Abstract
Maspin is a serine protease inhibitor that inhibits tumor invasion and metastasis in human breast cancer and is consistently expressed by mammary myoepithelial cells (MECs). To analyze the value of maspin as a marker of the MEC layer of the normal and tumoral canine mammary gland, the immunohistochemical expression of maspin was studied in formalin-fixed tissues from 55 benign and malignant tumors (40 tumors also contained the surrounding normal mammary gland) using a commercially available monoclonal antibody. Periacinar and periductal MECs of all 40 normal mammary glands were stained by the anti-human maspin monoclonal antibody, and immunoreactivity was observed in the nucleus and cytoplasm of these cells. In addition, maspin was found in 53 (98%) of the tumors studied, reacting with the MECs in 100% of benign tumors and 93% of malignant tumors and to the epithelial cells of 16% of benign and 73% of malignant tumors. In the MEC compartment, immunoreactivity was observed in the cytoplasm of hypertrophic MECs, fusiform MECs, stellate MECs, rounded (myoepithelial) cells, and chondroblasts. In the epithelial cell compartment, immunoreactivity was observed in the cytoplasm of cells with and without squamous differentiation. Stromal myofibroblasts were unreactive. Maspin appears to be a very sensitive marker of the normal and neoplastic myoepithelium that, contrary to smooth muscle differentiation markers, does not stain stromal myofibroblasts. In addition, a subset of neoplastic epithelial cells reacted with the maspin antibody. The relationship between maspin expression in different cellular compartments of canine mammary carcinomas and the biologic aggressiveness of the disease remains to be elucidated.
Canine mammary gland tumors show great histomorphologic heterogeneity and, in addition to epithelial cells, they frequently have spindle cells (complex tumors), as well as cartilage, bone, or fat (mixed tumors). 9 Spindle cells of complex and mixed tumors have been traditionally assigned a myoepithelial cell (MEC) origin. 4 Recent studies have demonstrated the myoepithelial origin of the cartilage and bone observed in canine mammary gland mixed tumors. 2 The high frequency of tumors showing myoepithelial or basal cell proliferation is one of the unique features of canine mammary tumors. 8 In contrast, similar lesions are considered rare in human breast. 8 In dogs, among the prognostic variables, the tumor type has been found to be an important independent factor in nearly all studies, and a range of increasing malignancy from complex carcinoma to simple carcinoma to sarcoma has been described. 8 MECs in human breast tumors are considered as natural paracrine suppressers of invasion and metastasis and may specifically inhibit the progression of precancerous disease states to invasive cancer in vivo. 18 Maspin is a unique member of the serpin family that inhibits tumor invasion and metastasis of human breast cancers. 11, 21 It is consistently expressed by mammary MECs. 14 The progressive loss of expression of maspin from in situ to invasive disease has been described in human breast cancer. 21 These findings prompted us to analyze the value of an anti-maspin antibody in selectively staining the MEC layer of the normal mammary gland of the dog and the MEC component of mammary gland tumors.
Materials and Methods
Case selection
Fifty-five mammary gland biopsy or mastectomy specimens were collected from the archives of the Departments of Comparative Pathology at the Universities of Las Palmas de Gran Canaria and Córdoba, Spain. The cases reported in this study were all examined histologically in hematoxylin and eosin (HE)–stained sections before being examined immunohistochemically to reclassify them according to the new World Health Organization classification of tumors of the mammary gland of the dog. 9 The 55 specimens comprised 25 benign and 30 malignant tumors (Table 1). Non-altered mammary gland tissue was found in the vicinity of the tumors in 40 cases (Table 1). All tissues had been fixed in 10% neutral buffered formalin and embedded in paraffin wax.
Characterization of cell types and expression of maspin in normal mammary gland and mammary tumors of the dog.
∗ MEC = myoepithelial cells; HMEC = hypertrophic myoepithelial cells; FMEC = fusiform myoepithelial cells; SMEC = stellate myoepithelial cells; RMEC = rounded myoepithelial cells; CHB = chondroblasts; EC = epithelial cells.
† Number of cases positive for specific cell type.
‡ Positive epithelial cells with squamous differentiation (in parentheses).
Maspin immunostaining
The monoclonal antibody used in this study was a mouse monoclonal anti-human maspin (clone G167-70) (Phar-Mingen International, San Diego, CA) diluted 1 : 400 in phosphate-buffered saline (PBS) pH 7.4 containing 10% normal rabbit serum. Sections 3 μm thick were cut from at least one representative block of each case and collected onto Vectabond-coated slides (Sigma Diagnostics, St Louis, MO). After this, the slides were deparaffined, rehydrated in graded alcohols, and incubated with 3% hydrogen peroxidase in methanol for 30 minutes to block endogenous peroxidase activity. Heat-induced antigen retrieval (water bath at 95° C) with 1 mmol/liter ethylenediamininetetraacetic acid (pH 8.0, 15 minutes) was used. 2 After cooling down, the slides were covered with 10% normal rabbit serum in PBS for 30 minutes before incubation with the primary antibody for 18 hours at 4° C, and the avidin–biotin–peroxidase complex (ABC) method was applied (Vector Laboratories, Burlingame, CA). The chromogen, 0.5% 3,3′ diaminobenzidine tetrahydrochloride (Sigma) diluted 1 : 10 in 0.05 M Tris containing 0.3% of hydrogen peroxide, was applied to the slides for 1–2 minutes at 20–22° C. Slides were counterstained with Harris's hematoxylin. Negative controls were obtained by omitting the primary antibody and by substituting the primary antibody for mouse ascites fluid diluted 1 : 400, whereas normal mammary gland tissue served as an internal positive control in every assay.
