Abstract
Equine arytenoid chondritis causes airway obstruction and abnormal upper airway noise due to a space-occupying lesion(s) and decreased abduction. Our objective was to compare clinical scores and ultrasonographic findings with gross and microscopic lesions of naturally occurring arytenoid chondritis, in order to guide surgical treatment. Seventeen naturally affected horses with advanced/severe chronic arytenoid chondritis and 4 control arytenoid cartilages were evaluated after partial arytenoidectomy. Cartilages were sectioned caudal to the corniculate process and the body of each arytenoid was measured. We assessed total gross area (TA), percentage of viable cartilage (VC), percentage of viable cartilage on the lateral wall, and medial expansion. Retrospectively, the gross lesions were used to suggest 2 preferred surgical management (SM) groups: those requiring partial arytenoidectomy and those amendable to focal medial resection (a conservative SM). TA of horses with arytenoid chondritis was significantly larger than controls (P = .005), due to a layered lesion composed of cavitation, granulation tissue, fibrosis, inflammation, hemorrhage, and edema, with relatively equal medial and lateral expansion that distorted the geometry of the affected cartilage. The increased TA paralleled the presence of immature cartilage with disorganized primitive mesenchymal cells. TA and SM were positively correlated (P = .01). All cases showed varying degrees of cartilage degeneration or necrosis, more severe medially; those appearing amenable to focal medial resection arytenoid group had significantly more viable cartilage on the lateral wall (P = .02). The gross and histopathologic findings suggest a new surgical approach—focal medial resection—that may save the lateral wall of the arytenoid.
The equine rima glottidis is delimited by the epiglottis, 2 arytenoid cartilages (arytenoids), and vocal folds (left and right) covered by mucosa. Horses affected with arytenoid chondritis develop a progressive inflammatory process resulting in enlargement of one or both arytenoids resulting in restrictive breathing and reduced airflow. 22 Clinically, it has been referred to by a variety of names: equine arytenoid chondritis (arytenoid chondritis), arytenoid chondroma, laryngeal chondritis, or arytenoid chondropathy. The condition is more common in North America compared to Europe, 7 and is common in racehorses, 8 with reports of one pony being affected. 13 The pathogenesis of arytenoid chondritis is unclear, with the current hypothesis involving initial mucosal trauma due to forced closing of the larynx during rapid breathing. This injury introduces viruses or bacteria with subsequent inflammatory response and cartilage degradation. 6
Arytenoid chondritis may be asymptomatic in its mildest form or associated with a decreased performance. Currently, 3 clinical presentations of arytenoid chondritis are seen, affecting one or both arytenoids with markedly different clinical impact on performance, treatment, and outcomes. Most commonly, horses have endoscopic evidence of mucosal ulceration, no significant cartilage involvement, normal abduction, and no statistically significant impact on racing performance. 12 A second clinical category presents with an intraluminal mass constituted by granulation tissue on the medial surface of the arytenoid, with mild or no reduction in abduction, and mild to moderate decline in racing performance. 10 The third clinical category involves the body of the arytenoid cartilage leading to various forms of cartilaginous deformity (sometimes with abscessation), marked reduction in abduction, and severe decline in racing performance. 6 Horses in this study (with the exclusion of the control) were all in the third clinical group (hereinafter referred to as Group 3).
The diagnosis of arytenoid chondritis is obtained via endoscopy, with ultrasound used to better define the lateral extent of the disease. 7,8 The current standard of care for arytenoid chondritis in Group 3 horses, with cartilage deformity and marked abduction deficit, is partial arytenoidectomy, which is thought to help the horse return to racing at a preoperative level of performance. 18 However, another study showed that although horses can return to racing after partial arytenoidectomy, the level of performance is not as high and the career length is reduced compared to age-matched controls. 23 Previous reports suggest that the musculature surrounding the cartilage is mostly unaffected by the disease. 8 The current understanding of the microscopic lesions of this disease is largely based on the detailed description of 7 lesions, 10 and another report describing 2 lesions. 5
Our objective was to describe the gross and microscopic lesions in Group 3 horses with advanced laryngeal chondritis and compare these to the clinical assessment of disease severity. Our aim is that this information could be used to guide the surgeon to a more targeted excision or treatment (ie, focal excision vs standard partial arytenoidectomy) with the goal of restoring some abductory function and/or increasing the diameter of the rima glottides without losing lateral wall stability.
Materials and Methods
Study Design
Arytenoid cartilages had been surgically removed as therapy from horses with naturally occurring arytenoid chondritis with marked airway obstruction associated with arytenoid enlargement and reduction in movement. Written informed consent to surgery and sample processing was obtained from the owners. The cases were all in Group 3 (advanced disease) based on their clinical presentation. Histopathologic evaluation of the samples was conducted by 2 pathologists (PB and MD), blinded to the sample identity and clinical condition of the horses. A clinical scoring system was designed to assign a numerical grade to the clinical condition, and this was compared to the histologic findings and to the surgical management (Table 1). For the chondritis cases, retrospective data collection was performed, including age, breed, sex, lesion location, and clinical scores (Table 2 and Supplemental Table S1).
Scoring System for Clinical Evaluation of Horses With Arytenoid Chondritisa,b
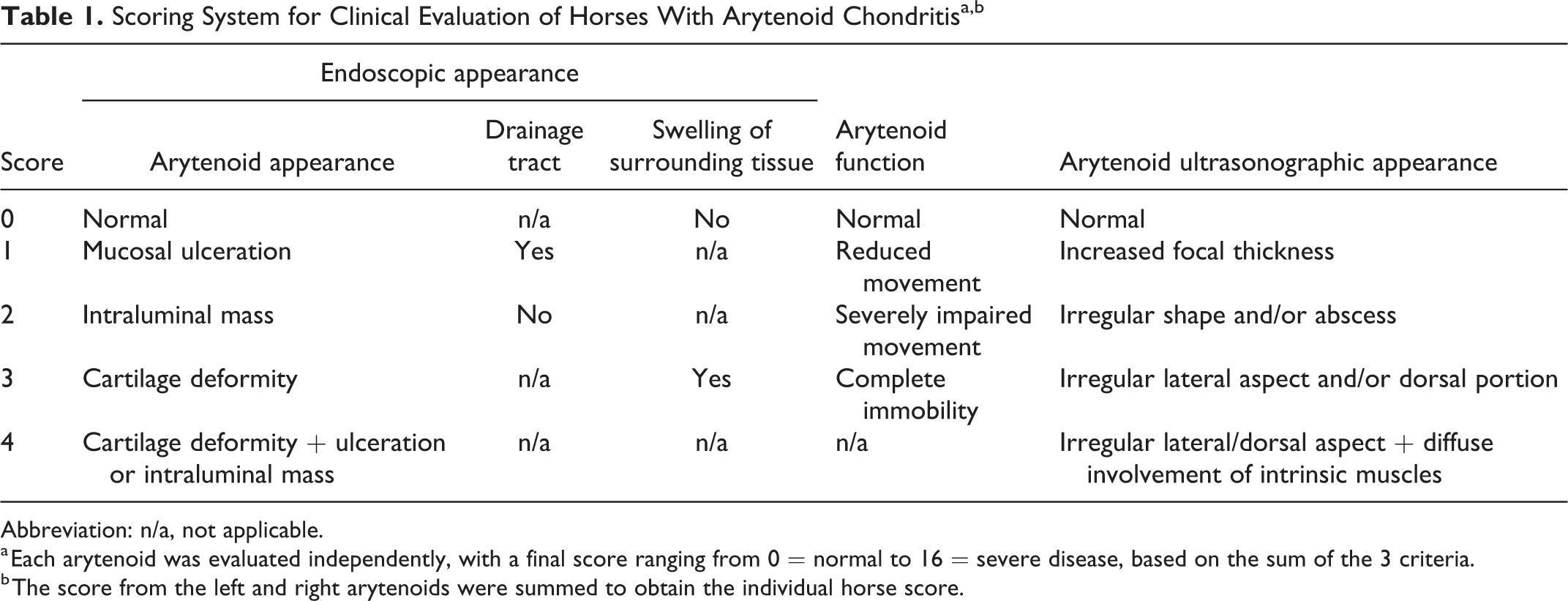
Abbreviation: n/a, not applicable.
a Each arytenoid was evaluated independently, with a final score ranging from 0 = normal to 16 = severe disease, based on the sum of the 3 criteria.
b The score from the left and right arytenoids were summed to obtain the individual horse score.
