Abstract
Canine cutaneous mast cell tumors (ccMCTs) have a highly variable biological behavior and accurate prognostication is essential for therapeutic intervention. Internal tandem duplications (ITD) of exon 11 are the most commonly detected c-kit mutation in ccMCTs and are associated with poor prognosis and increased cellular proliferation. The prognostic value of detecting mutations in other exons of c-kit has not been systematically examined. In this study, we analyzed the prognostic value of ITD mutations of exon 8 in c-kit of ccMCTs in comparison to ccMCTs with ITD mutations of exon 11 and ccMCTs without mutations of exon 8 or 11. The mutational status, histological grade, KIT expression pattern, Ki67 index, AgNOR (argyrophilic nucleolar organizing region) score, and Ag67 score were determined in 221 ccMCTs, and outcome was available for 101 dogs. ITD mutations of exon 8 were found in 73/221 (33%), of exon 11 in 100/221 (45%), and none of these mutations in 50/221 (22%) of ccMCTs. None of the dogs with mutations of exon 8 died due to suspected ccMCT-related cause, but 23% dogs with ccMCTs with mutations of exon 11 died due to suspected ccMCT-related cause. Prognostic parameters in ccMCTs with exon 11 mutations were commonly associated with a high proliferative activity and poor prognosis, while prognostic markers in ccMCTs with mutations of exon 8 had lower values similar to those observed in ccMCTs without mutations in exons 8 or 11 of c-kit. This study indicates that screening for ITD mutations in exon 8 in ccMCTs may be helpful to identify less aggressive ccMCTs and may be recommended as a supplementary prognostic test.
Keywords
Canine cutaneous mast cell tumors (ccMCTs) are one of the most common potentially malignant skin tumors in dogs. 24 They demonstrate highly variable behavior ranging from localized masses that are usually cured by surgical resection to highly aggressive tumors with local recurrence and multi-organ metastasis. Accurate prognostication of ccMCTs is essential for accurate therapeutic intervention and numerous studies have established multiple methods that are routinely used to predict biological behavior. Histological criteria of malignancy, such as the mitotic count, are an essential part of ccMCT diagnosis. 24 Although the mitotic count has been proposed as an independent prognostic measure, 21 it is known to have high interobserver variability. 1 Kiupel et al. 11 combined the mitotic count with 3 further criteria into a 2-tiered grading system that has been shown to predict favorable versus unfavorable outcomes more accurately and reproducibly than with either the mitotic count alone or the previously described 3-tiered grading systems. 10,11,22,27,34 However, a subset of ccMCTs do not behave as predicted and complementary prognostic markers are recommended. 10,22,26,34 Additional prognostic tests to supplement histological grading include measures of cellular proliferation such as the Ki67 index and the average number of argyrophilic nucleolar organizing region (AgNOR) within neoplastic mast cells. 24,38 Higher values of cellular proliferation are associated with an unfavorable prognosis. 10,24
Another parameter that has been shown to be of prognostic value in ccMCTs is the status of the KIT receptor protein (also known as CD117 or stem cell growth factor receptor), 10 which is a transmembrane type III tyrosine kinase receptor expressed in various cell types, including mast cells. 14,17,25 The receptor consists of various domains, which are encoded by 21 exons of the proto-oncogene c-kit. The extracellular part (encoded by exons 1–9) consists of 5 immunoglobulin-like domains. 14 The second and third immunoglobulin-like domains (encoded by exons 3–5) form the pocket to which the ligand, stem cell factor, can bind. 14 After binding to the stem cell factor, immunoglobulin-like domains 4 and 5 (encoded by exon 6–9) build an interaction between 2 monomeric receptors (dimerization). 14,25 The transmembrane domain is encoded by exon 10. 14 The juxtamembrane domain (encoded by exon 11) forms an inhibitory contact with the kinase domain in the inactive state of the (monomeric) receptor. 14,25 This inhibitory contact is disrupted by dimerization of 2 KIT receptors. Activation of the intracellular tyrosine kinase domain (encoded by exons 12–21) lead to signal transduction via various pathways and thereby to mast cell proliferation, differentiation, and survival. 14
Abnormal expression patterns of the KIT protein and mutations in the c-kit proto-oncogene have been linked to poor patient outcome in ccMCTs. 29 As a transmembranous protein, KIT has a perimembranous location in nonneoplastic mast cells and less aggressive ccMCTs. Aberrant localization in the cytoplasm is associated with an unfavorable prognosis. 12,38 Mutations in exon 11 of the proto-oncogene c-kit may lead to hyperactivity of the kinase domain of KIT (gain-of-function mutations) and have been shown to be associated with more aggressive biological behavior of ccMCTs. 40,41 In ccMCTs, internal tandem duplications (ITDs) are much more common than other types of mutations such as insertions, deletions, and single nucleotide substitutions. 3,15,37,40,42 The prevalence of mutations has been determined to range between 8.5% and 72.3% in different studies that greatly varied in the examined number of exons and type of mutations. 3,5,15,27,35 –37,41 Most previous studies identified ITDs in exon 11 to be the most common mutation in ccMCTs, 3,5,15,35 –37 while mutations in the kinase domain are rare. 15,37,39 One study identified mutations of exon 5 to be most common. 27 ITDs of exons 8 and 9 have been reported with prevalence of 2% to 7%. 3,5,15,18,27,35,37,41 Additionally, mutations of other exons including 2, 6, 7, 9, 10, 12, 14, 15, and 17 have been reported with low frequency. 2,5,15,20,27,36 Some studies have correlated mutations indirectly with histological grade, 3,18,27,28,31,35,36,40,42 other prognostic markers, 3,38,40 or directly with clinical outcome. 2,3,5,18,27,30,35 –37,40,41 The biological behavior of ccMCTs with mutations in comparison to ccMCTs wild type c-kit has been investigated for ccMCTs with mutations in exon 11 alone 27,31,35,37,38,40,42 or for groups of ccMCTs with mutations in different exons. 2,3,5,6,18,27,30,35,36,41 ITD mutations in exon 11 have been found consistently to be associated with more aggressive biological behavior. 34,35 Letard et al. 15 analyzed the function of 2 exon 11, 1 exon 8, and 1 exon 9 mutations in vitro and determined that all 4 mutations resulted in activation of the receptor (gain-of-function). Based on this result, some authors have justified their decision to combine c-kit mutations in different exons in their analysis. 5,27,41 The biological behavior of ccMCTs with exon 8 mutations has only been reported for individual cases. Vozdova et al 36 described an exon 8 mutation associated with a single high-grade ccMCT in a dog with a short survival time of 6 months and tumor recurrence. In 2 other studies involving 3 ccMCTs, the same investigators reported no association of exon 8 c-kit mutations with poor outcome. 35,37
The goal of the present study was to analyze ITD mutations in exon 8 and exon 11 of c-kit in ccMCTs with respect to their occurrence and their prognostic value. We hypothesize that mutations in exon 11 of c-kit are associated with more aggressive behavior of ccMCTs, as described previously, and that ITDs of exon 8 may result in a different patient outcome as they encode a different protein domain with a different function.