Evaluation of immunohistochemical data
Multiple fields of each slide were examined, and positivity was indicated by the presence of distinct brown nuclear or cytoplasmic staining. The immunostaining was evaluated separately, and results were based on the consensus of at least two observers. The amount of positive cells was determined as a percentage of the total number of cells observed in each slide. Thus, the semiquantitative scoring (SQS) was (−) when none of the cells were stained, (+) when positive cells represented less than 20% of cells observed, (++) when positive cells represented between 20% and 59% of cells observed, and (+++) when more than 60% of the cells observed were positive. The intensity of positivity was evaluated on a three-point scale (1i, 2i, 3i). Both the SQS system and the intensity scoring (IS) were analyzed in all different types of immunoreactive cells observed.
RESULTS
Normal mammary gland (n = 40)
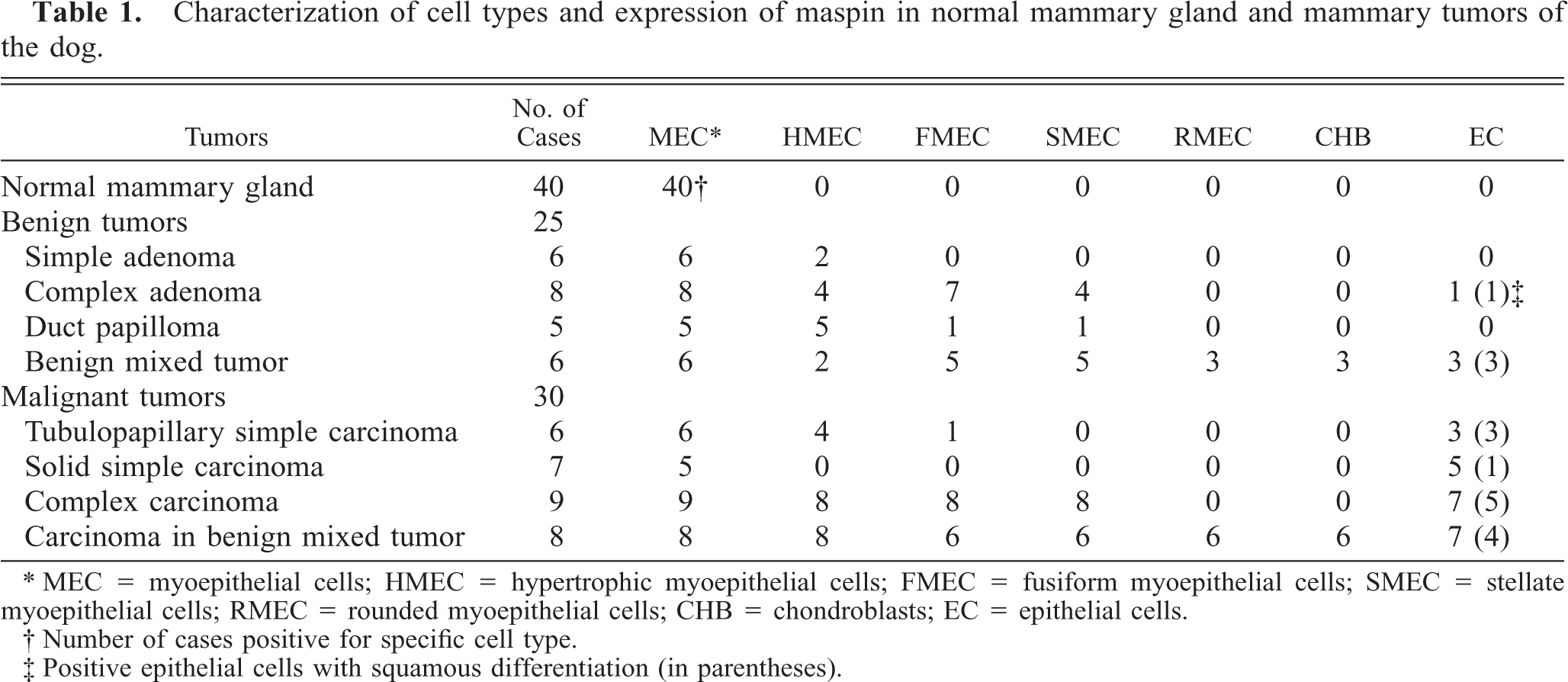
All MECs of ducts and acini reacted with the anti-maspin monoclonal antibody, whereas epithelial cells were unreactive. Immunoreaction was observed in the nucleus and throughout the cell cytoplasm (Fig. 1). SQS was +++ and IS was 3i. When the skin was included in the tissue section (n = 28), immunoreaction was observed in the epidermis, hair follicles, sebaceous, and apocrine sweat glands.
Normal mammary gland; dog. All the myoepithelial cells of ducts and acini are positive with the maspin antibody and the epithelial cell layer is unreactive. Maspin-immunoreactive products are observed in the nucleus and cytoplasm. ABC method, Harris's hematoxylin counterstain. Bar = 40 μm.
Benign tumors (n = 25)
Simple adenomas (n = 6)
Maspin-immunoreactive cells were found in two different locations: the continuous layer of periductal and periacinar, flattened spindle cells not visible in HE-stained sections (MECs) (n = 6) and large, polygonal cells with clear cytoplasm in HE-stained sections (hypertrophic MECs [HMECs]) that formed a monolayer around luminal epithelium (n = 2) (Table 1). MEC staining was nuclear and cytoplasmic, and HMEC reaction was mainly located in the cytoplasm. The SQS was +++, and the IS was 3i for MEC and variable (2i–3i) for HMEC. The inner epithelial cell layer was unreactive.