Subgross Measurements and Clinical Data From Horses With Advanced Naturally Occurring Arytenoid Chondritis (EAC) and Controls.
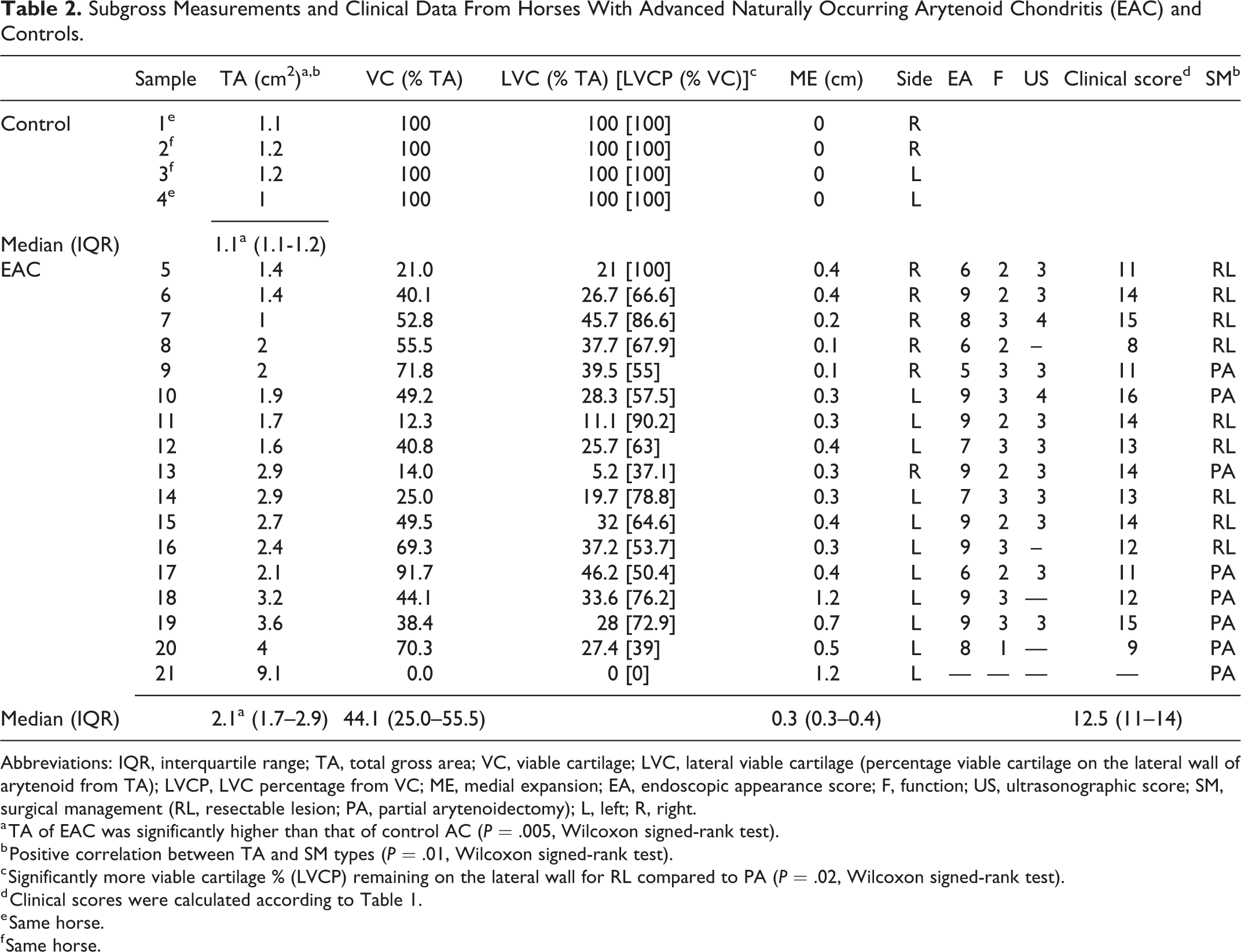
Abbreviations: IQR, interquartile range; TA, total gross area; VC, viable cartilage; LVC, lateral viable cartilage (percentage viable cartilage on the lateral wall of arytenoid from TA); LVCP, LVC percentage from VC; ME, medial expansion; EA, endoscopic appearance score; F, function; US, ultrasonographic score; SM, surgical management (RL, resectable lesion; PA, partial arytenoidectomy); L, left; R, right.
a TA of EAC was significantly higher than that of control AC (P = .005, Wilcoxon signed-rank test).
b Positive correlation between TA and SM types (P = .01, Wilcoxon signed-rank test).
c Significantly more viable cartilage % (LVCP) remaining on the lateral wall for RL compared to PA (P = .02, Wilcoxon signed-rank test).
d Clinical scores were calculated according to Table 1.
e Same horse.
f Same horse.
Sample Processing
Arytenoid cartilages (n = 17) surgically removed for treatment of advanced chondritis at the Cornell Equine Hospital between 2008 and 2016 were included in the study. Arytenoids of 2 unaffected horses, euthanized for reasons unrelated to this study, served as control (2 left and 2 right arytenoids). At the time of collection, all samples were fixed in 10% neutral buffered formalin and later shipped to the Pathology Department of Ross University School of Veterinary Medicine (RUSVM). Gross specimens were sectioned at the same level, caudal to the corniculate process, to include the rostral part of the arytenoids usually removed surgically during the partial arytenoidectomy procedure (Supplemental Fig. S1). The sectioned arytenoid samples (about 5 mm thick) were scanned using an Epson Perfection V850 scanner, including a scale for every image (Supplemental Fig. S2).
Following trimming and routine processing, arytenoid samples were stained with hematoxylin and eosin (HE), safranin O, Alcian blue, and Gram (Brown & Brenn and Brown-Hopps). The safranin O–stained slides were scanned (referred to as subgross measurements) using the same high-resolution scanner mentioned before, and all samples were analyzed histologically. Digital images (Supplemental Fig. S3) were then transferred to a computer linked with a conventional light microscope (Olympus BX 43) and equipped with an Olympus DP 26 digital camera and the cellSens Standard image analysis software for measurements.
Clinical Scores
The clinical scoring system (Table 1) was designed based on the author’s (NGD) clinical experience on most important characteristics to evaluate from a surgical point of view. For each horse, the system assigned points to each arytenoid based on 3 main criteria: endoscopic appearance, function/mobility, and ultrasound appearance. Each criterion was scored from 0 to a maximum of 4. The endoscopic appearance criterion included 3 subcriteria: arytenoid appearance, presence of a draining tract, and swelling of the peri-arytenoid structures. Based on the scoring system, an arytenoid with normal structure on endoscopy and ultrasound, normal function, and no abnormalities of the surrounding tissues would have a total score of 0. A maximum score of 16 would be attributed to arytenoids with structural deformity, intraluminal defect, signs of infection such as abscess, lesions of the surrounding tissues, and complete immobility. As each arytenoid was evaluated independently, a single horse with bilateral disease could score higher than 16, and up to 32 if the disease was severe in both arytenoids.
