Abstract
Laminitis in equids is a clinical syndrome usually associated with systemic disease. Endocrinopathies recently have been recognized as the most common cause of laminitis, with hyperinsulinemia playing a key role. Descriptions of laminitis-associated lesions have been confusing due to the wide range of experimental models used, failure of adequate clinical documentation for naturally occurring cases, lack of separate analysis of inflammatory and endocrinopathic laminitis, and uncertainty regarding normal morphological variation of lamellae. In this study, lamellar morphology and pathology were described in 14 laminitic horses and ponies that had hyperinsulinemia (>20 mIU/l), with reference to 25 age- and breed-matched controls. The type and severity of lesions noted had no correlation with reported clinical duration and in at least some cases must have preceded it. Lesions were largely localized abaxially within the lamellar tissue and included apoptotic cell death, as well as lamellar fusion, hyperplasia, and partial replacement with aberrant keratin containing nucleated debris and proteinaceous lakes. The lesions resulted in irregular margins between the inner horn and the lamellar tissue. Acute separation originated from the abaxial region, with minimal associated inflammation. Axially, epidermal lamellar tapering was the most frequent morphological observation. The lesions in these chronic cases of laminitis were similar to those described in some inflammatory laminitis models and contained features seen in developmental phases of hyperinsulinemic models. These findings support the theory that repeated episodes of subclinical laminitis occur prior to clinical presentation. In addition, the pathology does not include extensive basement membrane failure seen in some inflammatory models.
Laminitis is a common, debilitating condition of Equidae that has significant welfare and economic consequences. It is a clinical syndrome rather than a morphologic diagnosis and usually occurs in association with systemic disease. 1,8,16,17,34 Less frequently, it is associated with mechanical overloading, for example, due to lameness in the contralateral limb. 36 Systemic diseases that result in laminitis are either severe inflammatory conditions (often with concurrent gastrointestinal disturbances) or endocrine diseases (endocrinopathies). Until recently, endocrinopathic laminitis was underrecognized; however, a growing body of evidence now indicates its predominance in naturally occurring cases of laminitis in both Europe and the United States. 12,21 Naturally occurring endocrine diseases associated with laminitis include equine metabolic syndrome and pituitary pars intermedia dysfunction (PPID). 12,21 Equine metabolic syndrome affects young to middle-aged horses and ponies and is characterized by hyperinsulinemia and insulin resistance, with obesity as a major risk factor. 5 Equine PPID is a disease of aged horses and ponies characterized by overproduction of pituitary hormones, including adrenocorticotropin (ACTH) due to pars intermedia hyperplasia or neoplasia; basal ACTH concentration is commonly measured to diagnose that condition. 27,28
The pathology of laminitis associated specifically with hyperinsulinemia was first described in 2010. 2 Experimental ponies in which hyperinsulinemia (mean insulin >1000 mIU/l) was induced developed the disease approximately 55 hours later. 2,22 In contrast to the previously described (and frequently referenced) lesions in some cases of inflammatory laminitis, the hyperinsulinemic cases showed lack of widespread basement membrane disintegration/separation with prominence of epidermal apoptosis and mitotic activity, as well as minimal neutrophilic infiltration. 2,9,22 A time-series analysis of the lamellae of hyperinsulinemic experimental horses that were not allowed to progress to laminitis (ie, the preclinical, developmental phase) showed the initial lesions to be secondary epidermal lamellar (SEL) elongation and increased apoptosis (6 hours) followed by progression in severity and (reactive) proliferation over the next 18 hours. 11 Similar lesions were seen in horses exposed to hyperglycemia without insulin infusion (mean endogenous insulin 208 ± 26.1 mIU/l). 10
The lesions of naturally occurring laminitis in horses and ponies with hyperinsulinemia and/or PPID have not been described. Unfortunately, the few histological descriptions for naturally occurring laminitis have lacked detailed clinical information. 19,25,26,31 Additional challenges for the pathologist include distinguishing pathological changes in lamellar tissue from normal morphological variation (which may potentially be age or breed related) and lack of knowledge regarding preclinical/developmental phase lesions vs clinical phase lesions in the natural disease. Recently, a classification system for the morphology of primary and secondary epidermal lamellae (PEL and SEL) in healthy horses was proposed and described; however, some of the animals had been euthanized due to systemic inflammatory disease (eg, colic, pleuropneumonia, and placental retention). 23
The aim of this study was to describe the histomorphometry and pathological lesions in hoof lamellar tissue of horses and ponies with naturally occurring endocrinopathic laminitis. All measurements and descriptions were made with reference to control tissue from young (age <12 years) and old (age ≥12 years) horses and ponies.
Materials and Methods
Case Selection and Clinical History
The experimental protocol was approved by the Research Ethics Committee of the University of Helsinki, Finland. A total of 59 horses or ponies were included that presented to the Equine Teaching Hospital, University of Helsinki, Finland, or the Philip Leverhulme Equine Hospital, University of Liverpool, United Kingdom, for euthanasia or were humanely killed at local stables or abattoirs between March 2010 and September 2013. In addition, 4 archived samples from a previous study (control ponies) were used. 2 All animals were older than 1 year and had no current evidence or a known history of systemic inflammatory disease. Body condition was scored for all the horses using the Carroll and Huntington 4 0 to 5 body condition score (BCS). Laminitis was diagnosed if there was lameness involving both fore or all 4 hooves associated with weight shifting, a typical “sawhorse” stance, and/or reluctance to move. The onset of laminitis was recorded as the date it was first detected by the owner or the treating veterinarian. Consistent clinical signs included increased digital arterial pulses and sensitivity to hoof tester pressure applied at the toe of the affected foot/feet. Hoof alterations inclusive of divergent rings (horizontal lines seen in the outer hoof wall that are wider at the heel than the toe; Fig. 1) and flat or convex soles were also recorded as clinical signs. Radiographs were taken (where clinically indicated) to confirm evidence of lamellar disruption, including sinking or rotation of the distal phalanx (DP). Following euthanasia, the right forehoof was bisected sagittally and examined for evidence of laminitis, including further inspection of the outer hoof wall for divergent rings and lack of parallel orientation of the dorsal surface of the DP and the overlying hoof wall.

