Abstract
Since 2002, reports of deer with swollen muzzles from throughout the United States have resulted in significant interest by wildlife biologists and wildlife enthusiasts. The condition was identified in 25 white-tailed deer (Odocoileus virginianus) and 2 mule deer (O. hemionus). Microscopic lesions consisted of severe, granulomatous or pyogranulomatous inflammation of the muzzle, nasal planum, and upper lip, as well as similar but less severe inflammation of the hard palate. Lymphadenitis of regional lymph nodes was common and granulomatous pneumonia was present in one individual. Splendore-Hoeppli material was typical in the center of inflammatory foci. Other than the single instance of pneumonia, systemic disease was not evident. Various bacterial species were isolated in culture, most of which were not morphologically consistent with the colonies of small, gram-negative bacteria observed in the center of the granulomas. Amplification and sequencing of the bacterial 16S rRNA gene from tissues of affected deer resulted in the identification of Mannheimia granulomatis. Laser capture microdissection was used to confirm that the colonies in the inflammatory foci were M. granulomatis. The cases described here are reminiscent of a bovine disease in Brazil and Argentina, locally called lechiguana. Although the inflammation of lechiguana is mostly truncal, the microscopic lesions are very similar and are also attributed to M. granulomatis. It is unclear if this is an emerging infectious disease of deer, or if it is a sporadic, uncommon condition that has only recently been recognized.
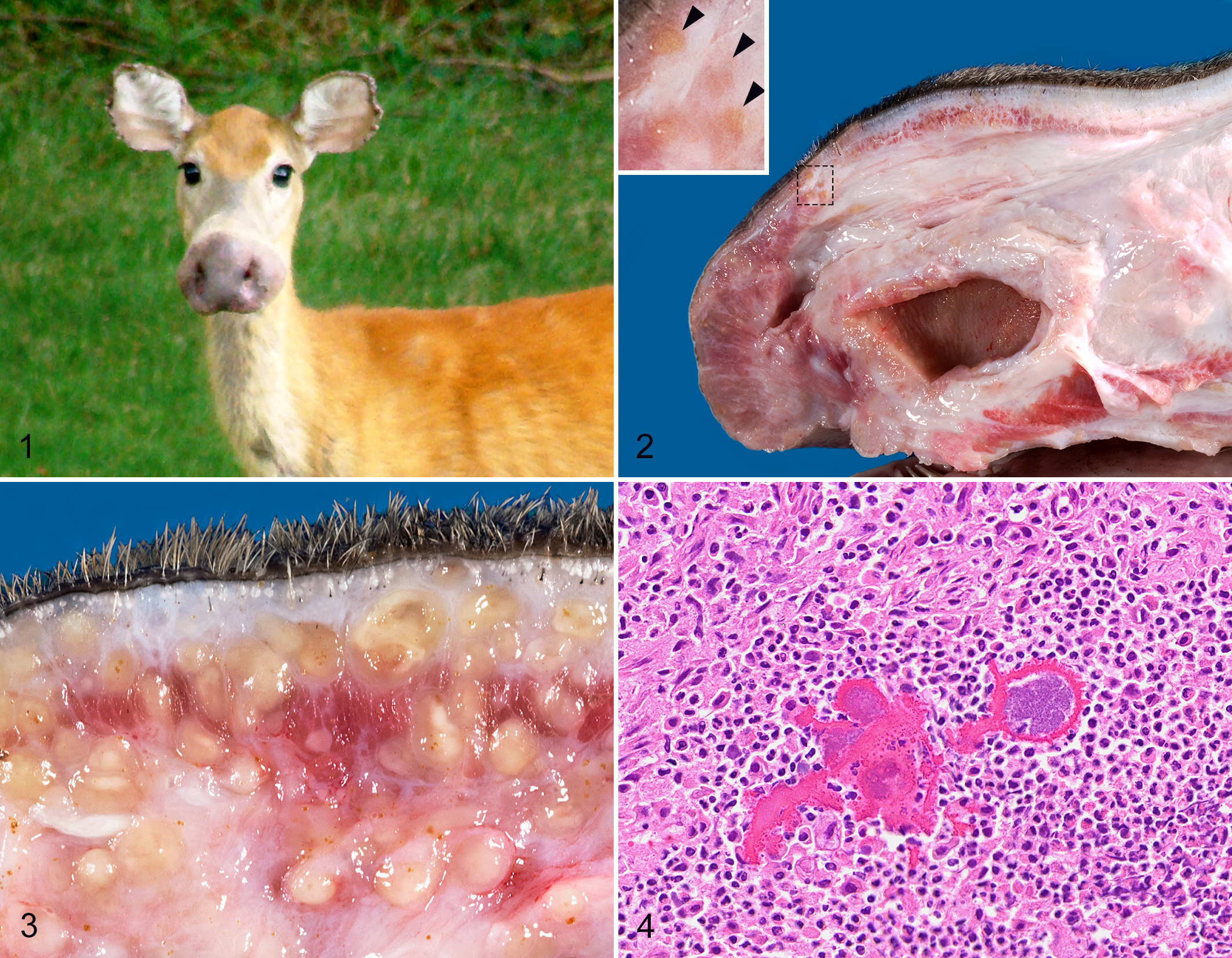
Since 2002, white-tailed deer (Odocoileus virginianus) from a wide region of the United States have been reported with an unusual condition involving marked swelling of the soft tissue of the muzzle (Fig. 1). This has typically been more severe on the distal aspect of the muzzle, suggesting a resemblance for some observers of the nose of a moose, and leading to the lay term “Bullwinkle deer,” after the cartoon character of that name. The total number of deer affected is unknown. Anecdotal evidence suggests it sporadically affects a small number of animals with no obvious implications for overall deer populations, although some deer biologists have reported 2 or more affected deer from a single county or adjacent counties in a given year. In most cases, affected deer were first discovered when harvested by hunters. However, the expanding use of trail cameras and backyard observations of deer have resulted in some diseased deer being observed for periods of months to a year or more.