The score, as designed here, was applied retrospectively to the clinical presentation of the horses whose arytenoids were assessed histologically for the study. Images from the presurgical endoscopic and ultrasonographic examinations were recovered and a consensus clinical score (Table 2) was assigned by two of the authors (MC and NGD), blinded to the histopathologic findings of the corresponding specimens.
Proposed Relevance to Surgical Management
All horses had the same surgical management consisting of standard partial arytenoidectomy, with resection of the entire arytenoid cartilage except the muscular process. 20 However, the results of this study lead to the hypothesis that removing only the medial wall in selected cases of arytenoid chondritis (ie, focal medial wall resection) may resolve the infectious process and improve the laryngeal diameter by both removing the physical enlargement associated with the disease and also allowing some degree of abduction. Indeed, preserving the lateral wall (cartilage) structure should allow the action of the muscular attachment (cricoarytenoid dorsalis and lateralis muscles) and thus is likely to preserve the respiratory and digestive functions of the larynx. To refine the hypothesis, the gross appearance (presence and severity of enlargement and status of the adjacent cricoarytenoid lateral muscle) of the lateral wall was reviewed for each case (Supplemental Fig. S3, Supplemental Fig. S6, and Table 2) and compared to its histologic findings. The refined hypothesis was the following: horses with arytenoid chondritis are amenable to the less invasive surgical procedure of focal medial wall resection if the arytenoid cartilage enlargement from the chondritis is confined to the medial surface of the cartilage, and have normal appearance of the intrinsic laryngeal muscles. In contrast, those cases requiring partial arytenoidectomy have severely enlarged arytenoid cartilage, abaxial bulging deformation of the lateral wall, and diffuse muscular involvement. 20 Given the morbidity of partial arytenoidectomy, we have been proceeding with removing the medial wall of a small number horse with chondritis. Further clinical experience is needed to see if this is a valid hypothesis.
Long-Term Outcome
Complete race records of the thoroughbred horses in the study population were obtained from an online database (equibase.com). Median earning over 3 races before surgery and 3 and 5 races after surgery were calculated. Return to racing, time to first race after treatment, and median earnings (in US dollars) before and after surgery were determined for each horse.
Gross and Subgross Quantification of Lesion Severity
Lesion severity was assessed at the gross and subgross levels by measuring the total cartilage area, percent viable cartilage, and medial expansion. Viable cartilage was defined based on the presence of safranin O staining. Medial expansion was defined as the extent of cartilage extending medially from the overlapped averaged controls size and shape (Supplemental Fig. S6). All dimensions were measured in pixels, performed in triplicate on both rostral and caudal sides of the arytenoid section, averaged and converted into metric units based on the scale bar used during the scanning process. Using the digitalized scanned gross images and the cellSens software, we measured the total gross area of arytenoids in cm2, based on grossly identifiable cartilage tissue (excluding nearby skeletal muscle, glands, or epithelial tissue). This was easier to achieve for the controls, where the cartilage was clearly outlined. For the samples from horses with arytenoid chondritis, total gross area was measured to the best of our ability and included areas of cartilage loss or sinus tracts (Supplemental Fig. S4). As a next step, using the safranin O–stained slides, we outlined the areas of visible viable cartilage (red and shades of red/orange; Supplemental Fig. S5). For viable cartilage, the total area of viable cartilage in cm2 was calculated and transformed into percentage (%) of the total gross area (considered 100%).
To verify the premise for an alternative surgical approach, 2 additional cartilage measurements were calculated focusing on the lateral wall of each arytenoid: lateral viable cartilage (LVC) and lateral viable cartilage percentage (LVCP). For these, we measured the visible viable cartilage of the lateral wall and expressed LVC as the percentage of total gross area represented by viable cartilage on the lateral wall, while LVCP was the percentage of the overall viable cartilage present on the lateral wall. We speculated that surgical management might differ based on the proportion of remaining viable cartilage present on the lateral wall of the arytenoid (LVCP).
ME was calculated from scanned gross images. A reference outline was created of the shape (wedge) of the cross section of the area of interest of the arytenoids (only cartilage) averaged between control animals. Cross sections of the area of interest of the arytenoids from arytenoid chondritis samples were superimposed over the reference outline. The most dorsal and most ventral recognizable cartilage areas for each specimen profile were used as points of reference (Supplemental Fig. S6). Thereafter, 5 measurements were taken (top, top-middle, middle, low-middle, and low) of the medial side of the arytenoids, with the points of origin maintained at the same sites for all 17 specimens. These measurements extended from the reference outline to the outer most recognizable cartilage area. They were averaged to give the medial expansion in centimeters.
Histopathology
The following histological changes were recorded for arytenoid chondritis cases: epithelial hyperplasia of the overlying mucosa on the medial aspect, ulceration, cartilage degeneration and/or necrosis, cavitation in the arytenoids, evidence of presence of hemorrhage and/or hemosiderin (free or within macrophages), presence of granulation tissue and/or fibrosis, presence of immature cartilage with disorganized mesenchymal cells associated with cartilaginous matrix, inflammation (type, severity and extent, using the ordinal method for grading 9 ), muscle involvement, vascular changes, thrombosis, and osseous metaplasia. Cartilage degeneration was defined as loss of safranin O staining. Cartilage necrosis was defined as also having necrotic cells or loss of matrix and cells. Any additional histological changes were described but not used in the statistical analysis.
Statistical Analysis
Correlations between each gross measurement (total gross area, percent viable cartilage, and medial expansion), and between each of these and the age, clinical score, and endoscopic appearance score were assessed using Spearman’s correlation coefficient. Association of gross measurements (total gross area, viable cartilage, medial expansion, and LVCP), clinical score, endoscopy score, proposed surgical management, and age with histological changes were tested using Wilcoxon signed-rank test. Performance before and after partial arytenoidectomy were compared using paired Wilcoxon test. All calculations and statistical tests were done in Excel (Microsoft) and R software (R Development Core Team, 2010).
Results
Clinical Findings
Arytenoids affected by advanced chondritis were collected from 17 horses (Table 2 and Supplemental Table S1). Data from one arytenoid could not be retrieved due to mislabeling at the time of collection; consequently, clinical data were available from only 16 horses. The remaining population included 15 Thoroughbred horses and 1 Quarter horse. Disease duration could be accurately determined by clinical history in 7 horses, and it ranged between 3 weeks and 5 months. Twelve horses at the time of hospitalization were evaluated by endoscopy and ultrasound, the other 4 horses only via endoscopy.
Arytenoid chondritis affected only the left side (n = 4), only the right side (n = 1), or bilateral but more severe on the left or right (n = 11, with various degrees of abnormality on the contralateral arytenoid). Eleven horses presented other upper airway abnormalities in addition to arytenoid chondritis, including recurrent laryngeal neuropathy, permanent dorsal displacement of the soft palate, subepiglottic ulcer, epiglottic entrapment, and fourth branchial arch defects. One horse had both perilaryngeal abscess and subepiglottic entrapment; another horse had both permanent dorsal displacement of the soft palate and epiglottic entrapment. Five horses received other treatments for the chondritis before undergoing the arytenoidectomy. The clinical signs were of 3 weeks to 5 months duration and included mainly respiratory noise, cough while eating or exercising, and labored breathing. The arytenoid chondritis had developed following laryngoprosthesis surgery in 2 horses (horses 12 and 7). Bacterial culture was performed in 6 cases from a portion of the removed arytenoid or the associated abscess.