Hyperinsulinemic laminitis, hoof, Equus caballus. Rings on the outer hoof wall of a horse that had a reported duration of laminitis of 548 days. These were noted in 13 of 14 of the laminitic animals. The rings are divergent (ie, the distance between them is wider at the heel [arrows]).
Endocrinopathic laminitis was defined as a clinical disease in horses or ponies that had elevated basal insulin (>20 mIU/l), with or without a concurrent elevated (seasonally adjusted) basal plasma ACTH concentration (>29 pg/ml from November to July and >47 pg/ml from August to October). 7,15 Animals were placed in the following groups based on their clinical and endocrine criteria (Table 1): Young (age <12 years) and old (age ≥12 years) control horses, young (age <12 years) and old (age ≥12 years) control ponies, and ponies and horses with endocrinopathic laminitis. Animals were excluded if they did not meet the criteria for any group, had pyrexia or illness, or had a recent history of gastrointestinal disease. Also excluded were horses and ponies with elevated basal ACTH and no laminitis (n = 11) and laminitic horses and ponies with normal or unmeasured endocrine values (n = 7). In addition, 1 horse was excluded because of fever and another because of suspicion of (iatrogenic) corticosteroid-induced laminitis.
Clinical and Laboratory Selection Criteria for Different Groups.
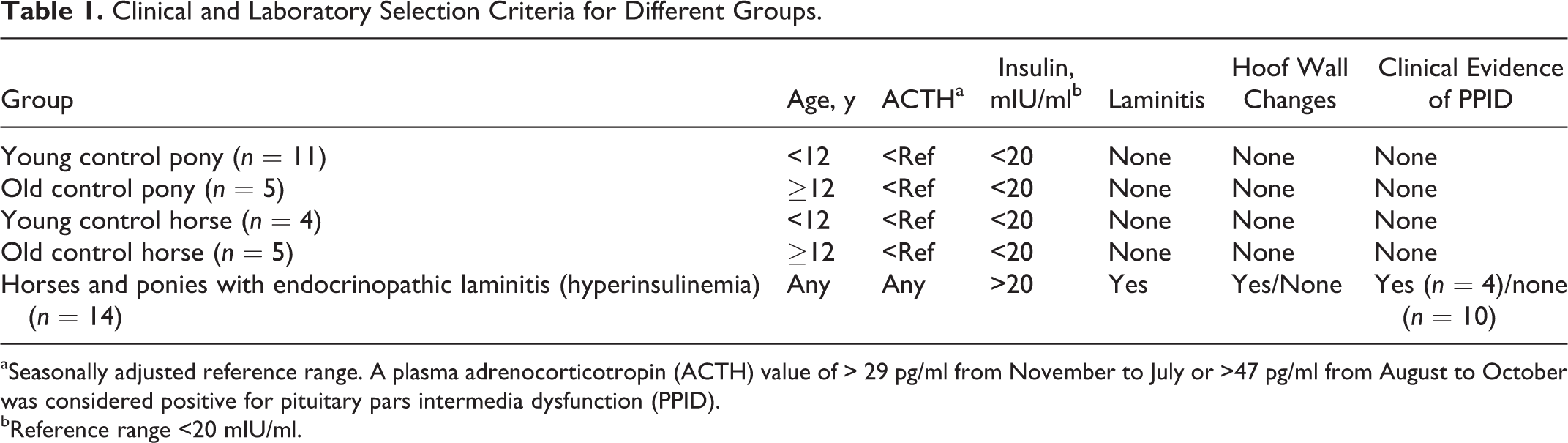
aSeasonally adjusted reference range. A plasma adrenocorticotropin (ACTH) value of > 29 pg/ml from November to July or >47 pg/ml from August to October was considered positive for pituitary pars intermedia dysfunction (PPID).
bReference range <20 mIU/ml.
Laboratory Assays
Blood samples for insulin concentration were collected in 6-ml serum sample tubes following at least 4 hours of fasting (Vacuette, 2-serum clot activator; Greiner bio-one, Kremsmünster, Austria). Insulin analysis was performed using a chemiluminescent immunoassay (Immulite; Liphook Equine Hospital, Liphook, UK). The samples were centrifuged and the serum was separated within 3 hours, frozen to –20°C, and sent by courier to the laboratory by next-day delivery. Blood samples for ACTH concentration were collected in 10-ml plastic EDTA tubes (Vacuette, EDTA K3; Greiner bio-one). Plasma was separated and frozen in plastic tubes within 1 hour and sent frozen by courier to the same laboratory. The samples were taken, when possible, in the morning when the stable environment was quiet and the horse was calm. Samples were not taken for this analysis during the acute phase of pain or from abattoir horses to reduce the possibility of stress-related elevation in plasma ACTH concentration. In those situations, animals with hirsutism were excluded from control groups, as that clinical sign is considered to be pathognomonic for PPID. 14 Quantitative plasma ACTH analysis was also performed using a chemiluminescent immunoassay (Immulite; Liphook Equine Hospital). Blood samples for glucose measurement were collected in 2-ml plastic lithium heparin tubes following at least 4 hours of fasting (Vacuette; Greiner bio-one). Glucose was measured immediately after blood collection with a glucometer (Ascensia Contour; Bayer HealthCare, Leverkusen, Germany).
Lamellar Sample Collection
For each horse/pony, the distal aspect of the right forelimb was disarticulated at the metacarpophalangeal joint within 1 hour of death and sectioned with a band saw as soon as possible. With the sole on the base plate of the band saw, the foot was first bisected sagittally (Fig. 2). Then, a 1-cm-thick parasagittal section was cut from half of the hoof; this specimen included the hoof wall, lamellar tissue, and most of the DP (Fig. 3). The midpoint between the coronary band and the ground-bearing surface was identified on the dorsal aspect of this section (Fig. 3). A square or rectangular segment including the dorsal cortex of the DP was excised and then a 0.5-mm-thick transverse section cut from its lower face (at the previously described midpoint) (Figs. 4, 5). From that transverse section (in which lamellar detail should be seen), the dense hoof wall (often pigmented) was removed to within approximately 1 mm of the abaxial aspect of the lamellar tissue (Fig. 5) using a single-edge razor blade (scalpel blades are not sufficient for this step). The phalangeal bone was then removed (Fig. 5) by running a scalpel blade along its dorsal edge. Each specimen was divided into 5-mm-wide (square) blocks that were fixed in 4% paraformaldehyde for 24 to 72 hours before processing by routine methods and embedding in paraffin wax. These blocks were oriented with the longitudinal axes of the PEL parallel to the cutting edge (Fig. 6); if oriented perpendicularly (ie incorrectly), the remaining horn at the abaxial aspect of the lamellar tissue was likely to fold over during section cutting (Fig. 7).