Mannheimia granulomatis cellulitis, white-tailed deer.
These cases generated considerable interest and concern among citizens and wildlife biologists due to the unusual nature of the lesions and because this previously unrecognized disease was suddenly reported over a large geographic area. The concern of wildlife managers prompted an investigation of the condition, and cases were actively solicited from wildlife biologists and hunters. From January 2002 until January 2019, 25 white-tailed deer and 2 mule deer (O. hemionus) were acquired. All 27 deer in this group had the characteristic swelling of the soft tissue of the muzzle. In all but 5 cases, only the head was available for examination. This report describes the correlation of these lesions with, Mannheimia granulomatis, a novel pathogen of Odocoileus spp., as well as the unique gross and microscopic lesions characteristic of the affected deer.
Materials and Methods
Case Material
Necropsy databases of the Southeastern Cooperative Wildlife Disease Study (SCWDS), University of Georgia, Athens, Georgia, were screened for deer submissions with a diagnosis of swelling or cellulitis of the muzzle. Submissions were reviewed from 1975 to January 2019. Inquiries regarding the presence of unusual lesions on the muzzle of deer often resulted in the intentional submission of additional cases, resulting in a potentially biased sample. The signalment, location, and descriptions of lesions were compiled for all individuals meeting our criteria.
Pathology and Immunohistochemistry
In 22 of 27 cases, only the head was available for postmortem examination. Age, if recorded, was based on tooth wear and attrition. The samples were frozen or refrigerated prior to submission for diagnostic evaluation. A thorough postmortem investigation was performed on all submissions and representative tissues from all available organs were preserved in 10% neutral-buffered formalin. Fixed tissues were dehydrated through baths in graded alcohol and xylene, followed by embedding in paraffin; 5-μm-thick sections of the paraffin-embedded tissues were rehydrated and stained with hematoxylin and eosin, Gram stains (Lilly-Twort or Brown & Brennen), Ziehl-Nielsen acid-fast stains, and Fites-furachrome acid-fast stains, as previously described. 6 Representative tissues from 5 cases were evaluated for the presence and distribution of bacteria using a published immunohistochemistry assay for the bacillus-Calmette-Guerin antigen. 5 This assay is nonspecific and confirms the presence of bacteria and fungi, indiscriminately.