Clinical Scores
The clinical scores calculated for the arytenoids ranged from 8 to 16 (16 was the maximal unilateral score), with a median value of 13, and only 2 arytenoids scoring <10. The contralateral arytenoid score had a median value of 1.5 (range 0–12), with only 3 arytenoids scoring >6.
On endoscopy, 15/16 arytenoids presented structural deformity (score ≥3), associated with intraluminal ulceration or mass in 11 cases. Severely impaired function or complete immobility (score ≥2) was recorded in 15/16 cases. No significant correlation was found between preexisting recurrent laryngeal neuropathy and impaired arytenoid function in these cases. All 9 cases without concurrent signs of recurrent laryngeal neuropathy had severely impaired function or complete immobility (score ≥2); of 7 cases with concurrent signs of recurrent laryngeal neuropathy, 4 had complete immobility. Ultrasonographic scores were assigned to 12 arytenoids and all scored ≥3/4, showing altered shape and an irregular lateral border or deformity extending dorsally toward the muscular process. Only 2 arytenoids showed signs on ultrasound that were compatible with diffuse severe inflammation of the intrinsic muscles, in the form of fluid collection and heterogenicity.
Long-Term Outcome
Long-term performance follow-up was available for 12 thoroughbred horses (Supplemental Table S1). Four horses never returned to race after surgery (one of these was concurrently affected by a musculoskeletal problem). Of the 8 that returned to racing, after surgery 2 were retired after a single race. The other 6 horses (50% of those with data) had successful race records after surgery. These 6 horses had minimal involvement of the contralateral arytenoid, with a median contralateral clinical score of 1 (range 1–6), while the 6 horses that were retired at the time or soon after the arytenoidectomy had a more significant lesions also of the contralateral arytenoid, with a median clinical score of 6 (range 0–12). The time to first race after surgery ranged from 69 to 281 days (median 175 days), with the longest intervals recorded in the 2 horses that never raced before surgery (mean 239 ± 42 days). Total earnings prior to surgery ranged from $4862 to 101 498 (median $55 217), while after surgery the range was $12 162 to $164 480 (median $23 445).
Gross and Subgross Lesions
Grossly, diseases arytenoids in this study were enlarged, deformed, and some had erosions or ulcers on the mucosal surface. On sectioning the formalin-fixed specimens caudal to the corniculate process, we noticed arytenoid cartilage deformity and irregularity, with various degrees of cartilage loss, and presence of central areas of necrosis and cavitation (Fig. 1). The measurements of total gross area, percent viable cartilage and medial expansion are summarized in Table 2. The total gross area of the control arytenoids (median: 1.1 cm2, interquartile range [IQR]: 1.1-1.2 cm2) was significantly lower (P = .005, Wilcoxon signed-rank test) than that of the arytenoid chondritis cases (median: 2.1 cm2, IQR: 1.7–2.9 cm2). The total gross area and proposed surgical management types were positively associated (P = .01, Wilcoxon signed-rank test); that is, arytenoids suggested to be suitable for focal medial resection had a significantly lower total gross area compared to arytenoids for which partial arytenoidectomy was suggested to be required based on the gross findings.
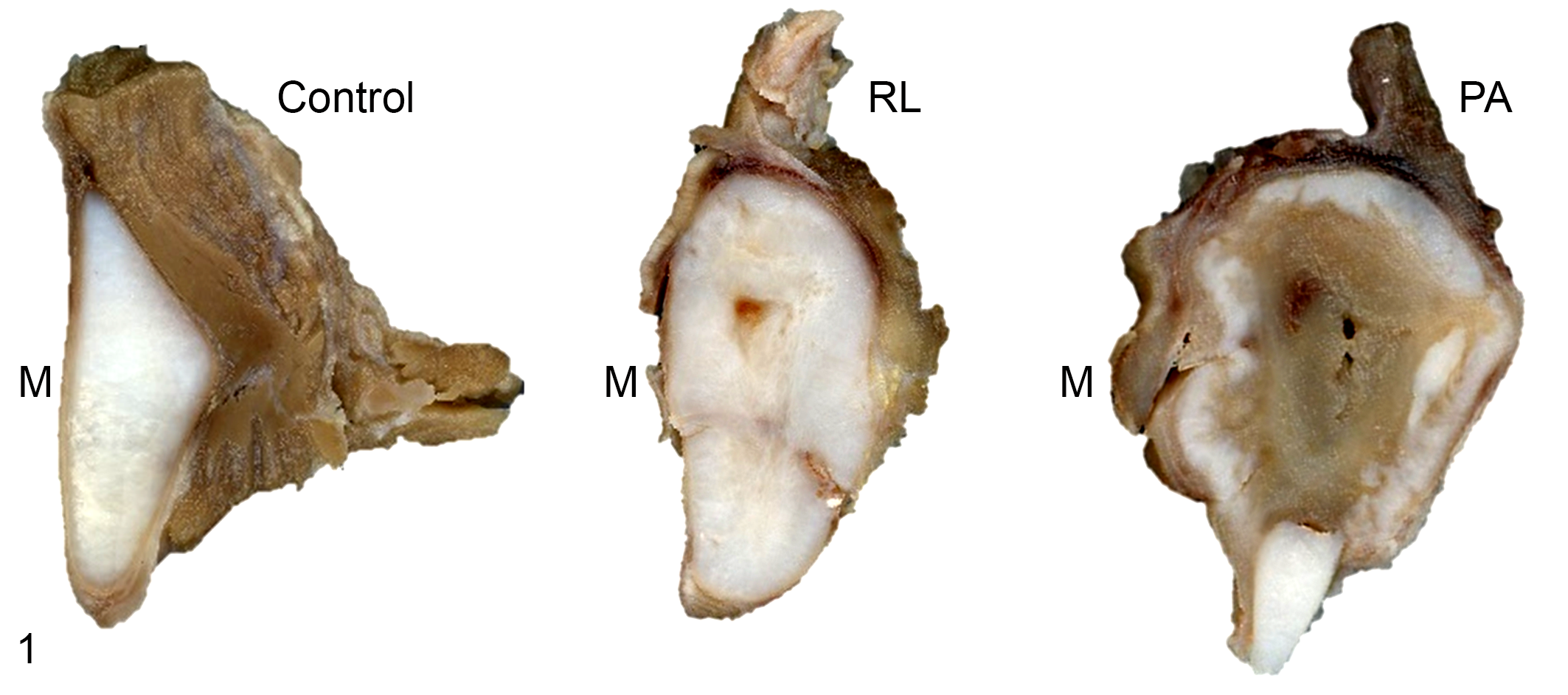
Equine arytenoid chondritis, larynx, horse. Formalin-fixed specimens. Scanned gross arytenoids from 1 control (case 4) and 2 chondritis cases: case 15, categorized as a possible candidate for resectable lesions (RL), and case 13, categorized as requiring partial arytenoidectomy (PA). Diseased arytenoid cartilages are enlarged, with irregular contour, medial expansion of cartilage, areas of cartilage degeneration, necrosis, replacement by fibrosis, and central cavitation. M, medial aspect.
Cartilage loss was present in 16/17 arytenoid chondritis cases. In these, the median percent viable cartilage was 44.1% (IQR 25–55.5%), whereas all control cases had percent viable cartilage values of 100%. Moreover, when comparing the medial to lateral distribution, we noted that most of the remaining viable cartilage was located laterally. LVCP was significantly higher in cases suggested to be suitable for focal medial resection compared to cases for which partial arytenoidectomy was suggested to be required based on the gross findings (median of 67.9% and 52.7, respectively, P = .02, Wilcoxon signed-rank test).