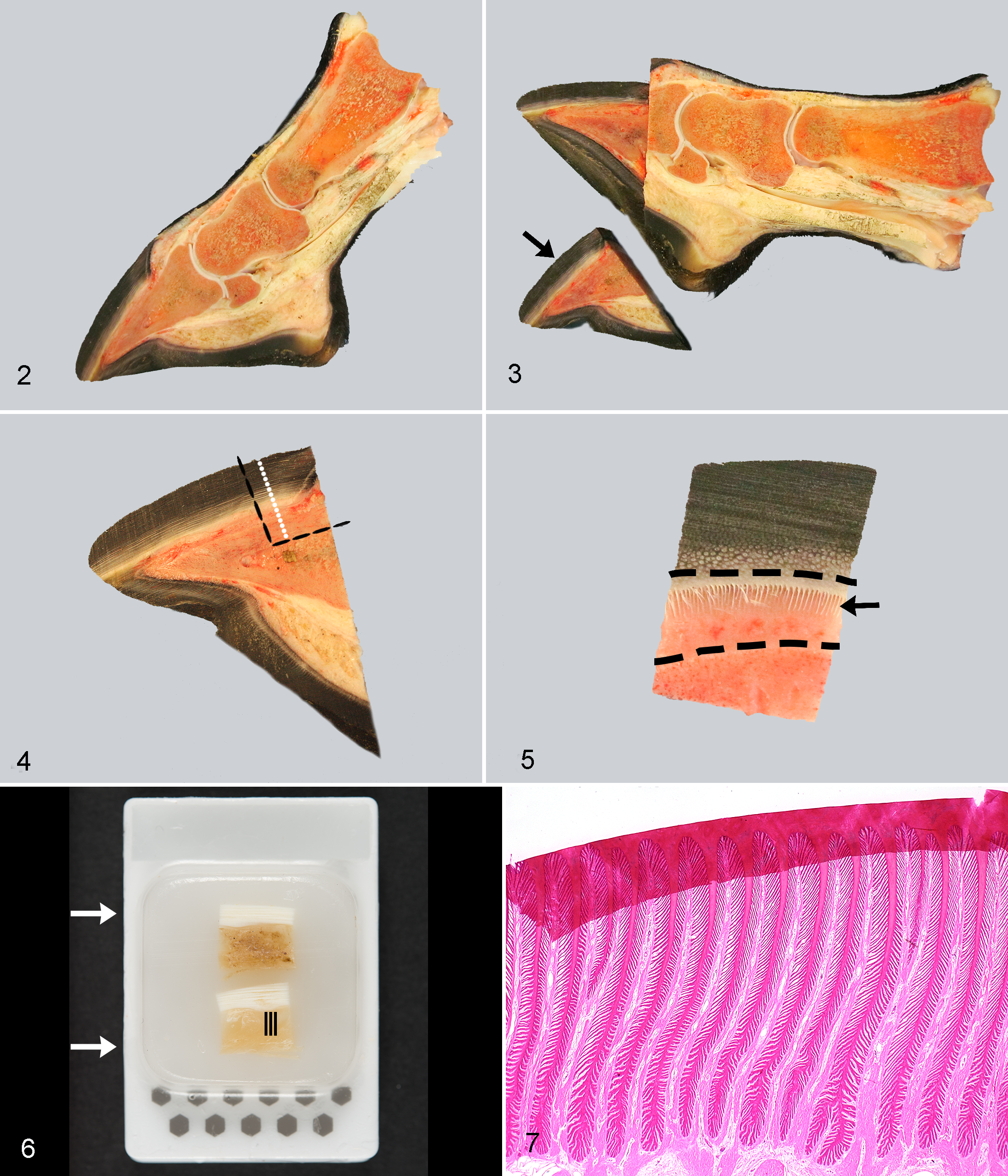
Technique for hoof lamellar sample collection, Equus caballus.
Histomorphometry and Categorization of Lamellae
Sections were stained using hematoxylin and eosin (HE) and periodic acid–Schiff (PAS) methods and then examined using a light microscope (N.P.K., J.C.P.-K.). The HE sections were used for detection of lamellar lesions and the PAS sections for morphometry. Digital images were taken using image capture software (Cell^D version 2.8; Olympus Soft Imaging Solutions, Münster, Germany) and a microscope-attached digital camera (Olympus DP71; Olympus Imaging America, Center Valley, PA). For PEL measurements, images were taken at 40× magnification and then merged (to image the complete PEL) (Adobe Photoshop CS5 version 12.0 x64; Adobe Systems, San Jose, CA). For SEL typing, images were taken at 200× magnification from axial (adjacent to keratinized hoof wall), middle, and abaxial (adjacent to DP) regions of each of the 10 randomly selected PEL (Suppl. Fig. S1). These regions were defined as 10%, 50%, and 90% of the length of the PEL, respectively. 22
Primary Epidermal Lamellar Length and Type
Measurements of primary epidermal lamellar length were made as described previously (Suppl. Fig. S1). 22 All of the PEL were numbered, and then 10 randomly selected PEL (http://www.randomizer.org/form.html) were marked in each image. The measurements of PEL length were performed using a drawing tool (Wacom Bamboo; Wacom Europe, Krefeld, Germany) and image analysis computer software (ImageJ version 1.4.3; National Institutes of Health, Bethesda, MD [rsbweb.nih.gow/ij/index.html]); a line was drawn from the axial to abaxial extent of each PEL (Suppl. Fig. S1). An adaptation of a previously documented PEL typing system was then used. 23 The selected PEL were classified as being standard (straight) or curving (with 2 or more bends), and bifurcations were reported when present.