Materials and Methods
Case Selection
A total of 221 cases of ccMCTs were included in this study. All cases were biopsies submitted to the Veterinary Diagnostic Laboratory at Michigan State University between June 2012 and March 2014 for routine histological examination. Cases were included in the study if they had a confirmed diagnosis of ccMCT, screening for ITDs in exons 8 and 11 of c-kit, as well as a prognostic panel (2-tier histological grading, Ki67 index, AgNOR score, combined AgNOR × Ki67 score, KIT expression pattern). Dogs with subcutaneous or oral/mucocutaneous MCTs were excluded from the analysis.
Follow-up Survey
A survey instrument was faxed to the referring clinicians. Veterinarians were asked to respond regarding the disease-free survival time, total survival time, cause of death if applicable, treatments used (chemotherapy, tyrosine kinase therapy, and radiation therapy), local tumor recurrence (MCT recurring at the site of previous surgery) and distant tumor recurrence (MCT developing at a cutaneous site other than the site of previous surgery), local and distant lymph node involvement, and metastasis in internal organs. Of the 221 cases, follow-up information for at least 24 months was received from 101 cases (46%). Indicated cause of death was grouped into (1) suspected to be related to ccMCT and (2) suspected to be unrelated based on the opinion of the referring veterinarian in the survey. Due to the retrospective nature of outcome assessment in the present study, confirmation of the cause of death through postmortem examination was not possible. Also, the approaches to staging varied among clinics, and the diagnostic methods used to determine tumor recurrence and metastasis were not provided for systematic analysis. None of the included dogs were euthanized as a result of a poor prognosis based on the information available in the pathology report, and all dogs had a minimal survival time of 1 month (for dogs with a suspected ccMCT-unrelated cause of death) or 2 months (for dogs with a suspected ccMCT-related cause of death).
Screening for Mutations in Exons 8 and 11 of c-kit
Polymerase chain reaction (PCR) amplification of exons 8 and 11 of the c-kit proto-oncogene was performed using previously described primer pairs. In regard to exon 8, the primers were located in intron 7 to 8 and amplified the 12-bp duplication mutation previously described in canine MCTs. 4 For exon 11 amplification, the primers flanked exon 11 and the 5′ end of intron 11, 9 which includes the previously described ITD region in canine MCTs. 40 In both primer sets, amplifications were performed using Type-it Mutation Detection PCR Kit (Qiagen). Amplification reactions were prepared in a 25-µL total reaction volume, with 10 to 25 ng of LCM-extracted DNA and 5 pmol of each primer. Cycling conditions were as follows: 95 °C for 5 minutes; 35 cycles at 95 °C for 30 seconds, 58 °C for 90 seconds, 72 °C for 30 seconds; followed by 68 °C for 10 minutes. PCR products were visualized on the QIAxcel Capillary Electrophoresis System (Qiagen).
Histological Grading
All ccMCTs were graded using the 2-tier grading system according to Kiupel et al. 11 Tumors were classified as high grade (less favorable prognosis) if any of the following criteria were reached: a mitotic count in 10 high-power fields (hpf, equivalent to 2.37 mm2) of at least 7, at least 3 multinucleated tumor cells in 10 hpf, at least 3 bizarre nuclei in 10 hpf, or karyomegaly. If quantification of these criteria were below the cutoff values, the ccMCT was classified as low grade (more favorable prognosis). Histological grading was available for all 221 dogs.
KIT Expression Patterns
For 211 dogs, the localization of the KIT protein was determined by immunohistochemical labeling as previously published. 12,38 KIT protein localization was classified into 3 patterns as follows: 12,38 (1) KIT pattern I: membrane-associated labeling; (2) KIT pattern II: focal to stippled cytoplasmic labeling with decreased perimembranous labeling; and (3) KIT pattern III: diffuse cytoplasmic labeling.
Ki67 Index
Immunohistochemistry for the Ki67 protein was performed as previously published. 38 Consistent with previous studies, we quantified Ki67-positive cells in a 10 mm2 grids within the tumor area with the highest density. A Ki67 index above 23 cells per grit area has been associated with significantly increased risk of local ccMCT recurrence, distant ccMCT occurrence, and ccMCT-related mortality. 38 In the present study, the Ki67 index was determined for 211 dogs.
Combined AgNOR × Ki67 (Ag67) Score
Tumor proliferation is related to the number of tumor cells undergoing cell division (growth fraction) and the rate of the cell cycle. 10 Therefore, it is best represented by multiplying the AgNOR score by the Ki67 index, resulting in the Ag67 score. 10 Histochemical staining of AgNORs was performed as previously published. 38 Consistent with previous studies, the average number of AgNORs were determined in 100 randomly selected neoplastic mast cells. An Ag67 score above 54 has been associated with a significantly increased risk of local ccMCT recurrence and ccMCT-related mortality. 38 In the present study, Ag67 was determined in 211 cases.
Statistical Analysis
Survival data are shown as Kaplan-Meier survival curves. Survival analysis was performed using a Cox proportional hazard model. For analysis, the overall death was used as ccMCT-related causes could not be confirmed through pathological examinations in this retrospective study. The proportional hazard assumption was checked using Schoenfeld residuals. Hazard ratios are reported with 95% confidence intervals (CIs). Hazard ratios for high values of prognostic parameters are given; hazard ratios for low values are the reciprocal of the stated values. P values were determined using the Cox model or the logrank test in the case where none of the dogs was suspected to have died due to a ccMCT-related cause. We tested for differences in treatment using a Fisher’s exact test and quantified the effect using Cramer’s V. An ANOVA with post hoc comparison against control group was used to determine the differences in tumor markers depending on exons 8 and 11 c-kit mutations against the control group without mutations of exons 8 and 11. Ki67, Ag67, and AgNOR values were logarithmized to achieve normal distribution and the coefficient of variation (CV; standard deviation divided by mean) is indicated. All P values <.05 were considered significant. Analyses was performed using R version 3.63 using the survival package (version 3.1-11). Figures were created using the package survminer (version 0.4.6). Based on the causal relationship of mutational status of c-kit and outcome with histological grade and proliferation markers (Suppl. Fig. S1), mutational status is likely not an independent prognostic marker. 29
Raw data of the included cases can be made available upon reasonable request to the corresponding author.