Complex adenomas (n = 8)
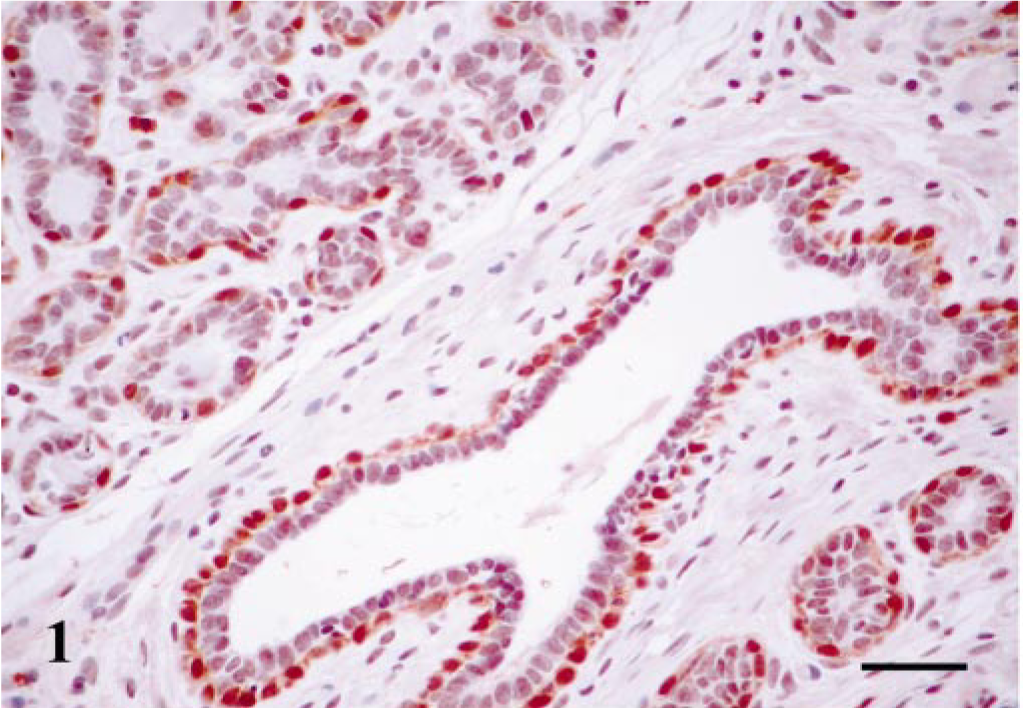
Maspin-immunoreactive cells were found in all cases (Table 1). 1) MECs formed a monolayer around luminal epithelial–type structures (n = 8 cases); immunoreactive products were homogeneous within the cell nucleus and cytoplasm; and SQS was +++ and IS was 3i. 2) HMECs formed a monolayer around luminal epithelial–type structures (n = 4 cases); immunoreactive products were homogeneous within the cell cytoplasm; and SQS was +++ and IS was 3i. 3) Fusiform MECs (FMECs) formed nodules either above or below of the layer of MECs (n = 7 cases); immunoreactive products were homogeneous within the cell cytoplasm; and SQS was + to ++ and IS was 2i to 3i (Fig. 2). 4) Stellate MECs (SMECs) were arranged in solid nests and intermingled with FMECs (n = 4 cases); immunoreactive products were homogeneous within the cell cytoplasm; and SQS was + and IS was 2i (Fig. 2). The staining was nuclear and cytoplasmic for MECs and mainly located in the cytoplasm for the other types of MECs. In one case, luminal epithelial cells showed intracytoplasmic immunoreactivity. These cells were located in areas of squamous differentiation and SQS was + and IS was 1i.
Mammary gland complex adenoma; dog. Nests of fusiform (FMEC) and stellate (SMEC) cells. Maspin-immunoreactive cells show cytoplasmic staining. Some scattered cells showed nuclear immunoreaction. ABC method, Harris's hematoxylin counterstain. Bar = 50 μm.
Duct papilloma (n = 5)
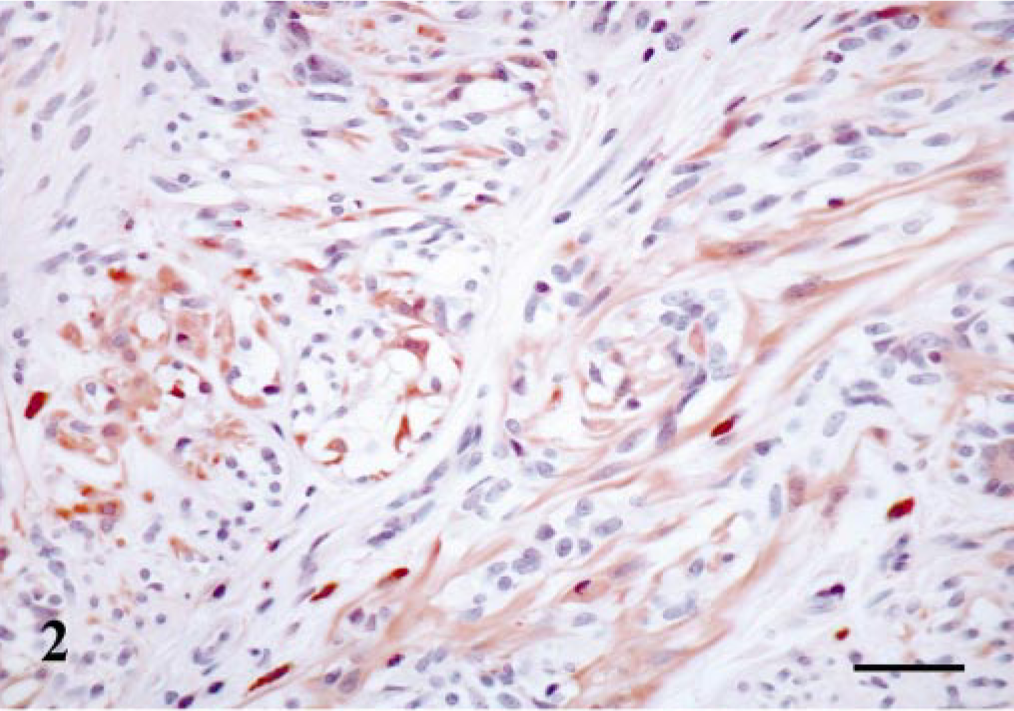
Histologically, three simple and two complex duct papillomas are included in this group. Maspin-immunoreactive MECs and HMECs were observed in all cases (Table 1). MEC staining was nuclear and cytoplasmic, and HMEC reaction was mainly located in the cytoplasm. The SQS was +++, and the IS was 3i for MECs and variable (2i–3i) for HMECs (Fig. 3). In one case of complex duct papilloma (Table 1), immunoreactive FMECs and SMECs were observed, and their immunoreactive products were homogeneous within the cell cytoplasm in both types of cells, and SQS was + to ++ and IS was 2i. The inner epithelial cell layer was unreactive.