Classification of Primary Epidermal Lamellae
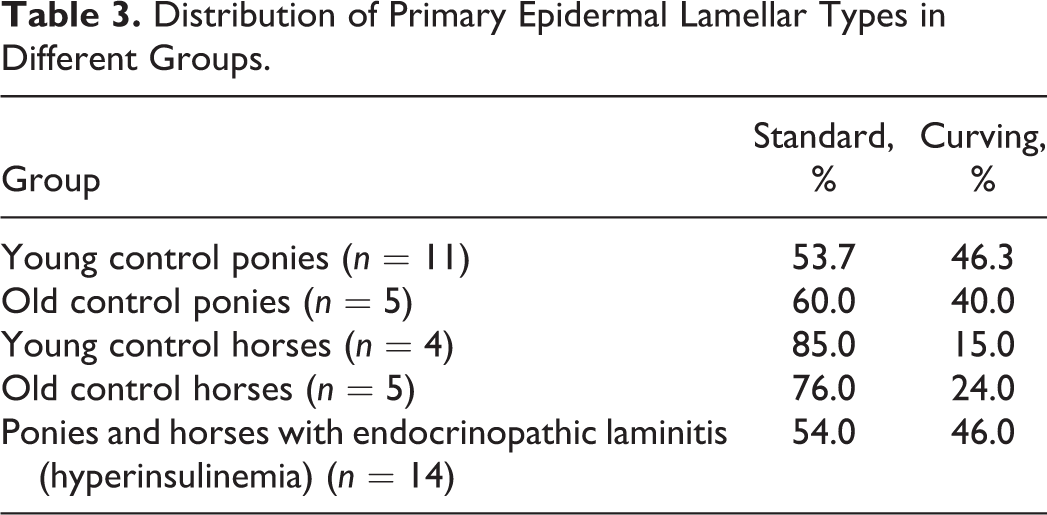
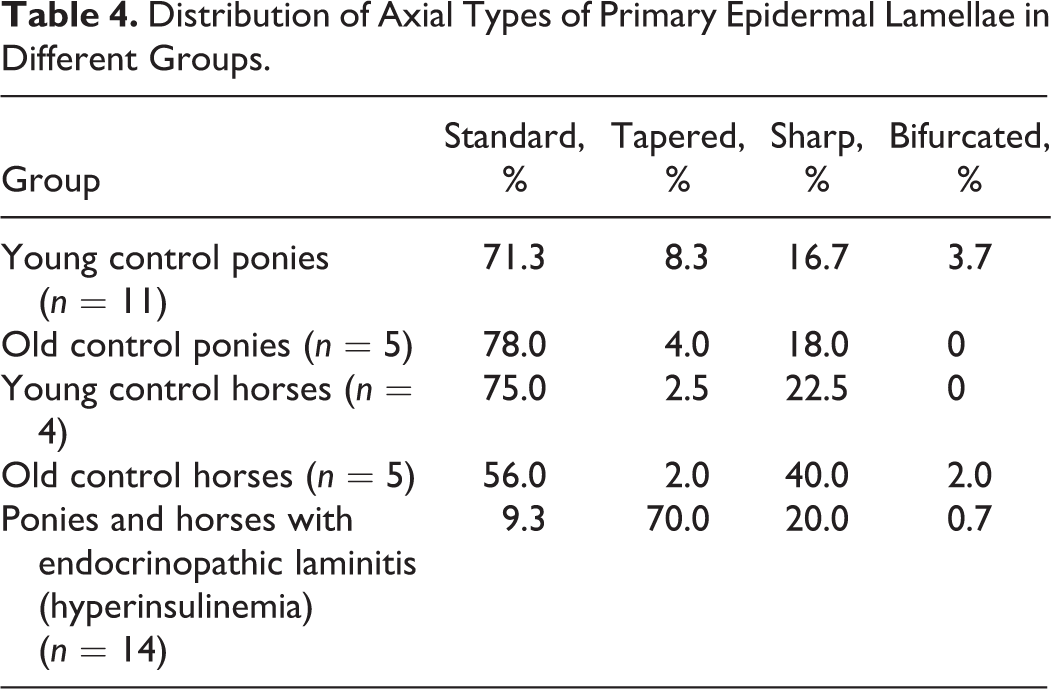
Primary epidermal axial region (tip) type was classified as in a previous study, that is, as standard, tapered, sharp (Suppl. Figs. S2–S4), or bifurcated. 23 The type of epidermis overlying the abaxial aspect of each primary dermal lamella (PDL) was defined as being standard, sharp, bifurcated, “proliferative” (dyskeratotic cells bridging dermal tissue), separated (ie, epidermal and dermal lamellae had torn apart), or keratinized (Suppl. Figs. S5–S9). The degree of abaxial keratinization (ie, in the “cap horn” region) was graded as normal, slightly increased (1–10 extra layers), moderately increased (10–30 layers), or markedly increased (>30 layers) (Suppl. Figs. S10–S13). 3 The amount of epidermal tissue bridging in a PDL at its abaxial aspect (ie, joining abaxial aspects of adjacent PEL) was defined as none, mild (1–2 epithelial bridges), moderate (>2 epithelial bridges; Suppl. Fig. S14), or extensive (abaxial region completely filled with epidermal cells). Epithelial islands were defined as isolated round or ovoid epithelial structures adjacent to abaxial, middle, or (more usually) axial aspects of the PEL (Fig. 8). Islands have been previously shown to lack direct attachment to their adjacent PEL (or other SEL), based on previous 3-dimensional reconstructions. 35 Lakes were defined as small to large cavities that were either empty (most likely due to processing artifact) or filled with homogeneous eosinophilic (proteinaceous) material (Suppl. Fig. S15). These lakes were either located between bridges of epithelial tissue extending across the abaxial aspect of the PDL or in the overlying cap horn.