Results
Study Population
Dogs had a median age of 7 years. Ten dogs were ≤2 years, 56 dogs had an age of >2 and ≤5 years, 98 dogs were >5 and <10 years of age, and 54 dogs were ≥10 years of age. The age for 3 dogs was unknown. Twelve dogs were intact female, 97 were neutered females, 16 were intact males, and 94 were neutered males. The sex was not reported for 2 cases. Sex and age groups were distributed similarly between cases with and without exon 8 or 11 mutations and were not associated with ccMCT-related death. Breeds of the included animals are given in Supplemental Table S1.
Of the 101 dogs with clinical follow-up data, 80 dogs had survived 24 months after surgery. The death of 14 cases was suspected to be related to ccMCT, and 7 died of suspected other causes.
c-kit Mutation Status
ITD mutations in exon 11 of c-kit were detected in 100/221 dogs (45.2%) and in exon 8 in 73/221 dogs (33.0%). Of these, mutations in both exons were identified in 2 dogs. For 50/221 dogs (22.6%), ITD mutations are not detected in either exons 8 or 11.
Dogs With Exon 8 Mutations Have Better Clinical Outcome
Clinical outcome was reported for 42/73 dogs with ccMCTs with exon 8 mutations and 59/148 dogs with ccMCTs without exon 8 mutations. Of 59 dogs with ccMCTs without exon 8 mutations, 42 (71%) survived longer than 24 months. Of the 17 that survived less than 24 months, 14 dogs were suspected to have died from the ccMCT and 3 were suspected to have died of other causes. In contrast, of the 42 dogs with ccMCTs with exon 8 mutations, 38 (90%) survived longer than 24 months (Table 1). The 4 dogs that survived less than 24 months died at 3, 5, 7, and 12 months after surgery; however, the cause of death was presumed to be unrelated to the ccMCT in 3 cases and unknown in 1 dog. Suspected ccMCT-related death was not reported in dogs with ITDs of exon 8 within 24 months. However, 2 dogs with exon 8 mutations had local tumor recurrence, 2 dogs had distant tumor recurrence, and local lymph nodes were involved in 3 dogs. The 2 dogs with ccMCTs having both exon 8 and 11 mutations were disease-free longer than 24 months without receiving additional treatment after surgery.
Overall Survival and Death and Suspected Canine Cutaneous Mast Cell Tumor (ccMCT) Related Death Within 24 Months Post Surgery, in 101 Dogs With ccMCTs With or Without Internal Tandem Duplications Mutations of Exons 8 and 11.
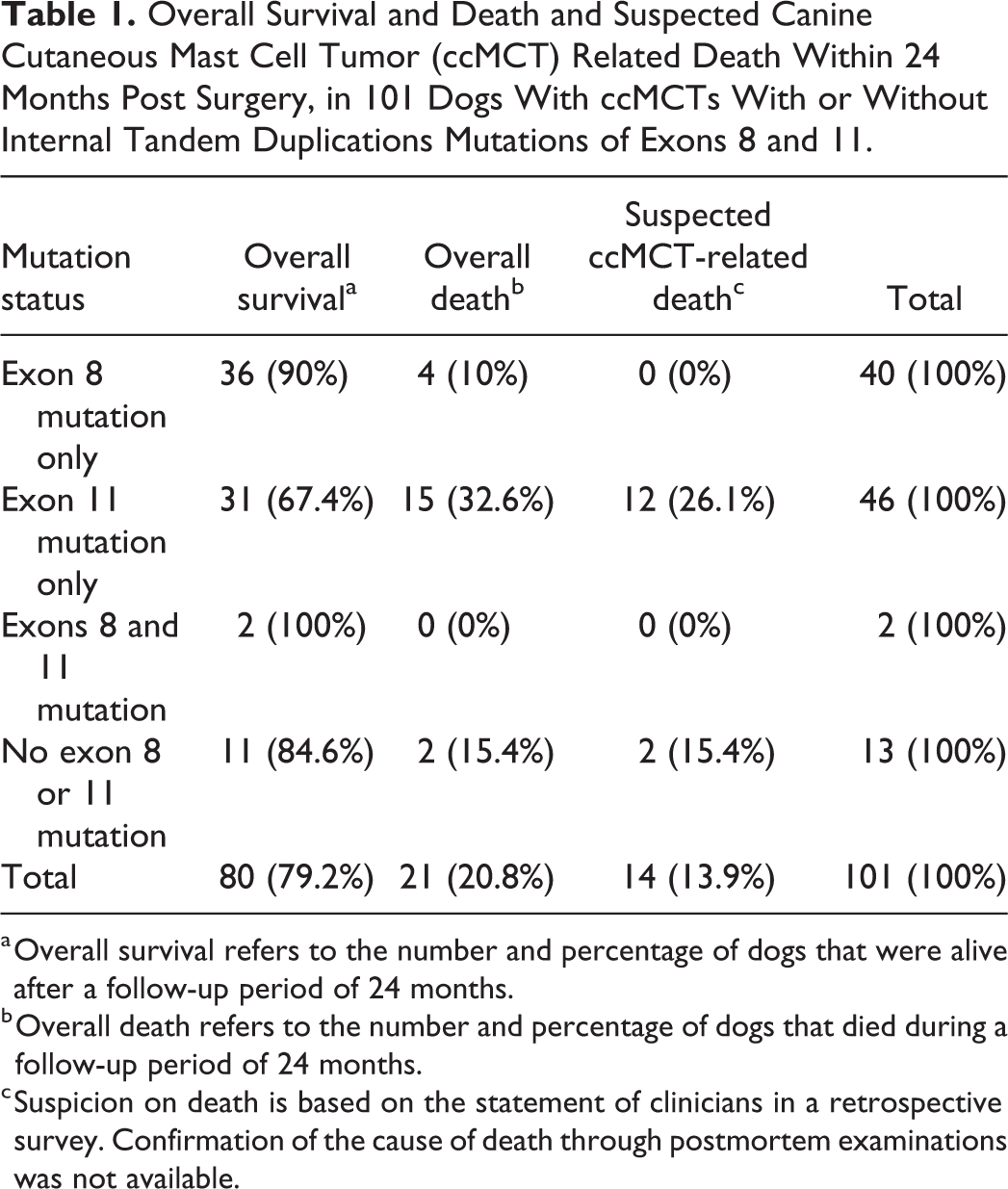
a Overall survival refers to the number and percentage of dogs that were alive after a follow-up period of 24 months.
b Overall death refers to the number and percentage of dogs that died during a follow-up period of 24 months.
c Suspicion on death is based on the statement of clinicians in a retrospective survey. Confirmation of the cause of death through postmortem examinations was not available.
