Abstract
Epitheliotropism is an important diagnostic feature of cutaneous epitheliotropic lymphoma and canine cutaneous histiocytoma; however, although noted in certain feline mastocytic diseases, it has not been considered a feature of canine cutaneous mast cell tumor. In this study, 3 canine cutaneous mast cell tumors had epitheliotropic invasion of neoplastic mast cells into the epidermis and follicular epithelium. This unusual histologic finding was characterized by infiltrates of individual and clusters of neoplastic mast cells in the stratum basale and stratum spinosum. The mast cell origin of these cells was documented by demonstration of metachromasia with Giemsa stain and positive immunoreactivity to KIT protein. On the basis of these findings, mast cell tumors should be included in the differential diagnosis for canine cutaneous round cell neoplasms that infiltrate the epidermis.
In dogs and cats, cutaneous mast cell tumors (MCTs) are solitary or multicentric tumors that are usually confined to the dermis and/or subcutis. 3 Invasion of the epidermis by mast cells is rare and has been described in only a few cases of mastocytic proliferative diseases in cats. Epitheliotropism was evident in 1 of 14 cats in a retrospective study of MCTs 1 and in a case of disseminated MCT with systemic mastocytosis. 5 In addition, mast cell invasion of the epithelium of the nictitans was a characteristic finding of feline conjunctival papillary mastocytosis. 2
In dogs, the major epitheliotropic round cell tumors of the integument are cutaneous epitheliotropic lymphoma and cutaneous histiocytoma. To the authors’ knowledge, epitheliotropic canine cutaneous mast cell tumors have not been reported. The purpose of this study is to report the clinicopathologic findings in 3 dogs with epitheliotropic cutaneous MCTs.
Clinical Histories
Case No. 1 was a 2-year-old, intact female, Norfolk Terrier that was presented to a private clinic for evaluation of an 11-mm-diameter, pink, discolored mass at the right ventrolateral aspect of the nasal mucocutaneous junction. An incisional biopsy specimen was submitted for histopathologic examination. Upon evaluation at a reference hospital, both submandibular lymph nodes were palpable but not significantly enlarged. Fine needle aspirates of the spleen and left submandibular node were unremarkable; rare, well-granulated mast cells were observed in the cytologic preparation from the right submandibular lymph node. Although the low number of mast cells in the cytologic preparation was consistent with a nonneoplastic process, the possibility of metastasis could not be excluded. Thoracic radiographs and abdominal ultrasound were normal. Given the tumor location, complete resection would have been disfiguring, so a course of chemotherapy and radiotherapy was prescribed.
Chemotherapy with weekly vinblastine injections and oral prednisolone commenced, and after 3 doses of vinblastine, the tumor was almost imperceptible. Radiotherapy (12 fractions on a Monday-Wednesday-Friday schedule over 4 weeks) was initiated at the time of the fourth weekly vinblastine treatment. Vinblastine injections were continued every other week throughout and subsequent to radiation, until a total of 8 treatments had been administered. No clinical signs of recurrence or metastasis were apparent 18 months after cessation of treatment.
Case No. 2 was a 3-month-old, castrated male, Maltese puppy, evaluated at a private clinic for a 5-mm-diameter, slightly raised mass just ventral to the anal mucocutaneous junction. The mass was aspirated and interpreted at the clinic as containing numerous, well-differentiated mast cells. The mass was resected and submitted for histopathological evaluation. The dog was considered disease free 6 months after surgical excision, the sole treatment for its tumor.
Case No. 3 was a 6-year-old, castrated male, Greyhound, evaluated at a private clinic for a 2-cm-diameter, firm cutaneous mass on the lateral aspect of the left metatarsus. The mass was resected and submitted for histopathological evaluation. This dog was considered disease free 7 months after surgical excision, the sole treatment for its tumor.
Histologic and Genetic Findings
In histologic sections, the neoplasms of case Nos. 1 and 2 were 5 mm in diameter and 3 × 1 mm, respectively. In both tumors, haired skin contained a non-delimited neoplasm that extended from the superficial dermis into the deep dermis. Neoplastic cells were arranged in loose aggregates in collagenous stroma, interspersed by eosinophils. Neoplastic cells had distinct cell borders and moderate amounts of pale, amphophilic to eosinophilic cytoplasm with variable numbers of fine basophilic granules. Nuclei were round or oval and had finely stippled chromatin and 1 to 2 small nucleoli. Anisokaryosis was mild; mitotic figures were fewer than one per ten 400 × fields in case No. 1 and two per ten 400× fields in case No. 2. Multifocally, neoplastic cells infiltrated the epidermis (Fig. 1) and the follicular epithelium, either individually or in small clusters that disrupted the stratum basale and stratum spinosum.