Classification of Secondary Epidermal Lamellae
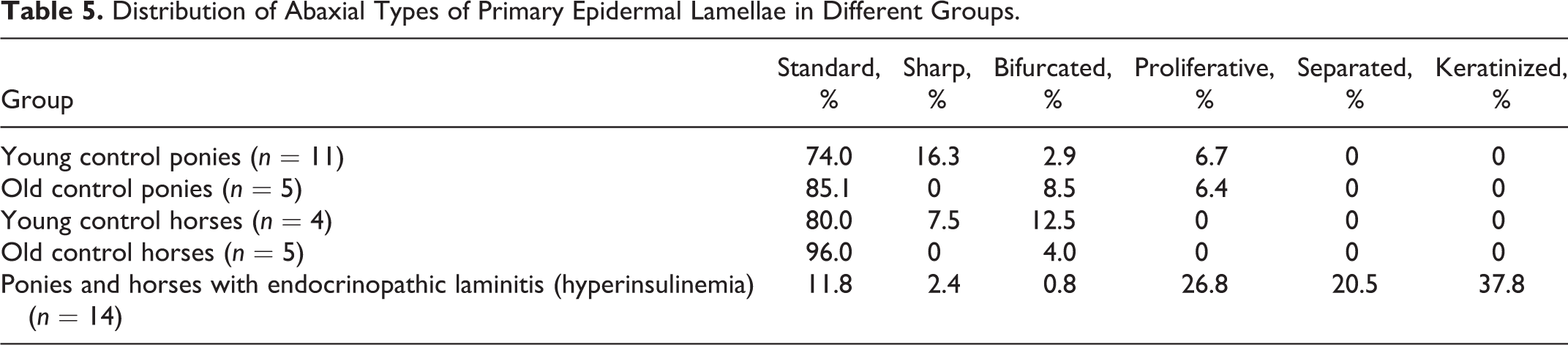
Secondary epidermal lamellar type was determined for 10 randomly selected SEL (5 from the left side and 5 from the right side) in each abaxial, middle, and axial region of each of the previously (randomly) selected PEL (ie, 30 in total per PEL; Suppl. Fig. S1). The SEL were classified as standard, tapered, club shaped, suprabasal layer hyperplasia, bifurcated, fused, separated, or keratinized (Suppl. Figs. S16–S21). 23
Immunohistochemistry
Further sections were deparaffinized (Histo-Clear; National Diagnostics, Hull, UK) and then rehydrated using graded alcohols. Antigen retrieval was required for anti–gamma-H2AX immunolabeling; this was performed by placing slides in sodium citrate buffer (pH 6.0) for 1 minute and 40 seconds using a pressure cooker (Antigen Access Unit, A. Menarini Diagnostics Ltd., Wokingham, UK). All slides were then loaded into a Dako-Autostainer (Dako UK Ltd., Ely, UK). Endogenous peroxidase and nonspecific staining were blocked using 3% hydrogen peroxide (in phosphate-buffered saline [PBS]) for 5 minutes. All tumors were incubated with a primary mouse monoclonal antibody against gamma-H2AX (clone JBW301 [1:500]; Millipore, Watford, UK) for 30 minutes at room temperature (RT). An anti–mouse secondary antibody conjugated to a horseradish peroxidase (HRP)–labeled polymer (K4003; Dako Envision + System-HRP; Dako UK Ltd.) was then applied for 30 minutes at RT. Visualization was achieved using 3,3′-diaminobenzidine (DAB+ Substrate Chromagen system; Dako UK Ltd.), 20 µl/ml for 10 minutes. Tissues were counterstained using Gill’s hematoxylin (made in house) for 27 seconds, then dehydrated, cleared, and mounted (DPX mounting medium; Cellpath, Newtown, Powys, UK).
Statistical Analysis
Comparisons between groups were made for all the parameters. The analysis was performed using computer software (SPSS for Windows, version 21; SPSS, Inc., an IBM Company, Chicago, IL). First, the normality of each parameter distribution was tested using the Shapiro-Wilk test. As no parameter was normally distributed, either a χ2 test or Kruskal-Wallis test with Bonferroni adjustment was used. Significance was set at P < .05. Results are presented as medians and interquartile range (IQR).
Results
Cases and Clinical Information
A total of 39 animals fulfilled the inclusion criteria and were divided into 5 groups: young control ponies (n = 11), old control ponies (n = 5), young control horses (n = 4), old control horses (n = 5), and endocrinopathic laminitis (n = 14; 4 horses, 10 ponies). Of the horses and ponies with endocrinopathic laminitis (all with hyperinsulinemia), 3 were diagnosed with PPID based on elevated seasonally adjusted basal plasma ACTH concentration and at least 3 clinical signs of PPID. Numbers, breed, sex distribution, median (IQR) age, BCS, fasting serum insulin, and fasting blood glucose for each group are presented in Table 2. The median reported duration of laminitis in horses was 120 days (range, 5–730 days).
Clinical Data of Different Groups.
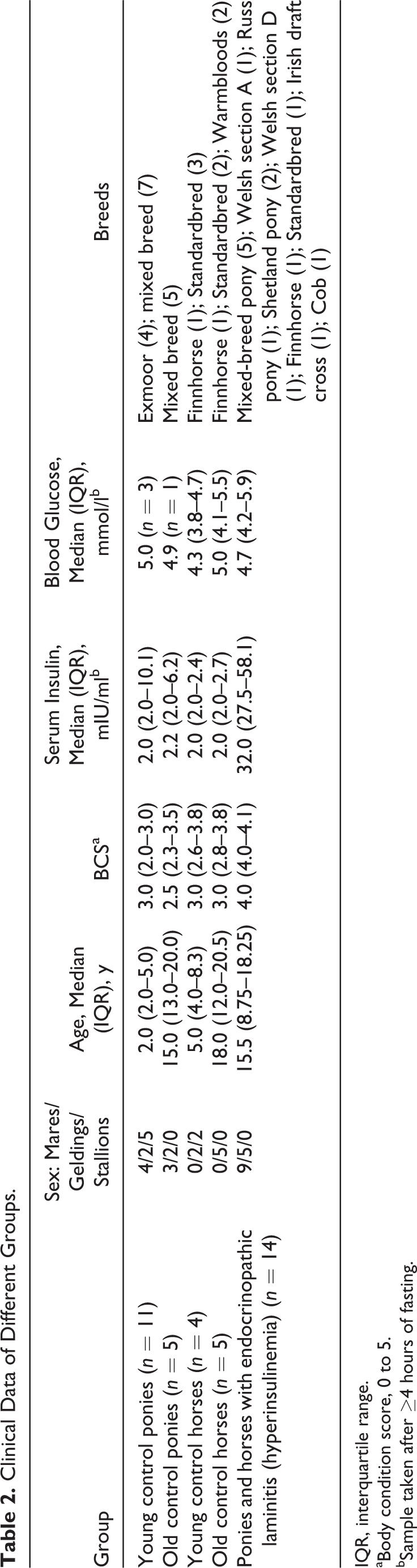
IQR, interquartile range.
