dot-like cytoplasmic immunoreactivity in MKL-1 cells (Fig. 24). AS-MCC01, FB-MCC01, and MKL-1 cells were positive for synaptophysin in a diffuse cytoplasmic pattern (Figs. 25–27). In contrast to the IHC results of original tumor tissue (cases 9 and 26), AS-MCC01 cells were negative for CK19 and positive for CD56, FB-MCC01 cells were positive for CK19 and negative for CD56, and MKL-1 cells were positive for CK19 and CD56. By immunofluorescence, AS-MCC01 and FB-MCC01 cells were both negative for the MCPyV LT antigen, whereas MKL-1 cells were positive. Specific PCR products of the MCPyV LT1, LT3, and VP1 genes were detected in MKL-1 cells, but not in AS-MCC01 or FB-MCC01 cells (Supplemental Fig. S2).
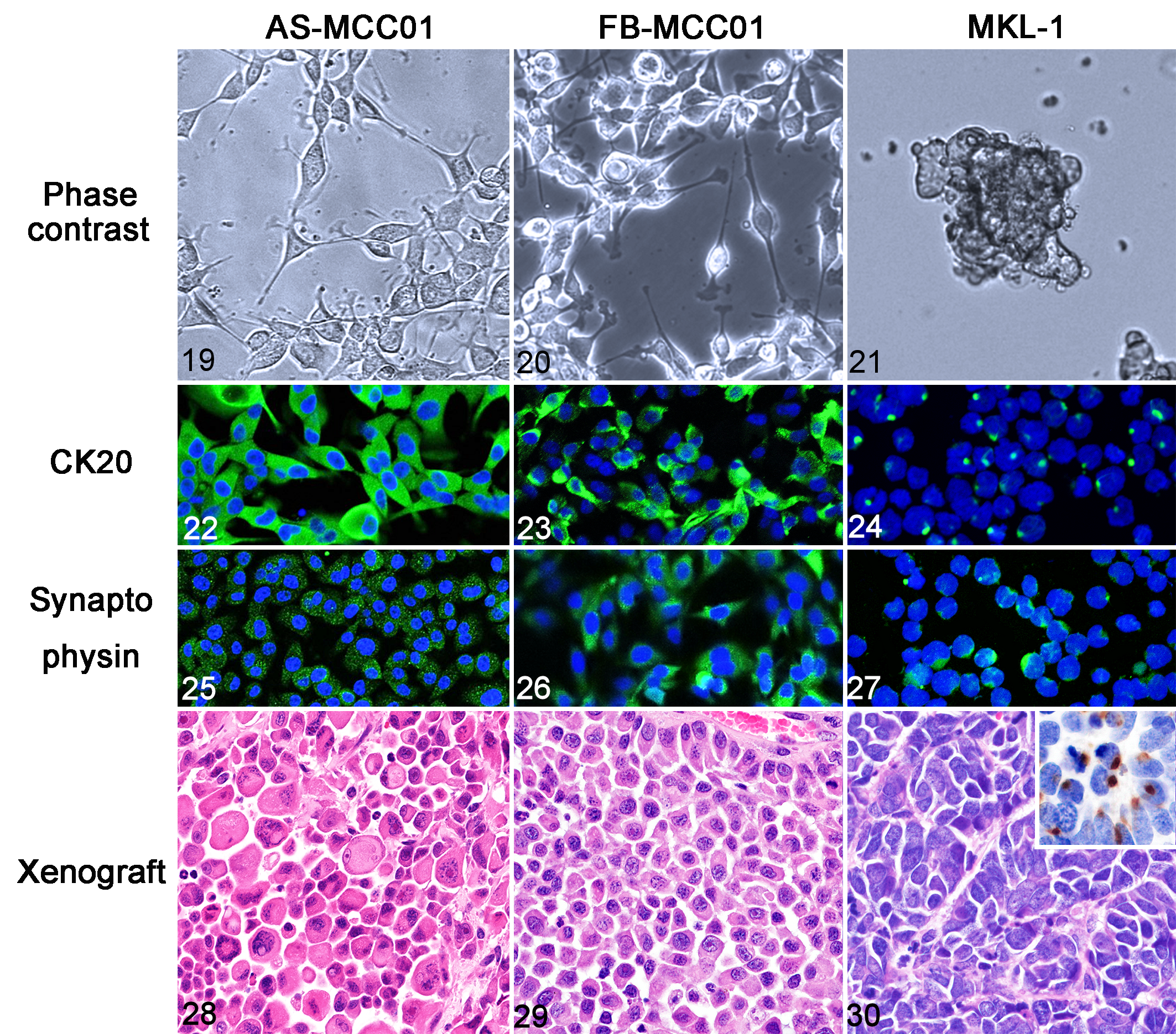
Comparison of characteristics of Merkel cell carcinoma cell lines of the cat (AS-MCC01), dog (FB-MCC01), and human (MKL-1).