Bacteriology
If the tissues were not significantly autolyzed or putrefied, uncontaminated samples of soft tissue were collected deep to the skin of the muzzle and these were submitted for bacterial culture. Samples from 16 deer were submitted to 1 of 4 independent bacteriology laboratories, and duplicate samples from 1 deer were submitted to 2 laboratories. All samples were evaluated for aerobic growth.
DNA Sequencing and Laser Capture Microdissection
Uncontaminated tissue samples were collected deep in the skin of the muzzle, and these were used for total DNA extraction with the DNeasy Blood and Tissue kit (Qiagen). Universal PCR primers targeting the bacterial 16S rRNA gene were used for PCR amplification and sequencing. The primers used have been described in previous publications and target overlapping regions of the 16S rRNA allowing for complete gene sequencing. 2,7,12 The primers are E334F (5′-CCAGACTCCTACGGGAGGCAGC), E939R (5′-CTTGTGCGGGCCCCCGTCAATTC), U1 (5′-ACGCGTCGACAGAGTTTGATCCTG GCT), U1R (5′-GGACTACCAGGGTATCTA AT), U2 (5′-CGCGGATCCGCTACCTTGTTACGACTT), U3 (5′-AGTGCCAGCAGCCGCGGTAA), U4 (5′-AGGCCCGGGAACGTATTCAC), and U5 (5′-TCAAAKGAATTGACGGGGGC). The primers were used in the following combinations under previously described conditions: E334F + E939R, U1R + U1, U3 + U4, and U5 + U4. 7,12
DNA extraction and PCR were performed in separate rooms with separate equipment, and all PCR reactions were run with a negative water control. The products were separated by electrophoresis on 2% agarose minigels and visualized with ethidium bromide. Amplicons were sequenced directly from the gels after purification with a gel extraction kit (Microcon, Millipore). BigDye terminator cycle DNA sequencing (ABI Prism) was performed. Sequences were evaluated using the BLAST algorithm from the National Center for Biotechnology Information. 1
A Leica LMD6000 laser-capture microscope was used to select isolated colonies of bacteria from within inflammatory foci. Sections of paraffin-embedded tissue were collected on PEN membrane slides and stained with hematoxylin and eosin. Each area of interest was collected in a cryotube, and the sample was rehydrated and processed for PCR and sequencing as described above, with the exception that DNA extractions from paraffin-embedded tissues were conducted using the QIAamp DNA FFPE Tissue Kit (QIAGEN Inc, Cat No. 56404) as prescribed by the manufacturer.
Results
Signalment and Geographic Distribution
Postmortem examinations were performed for 27 deer with this condition from 14 states. The states from which the deer were submitted included Alabama (n = 6), California (n = 1), Delaware (n = 3), Georgia (n = 1), Idaho (n = 1), Maryland (n = 4), Michigan (n = 1), Minnesota (n = 1), Mississippi (n = 1), New Jersey (n = 2), North Carolina (n = 2), Pennsylvania (n = 1), Rhode Island (n = 1), and South Carolina (n = 2). Of the 27 cases, 25 were white-tailed deer and 2 were mule deer (1 from Idaho and 1 from California). The deer from California was further characterized as a Columbian black-tailed deer (O. h. columbianus), a subspecies of mule deer.
The white-tailed deer included 8 does, 11 bucks, and 6 of unknown sex. The known ages of the 27 cases ranged from 0.5 years to 5.5 years with a median age of 2.5 years (n = 14). The median age for males was 3.5 years, and the median age for females was 2.5 years. Including those deer for which only an age class was recorded, there were 2 juvenile (8%) and 22 adult (92%) white-tailed deer. Samples were only collected during the period from September to January, and all but two were collected by hunters. Nutritional condition was only reported for 3 white-tailed deer: 2 deer were in poor nutritional condition and 1 deer was in good nutritional condition. One of the deer that was in poor nutritional condition was also diagnosed with pneumonia.