aBody condition score, 0 to 5.
bSample taken after ≥4 hours of fasting.
In all laminitis cases, there were gross pathological changes: divergent rings were seen in 13 of 14 cases and gross separation and rotation of DP in 13 of 14 cases (the horse with no DP rotation was not the same animal as that lacking divergent rings). There was no correlation between the severity of macroscopic lesions and the historical time frame for laminitis. For example, large divergent rings were seen extending all the way down the hoof wall in 1 pony with a reported duration of laminitis of only 10 days, while another pony with disease duration of 548 days had similar lesions (Fig. 1).
Histological Evaluation
Control horses and ponies
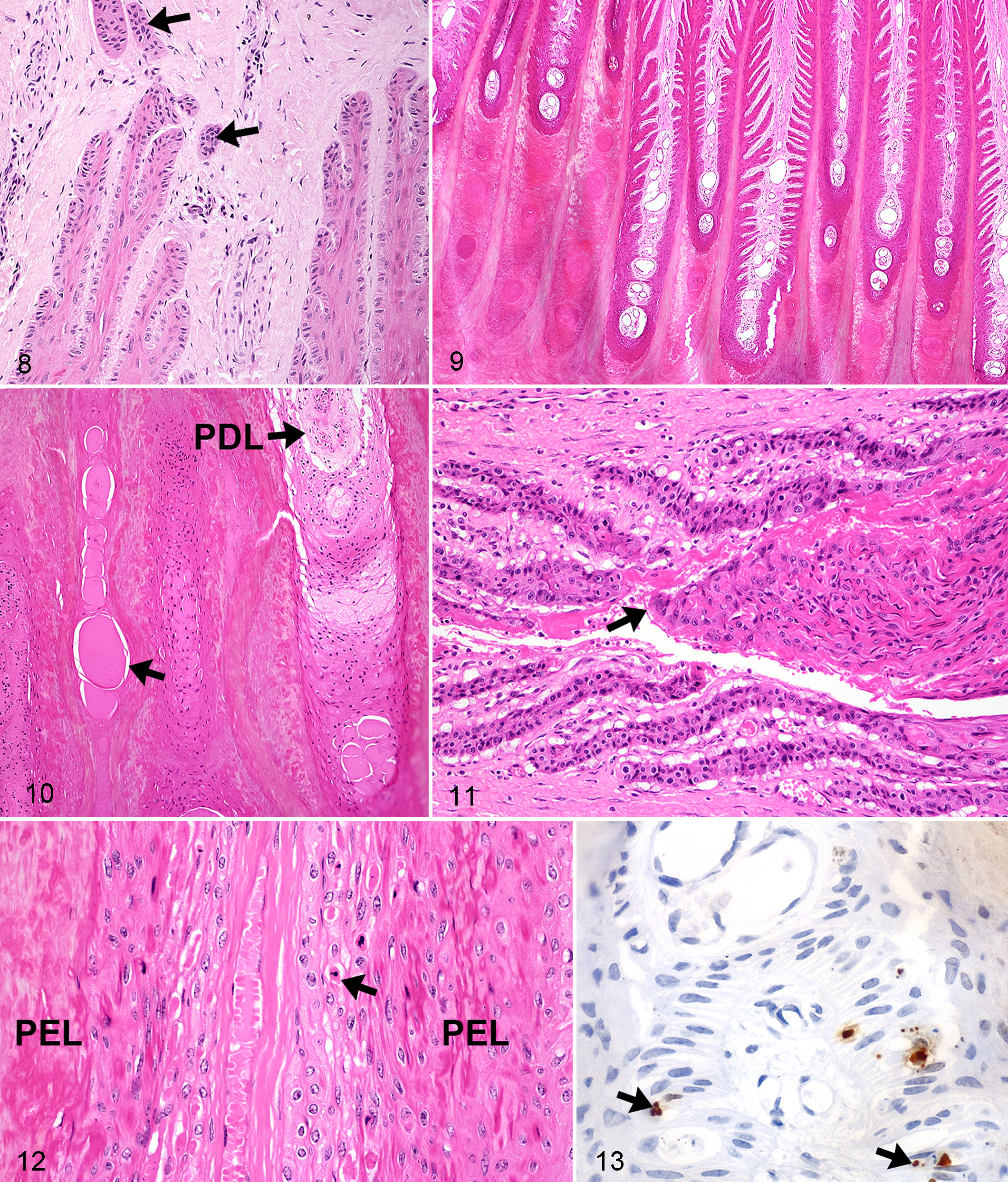
There was no difference in PEL length between young and old ponies (P = .99). However, old control horses had longer PEL than did young control horses (P = .04). Control horses in general had significantly longer PEL than did control ponies (P < .001). The most common PEL type in all control animals was standard, as were the most common PEL axial and abaxial epidermal classifications (Tables 3–5). There was no increase in keratinization at the abaxial aspects in any of these groups. The most common SEL type in all regions (abaxial, middle, axial) in all control groups was standard (Table 6). Occasional epithelial islands were noted in axial regions in old (3/5) and young (2/11) control ponies (Fig. 8), but these were not noted in control horses of any age. Epithelial islands were not noted in abaxial or middle regions in any animal. In 2 of 5 old control ponies, there were (multifocally) significantly increased numbers of apoptotic cells/bodies in SEL without regional localization (ie, not confined to axial, middle, or abaxial areas); up to 5 to 6 were noted per high-power field (hpf) (400×), and these showed the typical morphological features of this type of cell death (cytoplasmic rounding, shrinkage and hypereosinophilia, nuclear condensation and fragmentation, formation of apoptotic bodies, and phagocytosis by neighboring epithelial cells). 11,24
Distribution of Primary Epidermal Lamellar Types in Different Groups.