Medial expansion of various extents (median of 0.3 cm, IQR 0.3–0.4 cm) was present in all arytenoid chondritis cases, including the ones that had a total gross area within the range of the controls. Medial expansion was larger than lateral extension when the arytenoid chondritis samples were overlapped to the normal arytenoid shape of the control samples (Supplemental Fig. S6). One case (case 21) had the largest total gross area, 0% viable cartilage and the largest medial expansion.
We investigated potential associations between age, endoscopic appearance score, and clinical score; gross and subgross measurements (total gross area, percent viable cartilage, and medial expansion); and the main histological parameters (presence or absence of inflammation, muscle involvement, epithelial hyperplasia, hemorrhage or hemosiderin, cavitation, immature cartilage with primitive mesenchymal cells, ulceration, and bone metaplasia). Statistically, the presence of epithelial hyperplasia was associated with percent viable cartilage (P = .02, Wilcoxon signed-rank test), medial expansion (P = .02, Wilcoxon signed-rank test), and endoscopic appearance score (P = .0512, Spearman’s rank correlation test). ME was associated with presence of immature cartilage containing primitive mesenchymal cells, although not statistically significant (P = .08, Wilcoxon signed-rank test).
The clinical score and percent viable cartilage were negatively correlated, although not statistically significant (P = .06, Spearman’s rank correlation test), suggesting that lower amounts of viable cartilage might correspond to a high clinical score. Endoscopic appearance score was negatively correlated with age, although not statistically significant (P = .057, Spearman’s rank correlation test).
Histopathology
No microscopic abnormalities were observed in the control arytenoids. All samples from horses with arytenoid chondritis had multiple lesions. Even though bacteria are suspected to be incriminated in the pathogenesis of arytenoid chondritis, none of the histologic sections (stained with HE, Warthin-Starry, and Gram stains) had detectable bacterial colonies in the tissues or any overt infectious process. The most common lesions, affecting all cases, were layers of cartilage degeneration or necrosis and the presence of granulation tissue and fibrosis (Table 3).
Arytenoid Histopathology in 17 Horses With Advanced, Naturally Occurring Equine Arytenoid Chondritis.
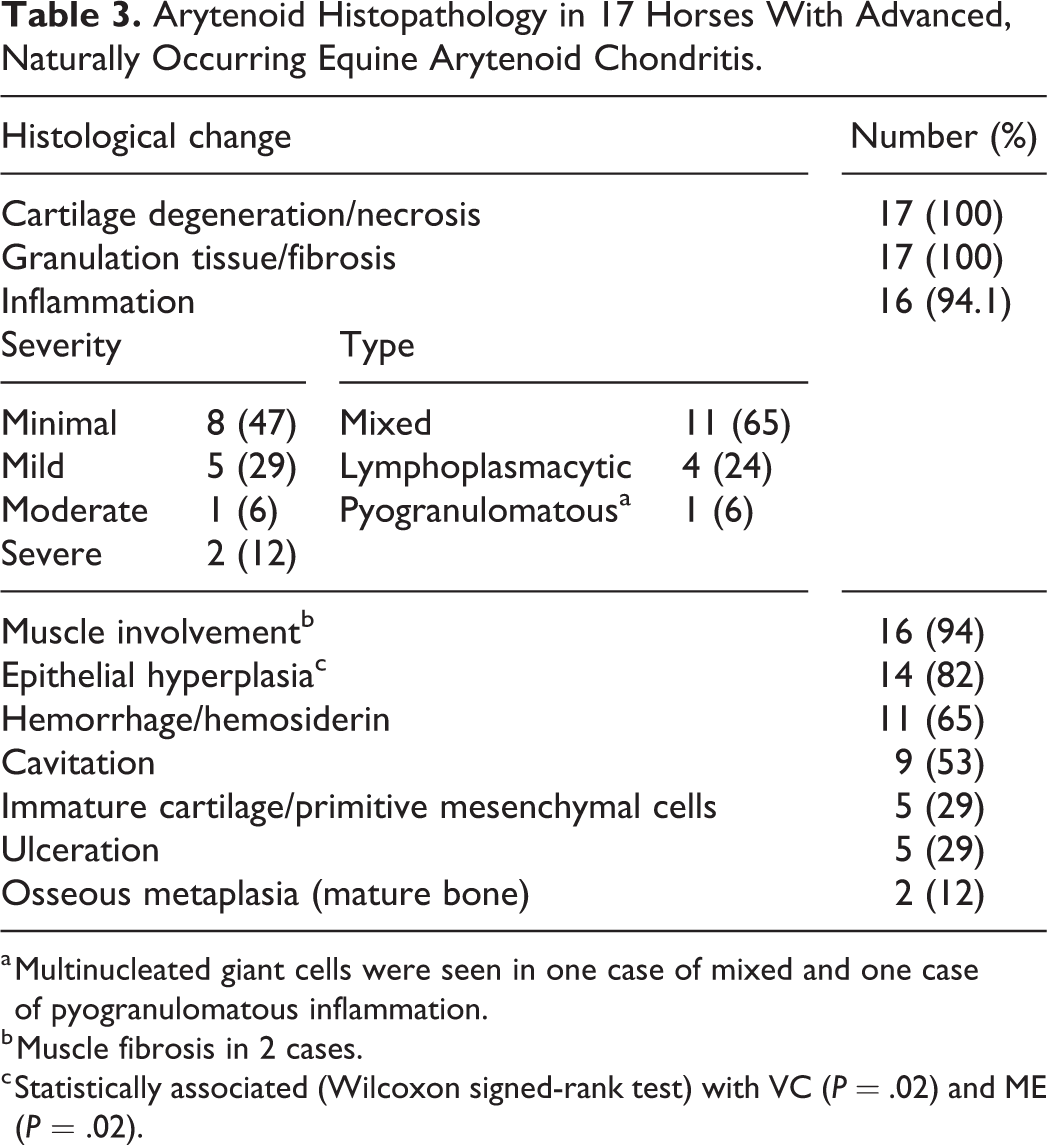
a Multinucleated giant cells were seen in one case of mixed and one case of pyogranulomatous inflammation.
b Muscle fibrosis in 2 cases.
c Statistically associated (Wilcoxon signed-rank test) with VC (P = .02) and ME (P = .02).
On safranin O stain, red to dark orange staining was observed only for viable cartilage and not the fibrosis or granulation tissue (Fig. 2). The viable cartilage in arytenoid chondritis, when present, was variably sized and shaped, whereas in the controls it was uniformly sized and shaped (Supplemental Fig. S3). Viable cartilage was bordered by areas of various degrees of cartilage degeneration, necrosis, or loss. Progressing from viable hyaline cartilage to areas of granulation tissue, fibrosis or inflammation, this cartilage lesion was characterized by progressively paler matrix staining (Figs. 2, 3, and 7) and decreased amounts of matrix, with small, shrunken chondrocyte nuclei (pyknosis) or empty lacunae. The cartilage degeneration and necrosis appeared to initiate at the level of chondrocytes and their territorial matrix, followed by degeneration of the interterritorial matrix with dissolution or decrease of glycosaminoglycans. The cartilage loss was more severe medially than laterally (Table 2). In less severe cases, lesions were confined to perivascular regions of the diseased arytenoids. The blood vessels were surrounded by a clear space (edema) with various degrees of multifocal cartilage degeneration and sometimes fibrosis without inflammation. In some areas, the interterritorial cartilage matrix was finely granular, surrounding degenerating or necrotic chondrocytes and their pale blue-gray degenerating territorial matrix on safranin O stain (Fig. 3); cartilage pallor (loss of matrix proteoglycans) or eosinophilia was visible in HE-stained sections.