Characteristics of AS-MCC01, FB-MCC01, and MKL-1 Cell Lines.
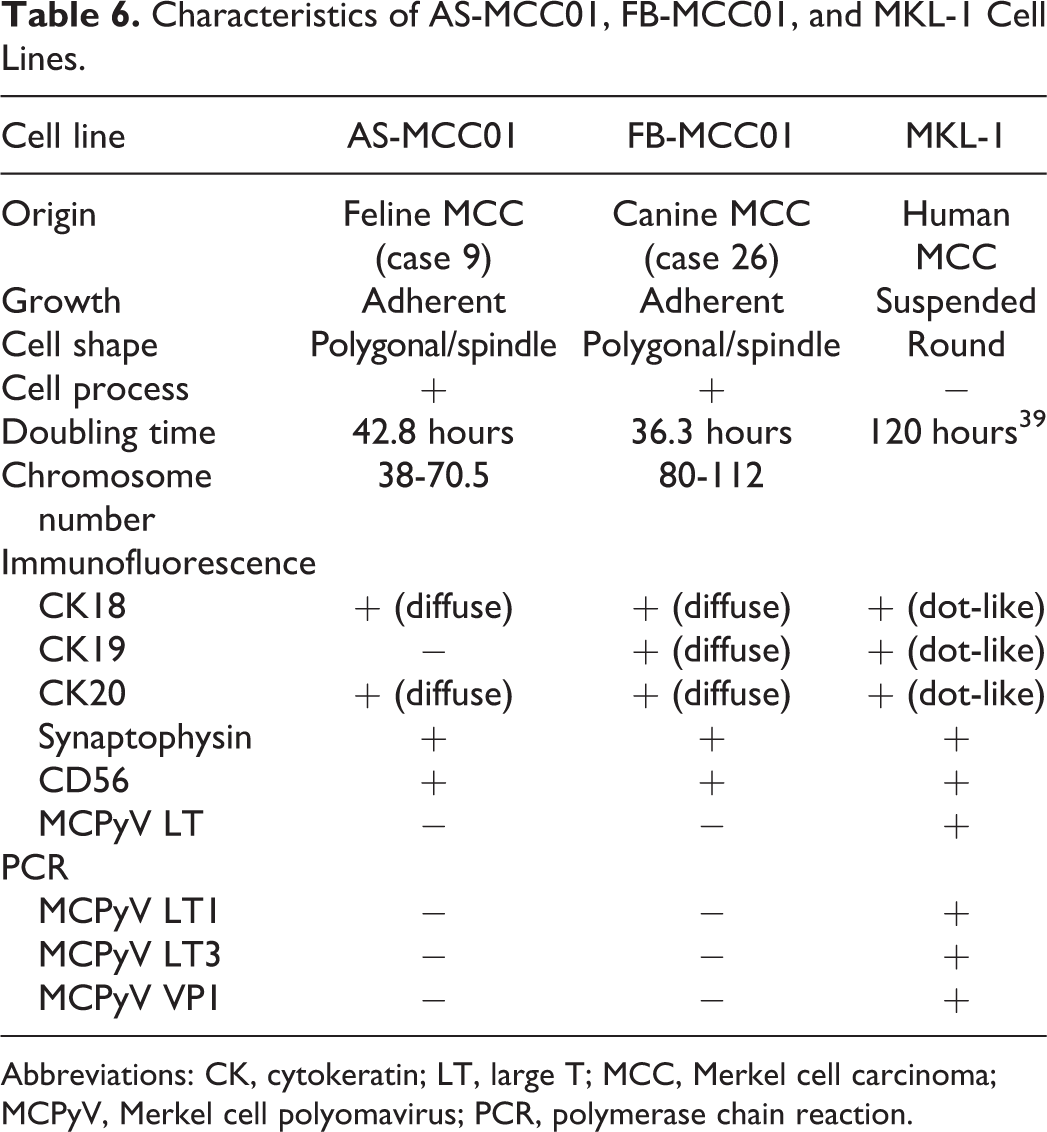
Abbreviations: CK, cytokeratin; LT, large T; MCC, Merkel cell carcinoma; MCPyV, Merkel cell polyomavirus; PCR, polymerase chain reaction.
Comparison of Xenograft Tissues of Feline, Canine, and Human MCC Cell Lines
After the subcutaneous injection of each cell line, a tumor mass developed at the injection site (flank) of all SCID mice. The average times from the injection to the detection of a tumor were 26 days in AS-MCC01, 14 days in FB-MCC01, and 38 days in MKL-1. On gross examination, all tumors were dome-shaped, and the cut surface showed a pink-gray solid mass with necrosis and hemorrhage at the center. Metastatic lesions were not found in any mice.
Xenograft tissues of AS-MCC01 were composed of solid sheets of polygonal cells with eosinophilic cytoplasm. In some regions, tumor cells exhibited marked anisocytosis and cellular pleomorphism (Fig. 28). The mean mitotic count was 60.8 per 10 HPF. Xenograft tissues of FB-MCC01 were composed of solid sheets of polygonal cells with eosinophilic cytoplasm (Fig. 29). The mean mitotic count was 63.1 per 10 HPF. Xenograft tissues of MKL-1 were composed of polygonal cells with scant basophilic cytoplasm arranged in solid packets separated by a fibrovascular stroma (Fig. 30). The mean mitotic count was 55.0 per 10 HPF. Immunohistochemically, AS-MCC01 and FB-MCC01 cells of the xenograft tissues were positive for CK18 and CK19 and negative for CK20, synaptophysin, CD56, and MCPyV LT antigen, whereas MKL-1 cells of the xenograft tissues were positive for CK18, CK19, CK20, synaptophysin, CD56, and MCPyV LT antigen.
Discussion
Feline and canine MCC were histologically characterized by trabeculae or solid packets of tumor cells infiltrating the dermis, similar to human MCC. Other proliferative cutaneous lesions, including acanthosis, carcinoma in situ, SCC, and BCC, were detected in 80% of feline MCC cases and mostly developed adjacent to MCC lesions (Table 1). In humans, actinic keratoses, Bowen’s disease, SCC, and BCC are incidentally found in 5% to 10% of MCC cases. 8,34,49 Previous studies reported that these combined MCC lesions were associated with chronic ultraviolet exposure. 8,19,34 Sun exposure and infection by Felis catus papillomaviruses (FcaPVs) are well-known factors associated with acanthosis, in situ carcinoma, and SCC in cats. 11,33,50 The high incidence of proliferative cutaneous lesions in cats with MCC may suggest a common etiology between MCC and these lesions. Nevertheless, these lesions are common in cats compared to MCC, and thus may be coincidental.