Distribution of Axial Types of Primary Epidermal Lamellae in Different Groups.
Distribution of Abaxial Types of Primary Epidermal Lamellae in Different Groups.
Distribution of Secondary Epidermal Lamellar Types in Axial, Middle, and Abaxial Regions of Primary Epidermal Lamellae.
Endocrinopathic laminitis
The changes/lesions noted were highly variable between individuals, and the severity of lesions could not be related to the reported duration of laminitis or whether PPID was concurrent. Horses and ponies with endocrinopathic laminitis had significantly longer PEL compared with controls (P < .001). In horses and ponies with endocrinopathic laminitis, PEL were either standard or curving (Table 3). Axially, increased numbers of tapered PEL ends were noted in laminitic horses and ponies compared with controls, with a corresponding reduction in the proportion of standard ends (P < .001, Table 4). Abaxially, increased numbers of proliferative, keratinized, and separated types of epidermis overlying PDL were noted compared with controls (P < .001, Table 5), with “separated” relating to acute tearing associated with primary or repeated laminitis episodes. The overall degree of keratinization of abaxial aspects of PEL was also increased (median degree of keratinization markedly increased vs normal in control ponies and horses) in laminitic horses and ponies (P < .001). Increased proportions of tapered SEL in axial regions (of the PEL) and of keratinized, separated, and fused SEL types in abaxial regions (Table 6) were seen in the laminitic animals (P < .001). In middle regions, horses and ponies with endocrinopathic laminitis had a higher percentage of fused, separated, and tapered SEL than did control animals (P < .001, Table 6).
Interlamellar epidermal bridging across the abaxial aspects of PDL (and keratinization) occurred in a variable fashion in the laminitis group only (median amount mild), resulting in a highly irregular margin between the inner horn and the lamellar tissue (Fig. 9). In some cases, up to half the lamellar length was lost (in some fields). In many fields, rows of isolated, rounded islands of dermal tissue were noted (entrapped) within the increased amounts of cap horn; some were still vascular, but in others, the blood vessels were degenerate or were not observed. Some of the dermal islands were partially filled with dyskeratotic cells and others with proteinaceous fluid (Fig. 10). The variably long areas of increased cap horn containing these lakes and/or large amounts of parakeratotic debris were clearly not as well organized as normal cap horn (Fig. 10). In addition to the proteinaceous material noted in lakes within cap horn, in some fields, smaller foci were associated with the dyskeratotic epidermal tissue overlying PDL bases; neither form was clearly associated with lamellar separation in the same section. Axial epithelial islands were seen in small numbers of animals, similar in location to those seen in specimens from control groups. However, abaxial islands were also noted in 5 of 14 cases with endocrinopathic laminitis.
The reported duration of laminitis did not correlate with the observation or the severity of acute lamellar separation (seen in one-third of laminitic cases). Where noted, this separation usually involved abaxial to middle regions of the lamellae, often with separation of epidermal from dermal tissue and axial displacement (ie, recoil) of the latter. In a few areas, the PEL were torn from the associated SEL, and in 1 horse, this type of separation occurred almost to the axial tip of the lamellar tissue (Fig. 11). Torn tissue was pale-staining with loss of cellular detail and large numbers of necrotic and apoptotic cells but (surprisingly) minimal hemorrhage and neutrophilic infiltration (Fig. 11). In some cases, toward and at the margins of the torn areas, there was vacuolar swelling of many SEL cells with marked increases in numbers of apoptotic cells; frequently, these SEL were elongated and difficult to discern from one another (Fig. 12). In sections where separation was not noted, increased and often large numbers of apoptotic cells (up to 30 per hpf) were still noted in SEL in the abaxial regions of the PEL, with more variable observation in more axial regions. This was confirmed to be apoptotic cell death by positive immunoreactivity for gamma-H2AX, this being a protein that responds to double-strand DNA breakage (ie, is specific for apoptotic cells) (Fig. 13). 20 The observation of mitotic figures in SEL cells was even more variable in terms of number and location, but they were most numerous in sections where separation had occurred or there was extensive abaxial fusion; in these sections, most of the mitotic figures were located axially (1–3 per hpf). In other sections, they were more occasionally noted (0–1 per hpf), again most frequently in axial regions (although they did occur throughout the lamellar tissue in 4 animals). No direct association was noted either quantitatively or in terms of localization between apoptotic cells and those showing mitotic activity.
Discussion
This article presents the first descriptions of macroscopic and microscopic lesions in appropriately defined cases of naturally occurring endocrinopathic laminitis. Chronic lesions were identified in all animals with laminitis, and these were most severe and consistent in abaxial regions of the lamellar tissue and associated inner hoof horn. The markedly increased numbers of tapered and lengthened SEL (50%–70% vs 5%–10% in controls) were in agreement with previous studies of acute (experimentally induced) hyperinsulinemic laminitis (ie, this is an indicative lesion whatever the stage, but it does not persist abaxially in chronic disease). 22 Abaxially, the most frequent changes included irregular SEL fusion and replacement with abnormal keratinized tissue (associated with dermal island isolation and vascular degeneration), separation of epithelial islands, and variably but often markedly increased numbers of apoptotic cells.