The Kaplan-Meier curve showed a higher survival probability over 24 months (P = .027) for dogs with ccMCTs with exon 8 mutations in comparison to dogs with ccMCTs without exon 8 ITD mutations (Fig. 1). Comparing these 2 groups, dogs with ccMCTs with exon 8 mutations had a more than 70% reduced hazard of death with a hazard ratio of 0.29 (CI: 0.09–0.87, P = .027) for any cause of death (overall deaths). Since none of the animals died due to ccMCT-related complications, the observed hazard ratio was 0 (P < .001) for ccMCT-related deaths. For suspected nontumor deaths (other deaths), we observed a slightly higher hazard ratio of 1.66 (CI: 0.37–7.44; P = .506), which can be attributed to the fact that the animals did not die due to the competing risk of tumor death.
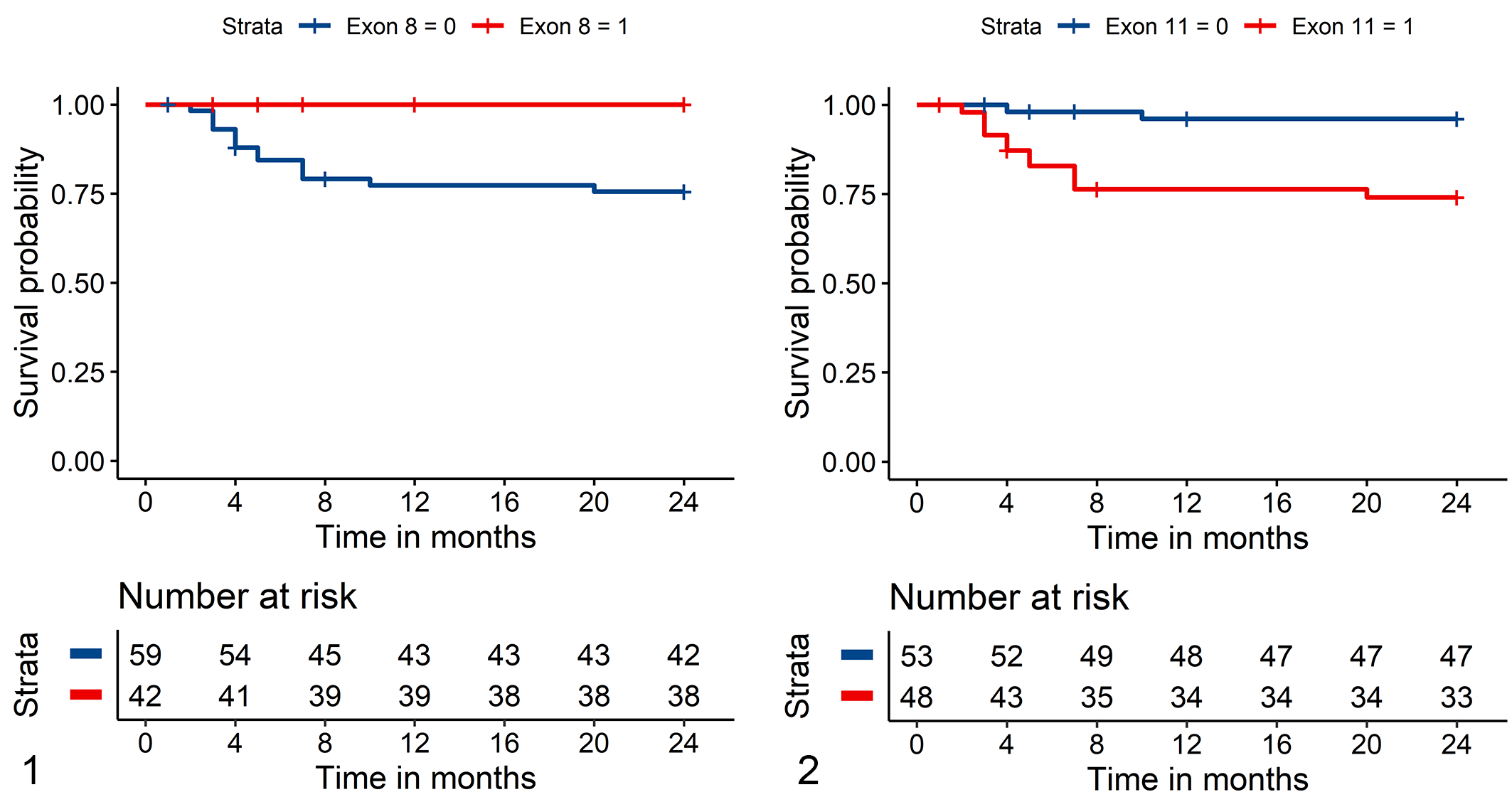
Kaplan-Meier survival curve for tumor-related death of dogs with canine cutaneous mast cell tumors (ccMCTs).
Outcome information was obtained by the survey for 48/100 dogs with ccMCTs with ITD mutations in exon 11 and for 53/121 dogs with ccMCTs without exon 11 mutation. Of the dogs with ccMCTs with exon 11 mutations, 33/48 (69%) survived more than 24 months. Of the 15 dogs that survived less than 24 months, 12 were suspected ccMCT-related deaths and 3 deaths were of presumed other causes. Twelve dogs had local recurrence, one had distant recurrence and two had both local and distant recurrence. Local lymph nodes were involved in 12 dogs and local as well as distant lymph nodes in 3 dogs. In contrast, of dogs with ccMCTs without ITD mutations in exon 11, 47/53 (85%) survived more than 24 months. Of the 6 dogs that survived less than 24 months, 2 were suspected ccMCT-related deaths and 4 other suspected causes of death. The Kaplan-Meier analysis showed a lower survival probability (P = .017) for dogs with ccMCTs with exon 11 mutations in comparison to dogs with ccMCTs without these mutations (Fig. 2). The hazard ratio for dogs with ccMCTs with exon 11 mutations were 3.18 for overall deaths (CI: 1.23–8.20; P = .017), 7.62 for suspected ccMCT-related deaths (CI: 1.70–34.05; P = .008), and 0.95 for other deaths (CI: 0.21–4.23; P = .942).
c-kit Mutations and Effect of Treatment on Outcome
Treatment of ccMCTs having exon 8 mutations were known in 39 cases. Whereas 18 dogs were treated by surgery alone, 21 dogs received additional treatment including radiation in 1 case, chemotherapy in 2 cases, chemotherapy and tyrosine kinase inhibitor in 5 cases, and tyrosine kinase inhibitor in 13 cases. There was only a minor difference in the treatment regime compared to dogs with ccMCTs without exon 8 mutations (Cramer’s V = 0.091, P = .551). Overall, the effect of additional treatment could not be determined for dogs with an exon 8 mutation, since none of the dogs died due to their ccMCT.