In the present study, normal Merkel cells of the cat and dog were detected in the basal layer of epidermis and sinus hair follicle. The present study revealed the CK profiles of normal feline and canine Merkel cells in the epidermis and sinus hair follicle. In addition to CK20, a well-known marker of Merkel cells, 37,38 feline and canine Merkel cells were positive for CK5/6, CK18, and CK19 (Table 4). Besides Merkel cells, immunopositivity for CK5/6 was confirmed in the basal cells and keratinocytes of the epidermis and adnexa; CK18 in apocrine gland luminal cells; CK19 in apocrine gland luminal and ductal cells and the outer root sheath basal layer of the hair follicle. 21,22 Immunopositivity for CK19, a stem cell marker, in Merkel cells suggests its common origin with basal cells of the outer root sheath, which possibly are multipotent stem cells in the hair follicle bulge. In epithelial cells of the skin, immunopositivity for CK20, synaptophysin, and CD56 was specifically restricted to Merkel cells, suggesting the potential of these markers in the diagnosis of Merkel cell tumors.
All feline MCC cases were immunopositive for at least two of the Merkel cell-specific markers, CK20, synaptophysin, and CD56. CK20 immunolabeling in tumor cells was absent in 20% and low (score +) in 50% of feline MCC cases. In human MCC, although CK20 is the most reliable marker, approximately 5% of cases lacked its expression. 16 In these cases, MCC was diagnosed according to histological features and immunopositivity for CKs coupled with positivity for at least one neuroendocrine marker. 32 Previous studies reported that synaptophysin, a synaptic vesicle protein, and CD56, a neural cell adhesion protein also known as NCAM, were consistently positive in human MCC. 7,18,23,25 Another study showed that more than 75% of MCC cells were positive for CD56 in all cases examined, including cases with low CK20 positivity (less than 25% of tumor cells were positive). 25 In feline MCC examined in the present study, 96% of cases were positive for synaptophysin, 88% were positive for CD56, and all cases were positive for at least one of these neuroendocrine markers (Tables 4, 5). These results suggest that, in conjunction with a histological examination, immunopositivity for synaptophysin and/or CD56 in combination with positivity for low-molecular-weight acidic CKs (ie, CK18, CK19, and CK20), supports the diagnosis of MCC in cats and dogs as well as in humans. However, the pattern of CK immunopositivity in feline and canine MCC cells differed from that in human MCC cells. Human MCC cells are characterized by a perinuclear dot-like positivity for CKs, 9,35 whereas feline and canine MCC cells showed diffuse cytoplasmic positivity, which was similar to the pattern of normal Merkel cells.
The cutaneous stem cell markers, CK19, CK15, and p63, are expressed in cutaneous epithelial cells that have self-renewal and multipotency. 2 In previous studies on human MCC, 34% to 100% were positive for CK19, 1,3,34 none were positive for CK15, 1 and 33% to 69% were positive for p63. 43,48 In the present study, 80% of feline cases were positive for CK19, and all were negative for CK15, similar to the findings of human MCC studies. Our previous study showed that feline MCC cells are consistently positive for p63. 44 In the present study, all feline and canine MCC were located in the dermis and/or subcutis without showing continuity with the overlying epidermis. Histological location of the tumor and positive immunoreactivity for stem cell markers, such as CK19 and p63, suggests follicular stem cells residing in the bulge region as a possible cell of origin for MCC.
The novel cell lines, AS-MCC01 and FB-MCC01, were established from feline (case 9) and canine (case 26) MCC tissues, respectively. Immunofluorescence revealed that both cell lines were positive for CK20, synaptophysin, and CD56, showing an MCC immunophenotype in vitro (Table 6). However, in contrast to the original tumor tissues, AS-MCC01 cells were negative for CK19 and positive for CD56, and FB-MCC01 cells were positive for CD56. In xenograft tissues, AS-MCC01 and FB-MCC01 cells lost immunoreactivity for CK20, synaptophysin, and CD56. These phenotypic alterations may be associated with the dedifferentiation of tumor cells during subculturing and selective cloning or differences in the microenvironments of cells between host tissues, culture medium, and recipient mouse tissues. 26,40 These results implicate the plasticity of MCC cells that exhibit various immunophenotypes among different cases (Table 5).