Mammary gland duct papilloma; dog. Papillas are shown on longitudinal section. Maspin-immunoreactive cells are seen in the papillomatous cores as large, polygonal cells (hypertrophic myoepithelial cell, HMEC-type) with a diffuse cytoplasmic staining pattern. Some scattered cells showed nuclear immunoreaction. ABC method, Harris's hematoxylin counterstain. Bar = 20 μm.
Benign mixed tumors (n = 6)
All four types of maspin-positive cells already described in complex adenomas and duct papillomas were seen in those areas of the tumors with a complex pattern of growth (Table 1). Thus, MECs were found in all cases forming a monolayer around luminal epithelial–type structures; immunoreactive products were homogeneous within the cell nucleus and cytoplasm; and SQS was +++ and IS was 3i. HMECs were observed in two cases, immunoreactive products were homogeneous, and mainly located within the cell cytoplasm, and SQS was ++ and IS was 2i. FMECs were found in five cases, and the immunostaining was homogeneous within the cell cytoplasm and sometimes in the nucleus, with an average SQS of ++ and an IS of 2i (Fig. 4). Finally, SMECs were found in five cases, immunoreactive products were homogeneous within the cell cytoplasm and rarely in the nucleus, and SQS was + and IS was 2i. In addition to these four types of maspin-positive cells, rounded MECs (RMECs) found in areas of early chondroid differentiation (n = 3) and chondroblasts (n = 3) (Fig. 5) were also stained (Table 1). RMECs were seen either beneath a peripheral band of FMECs or SMECs that surrounded the focus or intermingled with FMECs and SMECs within it. Their immunostaining was homogeneous within the cell cytoplasm, and SQS was + and IS was from 2i to 1i. Immunoreactive chondroblasts were usually located at the periphery of the cartilage focus, although sometimes they were found within it. The number of maspin-positive chondroblasts, as well as the intensity of staining, were low in the three cases (SQS and IS were + and 1i, respectively) and their pattern of staining was nuclear and/or cytoplasmic. One benign mixed tumor contained areas of bone, which was unreactive with the maspin antibody. In three of the benign mixed tumors, the epithelial cell layer showed focal immnunoreaction. These cells were located both lining neoplastic acini and in areas of squamous differentiation, the immunostaining was observed within the cell cytoplasm, and SQS was + and IS was 1i.
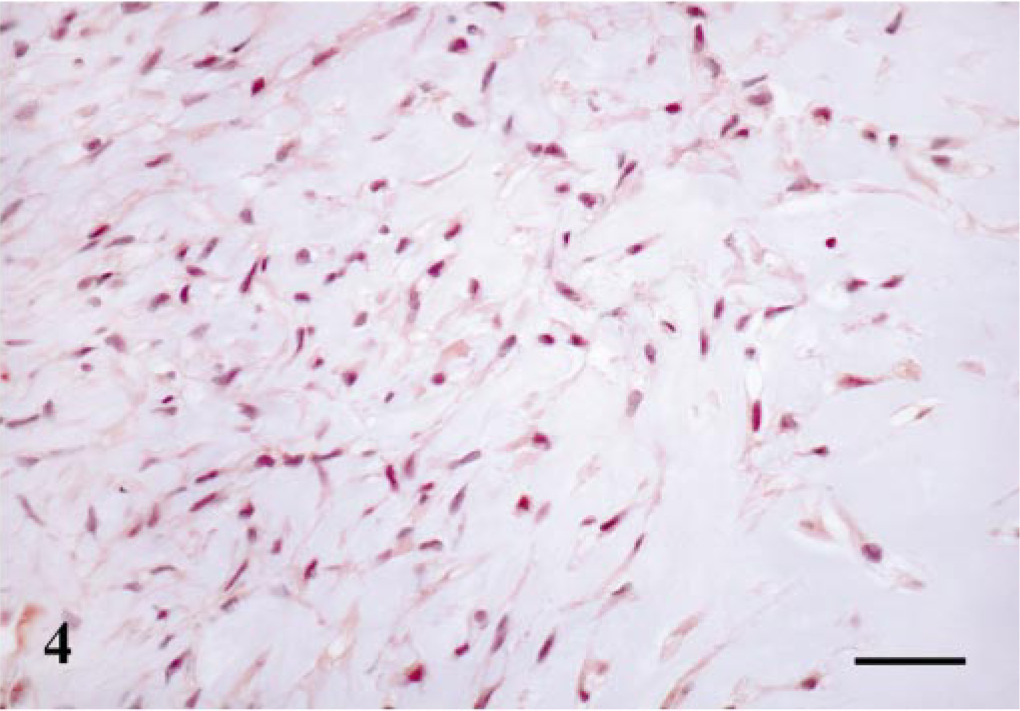
Mammary gland benign mixed tumor; dog. Fusiform (FMEC) and stellate (SMEC) myoepithelial cells in abundant matrix (early condroid differentiation area). Maspin-immunoreactive cells have a diffuse cytoplasmic staining pattern. ABC method, Harris's hematoxylin counterstain. Bar = 20 μm.