Thirty-two of 46 dogs with ITDs of exon 11 received additional treatment (radiation, chemotherapy, tyrosine kinase inhibitor; unknown in 2 cases). This included radiation in 1 case, chemotherapy in 6 cases, tyrosine kinase inhibitor in 15 cases, radiation and chemotherapy in 1 case, chemotherapy and tyrosine kinase inhibitor in 8 cases, and radiation, chemotherapy, and tyrosine kinase inhibitor in 1 case. There was a small difference in the treatment regime compared to dogs without exon 11 mutation (Cramer’s V = 0.166, P = .241). This can be explained by association of exon 11 mutations with higher grades and higher values of prognostic markers, which influences the treatment decision of the treating veterinarian. In comparison to surgical resection alone, dogs that had received additional treatment seemed to have no adverse outcome unless both systemic therapies (chemotherapy and tyrosine kinase inhibitors) were applied, which was only the case in tumors with poor prognosis. However, statistical analysis was hampered by the retrospective, nonrandomized, noncontrolled study design.
ITDs of Exon 8 Are Associated With Low Histological Grade
Of all 221 dogs with ccMCTs, low-grade tumors were diagnosed in 146 dogs and high-grade tumors in 75 dogs. Total survival was available for 101 of these dogs (low grade: 63; high grade: 38), and data analysis showed that high-grade ccMCTs had overall a lower survival probability (Fig. 3). Compared to low-grade ccMCTs, high-grade tumors had a 4.85 (CI: 1.52–15.48) times (P = .008) higher hazard for suspected ccMCT-related deaths.
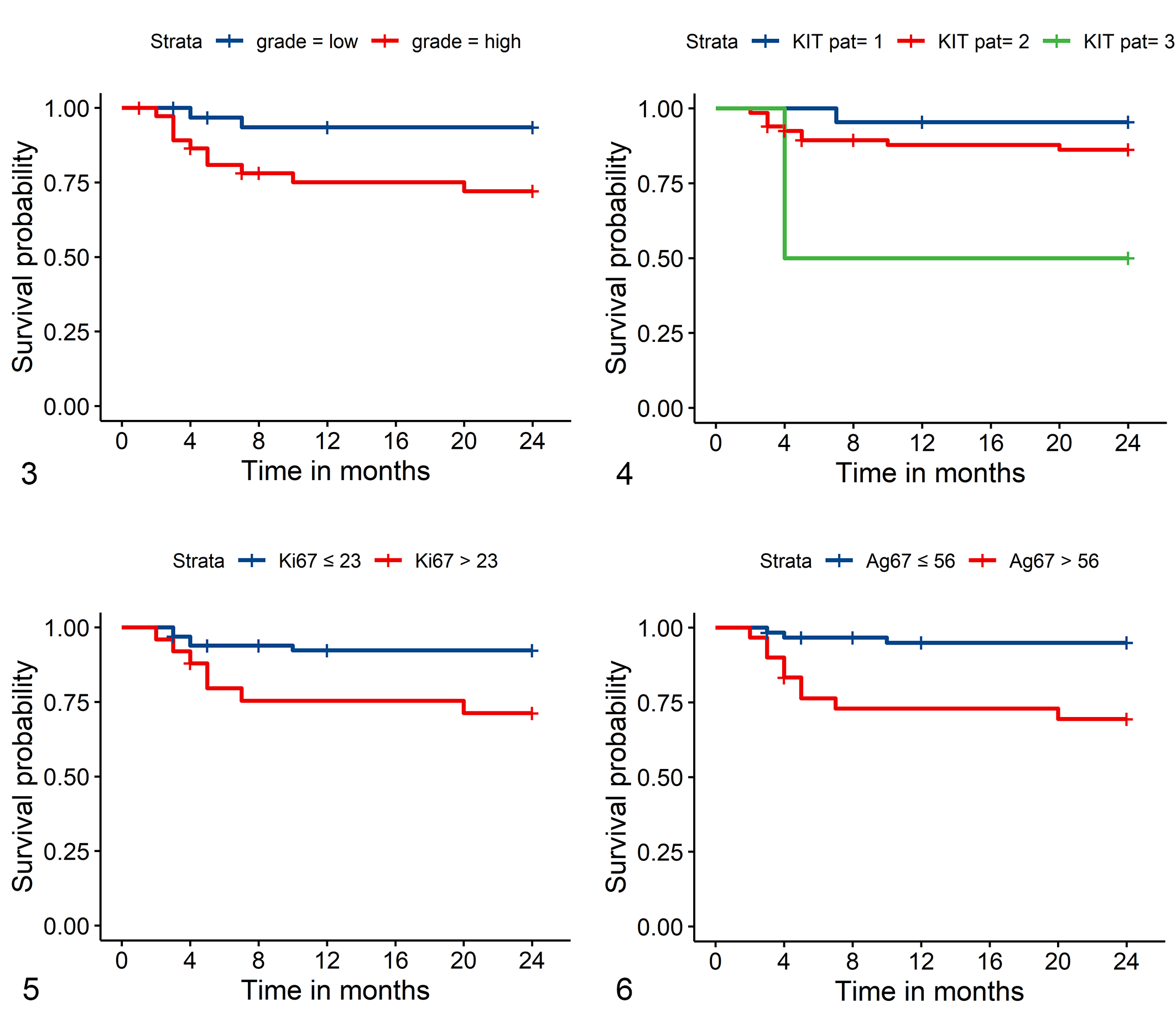
Kaplan-Meier survival curve for tumor-related death of dogs with canine cutaneous mast cell tumors (ccMCTs) based on cutoff values for different prognostic markers.
Of the 73 dogs with ccMCTs with exon 8 mutations, 64 dogs (88%) had low-grade ccMCTs and 9 dogs (12%) had high-grade ccMCTs, which was significantly (P < .001) different from cases without exon 8 mutation (82 low-grade, 55%; 66 high-grade, 45%) and cases with exon 11 mutations (38 low-grade, 38%; 62 high grade, 62%). Total survival was reported for 4/9 dogs with high-grade ccMCTs with exon 8 mutations. Of these 4 dogs, 3 dogs survived longer than 24 months, while one died 7 months after surgery due to suspected ccMCT-unrelated cause.
Tumors with ITD mutations in exon 11 were classified as low grade in 38 dogs (38%) and high grade in 62 dogs (62%), as opposed to 108 low-grade (89%) and 13 high-grade (11%) tumors without ITD mutations in exon 11 (P < .001). Total survival was reported for 33/62 dogs with high-grade ccMCTs with exon 11 mutation. Of these, 21 (64%) dogs survived 24 months and 12 dogs died earlier due to suspected (n = 9) ccMCT-related cause or suspected other causes (n = 3).
ccMCTs with no mutations in exon 8 or 11 were graded as low grade in 45 dogs (90%) and high grade in 5 dogs (10%). One of 2 dogs with a high-grade ccMCT and 1/11 dogs with a low-grade ccMCTs died before 24 months (10 and 4 months, respectively) presumably due to the ccMCT. The grade of ccMCTs with no mutations in exon 8 or 11 was significantly (P < .001) different compared to the grade of ccMCTs with exon 11 mutations and similar (P = .865) compared to the grade of ccMCT with exon 8 mutations.