Two white-tailed deer were reportedly observed with the rostral swelling for 3 years before they were collected. One was a buck, and antler characteristics provide a reasonable certainty that the deer collected was the same one observed during that 3-year period. The other individual was a doe. Although we suspected the reports were of a single animal due to the rarity of these cases, we could not confirm only one animal was affected during this time.
Gross Pathology
As with the anecdotal reports, all 27 deer available for postmortem examination were similarly affected by marked thickening of the soft tissues of the muzzle and upper lip (Fig. 1). The lower lip was also swollen, but to a lesser degree, in 3 deer (cases 2, 3, and 9). The swelling was always most severe at the nasal planum and continued proximally to the midpoint of the muzzle (Fig. 2). The swelling was isolated to the subcutis, underlying muscles and other soft tissues. There was typically no significant thickening of the epidermis, and alopecia was not evident or was minimal. In some individuals, the superior buccal papillae were variably thickened, as was the submucosa of the hard palate.
Antemortem photographs were available for case 7 (Fig. 1). The swelling at the nasal planum created a prominent bell-shaped flaring of the nose before death. After death the severity of swelling at the nasal planum was greatly reduced relative to the swelling of midrostral tissues, suggesting postmortem imbibition of edema fluid into more proximal soft tissues of the muzzle.
The cut surfaces of the muzzle appeared slightly wet, but they were very firm and did not bulge. In most cases, the soft tissues of the muzzle included widely scattered, tan foci that were occasionally more glossy than adjacent regions (Figs. 2, 3). These foci varied in firmness but were never soft. Similar lesions were present in various lymph nodes of the head, most notably the parotid, mandibular, and retropharyngeal lymph nodes.
The entire body was available for examination from only 5 of 27 deer. Cranial lesions in all 5 were as described for other cases, but the lungs of case 7 had multiple firm nodules grossly similar in appearance to those in the lymph nodes and muzzle.
Histopathology and Immunohistochemistry
Microscopic lesions were consistent among all individuals, with minor variation in severity and distribution. In general, the subcutaneous tissues of the face, and to a lesser extent, other regional soft tissues, were greatly expanded by multifocal inflammatory infiltrates and fibrous connective tissue that was variably rarefied, consistent with edema. Inflammatory foci in all cases included widely scattered infiltrates of macrophages, epithelioid macrophages, and variable numbers of neutrophils, with multifocal aggregates of Splendore-Hoeppli material containing colonies of small rod-shaped bacteria (Fig. 4). The Splendore-Hoeppli material was typified by radiating aggregates of club-shaped eosinophilic material that tended to coalesce in larger foci. This material was often surrounded by and partially engulfed by histiocytic multinucleated giant cells. Smaller eosinophilic, club-shaped fragments of the material were frequently present within the cytoplasm of giant cells.
The granulomas varied in number and size but were typically abundant. In cases with fewer granulomas, the dermis, subcutis, and, to a lesser extent, the skeletal muscle had more loosely aggregated infiltrates of lymphocytes, plasma cells, and variable numbers of neutrophils. In rare cases, smaller aggregates of epithelioid macrophages and multinucleated giant cells were surrounded by dense sheets of neutrophils but were still associated with Splendore-Hoeppli material and bacterial colonies (Fig. 5). Small to moderate numbers of lymphocytes and macrophages were frequently scattered in the margins of the inflammatory nodules and in the adjacent tissue. In many cases the macrophages had foamy cytoplasm, and some epithelioid macrophages or multinucleated giant cells contained small intracytoplasmic aggregates of Splendore-Hoeppli material.