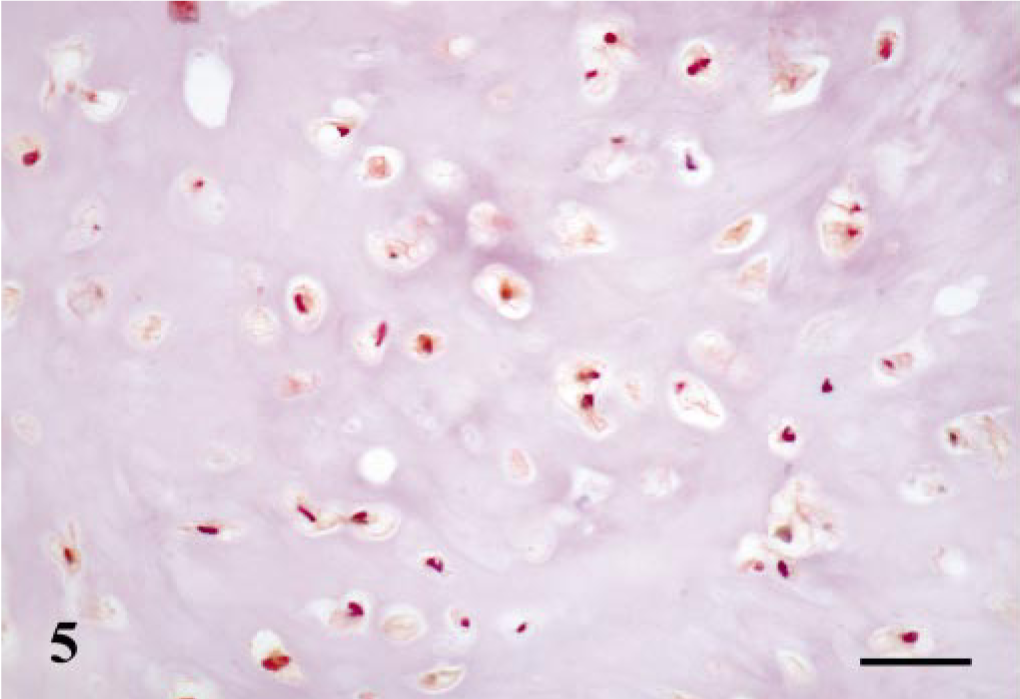
Mammary gland benign mixed tumor; dog. Well-differentiated cartilage. Maspin-immunoreactive products are observed within the cytoplasm of chondroblasts. ABC method, Harris's hematoxylin counterstain. Bar = 15 μm.
Malignant tumors (n = 30)
Tubulopapillary simple carcinomas (n = 6)
Maspin-immunoreactive cells were found in four cell types (Table 1). 1) MECs were observed in all cases (n = 6), and the staining was nuclear and cytoplasmic. SQS was +++ and IS was 3i. 2) Immunoreactive HMECs were identified in four cases, and the reaction was mainly located in the cytoplasm. SQS was +++ and the IS was variable (2i–3i). 3) Immunoreactive FMECs were observed in one case close to individual neoplastic tubules. The reaction was located in the cytoplasm. SQS was + and the IS was 1i. 4) Immunoreactive epithelial cells were observed in three cases. In two of them the number of positive cells was numerous, and in the third case the immnunoreactive cells were scarce. In all these three cases, the immunoreactive epithelial cells showed atypia or squamous differentiation and were identified in papillary growths within neoplastic ducts (Fig. 6) and in solid growths where these cells infiltrated into the connective tissue. In the epithelial cells, immunoreactive products were homogeneous within cytoplasm, SQS was + to ++, and IS was from 1i to 3i.
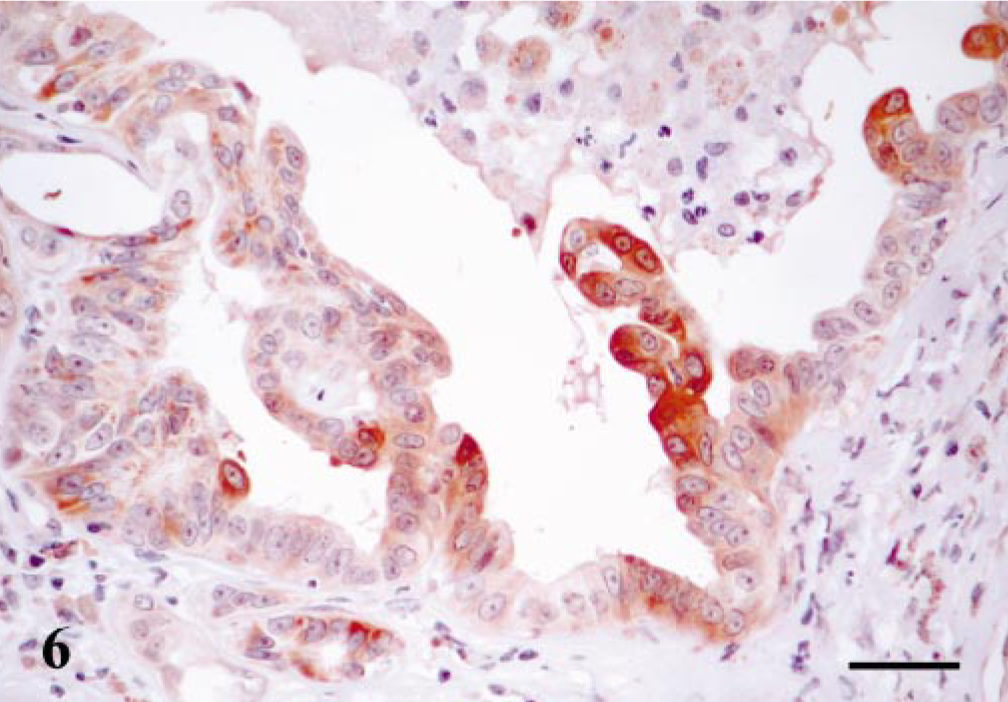
Mammary gland tubulopapillary simple carcinoma; dog. Neoplastic duct with papillary growths within its lumen. Maspin immunoreaction is observed in the cytoplasm of neoplastic epithelial cells, and some of them show atypia and squamous differentiation. ABC method, Harris's hematoxylin counterstain. Bar = 12 μm.