Of the 2 ccMCTs with mutations in both exon 8 and 11, one was low grade and the other high grade, and the affected dogs had a disease-free survival of at least 24 months.
ITDs of Exon 8 Is Less Often Associated With Aberrant KIT Pattern
The KIT immunohistochemical staining pattern distinguished overall patient outcome as shown in the Kaplan-Meier curve (Fig. 4). Compared to dogs with ccMCTs with a KIT pattern of 1, the hazard for ccMCT-related death was 3.2 (CI: 0.4–25.4, P = .268) times higher for dogs with ccMCTs with a KIT pattern 2, and 14.9 (CI: 1.34–165.5, P = .028) times higher for dogs with ccMCTs with a KIT pattern 3. While 2/22 dogs with ccMCTs with a KIT pattern 1 died within 24 months (9%; suspected ccMCT-related death in one case), 15/71 dogs with ccMCTs with a KIT pattern 2 or 3 died within 24 months (21%, suspected ccMCT-related death in 11 cases).
KIT pattern 2 or 3 was present in 47/69 ccMCTs with exon 8 mutations (68%) and in 83/95 ccMCTs with exon 11 mutations (87%). A KIT pattern 2 or 3 was detected in 38/50 ccMCTs with no mutations in exon 8 or 11 (76%); this percentage was slightly, but not significantly higher (P = .158) than for ccMCTs with exon 8 mutations, and slightly lower (P = .109) than for ccMCTs with exon 11 mutations.
ITDs of Exon 8 Are Associated With Low Ki67 Index
The Ki67 index in ccMCTs from dogs with survival data was of prognostic value as shown in the Kaplan-Meier curve (Fig. 5). The hazard ratio was 4.0 (CI: 1.3–12.8, P = .017) for dogs with ccMCTs with a Ki67 index above the cutoff value of 23 compared to dogs with ccMCTs with a Ki67 index below the cutoff value for ccMCT-related deaths. Within 24 months, 8/25 dogs with ccMCTs with a Ki67 index above the cutoff died (32%; suspected ccMCT-related death in 7 cases), and 9/66 dogs with ccMCTs with a Ki67 index below the cutoff died (14%, suspected ccMCT-related death in 5 cases).
ccMCTs with exon 8 mutations (mean: 12.8, CV: 0.8) had a lower Ki67 index than ccMCTs without exon 8 mutations (mean: 22.0, CV: 0.8). In contrast, ccMCTs with exon 11 mutations (mean: 27.6, CV: 0.6) had a higher Ki67 index than ccMCTs without exon 11 mutations (mean: 12.3, CV: 0.9). The mean Ki67 index for ccMCTs without mutations in exon 8 or 11 was 11.6 (CV: 0.9), which was similar (P = .450) to the mean Ki67 index of ccMCTs with exon 8 mutations and significantly lower (P < .001) than the mean Ki67 index of ccMCTs with exon 11 mutations (Fig. 7). One ccMCT with mutations in both exons 8 and 11 had a Ki67 index of 14.0. Eleven of 69 (16%) ccMCTs with exon 8 mutations, 52/93 (56%) ccMCTs with exon 11 mutations, and 6/50 (12%) ccMCTs without mutations in exon 8 or 11 had a Ki67 index above the cut-off of 23.
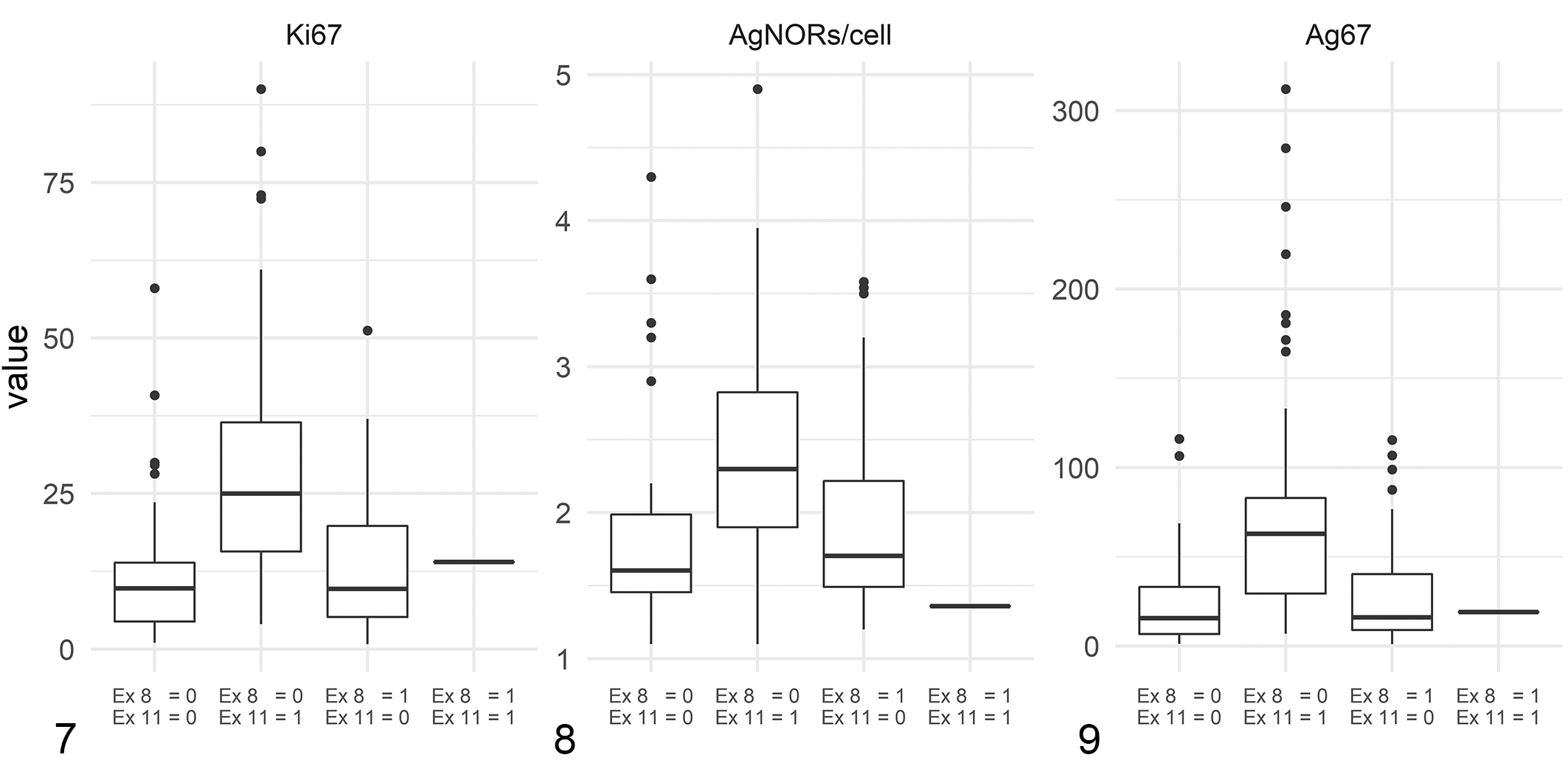
Comparison of prognostic values of canine cutaneous mast cell tumors without mutations in exon (Ex) 8 or 11 of c-kit (n = 13), with internal tandem duplications (ITD) mutations in exon 11 of c-kit (n = 40), with ITD mutations in exon 8 of c-kit (n = 37) and with ITD mutations in both exons 8 and 11 of c-kit (n = 1).