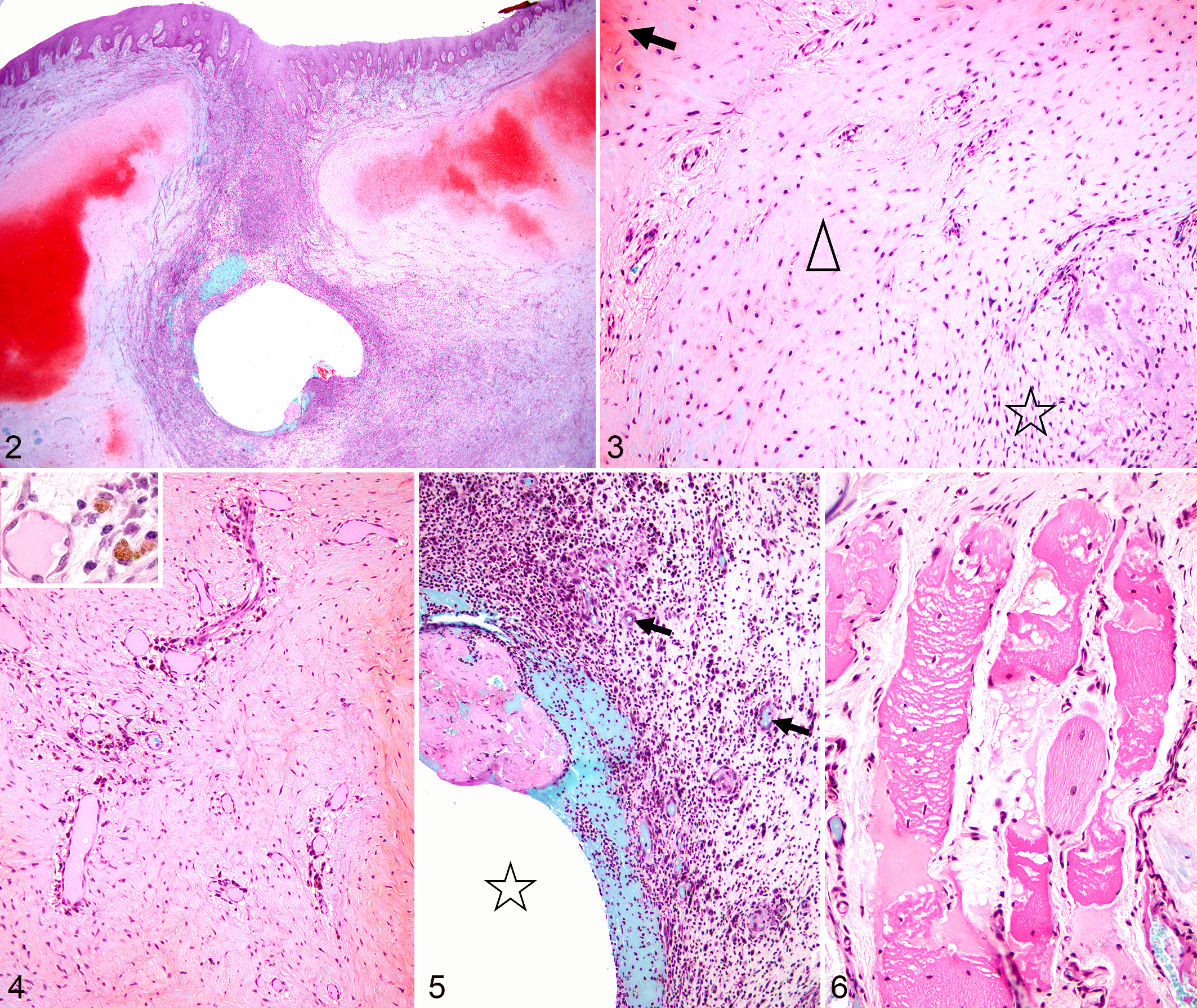
Equine arytenoid chondritis, larynx, horse.
Equine arytenoid chondritis, larynx, horse.
Multifocally in some cases, the arytenoid cartilage was dissected by fibrosis and/or inflammation into smaller irregular islands (Fig. 2). In 5 cases, this area of transition between the cartilage and the fibrous connective tissue and/or inflammation also contained cartilage remodeling that appeared as immature cartilaginous matrix admixed with primitive mesenchymal cells. In the peripheral areas of arytenoid chondritis, chondrocytes were spindle-shaped, unevenly spaced and haphazardly oriented, and were interspersed with areas of stellate primitive mesenchymal cells (Fig. 4).
Inflammatory cell infiltrates were seen in 16 of 17 cases, in various locations, mainly involving the lamina propria or extending into the nearby laryngeal muscles, but also surrounding the necrotic/cavitated area in the center of the arytenoid cartilage (Fig. 5). The inflammatory response was mixed in 11 cases (various combinations of neutrophils, macrophages, lymphocytes, and plasma cells), lymphoplasmacytic in 4 cases, and pyogranulomatous in 1 case. The severity of the inflammatory response was minimal (n = 8), mild (n = 5), moderate (n = 1), or severe (n = 2). The inflammation extended from the lateral aspect of the arytenoid into the adjacent muscle in 16 of 17 cases, accompanied by various degrees of myocyte degeneration, vacuolation, necrosis or loss, and fibrosis (Fig. 6).
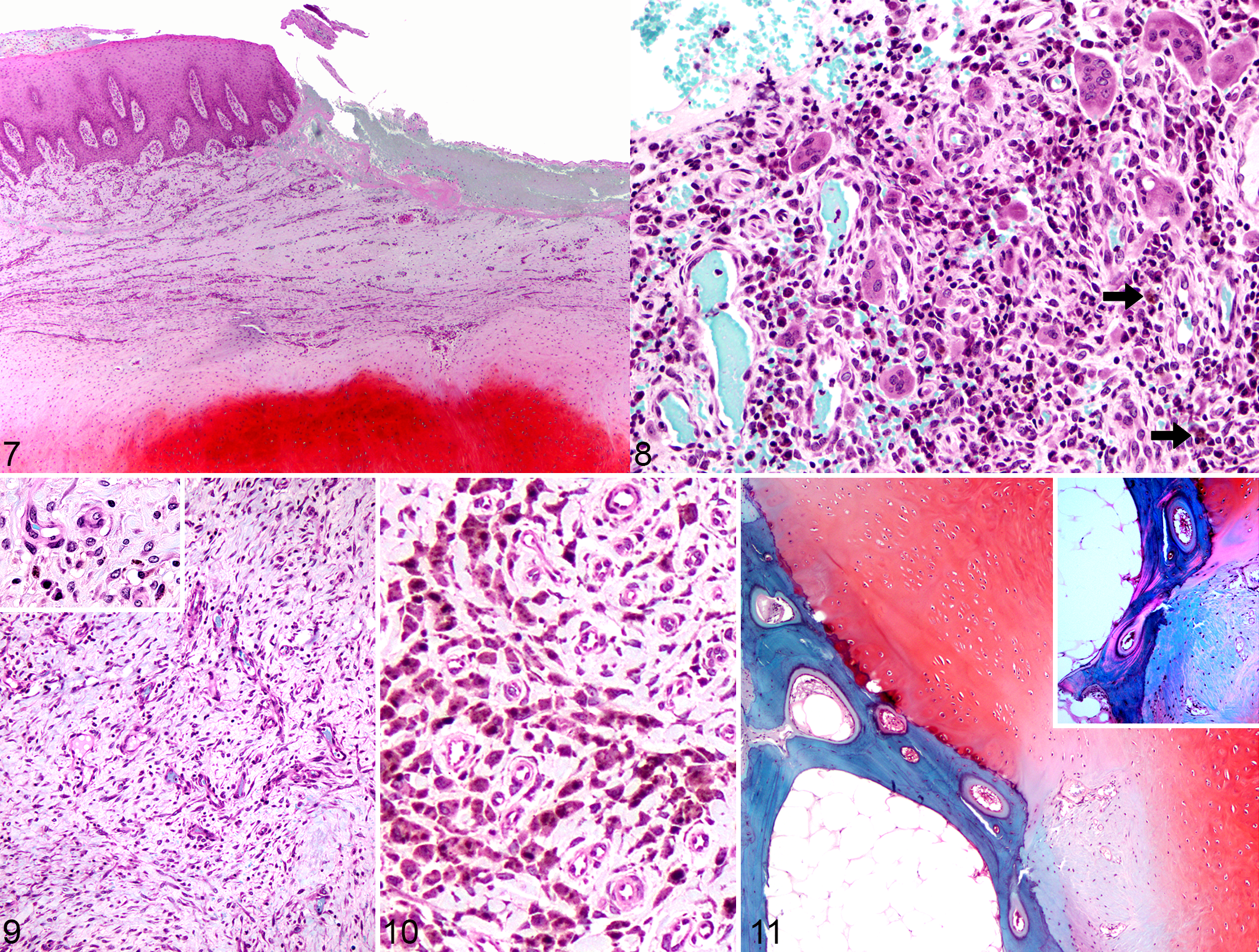
On the medial (luminal) surface of the arytenoids, epithelial hyperplasia was present in 14 of 17 cases and was associated with focal or multifocal erosion and/or ulceration (Fig. 7) in 5 cases. The lamina propria ranged from normal to being expanded by granulation tissue and fibrosis (all 17 cases) or infiltrated by mixed inflammatory cells (n = 16), that sometimes involved submucosal glands and sometimes included multinucleated giant cells (n = 2, Fig. 8), lymphangiectasis (n = 1), or marked perivascular edema (n = 1).