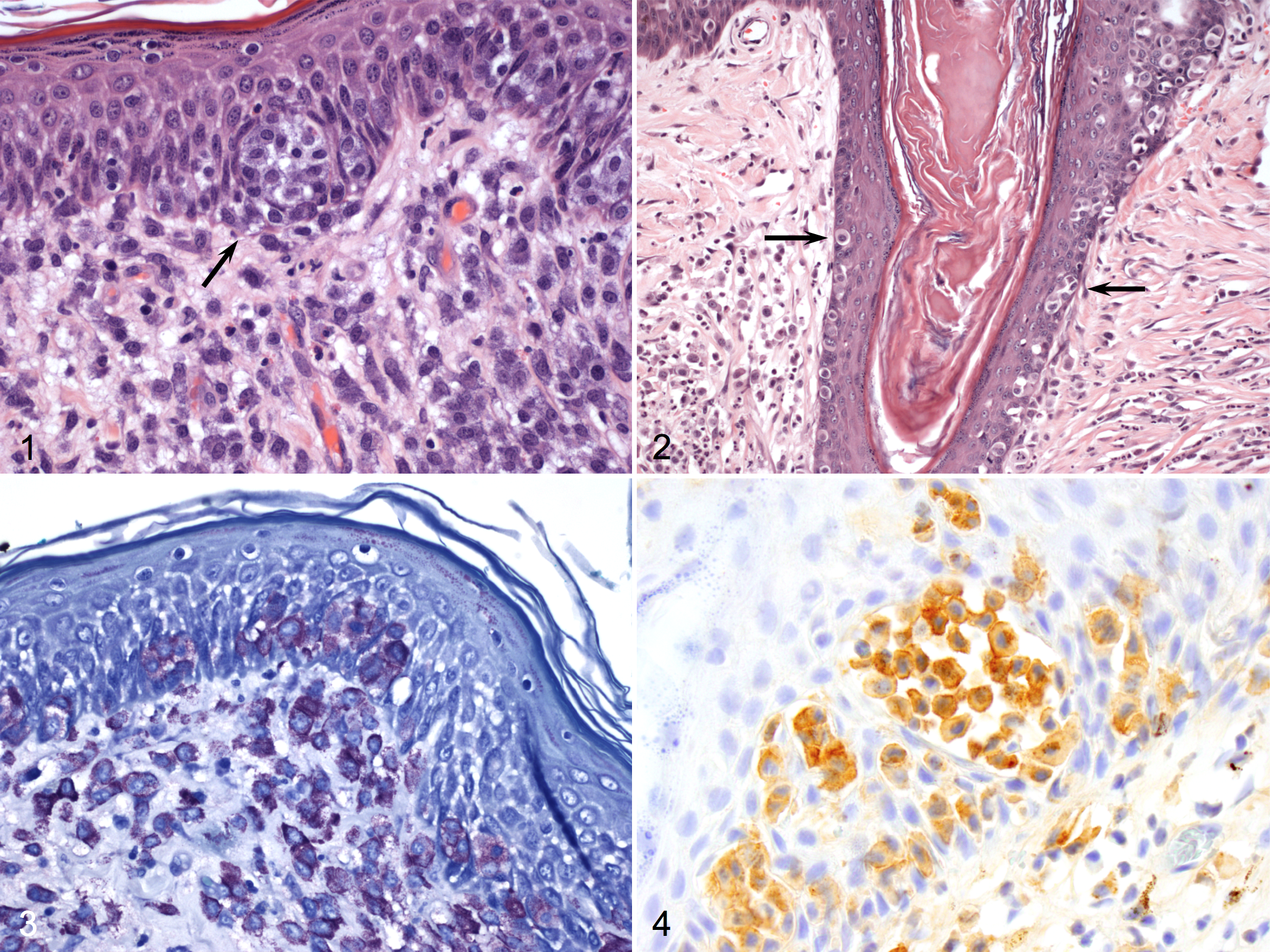
Haired skin; dog No. 2. A cluster of neoplastic mast cells disrupts the stratum spinosum (arrow). Hematoxylin and eosin (HE).
In case No. 3, haired skin was infiltrated by a non-encapsulated neoplasm measuring 1.6 × 0.6 cm in diameter and composed of dense sheets of round cells that extended from the superficial dermis into the subcutis. Neoplastic cells had variably distinct cell borders and moderate to large amounts of pale amphophilic cytoplasm containing few to moderate numbers of fine basophilic granules. Nuclei were oval and had finely stippled chromatin and 1 to 2 moderate-sized nucleoli. There were moderate anisocytosis and anisokaryosis; 12 mitotic figures were observed in ten 400 × fields. As in the other 2 tumors, neoplastic cells infiltrated the epidermis and the follicular epithelium, either individually or in small aggregates (Fig. 2). Numerous eosinophils were admixed with neoplastic cells.
In Giemsa-stained sections of the tumor of case Nos. 1 and 2, the cytoplasmic granules of the neoplastic cells, including those that infiltrated the epidermis and follicular epithelium, had strong metachromasia (Fig. 3). In case No. 3, metachromasia of the cytoplasmic granules was weak in both dermal and epidermal neoplastic cells. The morphologic features of the neoplastic cells in all 3 cases, coupled with the metachromasia of cytoplasmic granules in the Giemsa-stained sections, warranted the diagnosis of MCTs with epitheliotropism. According to Patnaik’s grading system for cutaneous mast cell tumors, 8 the masses of case Nos. 1 and 2 were diagnosed as grade II mast cell tumors and the third case as grade III. According to a more recently proposed grading system for MCTs, 4 the first 2 tumors were classified as low-grade MCTs; the third was classified as a high-grade MCT.