ITDs of Exon 8 Are Associated With Low Ag67 Score
The Ag67 score was a good prognostic marker in the examined dogs as shown in the Kaplan-Meier curve (Fig. 6). Dogs with ccMCTs with Ag67 scores above the cutoff had an increased hazard for ccMCT-related death by 7.0 times (CI: 1.9–25.8, P = .004). Within 24 months, 10/30 dogs with ccMCTs with an Ag67 score above 54 died (33%, suspected ccMCT-related death in 9 cases) and 7/61 dogs with an Ag67 score below the cutoff died (11%, suspected ccMCT-related death in 3 cases). The AgNOR score alone with a cutoff value of 2.25 23 had lower prognostic value (Fig. 8) and data are given in the Supplemental Materials.
ccMCTs with ITD mutations in exon 8 had a mean Ag67 score of 28.0 (CV: 1.0), which was lower than the Ag67 score of ccMCTs without mutations in exon 8 (mean: 53.6, CV: 1.0). In contrast, ccMCTs with exon 11 mutations had a higher Ag67 score (mean: 69.6, CV: 1.0) than ccMCTs without exon 11 mutations (mean: 26.0, SD: 26.6). ccMCTs with no mutations in exons 8 or 11 had a mean Ag67 score of 23.1 (CV: 1.0), which was similar (P = .322) to the Ag67 score of ccMCTs with exon 8 mutations and significantly (P < .001) lower than the Ag67 score of ccMCTs with exon 11 mutations (Fig. 9). One ccMCT with mutations in both exons 8 and 11 had an Ag67 score of 19.0. Ag67 scores were above the cutoff of 54 for 12/69 (17%) ccMCTs with exon 8 mutations, 52/93 (56%) ccMCTs with exon 11 mutations, and 5/50 (10%) ccMCTs with no mutations in exon 8 or 11.
Discussion
In this study, we analyzed the prognostic value of ITD mutations of exon 8 of c-kit in ccMCTs compared to dogs with ccMCTs with ITD mutations of exon 11 of c-kit and compared to dogs with ccMCTs without mutations in exon 8 or 11. The analysis was based on patient outcome in a large subset of dogs, and on the relationship to other prognostic parameters (histological grade, Ki67 index, AgNOR score, Ag67 score, and KIT expression patterns) within the whole study population. Our results corroborate that ITD mutations of exon 11 in c-kit are an indicator of a poor prognosis for ccMCTs and, in contrast, document that ITD mutations of exon 8 in c-kit were associated with less aggressive biological behavior of ccMCTs. 34,35 Dogs with MCTs with ITD mutations of exon 8 had longer overall survival, were associated with lower histological grade and proliferation score, and less often had aberrant KIT localization. Regardless, mutational status of c-kit is not an independent factor as the influence on patient outcome is explained through altered tumor cell proliferation and therefore influences tumor grade (includes mitotic count) and proliferation markers (Ki67, Ag67). 29 However, the hazard ratio for ITD mutations of exon 8 was lower than hazard ratios for low grade, proliferation markers below cutoff values, and KIT pattern 1. Our results suggest that the exon 8 mutational status is helpful for predicting overall good patient outcome and therefore may be recommended as a supplementary prognostic test. An advantage of molecular methods is objectivity, 29 whereas histological, 1,11 immunohistochemical, 19 and histochemical 7 prognostic parameters are prone to at least some intra- and interobserver variability.
The main limitation of the current study was that outcome information was obtained through a retrospective survey, and was based on a statement by the referring veterinarians of when the dog had died and an opinion of whether the dog exhibited ccMCT-related complications. Definitive confirmation of outcome criteria was not confirmed through postmortem examinations or histological examinations of metastasis. Due to the lack of true confirmation of the cause of death, we have calculated hazard ratios for overall death (including ccMCT-related and -unrelated death) as another cause of ccMCT-unrelated deaths should not be affected by the mutational status of the tumor. However, overall death may also be biased if the prevalence of unrelated deaths is unequally distributed in the groups evaluated. Future studies with prospective assessment of patient outcome are recommended to confirm our findings and should include confirmation of cause of death through postmortem examinations.
ITD mutations of exon 11 in c-kit play a crucial role in ccMCT tumorigenesis in some tumors. 40 It is likely that gain-of-function mutations of the negative regulatory juxtamembrane domain leads to decreased inhibition of the kinase domain and thereby to increased signal transduction and mast cell proliferation. 17 In contrast, the outcome of ccMCT with exon 8 mutations has been investigated in very few cases. 35 –37 While no association of an exon 8 c-kit mutation with poor outcome was observed in 3 ccMCTs, a mutation in exon 8 of c-kit was associated with a high-grade tumor, a short survival time of 6 months, and tumor recurrence in a single dog. 35 –37 Some studies combined ccMCTs with any c-kit mutation, including mutations in exon 8 and 11, and contrasted their behavior or prognostic parameters to ccMCTs without c-kit mutations. 3,5,6,18,27,30,35,36,41 As mutations in exon 11 were generally much more common than mutations in other exons, the effect of ITD mutations of exon 11 may potentially have superimposed the other mutations. This study describes patient outcome in a large number of dogs with ccMCTs with mutations of exon 8 for the first time. Of note, we have determined much higher prevalence of ITD mutations of exon 8 in comparison to previous studies. 5,27,37 Interestingly, we found a favorable outcome of affected dogs and relatively low scores of the various prognostic markers in these ccMCTs. Indeed, none of the dogs with ccMCTs with a mutation of exon 8 had died due to a confirmed or suspected ccMCT-related cause within 24 months postsurgery regardless of treatment regime or even high histological grade. While the 2 dogs with ccMCTs with concurrent mutations of exons 8 and 11 remained disease-free for at least 24 months, larger case numbers are necessary to determine if exon 8 mutations may have a protective effect in those cases. Notwithstanding, local and distant tumor recurrence was reported in few dogs that had ccMCTs with mutations of exon 8. The results of our study suggest that screening for mutations of exon 8 of c-kit in ccMCTs might be helpful to identify less aggressive tumors, while detection of mutations of exon 11 of c-kit is important to identify more aggressive ccMCTs. Future prognostic studies should analyze ccMCTs with mutations in different exons separately as ccMCTs with ITD mutations of exon 8 or 11 clearly have different biological behavior, and grouping such neoplasms may negatively impact prognostic evaluations.