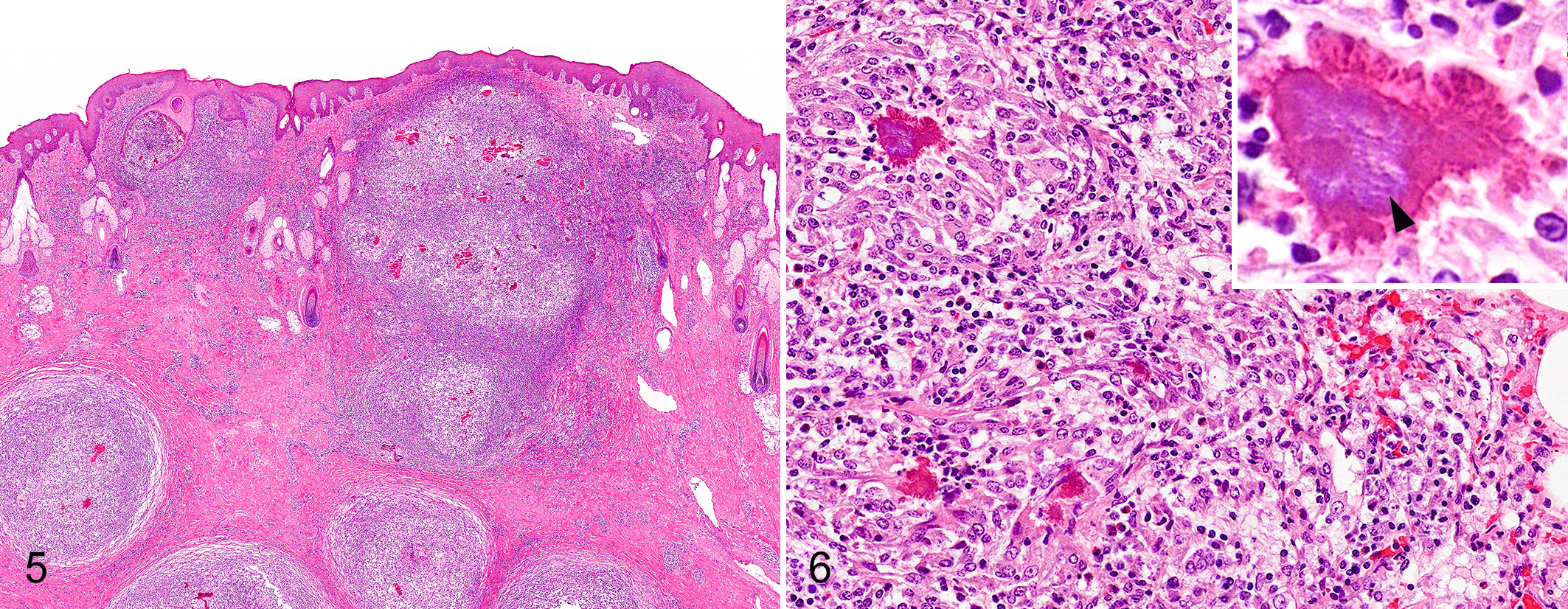
Mannheimia granulomatis cellulitis, skin, white-tailed deer. Granulomas containing Splendore-Hoeppli material are widely scattered in the subcutis and occasionally abut the epidermis. Hematoxylin-eosin.
Bacteria were observed in all 27 cases; they were consistently gram-negative and were not stained by the Fites-furachrome, an acid-fast stain. The bacterial colonies were typically evident only within the aggregated Splendore-Hoeppli material or closely adjacent to it. Special stains and immunohistochemistry for Bacillus-Calmette-Guerrin antigen did not indicate other bacteria or fungal elements within the granulomas. However, some samples had randomly distributed bacteria that were not correlated with inflammatory infiltrates (interpreted as postmortem growth).
One deer had granulomatous pneumonia in addition to the rostral cellulitis. Splendore-Hoeppli material was less abundant in the lungs than in the tissues from the head, but the cell populations and organization were consistent with those in the cranial tissues (Fig. 6). The infiltrates were scattered around small airways and did not uniformly affect each lobule. None of the other deer that received a complete necropsy had any evidence of organ involvement beyond the soft tissues of the head.