Solid simple carcinomas (n = 7)
Maspin-immunoreactive cells were found in two different locations: MECs and epithelial cells (Table 1). Immunoreactive MECs were found in five of seven tumors and appeared as complete or incomplete bands around solid nests of neoplastic epithelial cells. The immunostaining was nuclear and cytoplasmic, the SQS was +++, and the IS was 3i. Immunoreactive epithelial cells were found in five cases (Table 1); in three of them the number of cells was scarce and were located within the solid growths of neoplastic cells (with epithelial appearance in the HE-stained sections) or lining neoplastic tubules. In a fourth case, the immunoreaction was observed in epithelial cells both in solid growths and those located within lymphatic vessels constituting emboli of neoplastic cells. The intensity of reaction was stronger in the latter. Finally, the fifth case with positive epithelial cells showed immunoreaction in practically all cells. These cells were arranged in solid growths, without the limitation of a band of MECs (Fig. 7) or infiltrating within the connective tissue as anaplastic cells. The immunoreactive products for the epithelial cells in all these cases (n = 5) were located within the cytoplasm and, in addition, the fifth case showed some cells in the solid growths with nuclear reaction. Thus, SQS and IS for epithelial cells were very variable and ranged from + to +++ and from 1i to 3i, respectively.
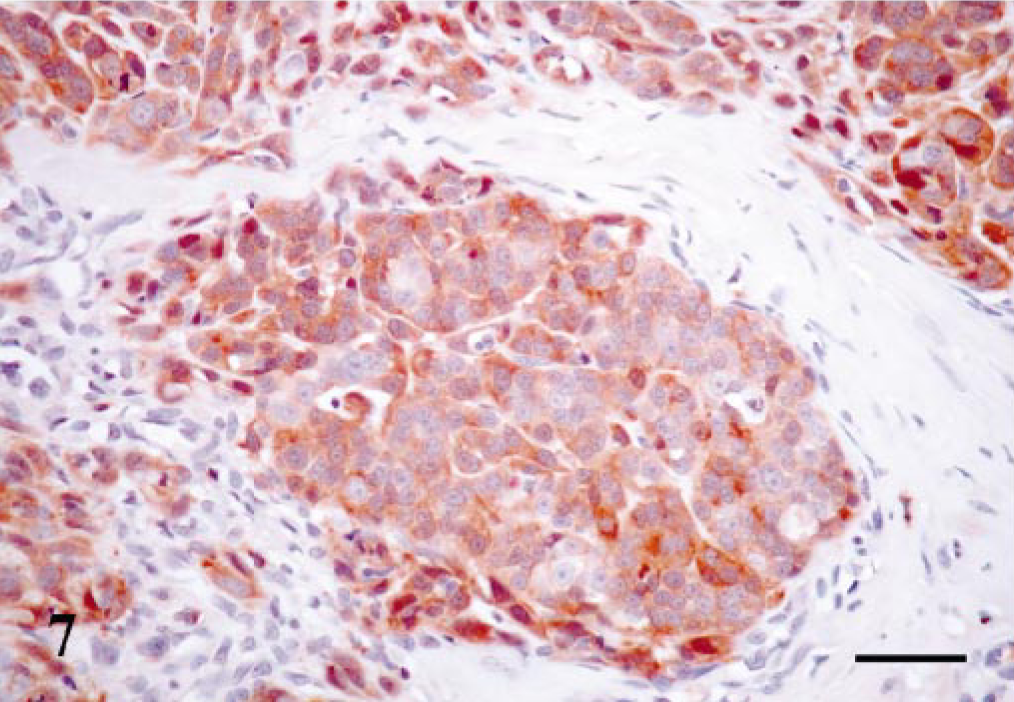
Mammary gland solid simple carcinoma; dog. Nest of neoplastic cells without the limitation of a band of MECs. All the cells within the nest show maspin immunoreaction in the cytoplasm. Some scattered cells showed nuclear immunoreaction. ABC method, Harris's hematoxylin counterstain. Bar = 15 μm.
Complex carcinomas (n = 9)
Types of cells reacting with the maspin antibody were the same as in complex adenomas, but their number and distribution were different (Table 1). Thus, MECs were found in all cases, and their immunostaining was both nuclear and cytoplasmic; the SQS ranged from ++ to +++, and the IS was variable from 1i to 3i. HMECs were found in eight out of nine cases and maspin immunoreactivity was mainly homogeneous within the cell cytoplasm, but some cells also showed nuclear reaction. SQS was ++ and IS was 2i. FMECs and SMECs were immunoreactive in eight out of nine cases, and their immunostaining was homogeneous within the cell cytoplasm. SQS was ++ for FMECs and + for SMECs, and IS was variable from 1i to 2i for both types of cells. Finally, epithelial cells showed immunoreaction in seven cases. These cells were located within the solid growths of neoplastic cells with the limitation of a band of MECs in some cases, lining neoplastic tubules or in papillary projections within these tubules. In all cases, the epithelial cells showed signs of atypia or squamous differentiation, and the immunoreactive products were located within the cytoplasm. SQS was + to +++ and IS was from 1i to 3i.