Similar to ITD mutations of exon 11 of c-kit, it has been proposed that ITD mutations of exon 8 are gain-of-function mutations based on an in vitro experiment. 15 Additionally, ligand-independent phosphorylation of KIT, consistent with a gain-of-function mutation, was reported in an intestinal MCT with an exon 8 mutation of a dog. 13 This is in contrast to the findings of the present study, as we were unable to demonstrate a more aggressive behavior of ccMCTs with exon 8 mutations, and there was no significant increase of proliferation markers in these tumors compared to ccMCTs without mutations of exons 8 and 11. Based on the outcome and proliferation marker data of this study, it seems unlikely that the detected mutation of exon 8 of c-kit results in a gain-of-function of KIT in ccMCTs, and that subsequently the hyperactivated receptor would cause increased tumor proliferation. In humans, both gain-of-function and loss-of-function mutations have been reported for exon 8 of c-kit in different diseases. 25 A gain-of-function mutation can be explained by increased stabilization of the dimerization between the 2 monomeric KIT receptors and is found in some human proliferative disease such as mastocytosis. Loss-of-function mutations, a hypoactivated receptor, can be explained by a weakened binding affinity of dimerization and accelerated ligand dissociation with shortened signal transduction. 25 They are usually not important for tumorigenesis, but have been reported with inherited absence of cutaneous melanocytes (piebaldism) in humans. 14,25 We speculate that the detected exon 8 mutations in ccMCTs have either no effect on KIT protein function or may even represent a loss-of-function mutation and may not directly be involved in the tumorigenesis of ccMCTs. This speculation highlights the importance of a future, more comprehensive analysis of c-kit mutations occurring in ccMCTs and their association with the biological behavior of ccMCTs. Although Letard et al 15 demonstrated in an in vitro study that a mutation of exon 9 of c-kit was activating the kinase domain, these results should be verified in a study with outcome data and appropriate case number, as was done for exon 8 ITD mutations in the present study. A limitation of the present study was that only ITD mutations of exons 11 and 8 were examined. Screening for other types of mutations or mutations in other exons was not performed. Therefore, positive or negative effects of potential concurrent gain-of-function or loss-of-function mutations cannot be excluded.
Besides the mutations in different exons, the histological location of the tumor mass, that is, cutaneous or subcutaneous MCT, may influence the associated prognosis. 32 Generally, c-kit mutations have been described to be less frequent in subcutaneous MCT. 31,33,37 While ITD mutations of exon 11 have previously not been found in subcutaneous MCTs, there is a report of a single subcutaneous MCT with an ITD mutation in exon 8 within a series of 11 examined subcutaneous MCTs. 35 Although we have found both exon 11 and exon 8 mutations in a few subcutaneous MCTs (unpublished data), we have excluded them from this study as they were too few to analyze separately, and we wanted to avoid analyzing them together with ccMCTs. For similar reasons, we also excluded a few oral MCTs in which we also found ITD mutations in exons 11 and 8 (unpublished data).
Another limitation of this study was that dogs had received different treatments that may have influenced survival. Although treatment was similar between groups with different mutation status, decision on treatment was done by the owners and referring veterinarians based on information of histological grade and prognostic markers, that is, not in a randomized manner. The results of the present study might suggest that additional therapeutic measures (radiation, chemotherapy, tyrosine kinase inhibitors) may not be necessary in ccMCT with ITDs of exon 8. However, as no dog with exon 8 mutations died due to ccMCT regardless of the treatment regime, the benefit of additional therapy is difficult to assess in the present cases, and future studies with a prospective, randomized, controlled study design are necessary. For ccMCTs with exon 11 mutations, it is assumed that specific tyrosine kinase inhibitors can reduce the hyperactivated status of the receptor and thus improve prognosis. However, there are conflicting study results on the effect of treatment success of tyrosine kinase inhibitors based on the presence or absence of the mutation. While some studies determined that tyrosine kinase inhibitors are especially beneficial for dogs with MCTs with exon 11 mutations, others did not find a difference comparing the mutation status. 5,8,16,41 As the present study suggests that ITD mutations of exon 8 are not gain-of-function mutations, the effect of tyrosine kinase inhibitors is unknown and needs to be evaluated in future studies.
Conclusion
Prognostic parameters have different accuracies for predicting poor or good patient outcomes. Therefore, a combination of these prognostic tests, including the mutational status of c-kit, is generally recommended. 10,24 While mutations in exon 11 of c-kit in ccMCTs predict a higher risk of aggressive behavior, ITD mutations in exon 8 in ccMCTs appear to be associated with better clinical outcomes regardless of treatment regimens and histological grade. Screening for ITD mutations of exon 8 might be helpful to identify less aggressive ccMCTs. The data of this study may suggest that additional treatment following complete surgical removal is likely unnecessary in cases of ccMCTs with exon 8 mutations, due to the overall good prognosis. Regardless, future studies are required to analyze the effect of ITD mutations of exon 8 on the receptor function (gain or loss of function) of KIT, and to determine the efficacy of different treatment regimens in ccMCTs with exon 8 mutations. Last, future prognostic and therapeutic studies should be conducted with the knowledge of a more complete c-kit mutation status of the analyzed population, and MCTs should be sorted into different groups based on their mutation status.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Bertram_et_al - Internal Tandem Duplication of Exon 8 of c-kit Is Associated With Longer Total Survival in Canine Cutaneous Mast Cell Tumors
Supplemental Material, Combined_supplemental_materials-Bertram_et_al for Internal Tandem Duplication of Exon 8 of c-kit Is Associated With Longer Total Survival in Canine Cutaneous Mast Cell Tumors by Bouvien A. W. Brocks, Christof A. Bertram, Alexander Bartel, Jolle Kirpensteijn, Alexandra Collins-Webb, Chuck Catlin, Tuddow Thaiwong and Matti Kiupel in Veterinary Pathology
Footnotes
Acknowledgement
The authors would like to thank the MSU VDL histology and virology laboratories for perfomring the histology, immunohistochemistry and PCR testing and the referring veterinarians for providing the cases and clinical information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