In 13/17 cases, sinus tracts connected the diseased arytenoids with the laryngeal lumen. In 9 of these, there was a central area of arytenoid cartilage necrosis and cavitation. The sinus tracts were surrounded by fibrosis or granulation tissue (Fig. 9), and less consistently by inflammation. Fibrin, hemorrhage, edema, and inflammatory cells (predominantly neutrophils and macrophages) surrounded the central necrotic areas in some cases. In more advanced cases, fibrous connective tissue at the margin of the sinus tract was variably infiltrated by inflammatory cells (pyogranulomatous, lymphoplasmacytic, or mixed inflammation), and sometimes hemosiderin-laden macrophages. Overall, hemorrhage and/or hemosiderin-laden macrophages (Fig. 10) were present in 11 cases. No vascular changes or thrombosis were observed.
Osseous metaplasia was characterized by mature lamellar bone that was birefringent under polarized light with marrow between the lamellae (Fig. 11). This was present on the lateral aspect of the arytenoids, close to the muscular attachment, in 2 cases: a 4-year-old female thoroughbred with a clinical duration of several weeks, and a 7-year-old female with a clinical duration of 2 to 4 months. Walls of arterial blood vessels in 5 cases (age range 2–8, median 3 years old) seemed expanded, thicker, folded, more tortuous, and had subintimal proliferation of a paler basophilic matrix.
Discussion
The results of this study should be interpreted with the consideration that only horses with obstructive enlargement and/or decrease abduction of the arytenoid were included in the study. Advanced arytenoid chondritis is considered by some authors the most debilitating upper respiratory tract disease in racehorses because treatments are not as consistently successful as those of recurrent laryngeal neuropathy. 23 The purpose of this study was to correlate clinical, gross, and histological features of arytenoid chondritis, and to identify parameters that could be further evaluated to guide the treatment of horses with severe arytenoid chondritis.
Grossly, the disease can be recognized based on laryngeal asymmetry, enlargement, erosions or ulcers, granulation tissue, or mucopurulent drainage. 1,8 Cartilage loss, increased total area and medial expansion, as described and evaluated in this study, represent important contributors to clinical disease in arytenoid chondritis. The significant deformation of arytenoid cartilages was previously evaluated in a report on arytenoid chondritis in 7 horses, involving decrease in length and height with increase in width 10 . We found that the larger size of diseased arytenoids, as measured by the medial expansion, paralleled the presence of immature cartilage with primitive mesenchymal cells. Thus, we consider that this immature cartilage and primitive cells are therefore a significant component of the remodeling and the increased in size of arytenoids. One sample in our study (case 21) was statistically an outlier and it was included in the analysis as we had a large enough number of arytenoid chondritis cases and was considered to represent biological variation.
We investigated the histopathologic changes in advanced cases of arytenoid chondritis, and their correlation with the gross features and the amount of normal and abnormal cartilage in their arytenoids. The enlargement reported in the literature is usually a reflection of endoscopic or ultrasound assessment. 2,10 In this study, a more representative view of the diseased cartilage is described. We attributed the increase in size of arytenoids to the presence of immature cartilage and primitive cells as well as various combinations and severities of cavitation, granulation tissue and fibrosis, inflammation, hemorrhage, and edema. It appears that once lesions extend through the medial cartilage into the core of the arytenoids, the combination of cartilage degeneration, necrosis, loss, and fibrosis present a mass effect, which results in the deformation of the medial and lateral wall of the arytenoid. Multifocal cartilage degeneration or necrosis affected all cases in our study, with an average loss of cartilage of over 50% in our cases. Importantly, cartilage degeneration and loss seem to progress from medial to lateral, possibly allowing for more conservative surgical approaches in the future (ie, focal medial resection). We observed that cartilage degeneration and necrosis seem to start at the level of chondrocytes and their territorial matrix followed by degeneration of the interterritorial matrix with dissolution or decrease of glycosaminoglycans. In HE-stained sections, this is characterized by loss of basophilia, with cartilage eosinophilia, 17 whereas with safranin O stain, cartilage degeneration is apparent as progressively paler red staining. The predominant and consistent presence of cartilage degeneration, necrosis, and loss among advanced arytenoid chondritis lesions suggests that this is principally a degenerative disease rather than infectious or inflammatory.
The role of inflammation in the pathogenesis of arytenoid chondritis is uncertain. Inflammatory conditions of cartilage are known to result in remodeling of extracellular matrix by chondrocytes, 14 as observed in this study. Previous studies reported that auricular chondritis is an immune-mediated disease as it may be induced in mice and rats following immunization with native type II collagen. 4,15 The current study of equine arytenoid chondritis shows that inflammation does not predominate in every case, and suggests that the inflammatory response develops secondarily to or in response to cartilage injury and degeneration. The findings may suggest that the inflammation in equine arytenoid chondritis was part of the repair process or resulted from secondary infections accompanying sinus tracts and ulceration. The limitation is that we cannot tell which pathological process (inflammation or degeneration) comes first in a cross-sectional study.