Carcinoma in benign mixed tumor (n = 8)
The pattern of staining was similar to that of their benign counterpart (Table 1). MECs and HMECs were observed in all cases, and the staining was nuclear and cytoplasmic for MECs and mainly located in the cytoplasm for HMECs. SQS was + to +++ and IS was from 1i to 3i for both types of cells. FMECs, SMECs, RMECs, and chondroblasts were immunoreactive in six cases, and all of them showed cytoplasmic reaction. Their SQS were + to ++ and IS varied from 1i to 2i. Osteoblasts were unreactive in the three tumors that contained areas of bone. Finally, epithelial cells showed immunoreaction in seven cases. These cells were located mainly in neoplastic tubules and in papillary projections within these tubules or, less frequently, within solid growths of neoplastic cells and in areas of squamous differentiation. In one case, immunoreactive epithelial cells, in addition to lining papillary projections, became spindle shaped and infiltrated adjacent connective tissue (Fig. 8). In all cases, the immunoreactive products were located within the cytoplasm. SQS was + to +++ and IS was from 1i to 3i.
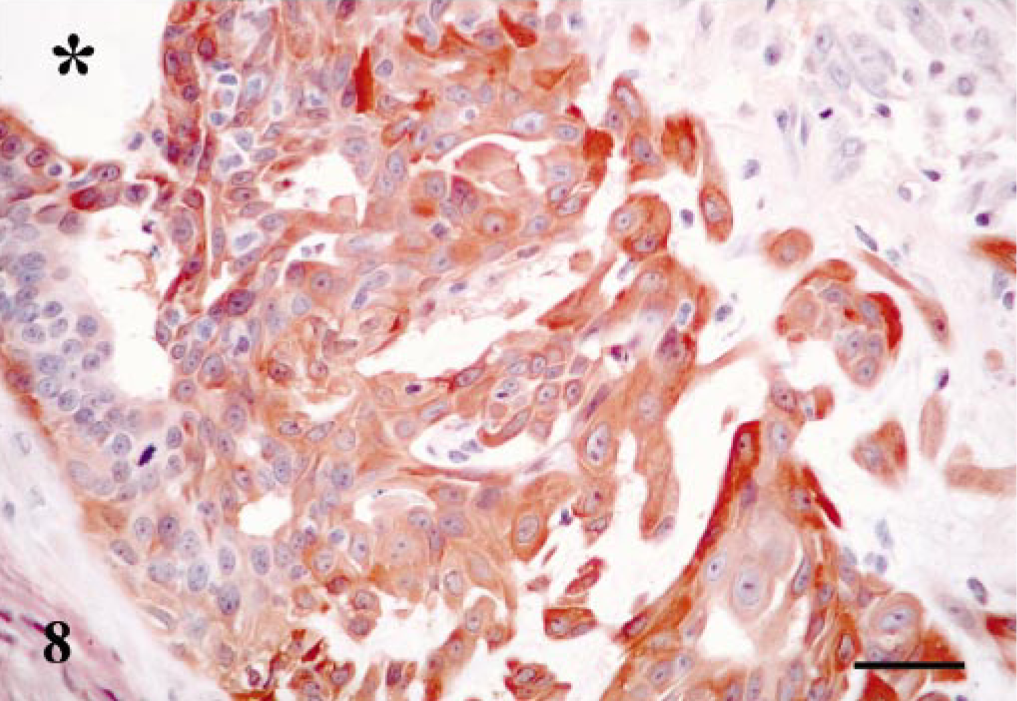
Mammary gland carcinoma in benign mixed tumor; dog. Neoplastic duct with cells infiltrating within connective tissue. Maspin-immunoreactive cells with luminal shape change the morphology to spindle shape cells and infiltrate from the lumen (star) to the connective tissue. Maspin-immunoreactive products are observed in cytoplasm. ABC method, Harris's hematoxylin counterstain. Bar = 15 μm.
DISCUSSION
Maspin is a unique serine protease inhibitor with a molecular weight of 42 kDa. It has been shown to inhibit tumor cell motility and invasion in cell culture and tumor growth and metastasis in animal models. 10, 12, 16, 20, 21 However, to date, the exact mechanisms underlying these effects of maspin and the overall schema of how maspin acts as a tumor-suppressor gene remain unknown. 10 Maspin is consistently expressed by mammary MECs, 14 a finding that was also observed in our study. Thus, periacinar and periductal MECs of all 40 normal mammary glands were diffusely stained by the anti-human maspin monoclonal antibody. In addition, maspin was found in 53 (98%) of the tumors studied, reacting with myoepithelal and epithelial cells.