Cellular growth, morphology, and immunophenotypes were compared among the feline (AS-MCC01), canine (FB-MCC01), and human (MKL-1) MCC cell lines. Although MKL-1 cells were round and grew in suspension clusters, AS-MCC01 and FB-MC001 cells were polygonal or spindle-shaped with cytoplasmic projections and grew in adherent monolayers. The cell lines of human MCC have been grouped into 4 types based on morphology. 27 Types I to III grow in suspension clusters, while type IV grows in adherent monolayers with cytoplasmic projections. Cell clusters of type I are tightly packed, type II are relatively dense, and type III, in which MKL-1 is classified, are very loose. Types III and IV show more aggressive growth behavior and these cells more closely resemble undifferentiated carcinoma. According to this classification, AS-MCC01 and FB-MCC01 were classified as type IV. The population doubling times of AS-MCC01 (42.8 hours) and FB-MCC01 (36.3 hours) were shorter than that of MKL-1 (120 hours), 39 which may be associated with the aggressive nature of feline and canine MCC. The in vitro immunophenotypes of the feline (AS-MCC01), canine (FB-MCC01), and human (MKL-1) MCC cell lines were similar, except for CK19 negativity in AS-MCC01 (Table 6). However, similar to tissues from MCC cases, feline and canine MCC cell lines showed diffuse cytoplasmic positivity for CKs, whereas the human MCC cell line showed perinuclear dot-like positivity. The patterns of immunoreactivity are associated with the microanatomical distribution of the antigen. 42 Electron microscopic examination of human MCC previously revealed perinuclear whorls of intermediate filaments in tumor cells. 15 The aberrant or decreased expression of E-cadherin, a cell adhesion molecule, has been reported in rhabdoid tumors showing the aggregation of CKs. 41 Therefore, the suspension growth of the human MCC cell line may be associated with the abnormal cytoplasmic aggregation of CKs.
All of the feline and canine MCC cases examined in the present study were negative for the MCPyV LT antigen by IHC. The established cell lines of feline (AS-MCC01) and canine (FB-MCC01) MCC were also negative for the MCPyV antigen and genes, while the human MCC cell line (MKL-1) was positive for the MCPyV LT antigen and genes (LT and VP1). The LT antigen of MCPyV has been detected in the tumor cells of approximately 80% of human MCC cases. 17 Liu et al demonstrated that MCPyV infected dermal fibroblasts isolated from mice, rabbits, rats, chimpanzees, and rhesus macaques. 28 Although it currently remains unclear whether MCPyV infects feline and canine cells, different mechanisms from MCPyV-positive human MCC may contribute to the tumorigenesis of feline and canine MCC. A high mutation burden associated with a UV-induced DNA damage signature similar to SCC and sun-damaged skin has been reported in human cases of MCPyV-negative MCC. 17
In the present study, novel cell lines of feline (AS-MCC01) and canine (FB-MCC01) MCC were established. In vivo and in vitro studies on immunophenotypic profiles suggested that stem cells in the hair follicle bulge may be the cell of origin of Merkel cells and MCC in the cat and dog. Negative results for the MCPyV antigen and gene detection in MCC tissues and cell lines indicate that the cause of feline and canine MCC differ from that of MCPyV-positive human MCC.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Ito_et_al - Comparative In Vitro and In Vivo Studies on Feline, Canine, and Human Merkel Cell Carcinoma
Supplemental Material, Combined_supplemental_materials-Ito_et_al for Comparative In Vitro and In Vivo Studies on Feline, Canine, and Human Merkel Cell Carcinoma by Soma Ito, James K. Chambers, Chikako Mori, Ayumi Sumi, Tetsuo Omachi, Hiroyuki Nakayama and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by our institutional review board.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