Bacteriology
Attempts were made to culture bacteria from the affected tissues of 17 deer. This yielded varied results, and the morphology of most bacteria cultured were inconsistent with the gram-negative, non–acid-fast colonies visible in tissue sections. The bacteria isolated included Trueperella pyogenes (n = 6), Pseudomonas aeruginosa (n = 4), α-hemolytic Streptococcus spp. (n = 3), Enterobacter sp. (n = 3), Pantoea sp. (n = 3), Pseudomonas fluorescens (n = 2), Serratia sp. (n = 2), Staphylococcus sp. (n = 2), non-hemolytic Streptococcus sp. (n = 2), Acinetobacter lwoffii (n = 1), Alcaligenes faecalis (n = 1), Corynebacterium divergens (n = 1), Corynebacterium jekeium-like sp. (n = 1), Escherichia coli (n = 1), Ewingella americana (n = 1), Klebsiella oxytoca (n = 1), Klebsiella pneumoniae (n = 1), Moraxella sp. (n = 1), and Psychrobacter sp. (n = 1). Unidentified gram-negative, rod-shaped bacteria were cultured from 3 deer, and an unidentified gram-positive bacterium was isolated from another.
The inconsistent culture results and a disparity between the expected staining characteristics of cultured bacteria and the bacteria visible in histologic sections suggested the etiologic agent was not included among the bacteria cultured from affected tissues. Therefore, molecular techniques were used to characterize the infection. Universal PCR primers for the 16S rRNA gene were used to amplify DNA from bacteria within tissues in a nonselective manner. Grossly visible granulomas were targeted from fresh tissue. Sequencing of PCR products from 8 deer for which fresh frozen tissues were available identified P. aeruginosa (n = 1) and M. granulomatis (n = 7).
Laser-capture microdissection was used to isolate bacterial colonies within inflammatory foci from formalin-fixed, paraffin-embedded tissues of 4 cases in which the sequence of the 16S rRNA PCR amplicons were similar to M. granulomatis. The laser-capture microdissection samples were similarly tested by PCR and the sequences matched M. granulomatis.
Discussion
There were no known reports of deer affected by granulomatous cellulitis of the muzzle, as described here, prior to 2002. However, it is uncertain if this represents an emerging infectious disease of deer, or an uncommon and widely distributed disease that has only recently been recognized. The ready availability of cellphone cameras, and the rapid dissemination of photographs through social media, could have influenced our awareness of uncommon events. A slight clustering of cases suggests they are not completely random, even though the prevalence is always relatively low.
Although there are no previous reports of granulomatous disease in deer due to M. granulomatis, it has been documented to cause a disease in cows called lechiguana. 10,11 These reports are predominately from Argentina and Brazil, and we are not aware of a similar disease being recognized in cattle from other regions. The bovine lesions are chronic panniculitis, typically distributed along the subcutis over the scapula. Early lesions in cattle are characterized by eosinophilic lymphangitis, but these may progress to pyogranulomatous foci, occasionally with Splendore-Hoeppli material. Lymphangitis was not evident in any of the deer examined in this study, but multiple deer had granulomatous lymphadenitis, suggesting some lymphatic involvement. The consistent granulomatous inflammation with Splendore-Hoeppli material suggests the lesions of the deer were more mature. We speculate that the severe lesions are chronic and, if earlier lesions are milder, they would likely be overlooked by casual observers.
It has been proposed that the distribution of lesions in cattle is due to the migration of Hypoderma bovis, which introduces M. granulomatis into subcutaneous tissues. Mannheimia granulomatis has been infrequently associated with other lesions in cattle, and the possible role as an opportunistic pathogen has not been completely evaluated. 3,4 This species of bacteria is occasionally isolated from the lungs of North American cattle with pneumonia; however, it is thought that it is a secondary invader of lesions in these cases, because the bacteria are not abundant and the culture results are inconsistent (Kristin Clothier, personal communication). It has been isolated from mucosal exudates and D. hominis larvae collected from cattle not affected by lechiguana, on premises where lechiguana has never been documented. 9
Mannheimia granulomatis has also been isolated from lesions in roe deer, including pneumonia, glossitis, and conjunctivitis. 3 However, that report did not discuss if this bacterium caused the lesions, nor did it eliminate the possibility of other primary infections. Another investigation of necrotizing glossitis in a single fallow deer attributed the lesions to M. granulomatis. 8 The authors reported that infections with fungi or bovine viral diarrhea virus were ruled out, but the possibility of M. granulomatis as a secondary contaminant is difficult to eliminate in a mucosal ulcer. In the cases described here, numerous other bacteria were cultured and some of them were initially considered to be possible causes of the disease. However, the morphology and staining patterns of the bacterial colonies in the lesions were inconsistent with many of the species cultured. In addition, PCR testing of bacterial colonies sampled by laser-capture microdissection indicated that the intralesional bacteria visible in histologic sections were M. granulomatis.