In the myoepithelium compartment, immunoreactivity was observed in the MECs of 100% of benign and 93% of malignant tumors; the HMECs of 52% of benign tumors (33% of simple adenomas; 50% of complex adenomas; 100% of duct papillomas and 33% of benign mixed tumors) and 67% of malignant tumors (67% of tubulopapillary simple carcinomas; 0% of solid simple carcinomas; 89% of complex carcinomas and 100% of carcinomas in benign mixed tumors); the FMECs of 52% of benign tumors (0% of simple adenomas; 88% of complex adenomas; 20% of duct papillomas and 83% of benign mixed tumors) and 50% of malignant tumors (17% of tubulopapillary simple carcinomas; 0% of solid simple carcinomas; 89% of complex carcinomas and 75% of carcinomas in benign mixed tumors); the SMECs of 40% of benign tumors (0% of simple adenomas; 50% of complex adenomas; 20% of duct papillomas; and 83% of benign mixed tumors) and 47% of malignant tumors (0% of tubulopapillary simple carcinomas; 0% of solid simple carcinomas; 89% of complex carcinomas; and 75% of carcinomas in benign mixed tumors); the rounded (myoepithelial) cells of 50% of benign mixed tumors and 67% of carcinomas in benign mixed tumors; and the chondroblasts of 50% of benign mixed tumors and 78% of carcinomas in benign mixed tumors. These findings are in agreement with other studies 11–3 and support the hypothesis of a progressive transformation from MECs to fusiform and stellate cells in canine complex mammary tumors and from fusiform and stellate cells to rounded cells of early chondroid differentiation areas in canine mixed mammary tumors. They also suggest a possible transformation from any of the former to chondroblasts. MECs and HMECs observed in simple carcinomas corresponded to remnants of the preexisting MEC layer. Thus, maspin may be useful in the identification of in situ and microinvasive forms of canine mammary carcinomas, as already shown with other MEC markers such as calponin. 2 In the epithelium compartment, immunoreactivity with the maspin antibody was found in 16% of benign tumors and 73% of malignant tumors. Maspin-positive epithelial cells showed squamous differentiation in all benign tumors and in 59% of malignant tumors. Maspin has been detected in normal cutaneous structures of human beings, including the suprabasal, prickle, and granular cell layers of the epidermis. 15 In our study, skin samples included in mammary tissue sections showed cytoplasmic expression of maspin in supra-basal, prickle, and granular cell layers of the epidermis; cells of the germinative hair matrix, Henle's and Huxley's layers, and cuticle of hair follicles; mature sebaceous cells and sweat gland secretory cells. In addition, nuclear expression was detected in some basal/MECs of the sweat glands and scattered mature sebaceous cells. It has been suggested that all novel myoepithelial markers, including maspin and cytokeratin 14, stain squamous cell islands. 13 However, epithelial cells without squamous differentiation also reacted with the maspin antibody in malignant tumors exclusively. Maspin expression has also been described in epithelial cells of human breast carcinomas, 13, 14 but the significance of this finding remains to be elucidated. 5 Zhang et al. (1998) noted that the level of maspin expression was significantly higher in node-positive carcinomas (as we have observed in one of our cases of solid simple carcinoma) and lymphocyte-rich invasive ductal carcinomas. However, there was no correlation of maspin expression with histologic grade and estrogen receptors status. In another study, it was suggested that the expression of maspin in epithelial cells could be upregulated during the progression of ductal carcinoma and that it correlated with the acquisition of an aggressive phenotype. 19 Taken together, these findings contradict the proposed function of maspin as a tumor suppressor with properties of inhibition of invasion and metastasis of mammary carcinoma. 16, 18, 21 In our study, no differences were observed in maspin expression in atypical epithelial cells of simple, complex, and mixed carcinomas of the canine mammary gland.
Maspin immunoreactivity was detected in the nuclear and/or cytoplasmic compartments of different types of cells. Thus, we have observed that immuno-reactivity with anti-human maspin monoclonal antibody was located diffusely both in the nucleus and cytoplasm of periacinar and periductal MECs of the normal and neoplastic canine mammary gland. The nuclear reactivity progressively disappeared in the other types of MECs described (HMECs, FMECs, SMECs, RMECs, and chondroblasts) and was absent in all but one solid simple carcinoma that showed immunoreaction in practically all the cells. These cells had morphologic features of epithelial cells with HE staining and were arranged in solid growths, without the limitation of a band of MECs, or infiltrating within the connective tissue as anaplastic cells. The positivity was cytoplasmic, but some cells showed nuclear reaction. Because maspin is related to the serpin family of proteins, its expression was always regarded to be limited to the cytoplasmic compartment of the cell; however, Pemberton et al. (1997) first demonstrated, by western blotting and immunohistochemistry, that maspin can be detected in the nucleus of cancer cells. More recently, a few other investigators have documented the presence of nuclear maspin in human breast, ovary, prostate, skin, and pancreatic tumors. 6, 7, 10, 15, 17 Nuclear immunoreactivity observed with maspin antibodies is now considered specific, but the function and significance of this pattern of expression is unknown. 10 A recent study has shown that maspin nuclear staining in the MECs of human breast carcinomas was associated with good prognostic factors, whereas cytoplasmic staining was associated with poor prognostic markers. 10 However, the possibility exists that simple solid carcinomas (as defined on HE-stained tissue sections) showing maspin expression in “epithelial cells” are in fact simple solid MEC-type carcinomas.One disadvantage of antibodies to smooth muscle–restricted proteins used as MEC markers is that they also stain myofibroblasts (cells with a partial smooth muscle phenotype). 2 In the current series, maspin did not react with cells located in the stromal compartment of the tumors analyzed, which is specially useful in those cases where it is difficult to distinguish true MECs from stromal myofibroblasts lying just beneath the basement membrane.
In conclusion, maspin appears to be a very sensitive marker of normal and neoplastic myoepithelium in the canine mammary gland whose identification in different cell types of complex and mixed histologic types of tumors further supports a major histogenetic role for MECs. Neoplastic epithelial cells with squamous differentiation were also stained in both benign and malignant tumors, whereas stromal myofibroblasts did not react with the maspin antibody, which is an advantage over other MEC markers, particularly with respect to the identification of in situ and microinvasive forms of canine mammary carcinomas. Finally, a subset of epithelial cells without squamous differentiation also reacted with the maspin antibody in malignant tumors exclusively. The relationship between maspin expression in different cellular compartments of canine mammary carcinomas and the biologic aggressiveness of the disease remains to be elucidated.
Footnotes
Acknowledgements
This work has been supported by CVI-287, PAI, Consejería de Educación y Ciencia, and Junta de Andalucía. We thank A. Sosa for technical assistance.