The presence of multinucleated giant cells in the arytenoid chondritis lesions of our study could best be explained by the presence of necrotic host cartilage requiring further degradation. Alternatively, their presence could be instigated by persistent pathogens 19 or foreign material (ingesta), which were not identified in our cases. An overt infectious process was not apparent histologically in our 17 arytenoid chondritis cases, although according to the clinical histories a single or a combination of the following bacteria were isolated from 6 horses: Actinobacillus spp. (2), Fusobacterium necrophorum (2), Klebsiella oxytoca (1), Streptococcus zooepidemicus (Supplemental Table S1). Neutrophils were variably present in our arytenoid chondritis lesions and could reflect a response to cartilage necrosis or possible previous bacterial infection. The absence of bacterial colonies in tissues with arytenoid chondritis could be explained by lesion chronicity, previous antibiotic treatment, or insensitivity of histology with the HE, Warthin Starry, and Gram stains. A previous report demonstrated mixed bacterial colonies in lesions of arytenoid chondritis from 2 of 3 horses examined, suggesting a significant involvement of bacteria in perpetuating and/or exacerbating the inflammatory process. 5 This would be somewhat similar to feedlot cattle with acute laryngitis from bacterial infections, where coughing and swallowing are thought to contribute to swelling and formation of contact ulcers on the medial aspect of the arytenoids. 11
Many of our arytenoid chondritis specimens presented degenerate cartilage converging centrally within a layer of viable cartilage, leading to a cavitated lesion in the most advanced cases. Most recently, Garrett et al described an ultrasonographically apparent layered appearance of arytenoid chondritis with central fibrosis, surrounded by hyaline-like cartilage. 8 We believe that the cavitation seen grossly and histologically in our cases may correspond to ultrasound lesions in which gas within tissue appear as small multiple hyperechogenic foci, similar to the appearance of gas in abscesses. 8 Areas of cartilage cavitation and replacement with connective tissue were previously interpreted as fibrotic centers of an abscess. 5 However, the lack of arytenoid abscesses in the present study suggests that cavitation is more likely a consequence of cartilage degeneration and necrosis. A possible explanation for the large size of one arytenoid chondritis case in our study (#21) is that complete loss of cartilage may allow granulation tissue and fibrosis to expand peripherally, assuming that viable cartilage acts as a mechanical barrier.
Epithelial hyperplasia correlated with loss of viable cartilage as well as with medial expansion in our study. These findings suggest that epithelial hyperplasia indicates more severe arytenoid chondritis lesions. Epithelial hyperplasia may be endoscopically evident as a roughened mucosal surface and further study of its value as a prognostic factor would seem justified.
A previous histopathologic study of 7 arytenoid chondritis cases showed marked fibrous connective tissue lamination and immature fibrocartilage in all cases, focal osseous metaplasia (considered incidental), and irregular bands of eosinophilic cartilaginous matrix at the junctional zone in 4 cases, and granulating sinus tracts in 3 cases. Another case had fibrosis, with cartilaginous and osseous metaplasia of the nearby cricoarytenoideus dorsalis muscle. 10 Presence of mature bone (osseous metaplasia) in two of our cases might be due to the chronicity of the lesion, based on the abrupt transition from viable cartilage to bone. Ossification of the laryngeal cartilages (cricoid, thyroid, and arytenoid) was previously reported in a horse, and histologically described as trabecular bone with minimal osteoblastic activity separated by adipose and hematopoietic tissue. 21 The previous reports of osseous metaplasia in arytenoid chondritis were believed to be incidental finding, and most likely associated with normal aging changes of equine laryngeal cartilages. 10 In our study, the 2 cases with osseous metaplasia associated with arytenoid chondritis were 4 and 7 years old; thus, we attributed this change to chronic inflammation and disease duration rather than age. Osseous metaplasia is thought to arise from fibroblasts differentiating into osteoblasts in areas of chronic inflammation or tissue damage, or in neoplastic processes following release of bone morphogenetic proteins by neoplastic cells. 16 The presence of tortuous, irregular arteries with subintimal proliferation in 5/17 cases in our study was considered an incidental finding, probably unrelated to arytenoid chondritis.
The structural medial expansion is the obvious component of the airway obstruction to clinicians; however, lateral extension of the arytenoid seems to worsen the clinical condition by restricting abduction, leading to the need for partial arytenoidectomy (unpublished observation, NGD and MC). The clinical scoring system that we developed found a correspondence between loss of arytenoid function and alteration of the lateral aspect of the arytenoid on ultrasound examination. This supports that ultrasound evaluation needs to be a consistent part of the clinical evaluation in cases of chondritis, with a particular attention to the smoothness and shape of the lateral border of the cartilage. Based on the scores recorded in these advanced cases, we hypothesize that a total score ≥6 and an ultrasound score ≥3 represents a poor prognostic indicator for return to function or response to medical treatment. Moreover, the histopathologic findings in cases with ultrasonographic score ≤2 and with gas within tissue previously attributed to abscess might indicate a focal debridement as therapy. The validity of these interpretations requires further prospective studies on application of the scoring system.
If one could intervene surgically to remove the medial wall of the arytenoid before the mass effect has deformed the lateral wall, then restoration of some cross-sectional area of the rima glottidis may be possible because native abduction could be restored. Clinically, the absence of significant abduction diagnosed endoscopically and enlargement or deformation detected on ultrasound have dictated partial arytenoidectomy as the treatment of choice for arytenoid chondritis. However, based on gross and histologic findings, we identified 9 of our 17 cases which retrospectively might have been amendable to excision of only the medial wall of the body of the arytenoid. We speculate that if the medial abnormal cartilage could be removed, leaving the framework of lateral normal cartilage including the muscular process, then return to full function might be possible. If the core lesion size could be better estimated with ultrasound, as well as the viability of the surrounding cartilage, a focused debridement of the degenerated cartilage and cavitated lesion could be attempted, followed by filling of the defect with a cartilaginous graft. A cartilaginous graft has the potential to integrate within the native tissue, inducing cartilage remodeling and repair of structural defects within the arytenoid. 3 This kind of regenerative approach might help restoring the cartilage integrity, but the arytenoid function depends also on the condition of the intrinsic laryngeal muscles and absence of involvement of the cricoarytenoid articulation. On clinical evaluation, 94% of the arytenoids in this case series showed severely compromised mobility. In these cases, the only common concurrent ultrasonographic feature was diffuse abnormality of the lateral surface of the arytenoid, which had rough edges, was broadly convex, had a cartilage/muscle interface that was thickened and echoic (data not shown). These lateral changes extended dorsally in most of the arytenoid chondritis and seemed to involve also the muscular process. More than a primary muscle dysfunction, impaired abduction seems to be related to the abnormal lateral shape and increased size of the arytenoid that represents a mechanical restriction to reach adequate abduction. Finally, restriction of abduction is likely to be proportional to the medial enlargement of the arytenoid rather than neuromuscular involvement. Limitations to the validity of such conclusions are the lack of histology of the cricoarytenoideus dorsalis muscle, as well as the retrospective nature of the ultrasonographic evaluation.
In conclusion, we describe the main subgross and histological features of advanced arytenoid chondritis. The most important and consistent features of arytenoid chondritis are varying degrees and combinations of increased arytenoid cartilage size, degeneration and loss of cartilage, replacement with fibrosis, inflammation, and involvement of muscle on the lateral side. The findings suggest that it may be possible to remove only the medial wall in some cases, which would offer the possibility of improving the cross-sectional diameter of the airway despite some degree of restriction in abduction. This remains to be validated by futures studies using ultrasound and endoscopy in combination. Improvement in abduction would be most likely if the focal medial resection procedure were done prior to lateral bulging of the arytenoid cartilage and extension into the adjacent musculature. It is also possible that some of the arytenoid cartilage inflammation results from cartilage necrosis and the subsequent reparative or immune response rather than an infectious cause.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Bolfa_et_al - Clinical and Pathological Features in Horses With Advanced Arytenoid Chondritis
Supplemental Material, Combined_supplemental_materials-Bolfa_et_al for Clinical and Pathological Features in Horses With Advanced Arytenoid Chondritis by Pompei Bolfa, Marta Cercone, Michelle M. Dennis, Anne Conan, Bernard Grevemeyer and Norm G. Ducharme in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to RUSVM Research Center “One Health Center for Zoonoses and Tropical Veterinary Medicine” for funding, to master Paul Orchard “the Photoshop wizard,” to Mr David Hilchie, RUSVM Histology lab manager, and to Dr Lusan DellaGrotte for their technical support and assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RUSVM Research Center “One Health Center for Zoonoses and Tropical Veterinary Medicine.”
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