Although the lesions associated with this infection were dramatic, mortality has not been attributed to the disease. Most cases are identified by hunters, and there is limited anecdotal evidence suggesting that deer may survive for a year or more with severe muzzle swelling. Information on nutritional condition or intercurrent disease is lacking for most cases due to the limited submission of cranial tissues only. One of the 2 deer for which we could document poor nutritional condition received a full necropsy examination, and it was also diagnosed with granulomatous pneumonia. The chronic lung inflammation was a likely cause of muscle and fat atrophy in that case. Lechiguana of cattle is associated with progressive weakness and death in untreated cases. 10
The reason or mechanism for the tissue tropism of this lesion in deer is uncertain. None of the deer examined had grossly evident lesions in other regions of the skin. Furthermore, we have not identified cases that lacked muzzle inflammation but had this form of pyogranulomatous infiltrates in other tissues. It is possible that small abrasions on the muzzle, superior palpebrum, or dental pad inoculate bacteria into subcutaneous tissues. There has been an anecdotal association between feeding deer and the presence of lesions. However, incomplete historical information and the small number of affected individuals preclude an accurate assessment of the correlation between cellulitis involving the muzzle of deer and artificial food sources. To conclude, pyogranulomatous bacterial infection associated with M. granulomatis should be considered as a cause of extensive soft tissue muzzle swelling in deer.
Footnotes
Acknowledgements
We thank the staff of the Athens Veterinary Diagnostic Laboratory, the California Animal Health and Food Safety Laboratory, the Kansas State University Veterinary Diagnostic Laboratory, and the UC Davis School of Veterinary Medicine for bacterial cultures. Pattie Edwards at the Cellular and Molecular Imaging Core, The Center for Health and the Environment at UC Davis was instrumental in the laser-capture microdissection. Thanks to Barbara Shock, Piyaporn Eiamcharoen, and Arturo Oliver for their feedback and assistance. We owe a special thanks to all the individuals who took the time to assist in the submission of case material, including Cindy Driscoll and George Timko of the Maryland Department of Natural Resources; Tricia Hosch-Hebdon of the Idaho Fish and Wildlife, Health/Forensics Lab; Dale Wallace of Goshen, Indiana; Tom Cooley and Steve Schmitt of the Michigan Department of Natural Resources; Brian Eyler at the Maryland Department of Natural Resources; Chris Nix, Chris Cook, Chuck Sharp, and Fred Pringle with the Alabama Department of Conservation and Natural Resources; Charlie Killmaster from the Georgia Department of Natural Resources; Gerald Moore and Mark Carroll of the South Carolina Department of Natural Resources; Brian Teft of the Rhode Island Division of Fish and Wildlife; Joe Rogerson from the Delaware Division of Fish and Wildlife; Danny Ray and Evan Stanford of the North Carolina Wildlife Resources Commission; Justin Thayer of the Mississippi Department of Wildlife Fisheries and Parks; and Bruce Lynch of Sussex County, Delaware. The assistance of Kevin Niedringhaus in performing necropsies and collecting samples is greatly appreciated. We thank Dennis Wilson for his opinions regarding the pathogenesis of pulmonary lesions observed in association with M. granulomatis. The photograph in ![]() was provided courtesy of the Maryland Department of Natural Resources.
was provided courtesy of the Maryland Department of Natural Resources.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work was provided, in part, by the following agencies: the US Department of the Interior through Grant Agreement 06ERAG0005 with the Biological Resources Division of the US Geological Survey; the Veterinary Services branch of the US Department of Agriculture, Animal and Plant Health Inspection Service, through Grant Agreement 07-9613-0032-CA; and the wildlife management agencies of Alabama, Arkansas, Florida, Georgia, Kansas, Kentucky, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia. SCWDS support from the states was provided by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