Histochemistry for argyrophilic nucleolar organizing regions (AgNORs), immunohistochemistry for Ki67 and KIT, and polymerase chain reaction analysis for activating (internal tandem) duplication mutation in exon 11 of the c-KIT gene were performed as described. 10 AgNOR-stained slides had an average of 2.71, 2.24, and 1.79 positive AgNORs per nucleus in neoplastic cells of case Nos. 1, 2, and 3, respectively. The growth fraction of neoplastic cells, measured by immunoreactivity with Ki67, was 13, 14.2, and 11.2 in case Nos. 1, 2, and 3, respectively. The proliferation index of the neoplastic cells (AgNOR × Ki67) was 28.21, 31.81, and 20.05 in case Nos. 1, 2, and 3, respectively. All 3 tumors had low proliferative activity as measured by Ki67, AgNOR, and AgNOR × Ki67 indices. 10 All 3 tumors had immunohistochemical reactivity for c-KIT (Fig. 4) with a perinuclear or stippled cytoplasmic pattern and loss of perimembrane reactivity, consistent with a KIT pattern 2. 10 Mutation in the c-KIT gene was detected only in the tumor of case No. 3.
Discussion
The differential diagnosis for canine cutaneous tumors with an intraepidermal component includes epitheliotropic lymphoma (CEL), cutaneous histiocytoma (CH), melanocytoma, and melanoma. 3 The mast cell origin for the neoplastic cells in the cases reported here was documented by demonstration of metachromatic cytoplasmic granules and immunoreactivity for KIT in neoplastic cells of the dermis, as well as in those that infiltrated the epidermis.
Epitheliotropism is not a recognized feature of canine cutaneous MCTs. However, in cats, epitheliotropism was observed in a MCT of the lip 1 and in a case of disseminated MCT with splenic, hepatic, and bone marrow involvement. 5 Infiltration of mast cells into surface epithelium is also characteristic of the nonneoplastic condition, feline conjunctival papillary mastocytosis (FPCM). 2 Notably, in the cat with MCT of the lip and in cats with FPCM, the epitheliotropism involved the mucocutaneous junction or the mucosal surface.1,2 The tumors of case Nos. 1 and 2 were also near mucocutaneous junctions, which may suggest a preference of epitheliotropic MCTs for areas near or in mucosal surfaces.
The mechanisms by which neoplastic cells in CEL and CH infiltrate the epidermis are not well understood. In CEL, epitheliotropic malignant T cells express skin-homing receptors on their surface, which could enable them to bind to ligands on keratinocytes. 3 The expression of intercellular adhesion molecule 1 (ICAM-1) on keratinocytes is correlated with that of lymphocyte associated antigen 1 (LFA-1) on neoplastic T cells in human cases of CEL; however, these parameters were not statistically significant in canine CEL. 7 In canine CH, the invasion of dendritic Langerhans cells into the epidermis is speculated to be secondary to the expression, by neoplastic cells, of adhesion molecules such CD44, ICAM-1, and very late activation antigen 4 (VLA-4). 6 It is undetermined whether the epitheliotropism of the neoplastic mast cells is the result of neoexpression of adhesion molecules on these cells or due to other mechanisms.
The cytoplasmic granules of mast cells contain many biologically active substances, including proteolytic enzymes such as matrix metalloproteinases (MMPs) that degrade connective tissue matrix proteins. 9 The infiltration of lymphocytes into the epithelium has been attributed to basement membrane degradation by MMPs from degranulated mast cells in human oral lichen planus. 9 The epitheliotropism of the neoplastic mast cells in the canine cases may have been the result of damage to the epithelial basement membrane secondary to release of MMPs. Histologic evidence of active degranulation and damage of the basement membrane (evaluated with periodic acid-Schiff) was not detected in the tumors of this study, however.
The prognostication of canine MCTs currently is based on histologic grading, as well as proliferation indices (eg, Ki-67 activity, AgNOR count, and AgNOR × Ki67 index), the pattern of KIT immunoreactivity, and the presence of mutations in the c-KIT gene.3,4,8,10 All 3 dogs in this study responded well to the chosen treatment protocol, regardless of the histologic grade or other prognostic assessments. Despite the good outcome to date in all 3 cases, the low number of cases and the short follow-up period in case Nos. 2 and 3 are insufficient to document an association between epitheliotropism and prognosis.
Although rare, MCTs should be included in the differential diagnosis for epitheliotropic canine cutaneous round cell neoplasms. Epitheliotropism was recognized in both grade II/low-grade and grade III/high-grade MCTs, as well as in MCTs with or without mutations of the c-KIT gene.
Footnotes
Acknowledgement
We thank Dr Wayne Corapi for his assistance with photomicrographs.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