These descriptions differ from several classic and often-cited descriptions of laminitis typically referred to by pathologists, which were actually derived from carbohydrate overload models of inflammatory disease and a few naturally occurring cases. 18,30,31,34 They indicated a catastrophic and ultimately global failure of the basement membrane, resulting in a “de-gloving” injury in which epidermal and dermal lamellae separate. We propose that this is an extreme and acute situation relating to systemic inflammatory disease as a crisis event (ie, something that is unlikely in cases of hyperinsulinemia). However, not all inflammatory models have supported the basement membrane disruption theory. In one carbohydrate overload model study, vacuolar degeneration and death of epidermal cells in abaxial regions of PEL without significant basement membrane separation were observed in developmental stages, which progressed in clinical stages to lamellar separation in abaxial to middle regions with minimal associated inflammation. 29 Although lamellar damage in endocrinopathic laminitis may occur more slowly than in the inflammatory model, the abaxial location and nature of the lesions (including the initiation of separation) in our case series are similar, potentially indicating that lamellar tissue has a limited repertoire of pathological responses to multiple systemic stresses. In part, this may relate to various abaxial-axial gradients in the hoof wall, including the distribution of biomechanical forces, greater distances of abaxial lamellar cells from the incoming vascular supply, and a previously suggested higher metabolic demand or toxin/cytokine sensitivity of those abaxial cells. 29 In terms of biomechanics, the relationships between hoof loading, external hoof shape, and biological responses of the lamellar tissue are extremely complex. 33 The transformation noted in many of the current specimens, to a highly irregular lamellar–hoof wall interface (including increased amounts of poor-quality cap horn containing debris and fluid), would be expected to compound any biomechanical damage cycle.
Another morphological change seen in developmental and acute phases of hyperinsulinemic models was the appearance of increased numbers of apoptotic cells in the SEL, beginning axially and spreading abaxially in a “wave,” followed by reactive proliferation. 2,11 In our natural cases, large numbers of apoptotic cells tended to be concentrated in abaxial regions along with the other lesions. This suggests that epidermal cellular damage and death continue in this abaxial region, following initial reactive proliferation that would be required to bridge dermal tissue. In our chronic lesions, mitotic activity was no longer noted in the abaxial region, implying that beyond a certain point, this epithelium is in a dyskeratotic/hyperkeratotic and terminally differentiated stage. This is in keeping with previous findings in chronic laminitic cases (with variable causes, including inflammatory and endocrine disease) where abaxial epidermal cells were found to have reduced expression of p63, a protein that plays a key role in regulating epithelial proliferation and differentiation. 6 Axially in many of our cases, mitotic activity was still occurring and/or increased in conjunction with lengthening and tapering of the SEL and the PEL tips. This is in agreement with previous analysis of the acute phase of hyperinsulinemic laminitis (6–12 hours) that indicated that SEL lengthening occurs due to a combination of cell stretching and increased cell cycle entry. 11,22 Any mechanical imbalances acting on mid to axial aspects of the lamellar tissue may be compounded by the irregular abaxial fusion. The exact role of the high insulin levels in any of this lesion development is not known. Insulin signaling is mediated by highly complex intracellular networks with the capacity to control multiple processes, including growth and differentiation; the specifics and balances of these pathways in equine lamellar tissue have not yet been explored. 32
The finding of significant numbers of apoptotic cells in lamellae of 2 “old” control ponies was unexpected. Clearly, these animals did not meet any of the criteria of the laminitic groups (including evidence of endocrine disease), and their lamellar tissue did not contain any other histological lesions. Increased levels of apoptosis have been described previously in developmental and acute phases of hyperinsulinemic laminitis, but in those studies, there were other, associated changes in all affected ponies/horses, including increased mitotic activity, minor basement membrane separation, mild inflammation, and/or SEL stretching/tapering. 2,11 The absence of further lesions in these 2 control animals does not rule out other initiating causes of minor lamellar epithelial damage, including excessive mechanical loading. However, it does indicate that if increased numbers of apoptotic cells are noted in equine lamellar tissue in the absence of any other lesion (macroscopic or microscopic), a diagnosis of laminitis cannot be justified based on our current knowledge.
Importantly, the nature and severity of the lesions had no correlation with the reported duration of disease (that varied from days to years). The lack of correlation between histological lesion severity and duration of clinical disease might be due to inaccuracy of owner/referring veterinarian reporting or association of chronic abaxial lesions with low-grade pain or discomfort difficult to detect purely by observation. 29 It has previously been proposed that horses and ponies have repeated episodes of “subclinical” lamellar disease prior to an acute clinical presentation. 23,29 However, although the owners may have not noted any pain/lameness, our results suggest that the macroscopic hoof lesions would be a better focus. Divergent, nonconcentric rings often ignored or misinterpreted as “normal growth rings” were noted in this study in all but 1 laminitic animal. However, growth rings are always concentric and often seen in horses with healthy hooves. 13 It takes approximately 3 months for divergent rings to become evident once the dorsal hoof wall growth has been disturbed, and in the present study, in 6 of 14 cases, the duration of the (recognized) disease was significantly less. If divergent rings occur prior to clearly recognizable clinical signs of laminitis in at least some animals, their recognition should allow early detection and the adaptation of strategies to slow or reverse the lamellar disease, since equine metabolic syndrome can be treated successfully. Horse/pony owners should be educated to seek veterinary assistance and have blood insulin levels tested when divergent hoof rings are observed in animals that are not yet observably lame. It is interesting to note that 1 excluded horse in which the laminitis was historical (not current) did not have abaxial lesions or an externally abnormal hoof. This supports the potential for reversing hoof lesions and warrants further investigation. From the point of view of the pathologist, divergent hoof rings noted at necropsy examination should indicate the need to examine internal hoof structures both macroscopically (for DP deviation) and microscopically, even when this is not part of a routine examination.
Conclusions
In conclusion, this article provides a guide for effective sampling and recognition of normal and abnormal morphological variation of the equine hoof lamellae. All cases of endocrinopathic laminitis, regardless of their clinical duration, showed chronic pathology in the abaxial region of the hoof wall that in all but 1 horse was correlated with divergent hoof rings. The latter association suggests a genuine possibility for disease prevention.
Footnotes
Acknowledgements
We thank Lynn Stevenson and Lynn Oxford from Veterinary Diagnostic Services of University of Glasgow for developing the histological techniques and Dr Tina Rich, University of Glasgow, for advice on morphological identification and immunolabeling of apoptotic cells.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Finnish Foundation of Veterinary Research and Erkki Rajakoski foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
